Urolithin A and human cellular aging
Mitochondrial dysfunction and the progressive decline of cellular quality control mechanisms are primary biological drivers of the human aging process. Throughout the lifespan, cells accumulate damaged, inefficient mitochondria, leading to impaired energy production, elevated oxidative stress, and the onset of systemic, low-grade chronic inflammation frequently termed "inflammaging" 123. Urolithin A is a gut microbiome-derived postbiotic metabolite that has emerged in recent geroscience research as a targeted interventional compound for counteracting these age-related cellular deficits. Synthesized by specific human gut bacteria from dietary polyphenols known as ellagitannins and ellagic acid, urolithin A acts as a potent, specific activator of mitophagy - the selective autophagic clearance and recycling of damaged mitochondria 423.
Clinical and preclinical research indicates that urolithin A can enhance mitochondrial bioenergetics, improve skeletal muscle endurance, modulate age-related immune decline, and protect against joint tissue degradation 145. However, the endogenous production of urolithin A is highly variable across populations and heavily dependent on individual microbiome composition, prompting widespread investigation into the efficacy and pharmacokinetics of direct urolithin A supplementation 67. This report provides an exhaustive analysis of the molecular mechanisms, bioavailability dynamics, comparative efficacy against other gerotherapeutics, and clinical trial data surrounding urolithin A.
Molecular Mechanisms of Mitophagy
Urolithin A exerts its geroprotective effects through a highly coordinated network of interconnected cellular signaling pathways. The primary mechanism involves the restoration of mitochondrial quality control via the targeted degradation of defective organelles, supported by the concurrent modulation of energy-sensing kinases and inflammatory cascades.
The PINK1-Parkin Dependent Pathway
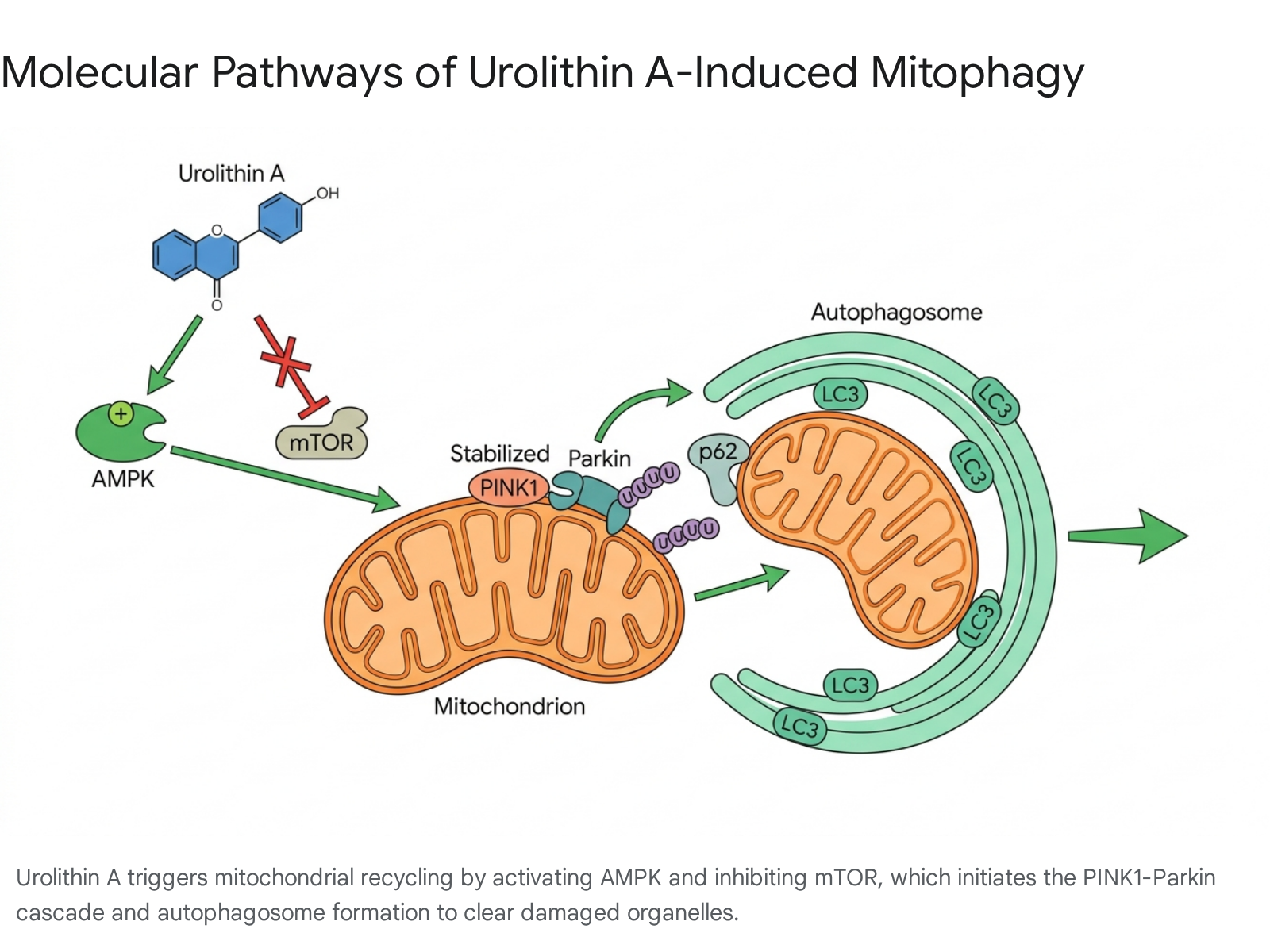
Mitophagy is a specialized form of macroautophagy dedicated to identifying, isolating, and degrading dysfunctional mitochondria, thereby preventing the accumulation of reactive oxygen species (ROS) and mitigating cellular toxicity 3411. The most thoroughly characterized mechanism by which urolithin A induces this process is the PINK1-Parkin-dependent pathway 2411.
Under normal physiological conditions in a healthy mitochondrion, the PTEN-induced putative kinase 1 (PINK1) is rapidly imported into the inner mitochondrial membrane and subsequently degraded. However, when a mitochondrion sustains damage and loses its membrane potential, PINK1 stabilizes and accumulates on the outer mitochondrial membrane (OMM) 12138. Urolithin A promotes the stabilization of PINK1, which subsequently acts as a molecular beacon signaling mitochondrial dysfunction 4.
Once stabilized, PINK1 recruits Parkin, a cytosolic E3 ubiquitin ligase, to the mitochondrial surface 49. Activated by PINK1-mediated phosphorylation, Parkin ubiquitinates various outer membrane proteins, including voltage-dependent anion channel 1 (VDAC1) and mitofusin 2 (Mfn2) 349. These poly-ubiquitin chains serve as high-affinity docking sites for specific autophagy adaptor proteins, such as p62/SQSTM1, OPTN (optineurin), TAX1BP1, and NDP52 3139. These adaptors tether the damaged mitochondrion to the microtubule-associated protein 1A/1B-light chain 3 (LC3) situated on the expanding phagophore membrane. The phagophore ultimately engulfs the organelle to form a double-membraned mitophagosome, which fuses with a lysosome, leading to the enzymatic degradation and recycling of the mitochondrial components into basic cellular building blocks 3411.
Ubiquitin-Independent Mitophagy Pathways
In addition to the classical PINK1-Parkin cascade, evidence indicates that urolithin A influences ubiquitin-independent mitophagy pathways. These alternative pathways rely on specialized mitophagy receptors localized on the outer mitochondrial membrane that contain LC3-interacting regions (LIRs), allowing them to directly bind the autophagosomal machinery without the prerequisite of ubiquitin priming 121011.
Key receptors in this pathway include BNIP3 (BCL2-interacting protein 3), NIX (also known as BNIP3L), and FUNDC1 (FUN14 domain-containing 1) 410. Research indicates that urolithin A can activate mitophagy in specific tissues independent of Parkin recruitment 912. For instance, in certain progenitor cells and within specific neurodegenerative models, the clearance of depolarized mitochondria is mediated primarily by BNIP3L and FUNDC1 rather than the PINK1-Parkin axis 912. The dual activation of both ubiquitin-dependent and ubiquitin-independent pathways highlights the broad physiological capacity of urolithin A to enforce mitochondrial quality control across diverse cellular and tissue environments.
Energy Sensing via AMPK Activation and mTOR Inhibition
The induction of mitophagy by urolithin A is tightly regulated by upstream nutrient and energy-sensing pathways, specifically the activation of AMP-activated protein kinase (AMPK) and the inhibition of the mechanistic target of rapamycin (mTOR) 21113.
AMPK functions as the central energy sensor of the cell. When cellular energy levels drop - typically indicated by an increased AMP/ATP ratio - AMPK is activated to stimulate catabolic processes that generate ATP while simultaneously inhibiting energy-consuming anabolic processes 1113. Urolithin A acts as a robust AMPK activator, signaling the cell to initiate mitophagy to clear inefficient mitochondria and restore energy homeostasis 211. This AMPK-boosting effect is particularly significant in the context of aging, where diminished endogenous AMPK activity contributes heavily to progressive mitochondrial dysfunction and oxidative stress 11.
Conversely, mTOR is a primary regulator of cell growth and protein synthesis. Hyperactive mTOR signaling is a well-documented hallmark of aging that actively suppresses autophagy and mitophagy 112014. Urolithin A suppresses the mTOR pathway, relieving this physiological blockade and allowing the autophagic machinery to proceed unhindered 23. Furthermore, the activation of AMPK by urolithin A stimulates the expression of PGC-1α (Peroxisome proliferator-activated receptor-gamma coactivator 1-alpha), the master transcriptional regulator of mitochondrial biogenesis 1312. This creates a highly efficient, dual-action cycle of mitochondrial renewal: urolithin A clears damaged organelles via mitophagy while simultaneously stimulating the production of new, healthy mitochondria via PGC-1α 112.
Immunomodulation and Anti-Inflammatory Pathways
Systemic low-grade inflammation is inextricably linked to mitochondrial dysfunction. Severely damaged mitochondria release mitochondrial DNA (mtDNA) and highly reactive oxygen species into the cytosol, triggering innate immune sensors such as the cGAS-STING pathway and the NLRP3 inflammasome 1516.
By efficiently clearing damaged mitochondria before they can rupture or leak their contents, urolithin A removes the primary intracellular source of these inflammatory triggers. Beyond this indirect clearance, urolithin A directly modulates inflammatory signaling by inhibiting the NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells) pathway, resulting in the decreased downstream production of pro-inflammatory cytokines such as IL-6, TNF-α, and IL-1β 121317. Concurrently, it stimulates the Nrf2 (Nuclear factor erythroid 2-related factor 2) signaling pathway. The activation of Nrf2 upregulates the expression of endogenous antioxidant enzymes and glutathione S-transferases (GSTs), thereby neutralizing oxidative stress at the cellular level and fortifying cellular resilience against future damage 1218.
The Gut Microbiome and Urolithin Metabotypes
Urolithin A is not naturally abundant in food sources in its active, bioavailable form. Rather, it is a postbiotic metabolite generated exclusively through the complex enzymatic transformation of dietary precursors by the human gut microbiome 1920. The primary dietary precursors are ellagitannins (hydrolyzable tannins) and ellagic acid, which are highly concentrated in foods such as pomegranates, walnuts, pecans, raspberries, and strawberries 192122.
Following ingestion, ellagitannins are hydrolyzed in the acidic environment of the stomach and upper gastrointestinal tract to yield ellagic acid. Upon reaching the colon, ellagic acid undergoes a sequential series of lactone-ring cleavages, decarboxylations, and dehydroxylations facilitated by specific microbial consortia. This metabolic cascade produces various urolithin intermediates, such as Urolithin D and Urolithin C, before culminating in the production of the terminal metabolites Urolithin A, Iso-Urolithin A, and Urolithin B 202122.
Classification of Urolithin Metabotypes
The capacity to produce urolithin A is not a universal human trait. Populations exhibit profound inter-individual variability in their colonic microbiome composition, leading to the categorization of individuals into specific "urolithin metabotypes" based on their end-stage metabolite production profile following the consumption of ellagitannins 67.
The specific gut bacteria responsible for these critical biotransformations include species primarily from the Eggerthellaceae family (such as Gordonibacter urolithinfaciens and Gordonibacter pamelaeae), as well as specific strains like Enterocloster bolteae, Bifidobacterium pseudocatenulatum, and Streptococcus thermophilus 7232425. Individuals lacking adequate populations of these specific microbial strains are functionally incapable of converting dietary ellagic acid into circulating urolithin A, regardless of the quantity of precursor-rich foods they consume 722.
| Metabotype Classification | Metabolite Production Profile | Clinical and Microbiome Characteristics |
|---|---|---|
| Metabotype A (UM-A) | Produces exclusively Urolithin A | Characterized by a higher Firmicutes/Bacteroidetes ratio and greater overall microbial diversity. These individuals exhibit the most pronounced therapeutic responses to natural dietary ellagitannins 723. |
| Metabotype B (UM-B) | Produces Urolithin A, Iso-Urolithin A, and Urolithin B | Often features distinct microbial taxa compared to UM-A. Individuals may require substantially higher dietary intakes or direct supplementation to achieve optimal circulating Urolithin A levels 7. |
| Metabotype 0 (UM-0) | Non-producers; unable to synthesize Urolithins A, Iso-A, or B | Lacks the necessary Gordonibacter or Enterocloster species. These individuals derive minimal targeted mitochondrial benefit from dietary ellagitannins and rely entirely on direct supplementation for exposure 67. |
Dietary Influences and Global Prevalence
The distribution of these urolithin metabotypes varies significantly across global populations and is heavily influenced by long-term dietary patterns. Clinical studies reveal that only 30% to 40% of the general population in the United States and other Western nations possess the UM-A phenotype capable of producing meaningful levels of urolithin A from diet alone 2126. In a cohort of healthy American adults, only 12% exhibited detectable circulating urolithin A at baseline, which increased to approximately 40% following a controlled dietary challenge with pure pomegranate juice 23. Consequently, up to 60% of individuals consuming a standard Western diet may be functional non-producers (categorized as UM-0 or extremely low-yield producers) 734.
Conversely, populations adhering to plant-rich, high-fiber diets, particularly the Mediterranean diet, demonstrate a significantly lower prevalence of the UM-0 metabotype. Epidemiological data indicate that only about 10% of individuals adhering to a traditional Mediterranean diet in Spain are classified as non-producers, while roughly 14% of Chinese subjects exhibit the UM-0 phenotype 726. The Mediterranean diet promotes a highly diverse microbiome enriched with fiber-degrading taxa such as Faecalibacterium prausnitzii, Prevotella, and Bifidobacterium. This creates a favorable, robust microbial ecosystem that facilitates efficient ellagic acid metabolism 272829. The Western diet, characterized by high intakes of saturated fats, refined sugars, and low dietary fiber, actively suppresses this required microbial diversity, leading to a much higher prevalence of urolithin non-producers 2829.
Bioavailability and Pharmacokinetics of Supplementation
Due to the unreliability of endogenous urolithin A production and the high prevalence of the UM-0 metabotype, clinical and longevity research has largely shifted focus toward the use of direct oral supplementation. Synthesized urolithin A is administered as a direct postbiotic, bypassing the prerequisite for microbial conversion entirely and standardizing systemic exposure across all human metabotypes 2334.
Dietary Precursors Versus Direct Supplementation
Relying solely on dietary sources to achieve therapeutic levels of urolithin A presents significant pharmacokinetic limitations. Ellagitannins and ellagic acid possess notoriously poor bioavailability; ellagic acid reaches maximum plasma concentrations of only roughly 120 ng/mL in humans due to poor gastrointestinal absorption and adherence to the Lipinski rule of five 21. While the resulting microbially produced urolithins are estimated to be 25 to 80 times more bioavailable than their parent precursors, their synthesis remains entirely bottlenecked by the host's specific microbial metabotype 2021.
Direct supplementation ensures consistent, reproducible, and measurable delivery of the active molecule into systemic circulation. Rigorous pharmacokinetic profiling reveals a stark contrast in bioavailability between food sources and direct supplementation. In a cohort trial comparing interventions, direct administration of 500 mg of formulated urolithin A resulted in peak plasma levels that were greater than six-fold higher than those achieved by consuming 8 ounces (240 mL) of 100% pure pomegranate juice 2338.
| Administration Method | Baseline UA Glucuronide (ng/mL) | Peak UA Glucuronide (ng/mL) | Time to Peak Concentration | Efficacy Across Metabotypes |
|---|---|---|---|---|
| 8 oz Pomegranate Juice | 5.48 ± 19.97 | ~110.47 ± 131.6 | 24 Hours | Highly variable; effective in ~40% of subjects 23. |
| 500 mg Urolithin A Supplement | 9.57 ± 47.78 | ~480.75 ± 238.0 | 6 Hours | Consistent exposure independent of gut microbiome 23. |
Dosing Efficacy and Metabolism
Upon oral administration, direct urolithin A is rapidly absorbed through the intestinal epithelium. In healthy human cohorts, single and repeated daily doses ranging from 250 mg to 1,000 mg produce reliable, dose-dependent plasma concentrations 2630. As noted in the comparative data, the peak plasma concentration ($T_{max}$) occurs approximately 6 hours post-ingestion 2326.
Once in systemic circulation, urolithin A undergoes extensive phase II metabolism in the liver and intestine, circulating primarily as urolithin A-glucuronide and urolithin A-sulfate 2640. The elimination half-life of these metabolites is approximately 17 to 24 hours, suggesting that a once-daily dosing regimen is sufficient to maintain steady-state therapeutic concentrations 2631. Mechanistic studies indicate that the compound readily dissociates back into its active aglycone (monomer) form upon reaching target tissues, where it penetrates the cellular membrane to exert its biological activity on the mitochondrial network 2630.
Clinical Efficacy in Skeletal Muscle and Physical Endurance
Sarcopenia - the progressive, age-associated loss of skeletal muscle mass and function - alongside general declines in muscular endurance, are intimately linked to compromised mitochondrial bioenergetics within muscle tissue. As mitochondrial turnover slows with age, muscle cells lose their primary capacity for efficient ATP generation. Urolithin A has been extensively evaluated in randomized controlled trials for its capacity to reverse these specific declines.
In a hallmark 2022 randomized, double-blind, placebo-controlled trial published in Cell Reports Medicine (Singh et al.), researchers investigated 88 healthy, overweight, middle-aged adults. Participants received either 500 mg or 1,000 mg of urolithin A daily for four months 52225. The intervention resulted in a statistically significant increase in hamstring muscle strength (yielding up to a 12% improvement in the 1,000 mg group) and enhanced overall aerobic exercise performance 225. Crucially, skeletal muscle biopsies collected during the trial confirmed a molecular signature of active mitophagy, demonstrating significant upregulations in PINK1, Parkin, and associated proteasomal system proteins compared to the placebo group 425.
A parallel trial published in JAMA Network Open (Liu et al., 2022) investigated a cohort of 66 older adults (aged 65 to 90) over a four-month period. While the primary predefined endpoint of increased 6-minute walking distance (6MWT) was not statistically significant when compared directly to the placebo group, the urolithin A cohort demonstrated notable, significant improvements in skeletal muscle endurance and resistance to fatigue, alongside significant reductions in systemic inflammatory markers such as C-reactive protein (CRP) 62232.
Further validating its role beyond aging populations and into sports nutrition, the 2025 ENDURO trial evaluated urolithin A in elite athletes. The study observed that four weeks of 1,000 mg daily urolithin A supplementation in 42 highly-trained male distance runners significantly improved endurance performance capacity and optimized markers of mitochondrial efficiency during intense physical exertion 543.
Immunomodulation and the MitoImmune Trial
As human populations age, the immune system undergoes a profound process of senescence, commonly referred to as immunosenescence. This decline is characterized by a drastic reduction in the production of naive immune cells, the accumulation of exhausted and dysfunctional memory cells, and a persistent state of chronic low-grade inflammation 12. The 2025 "MitoImmune" randomized, placebo-controlled trial, published in Nature Aging (Denk et al.), provided rigorous clinical evidence that urolithin A can actively counteract this immune decline by directly targeting the underlying mitochondrial architecture of immune cells 13.
In this trial, 50 healthy middle-aged adults (aged 45 - 70) received 1,000 mg of urolithin A daily for 28 days 144. The intervention resulted in profound phenotypic, functional, and metabolic remodeling of the immune system:
- Expansion of Naive T Cells: Urolithin A supplementation significantly increased the peripheral circulating levels of naive-like CD8+ T cells ($CD8^+ T_N$). These fresh, adaptable cells are critical for recognizing and mounting defenses against novel pathogens and typically dwindle in older populations 13.
- Reversal of Immune Exhaustion: The expression of TOX, a master regulatory gene that drives T cell exhaustion, was notably reduced. Concurrently, researchers observed an increase in the expression of Ki-67, an established marker of cellular proliferation and reinvigoration 1.
- Enhancement of Innate Immunity: The urolithin A cohort exhibited an expansion of CD56$^{dim}$CD16$^{bright}$ Natural Killer (NK) cells, the immune system's first responders against infected and malignant cells. Furthermore, monocytes demonstrated significantly improved phagocytic capabilities, shown by an enhanced ability to engulf Escherichia coli bacterial particles in laboratory assays 13.
Beyond cellular counts, the MitoImmune trial revealed that urolithin A induces a fundamental metabolic reprogramming within the immune cells themselves. Data indicates that T cells from the urolithin A group shifted away from an inefficient reliance on glucose metabolism. Instead, they demonstrated a significantly enhanced capacity for mitochondrial fatty acid oxidation (FAO) and amino acid oxidation (AAO) 13.
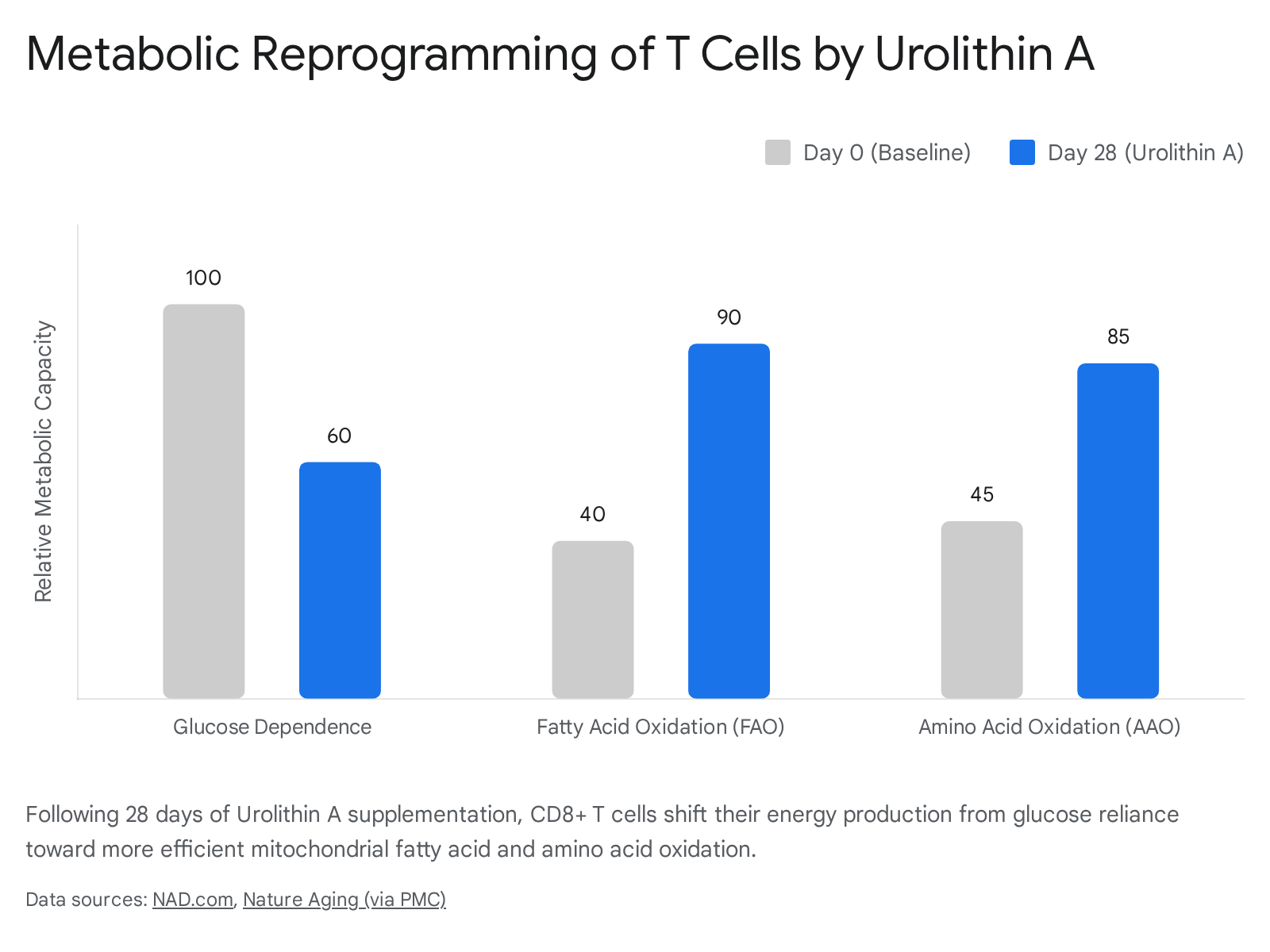
This metabolic flexibility is a hallmark of youthful, highly resilient immune cells. The shift was accompanied by the increased expression of PGC-1α, confirming that the initiation of mitophagy by urolithin A was actively followed by the biogenesis of new, metabolically superior mitochondria 13.
Joint Health, Osteoarthritis, and Systemic Horizons
Mitochondrial dysfunction within chondrocytes (cartilage-producing cells) is a major driver of joint aging and the pathological progression of osteoarthritis (OA). As chondrocytes accumulate damaged mitochondria, they lose the energy necessary to maintain the cartilage matrix and begin secreting inflammatory, degradative enzymes.
In a pivotal 2022 study published in Aging Cell (D'Amico et al.), researchers demonstrated that urolithin A effectively enhances mitochondrial respiration and ATP production in primary human chondrocytes sourced from both healthy donors and patients with active OA 53334. When tested in highly controlled in vivo murine models of knee osteoarthritis (destabilization of the medial meniscus models), two months of urolithin A administration significantly increased mitophagy flux within the joint tissue 3334.
This physiological cellular upgrade resulted in the preservation of cartilage structural integrity, reduced chondrocyte apoptosis, and notably decreased subjective joint pain 3334. Furthermore, urolithin A exhibited localized anti-inflammatory properties within the joint capsule, reducing synovitis and suppressing the production of cartilage-degrading enzymes and circulating OA biomarkers, such as MMP1 1834.
Beyond muscle and joint health, preclinical data suggests urolithin A possesses strong neuroprotective capabilities. In murine models of Alzheimer's disease, the restoration of mitophagy via urolithin A supplementation reduced the accumulation of pathological amyloid-beta and hyperphosphorylated tau proteins, subsequently improving cognitive performance and memory in aged mice 119. As clinical research accelerates, ongoing human trials are currently investigating urolithin A's efficacy in managing metabolic syndrome (NCT07377136), improving endothelial function (NCT05921266), and lowering oxidative stress in prostate tumor tissue prior to radical prostatectomy (NCT06022822) 353637.
Comparative Analysis with Other Gerotherapeutics
In the rapidly expanding landscape of geroprotective pharmacology, urolithin A is frequently compared to other advanced, pathway-specific supplements. Most notably, it is evaluated alongside NAD+ precursors (NMN, NR), mTOR inhibitors (Rapamycin), and senolytics (Quercetin, Fisetin). Each of these compounds addresses distinct, albeit overlapping, cellular hallmarks of aging.
Urolithin A Versus NAD+ Precursors (NMN and NR)
Nicotinamide mononucleotide (NMN) and nicotinamide riboside (NR) function as direct biochemical precursors to NAD+ (Nicotinamide adenine dinucleotide), a critical cellular coenzyme required for sirtuin activation, DNA repair, and fundamental oxidative metabolism 414. Intracellular NAD+ levels decline precipitously with age, fundamentally impairing the efficiency of the mitochondrial respiratory chain.
While both urolithin A and NAD+ boosters heavily target mitochondrial function, their biological roles are mechanically distinct but highly complementary. NMN provides the essential chemical "fuel" necessary to drive oxidative phosphorylation and support metabolic enzymatic reactions 1950. Urolithin A, conversely, focuses entirely on the structural and functional "hardware" of the cell. It triggers mitophagy, destroying physically degraded and inefficient mitochondria to make room for structurally sound replacements 419.
Current clinical analysis suggests that utilizing both compounds concurrently may yield a synergistic effect: urolithin A ensures the cellular mitochondrial "engines" are physically renewed and optimized, while NMN ensures those upgraded engines possess the necessary fuel substrate to operate at maximum capacity 1951. Furthermore, a 2024 study published in Frontiers in Aging Neuroscience demonstrated that while both urolithin A and NR effectively reduce cellular senescence in human microglial cells, they achieve this through distinct neuroinflammatory pathways, reinforcing the argument for their complementary, rather than interchangeable, nature 1551.
Urolithin A Versus Rapamycin and Senolytics
Rapamycin is a potent, FDA-approved pharmacological inhibitor of the mTOR pathway. In longevity research, it acts as a robust calorie restriction mimetic that broadly enhances macroautophagy and completely suppresses cellular growth signaling 1452. While rapamycin is widely considered the gold standard for lifespan extension in preclinical models - extending murine lifespan by up to 25% - its translation to healthy, aging humans is highly complicated 3839. Systemic mTOR inhibition via rapamycin presents a narrow therapeutic window, with significant risks of dose-dependent immunosuppression, insulin resistance, and metabolic dysregulation, limiting its viability for broad off-label anti-aging use 143839. Urolithin A shares the mTOR-inhibiting characteristic of rapamycin but acts highly selectively. It localizes its autophagic effects to the mitochondria (specifically mitophagy) without inducing the severe systemic immunosuppressive side effects seen with high-dose rapalogs 220.
Senolytics, such as Dasatinib and the naturally occurring flavonoid Quercetin, target an entirely different hallmark of aging: cellular senescence. These compounds selectively induce apoptosis in "zombie" cells that have ceased dividing and continuously secrete toxic inflammatory signals (the senescence-associated secretory phenotype, or SASP) 14. While senolytics operate at the tissue level to clear toxic, non-functional cells entirely, urolithin A operates at the sub-cellular level to clear toxic, non-functional organelles within surviving, otherwise healthy cells 2014.
| Gerotherapeutic Compound | Primary Biological Target | Mechanism of Action | Clinical Safety Profile (Healthy Adults) |
|---|---|---|---|
| Urolithin A | Mitochondrial Quality Control | Induces targeted mitophagy; activates AMPK; selectively inhibits mTOR 214. | Excellent; highly well-tolerated in trials up to 1,000 mg/day with no serious adverse events 1730. |
| NMN / NR | Cellular Metabolism & DNA Repair | Boosts intracellular NAD+ levels; provides substrate for sirtuin activation 1419. | High safety profile; rapidly alters metabolomic biomarkers without major toxicity 1451. |
| Rapamycin | Macroautophagy & Growth Signaling | Broad systemic mTOR inhibition; acts as a potent calorie restriction mimetic 2014. | Narrow therapeutic window; carries risks of immunosuppression and metabolic dysregulation 1439. |
| Senolytics (e.g., Quercetin) | Cellular Senescence | Induces targeted apoptosis specifically in senescent cells 1455. | Variable; generally safe for natural compounds, but carries high toxicity risks for pharmaceutical options (e.g., Dasatinib) 14. |
Safety Profile, Interactions, and Calibrated Uncertainty
As urolithin A transitions from specialized preclinical research to broader clinical applications and consumer availability, rigorous safety evaluations have been paramount. Given its origin as a natural, gut-derived microbial metabolite, the compound possesses a highly favorable toxicological profile, though understanding of its long-term interactions is still maturing.
Clinical Tolerability and Side Effects
Across multiple human clinical trials involving both acute single doses and chronic administration over periods of up to six months, urolithin A has consistently demonstrated excellent tolerability. In clinical interventions delivering doses of 500 mg to 1,000 mg per day to healthy elderly and middle-aged cohorts, participants experienced no significant adverse events attributed directly to the compound 173056. Blood and urine panels confirmed no evidence of hepatotoxicity (normal liver function markers), and no disruptions to renal or cardiovascular homeostasis 1730. Based on this extensive toxicological data, the U.S. Food and Drug Administration (FDA) has granted synthetic urolithin A "Generally Recognized as Safe" (GRAS) status for use as a dietary supplement ingredient 4.
Furthermore, extensive microbiome analysis confirms that administering high-dose synthetic urolithin A does not negatively alter the delicate balance of the host's existing gut microbiota. Direct supplementation does not cause bacterial die-offs, trigger radical microbial reconfigurations, or prompt endogenous microbial pathways to downregulate their natural ellagitannin-converting capabilities 17.
Despite this robust short- and medium-term safety data, calibrated uncertainty must be applied to the current state of the research. Multi-year longitudinal studies regarding the systemic effects of sustained, elevated mitophagy rates in humans are not yet available 1756. Furthermore, as highlighted by Kuerec et al. in a 2024 systematic review published in Ageing Research Reviews, several of the landmark clinical trials evaluating urolithin A have been funded or co-authored by researchers affiliated with the companies manufacturing the synthetic compound, indicating an inherent potential for industry bias that requires further independent validation through large-scale, third-party clinical trials 225556.
Potential Pharmacological Interactions
While formal contraindications for urolithin A have not been established on its FDA label, its physiological metabolic pathways suggest theoretical areas for caution, particularly for populations managing complex chronic diseases or polypharmacy.
Because urolithin A undergoes extensive phase II metabolism in the liver to form glucuronides and sulfates, there is the potential for competitive interactions with the Cytochrome P450 (CYP450) enzyme system. The concomitant use of drugs that heavily induce or inhibit CYP450 - such as certain statins (e.g., atorvastatin), specific antibiotics (e.g., clarithromycin), antifungals (ketoconazole), or immunosuppressants (cyclosporine) - could theoretically alter the clearance rate, peak plasma levels, and half-life of urolithin A, though the clinical significance of these interactions remains unproven 134058.
Regarding cardiovascular medications, particularly anticoagulant and antiplatelet therapies (e.g., warfarin, apixaban, clopidogrel), there is currently no clinical or pharmacological evidence indicating that urolithin A possesses intrinsic blood-thinning properties 5841. It does not increase perioperative bleeding risks and does not mandate discontinuation prior to surgical procedures or regional anesthesia under current medical guidelines 41. Nonetheless, due to the limited scope of specific, long-term drug-drug interaction trials, calibrated medical oversight is advised when combining urolithin A with complex, systemic prescription regimens.
Conclusions
Urolithin A represents a significant biological advancement in the field of nutritional geroscience, functioning as the most rigorously validated, direct inducer of mitophagy currently available for human application. By bypassing the genetic and dietary constraints of the gut microbiome through direct supplementation, the compound achieves high, reliable systemic bioavailability regardless of individual metabotype. At the molecular level, its ability to activate energy-sensing AMPK, suppress the growth-promoting mTOR pathway, and stimulate the PINK1/Parkin cascade results in the highly efficient recycling of damaged mitochondria.
Clinical trial data continuously confirms that this localized, sub-cellular cleanup translates into profound systemic physiological benefits, including increased skeletal muscle strength, enhanced aerobic endurance, the structural preservation of joint cartilage, and the metabolic rejuvenation of the aging immune system. With a highly favorable safety profile and distinct, complementary mechanisms to other longevity interventions like NAD+ precursors, urolithin A offers a targeted, biologically sound intervention to mitigate the mitochondrial and inflammatory hallmarks of human aging. Continued independent, long-term clinical research will be essential to fully elucidate its optimal dosing protocols and multi-year efficacy across diverse populations.