Transmission and genomics of 2025-2026 Andes orthohantavirus clusters
Environmental Drivers and Terrestrial Ecology
The emergence and spillover of zoonotic pathogens are fundamentally tied to the ecological dynamics of their terrestrial reservoirs. In the case of the Andes orthohantavirus (ANDV), the primary recognized reservoir is the long-tailed pygmy rice rat (Oligoryzomys longicaudatus), a species deeply integrated into the temperate Valdivian rainforests and Andean-Patagonian forests of southern Chile and Argentina 123. The epidemiological trajectory of the 2025-2026 ANDV clusters was heavily influenced by regional climatic extremes and the ecological cycles of local vegetation, which collectively drove increased rodent density and human-wildlife interface.
Bamboo Masting Cycles and Rodent Dynamics
The population density of O. longicaudatus is highly susceptible to "pulsed resources" in the environment. The most significant of these ecological pulses is the gregarious, synchronous flowering (masting) of native woody bamboo species, primarily Chusquea culeou and Chusquea quila 456. These bamboo species function as keystone elements in the forest understory and exhibit monocarpy, meaning they remain in a vegetative state for decades before a massive, synchronized blooming event occurs, followed by widespread plant death and seed drop 278.
The reproductive cycles of these bamboos vary significantly by species. C. quila typically exhibits a modal flowering cycle of 12 to 15 years, while C. culeou possesses a much longer cycle, historically recorded between 30 and 60 years 679. When these masting events occur, the sudden, overwhelming availability of highly nutritious seeds triggers an explosive reproductive response in granivorous rodents. This sudden population irruption is locally termed a ratada 4610. Historical data confirms that numerous ratadas in southern South America over the past century have directly followed Chusquea blooms, leading to sharp increases in hantavirus spillover events as starving, hyper-abundant rodents subsequently invade human settlements and agricultural areas in search of alternative food sources during the winter die-off 41011.
While comprehensive botanical field confirmation of a pan-Patagonian Chusquea masting event specific to 2025 is not definitively established in the literature, environmental monitoring indicates that the ecological preconditions for severe rodent displacement were highly active 1112. Additionally, historical records note that the aftermath of bamboo flowering and death leaves vast quantities of dry, highly flammable biomass in the forest understory, drastically altering soil properties and compounding the risk of environmental disturbances 13.
Climate Anomalies in Patagonia
Compounding the biological cycles of the reservoir host, the late 2025 and early 2026 seasons featured severe climate anomalies that significantly disrupted the Patagonian ecosystem. Driven by a persistent La Niña phase and the Southern Annular Mode, the region experienced extreme anticyclonic anomalies that established warm and profoundly dry conditions 1416.
Meteorological data indicates that precipitation levels from November 2025 to January 2026 dropped by approximately 20% in Argentine Patagonia and 25% in central Chile compared to pre-industrial baselines 1416. This acute moisture deficit led to severe soil stress and vegetation die-off. Utilizing the heat-dryness-wind index (HDWI), climatologists determined that the likelihood of extreme fire conditions increased by a factor of 2.5 in Argentine Patagonia and 3.0 in central Chile due to anthropogenic global warming and these acute cyclical anomalies 1415.
The resulting environmental degradation - marked by over 3,800 focal forest fires recorded in Chile since July 2025 and severe outbreaks in Argentina's Los Alerces National Park - destroyed vast tracts of primary O. longicaudatus habitat 1416. This widespread habitat destruction is an established driver of zoonotic spillover, as displaced rodent populations are forced into peri-urban and rural human environments, thereby escalating the probability of human exposure to aerosolized rodent excreta 316.
Regional Epidemiology in South America
The confluence of ecological disturbances and displaced rodent populations resulted in a highly active and lethal epidemiological season for hantavirus cardiopulmonary syndrome (HCPS) in both Argentina and Chile during 2025 and early 2026.
Incidence Trends in Argentina
The Argentine public health apparatus reported a historically severe outbreak period. According to the National Epidemiological Bulletin, Argentina recorded 101 to 102 confirmed cases of hantavirus between the beginning of the epidemiological season in early July 2025 and the end of April 2026 17181920. This figure maintained the national incidence rate at 0.21 cases per 100,000 inhabitants, keeping the country above the official outbreak threshold for nearly the entire analyzed period 1720.
The 2025-2026 case count represents a 77% increase over the preceding 2024-2025 season (which saw 57 cases) and easily surpassed recent elevated seasons in 2023-2024 (75 cases) and 2019 2021. Within the calendar year of 2026 alone, 41 to 42 new infections were recorded by May 171820. The geographic distribution of the virus was widespread but concentrated in specific regions. The Central Region accounted for 54% of national cases, heavily driven by the province of Buenos Aires 1718. However, the Northwest Region exhibited the highest per capita incidence rate at 0.60 cases per 100,000 inhabitants, with the province of Salta accounting for 83% of the cases in that specific zone 1718.
Despite a lower absolute case count in the Southern Region (Patagonia), the epidemiological profile of the cases in provinces like Chubut, Río Negro, and Neuquén was critical due to the endemicity of the highly virulent Andes orthohantavirus 111721. During the 2025-2026 season, authorities closely monitored an intrafamilial cluster in the locality of Cerro Centinela (Chubut) involving three cohabitating individuals. Subsequent environmental focus studies in the area captured wild rodents that tested positive for ANDV, confirming active local zoonotic transmission 19. Nationally, the outbreak resulted in 32 confirmed deaths, pushing the case fatality rate (CFR) for the Argentine season to 31.7%, a figure matching the upper bounds of historical lethality for the Andes strain 1820.
Epidemiological Situation in Chile
Parallel viral activity was documented in Chile, which shares the Patagonian ecosystem and the O. longicaudatus reservoir. The Chilean Ministry of Health (MINSAL) reported a sharp increase in lethality and widespread geographic distribution of the virus 162223.
By the first week of May 2026, Chile had confirmed 39 to 44 cases of HCPS since the beginning of the year 22232425. The infections were distributed across nine of the country's regions, spanning from the Metropolitan area down to Aysén, including O'Higgins, Maule, Ñuble, Biobío, La Araucanía, Los Ríos, and Los Lagos 2224. The outbreak claimed 13 lives, establishing a CFR of 33% 2223. This represents a significant escalation in virulence or a shift in diagnostic capture compared to the prior year; in 2025, Chile recorded 44 cases total with 8 deaths, yielding a CFR of 18% 161823.
Public health officials maintained a continuous epidemiological alert, noting that the Andes variant is the sole etiologic agent of human hantavirus confirmed in Chile 2224. To mitigate exposure in the context of the elevated case counts, authorities advised strict environmental controls, including ventilating enclosed rural spaces for at least 30 minutes prior to entry and disinfecting surfaces with a chlorine-water solution to neutralize viral particles 1622.
Table 1 summarizes the regional epidemiological statistics for the 2025-2026 season prior to and concurrent with the maritime cluster.
| Jurisdiction | Timeframe | Confirmed Cases | Confirmed Deaths | Case Fatality Rate (CFR) | Notes |
|---|---|---|---|---|---|
| Argentina | July 2025 - April/May 2026 | 101 - 102 | 32 | 31.7% | 77% case increase over previous season; high incidence in Central/NW regions. |
| Chile | Jan 2026 - May 2026 | 39 - 44 | 13 | ~33.0% | Distributed across 9 regions; active epidemiological alert due to increased CFR. |
| MV Hondius | April 2026 - May 2026 | 8 - 11 | 3 | ~27.0% - 37.5% | Multinational maritime cluster; initial zoonotic exposure hypothesized in Argentina. |
The MV Hondius Cruise Ship Outbreak
Against the backdrop of heightened regional endemicity in South America, an anomalous and severe cluster of ANDV emerged in April 2026 aboard the Dutch-flagged expedition cruise ship, the MV Hondius. The event represents one of the most operationally complex hantavirus outbreaks in history, demonstrating how long-incubation pathogens can intersect with maritime environments and global travel networks 262927.
Outbreak Timeline and Case Progression
The MV Hondius is an expedition vessel specialized in navigating remote polar and sub-polar environments. On April 1, 2026, the ship departed from Ushuaia, Argentina - the southernmost city in Tierra del Fuego - carrying between 147 and 149 passengers and crew members representing 23 different nationalities 26282930. The vessel's transatlantic itinerary included stops across isolated environments in the South Atlantic, including mainland Antarctica, South Georgia, Nightingale Island, Tristan da Cunha, Saint Helena, and Ascension Island, with a scheduled route toward Cabo Verde 263031.
The index case (Case 1), an adult male, began exhibiting symptoms on April 6, 2026, a mere five days after the vessel's departure from Argentina 2729. The initial clinical presentation included fever, headache, abdominal pain, and diarrhea - symptoms that align with the non-specific prodromal phase of hantavirus infection 2729. Driven by the rapid cardiopulmonary deterioration characteristic of ANDV, the index case died onboard on April 11 2729. At the time, the specific etiology of the illness remained unknown to the ship's medical staff.
The outbreak remained epidemiologically opaque for several weeks. During a scheduled stop in Saint Helena on April 24, approximately 30 passengers disembarked, including the wife of the index case (Case 2), while the body of Case 1 was removed from the vessel 2729. Most of these disembarked passengers boarded commercial flights, transiting through Johannesburg, South Africa, to reach their home countries 2932. Shortly after arriving and collapsing at the airport in Johannesburg, Case 2 died 2729. A third passenger (Case 3), who had fallen ill aboard the ship on April 22, was subsequently medically evacuated to South Africa on April 27, where he was admitted to an intensive care unit (ICU) 293133.
Diagnostic clarity was finally achieved in early May. On May 2, polymerase chain reaction (PCR) testing of serum and blood samples from Case 3 confirmed hantavirus infection 2933. The following day, May 3, testing confirmed the presence of hantaviral RNA in the post-mortem samples of Case 2 2931.
Evacuation and Contact Tracing Operations
Genomic sequencing conducted by the National Institute for Communicable Diseases (NICD) in South Africa, alongside affiliated institutes in Switzerland and the Netherlands, rapidly identified the pathogen as the Andes orthohantavirus 2934. Because ANDV is the only hantavirus strain with documented human-to-human transmissibility, the confirmation prompted an immediate international health emergency response coordinated by the World Health Organization (WHO) and the European Centre for Disease Prevention and Control (ECDC) 29283031.
By May 12, 2026, the cluster had grown to include between eight and eleven suspected or confirmed cases, including three fatalities 17272835. Due to the severe nature of the illness, the MV Hondius was held stationary off the coast of Cabo Verde, as local facilities lacked the capacity for safe medical evacuation 2728. Once clearance was granted by the Spanish Ministry of Health, the vessel sailed to Tenerife in the Canary Islands, where final medical evacuations and repatriations were conducted under stringent biocontainment protocols on May 10 2728.
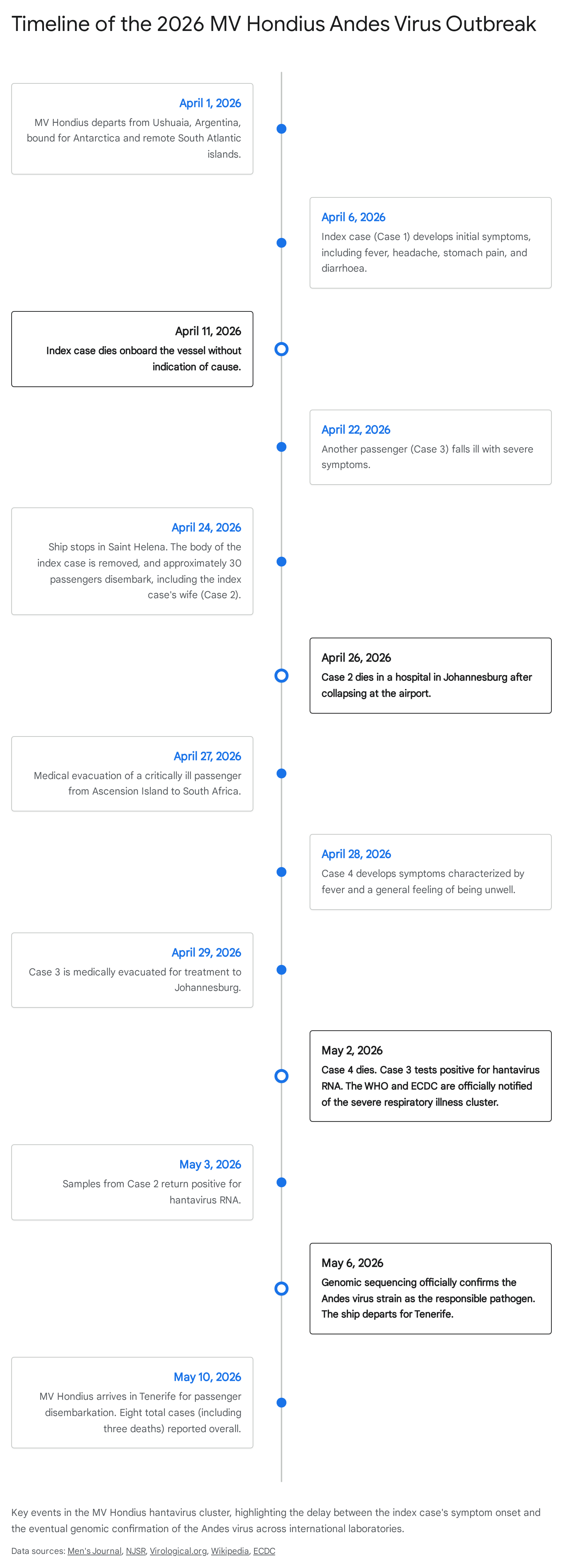
Epidemiological consensus suggests that the index case was exposed to the virus via environmental zoonotic spillover in Argentina prior to embarkation 2330. The incubation period for ANDV ranges from 7 to 42 days, making the April 6 symptom onset perfectly aligned with exposure on the South American mainland in late March 283936. However, the subsequent secondary cases aboard the vessel - including the spouse of the index case, another passenger, and the ship's doctor - strongly indicated instances of onboard human-to-human transmission facilitated by the confined maritime environment 262732. Consequently, an extensive international contact tracing operation was launched to monitor the 30 passengers who had disembarked early and utilized commercial flights (such as Airlink flight 4Z 132 from Saint Helena), highlighting the capacity of a virus with a long incubation period to infiltrate global transit networks 2732.
Genomic Characteristics of the 2026 Strain
The successful identification of the MV Hondius cluster was the result of a coordinated multinational effort utilizing advanced genomic techniques. Participating laboratories in South Africa, Senegal, Switzerland, and the Netherlands employed sequence-independent single-primer amplification (SISPA), unbiased metagenomics, and TWIST capture-based enrichment using Illumina and Oxford Nanopore platforms to secure full viral genomes directly from clinical samples 34.
Sequence Identity and Phylogenetic Relationships
Analysis of the viral RNA definitively confirmed the pathogen as Orthohantavirus andesense. The consensus sequences from the 2026 maritime cluster demonstrated an exceptionally high degree of sequence identity with historical ANDV strains known for severe lethality and interhuman transmissibility in Argentina.
Specifically, when compared to the well-documented human-isolated strains from the 1997 and 2018 Argentine outbreaks, the 2026 sequences shared over 98.7% identity across all three viral segments 2934. The virus's tripartite RNA genome consists of the L segment (encoding the RNA-dependent RNA polymerase), the M segment (encoding the Gn and Gc envelope glycoproteins), and the S segment (encoding the nucleocapsid protein and small nonstructural protein, NSs) 3738.
Table 2 illustrates the precise nucleotide sequence identity of the 2026 MV Hondius strain segments relative to the 1997 and 2018 benchmark sequences.
| Viral Segment | Identity to 1997 Sequence | Identity to 2018 Sequence | Key Segment Function |
|---|---|---|---|
| L Segment | 98.76% | 98.75% | Encodes the RNA-dependent RNA polymerase (RdRp). |
| M Segment | 98.71% | 98.68% | Encodes the Gn and Gc envelope glycoproteins. |
| S Segment | 98.73% | 98.73% | Encodes the nucleocapsid (N) protein and NSs. |
Nucleotide Polymorphisms and Viral Stability
A defining genomic hallmark of the MV Hondius cluster was the profound stability observed across the infected individuals. Genomic sequencing of the S and M segments revealed that they were entirely identical across all tested patients within the cluster, indicating a stark lack of viral diversity 2939. This genetic uniformity strongly supports the epidemiological hypothesis that the outbreak originated from a single zoonotic spillover event, or a very limited number of closely related events, which then propagated via an unbroken chain of human-to-human transmission 2939.
Detailed comparative alignment of the L segment (relative to the reference sequence MN258159) identified only two "true" single nucleotide polymorphisms (SNPs) distinguishing specific cases within the outbreak: a transition at position C2139T (detected in Case 5) and G576A (detected in Case 7) 2934. Both of these isolated SNPs were synonymous mutations, meaning they did not alter the amino acid sequence of the RNA-dependent RNA polymerase 2934.
This absence of coding mutations confirms that the virus did not require novel or rapid phenotypic adaptation to sustain human-to-human transmission aboard the ship 39. This observation aligns with experimental in vivo research utilizing the Syrian hamster model, which faithfully recapitulates human HCPS. Studies tracking the human-isolated ANDV strain CHI-7913 demonstrated that even over 25 serial passages in mammals, the virus accumulated only a single coding mutation 4041. The data suggests that wild-type ANDV circulating in the rodent reservoir is already fully competent for human infection and limited interhuman spread, requiring no intermediate evolutionary pressure 3941.
Structural Biology and Glycoprotein Architecture
Recent advances in structural virology have provided deep insights into the mechanical functionality of the highly conserved ANDV genome. By leveraging virus-like particle (VLP) systems, researchers have successfully established cryo-electron microscopy workflows capable of determining the structure of the membrane-embedded ANDV glycoprotein tetramer (Gn-Gc) at a 2.35 Å resolution 3742.
These high-resolution structures reveal previously uncharacterized features of glycoprotein organization, lattice formation, stability, and pH-sensing - critical components for viral assembly and cellular entry 3742. While these structures define the baseline mechanics of hantavirus infection, comparative genomic studies point to specific amino acid configurations within these glycoproteins and the small nonstructural protein (NSs) that dictate virulence. Research has identified discrete amino acid substitutions in the NSs and glycoprotein open reading frames that correlate specifically with the clade of ANDV strains capable of efficient person-to-person spread, distinguishing them from purely zoonotic strains 38. Immunological research utilizing self-amplifying replicon RNA (repRNA) encoding these ANDV-VLPs has shown promise in eliciting robust glycoprotein-binding antibodies, laying the groundwork for future structure-based vaccine designs, though currently no approved vaccines exist in the Americas 393742.
Transmission Dynamics and Interhuman Spread
The global landscape of hantaviruses encompasses dozens of distinct species, the vast majority of which - including the Sin Nombre virus in North America and the Puumala and Hantaan viruses in Eurasia - are strictly zoonotic 394348. The Andes orthohantavirus is unique within the Bunyavirales order as the sole agent with a verified capacity for direct human-to-human transmission 293644.
Zoonotic Aerosolization and Viral Entry
The transmission architecture of ANDV relies on dual pathways: primary zoonotic spillover followed by secondary interhuman spread. Primary infection occurs predominantly via the inhalation of aerosolized excreta (urine, feces, and saliva) from the O. longicaudatus reservoir 293943. This typically occurs when humans encroach upon infested environments or disturb desiccated rodent droppings during activities such as sweeping enclosed cabins or agricultural sheds, which lofts infectious virions into the air 394345.
Following the primary zoonotic introduction, ANDV allows for secondary human-to-human transmission. Unlike highly contagious respiratory pathogens such as SARS-CoV-2 - which utilize ACE2 receptors in the upper respiratory tract for rapid replication and easy exhalation - hantaviruses bind to β3 integrins 35. These integrin receptors are located deep within the pulmonary architecture and on the inner lining of vascular endothelial cells 335. Consequently, simple exhalation, sneezing, or casual social contact sheds very little virus. Instead, secondary transmission requires sustained close proximity to an infected individual, facilitating the transfer of deeper respiratory droplets or salivary aerosols 353946.
Secondary Attack Rates and Superspreading
The kinetics and efficiency of ANDV's interhuman spread differ fundamentally from viruses that cause mass pandemics. The most exhaustive baseline for ANDV transmission dynamics is derived from the 2018-2019 outbreak in Epuyén, Argentina. Triggered by a single rodent-to-human infection, the virus spread through 34 confirmed cases due to three symptomatic individuals attending crowded social gatherings (a birthday party and a funeral) 2847.
Prior to the implementation of aggressive public health interventions, including strict quarantine and isolation, the estimated basic reproduction number (R0) in Epuyén was 2.12 2847. Subsequent contact tracing and reanalysis of the cluster revealed that household members exposed during the late prodromal phase exhibited a secondary attack rate (SAR) of approximately 8%, while sexual partners faced a significantly higher transmission risk of 17.6% 2846.
A critical limiting factor in ANDV's spread is the timing of patient infectivity. COVID-19 thrives on presymptomatic transmission, with estimates suggesting up to 44% of secondary infections occur before the index case feels ill 28. ANDV, however, requires the patient to reach the late prodromal phase - the period marked by high fever and intense viremia just prior to cardiopulmonary collapse - to become infectious 28464849. Because patients rapidly deteriorate into severe respiratory distress, their ability to circulate in the community and act as superspreaders over an extended period is severely curtailed 28.
On the MV Hondius, the transmission efficiency proved lower than in severe terrestrial clusters but functionally lethal in the confined maritime setting. Generating roughly 8 to 11 infections from a pool of 147 passengers over five weeks, the attack rate aboard the ship hovered near 5% to 7% 28.
Table 3 compares the transmission metrics of historical ANDV outbreaks against a benchmark respiratory virus.
| Pathogen / Outbreak | Estimated Basic Reproduction Number (R0) | Secondary Attack Rate (SAR) | Primary Transmission Window |
|---|---|---|---|
| ANDV: Epuyén 2018 | 2.12 (pre-intervention) | ~8.0% (household); 17.6% (spousal) | Late prodromal phase; high viremia 2846. |
| ANDV: MV Hondius 2026 | Data currently insufficient for R0 | ~5.0% - 7.0% (overall ship cohort) | Late prodromal phase; sustained contact 28. |
| SARS-CoV-2: Cruise Benchmark | Up to 11.0 (pre-intervention) | ~17.0% (overall ship cohort) | Heavy presymptomatic shedding 2850. |
Airborne Transmission and Ventilation in Confined Settings
The emergence of a cluster on a modern cruise ship prompted intense debate regarding the mechanics of airborne versus droplet transmission and the role of HVAC systems. The WHO traditionally frames hantavirus interhuman spread around droplet and contact precautions 4851. However, the physical realities of the MV Hondius challenged this paradigm.
Epidemiological experts and the ECDC argued that in enclosed spaces - where individuals share cabins, dining rooms, and corridors for extended periods - the concentration of exhaled breath effectively acts as an aerosol hazard, blurring the operational distinction between large droplets and true airborne transmission 2648. Consequently, public health recommendations for managing the maritime cluster shifted toward treating ANDV as an airborne threat. Stringent interventions were mandated, prioritizing the optimization of ventilation, the absolute avoidance of unfiltered air recirculation, the deployment of portable High-Efficiency Particulate Air (HEPA) filters in quarantine zones, and the required use of N95 or FFP2 respirators for all healthcare workers and high-risk contacts during medical evacuations 3148.
Clinical Profile and Pathogenesis
Once human infection is established, the pathogenesis of ANDV is exceptionally severe, governed by the virus's specific cellular tropism and the profound systemic immune response it provokes in the host.
Symptomatology and Prodromal Shedding
The clinical progression of ANDV makes early detection extraordinarily difficult. Following an incubation period averaging 2 to 4 weeks (but ranging anywhere from 7 to 42 days), patients enter a non-specific prodromal phase 283936. Initial symptoms mimic severe influenza or gastrointestinal infections: sudden onset high fever, severe myalgia (particularly in the large muscle groups of the thighs, hips, and back), intense headache, dizziness, nausea, vomiting, and abdominal pain 26303648.
This lack of distinctive early symptoms often leads to misdiagnosis in non-endemic areas 3043. Crucially, it is during this late prodromal window - when viremia peaks and viable virus becomes detectable in saliva and respiratory secretions - that patients are most infectious to their close contacts 284648.
Cardiopulmonary Phase and Therapeutic Interventions
Within one to seven days of the prodromal onset, the disease rapidly transitions into the cardiopulmonary phase 2833. The ANDV glycoproteins target β3 integrins, facilitating viral entry into vascular endothelial cells 335. Rather than causing direct cytopathic cell death, the virus triggers a massive, dysregulated inflammatory cascade - often described as the immune system going into "overdrive" 5253.
This robust host immune response induces extreme vascular permeability, causing blood plasma to leak uncontrollably from the capillaries into the alveolar spaces. This non-cardiogenic pulmonary edema effectively fills the lungs with fluid, precipitating acute respiratory distress syndrome (ARDS), profound hypotension, hemodynamic instability, and shock 354853.
Currently, there are no approved targeted antiviral therapeutics for ANDV (while ribavirin has shown promise in HFRS, evidence for HCPS remains inconclusive), nor are there any licensed vaccines available in the Americas or Europe 293948. Patient survival hinges entirely on early admission to an intensive care unit for aggressive supportive care. Treatment protocols mandate fluid and electrolyte management, vasopressors, mechanical ventilation, and, in severe cases of cardiopulmonary failure, early referral for extracorporeal membrane oxygenation (ECMO) 32939.
Public Health Implications
The 2025-2026 Andes orthohantavirus clusters vividly illustrate the fragile boundaries separating shifting ecological systems from modern global transit networks. In Patagonia, acute climate extremes exacerbated by La Niña and widespread environmental disturbances disrupted the equilibrium of terrestrial rodent reservoirs, precipitating a historically severe epidemiological season characterized by mortality rates exceeding 31%.
When this heightened regional endemicity intersected with the enclosed, high-density environment of an international cruise ship, it provided a highly visible testing ground for ANDV's unique biological capability: human-to-human transmission. Genomic analyses confirm that the outbreak was not the product of a novel viral mutation, but rather the exploitation of an enclosed environment by a wild-type virus inherently capable of interhuman spread during its prodromal phase.
The MV Hondius event serves as a critical warning regarding zoonotic spillover. It demonstrates that public health paradigms must proactively address pathogens possessing latent interhuman transmission capabilities. However, the successful containment of the cruise ship cluster via rapid international genomic sequencing, the cessation of air recirculation, and rigorous contact tracing confirms that while ANDV is highly lethal, its transmission chains can be broken through classical epidemiological interventions. Moving forward, integrating ecological surveillance of terrestrial reservoir populations with heightened awareness in the global travel and maritime sectors will be essential to anticipating and neutralizing future viral incursions before they escalate into uncontrolled regional emergencies.