Tradeoffs between human reproduction and longevity
The intersection of evolutionary biology and biogerontology is dominated by the paradox of human life history: the existence of a prolonged post-reproductive lifespan juxtaposed against the profound physiological costs of childbearing. Unlike most terrestrial mammals, which typically experience somatic senescence in tandem with reproductive decline, human females routinely survive for decades following the cessation of ovarian function 123. This divergence raises fundamental questions regarding the evolutionary mechanisms that govern human life history, physiological resource allocation, and somatic maintenance.
Demographic, centenarian, and molecular data provide a complex picture of the reproductive-longevity tradeoff. Early evolutionary frameworks posited a strict zero-sum game between reproductive effort and lifespan. However, modern genomic analyses, epigenetic clock metrics, and extensive epidemiological meta-analyses reveal a highly nuanced, non-linear relationship. Current research indicates that the tradeoff is not a fixed biological law but a highly plastic phenomenon mediated by genetic architecture, cellular senescence patterns, and environmental resource abundance.
Evolutionary Theories of Senescence
The foundational theories of biological aging suggest that senescence is not a rigidly programmed biological imperative, but an evolutionary byproduct of declining selection pressure following the age of reproductive maturity 45. The theoretical frameworks addressing this phenomenon primarily diverge into models of metabolic resource allocation and models of genetic temporal tradeoffs.
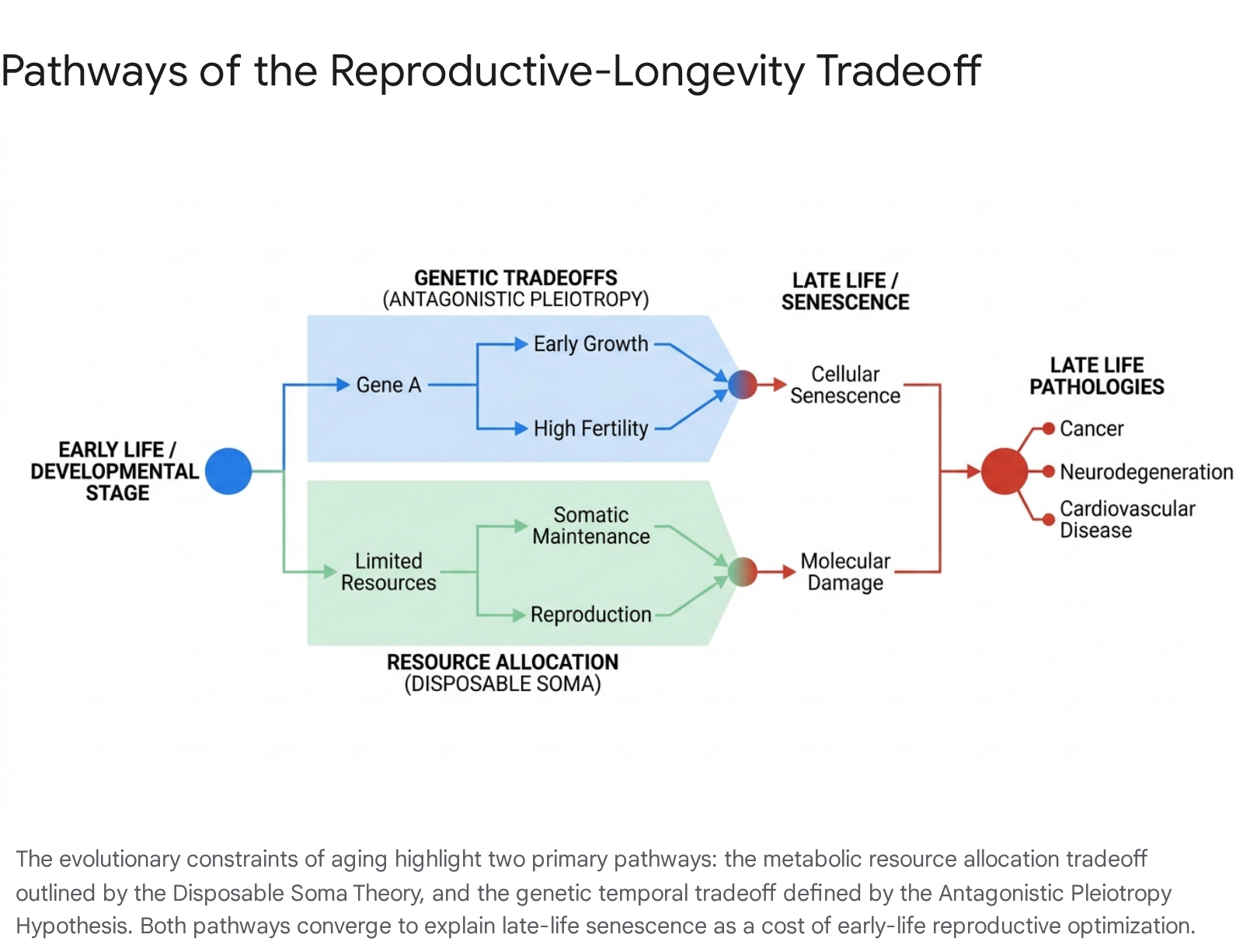
The Disposable Soma Theory
Formulated by Thomas Kirkwood, the Disposable Soma Theory posits that organisms must dynamically allocate finite metabolic resources among somatic maintenance, growth, and reproduction 678. Because natural environments present unavoidable extrinsic mortality risks - such as predation, disease, and starvation - investing heavily in indefinite somatic repair yields diminishing evolutionary returns. Consequently, natural selection favors allocations that ensure the organism survives long enough to reproduce, effectively rendering the non-reproductive body disposable after genetic propagation 6910.
Under this framework, high reproductive investment, manifested through multiple pregnancies and the energetic demands of lactation, theoretically drains resources that would otherwise be utilized for DNA repair, cellular waste clearance, and immune system maintenance 4811. However, empirical observations in humans complicate this strict allocation model. Despite bearing the overwhelming physiological burden of gestation, human females consistently outlive males 121314. Some animal models further challenge the residual impact of this tradeoff; studies on murine models indicate that while breeding incurs immediate mortality risks due to parturition complications, there is no evidence of a residual penalty on long-term survival once breeding ceases, suggesting that reproduction does not always incur permanent physiological debt 15.
The Antagonistic Pleiotropy Hypothesis
Proposed by George C. Williams, the Antagonistic Pleiotropy Hypothesis provides a genetic mechanism for the evolution of senescence 51516. It argues that natural selection will strongly favor alleles that confer substantial fitness benefits early in life, such as accelerated sexual maturation or robust fertility, even if those same alleles cause profound physiological degradation in later life 41017. Because late-life deleterious effects occur in the "selection shadow," when the organism has already passed on its genes and is increasingly likely to have died from extrinsic causes, evolution cannot easily select against them 15.
Modern genomic research provides compelling empirical support for this hypothesis in humans. Mendelian Randomization studies leveraging massive datasets have demonstrated a negative genetic correlation between early reproductive timing and late-life health 410. Individuals carrying polygenic profiles optimized for early menarche (before age 11) and early first childbirth (before age 21) exhibit significantly higher risks for type 2 diabetes, essential hypertension, heart failure, and late-onset Alzheimer's disease 4518. These individuals also demonstrate accelerated biological aging metrics, including advanced epigenetic clock scores and higher frailty indices, demonstrating that genetic factors favoring early reproduction come with significant biological costs later in life 4518.
Hyperfunction Theory
Recent theoretical critiques, such as the hyperfunction theory, challenge the premise that aging is driven purely by the stochastic accumulation of molecular damage resulting from resource scarcity 913. Hyperfunction theory posits that aging is a quasi-programmed, unintended continuation of developmental programs. For example, nutrient-sensing pathways like the Mechanistic Target of Rapamycin (mTOR) drive vital growth and reproductive maturation early in life. However, if this pathway remains inappropriately active in post-reproductive stages, it drives cellular hyperfunction, ultimately causing tissue exhaustion and age-related pathologies 912. This model seamlessly bridges Antagonistic Pleiotropy and the Disposable Soma Theory by explaining how early-life developmental programs become deleterious when operating outside their evolutionary scope.
Post-Reproductive Survival and Kin Selection
If evolutionary models predict that somatic maintenance should decline following the cessation of reproduction, the human female's extended post-menopausal lifespan presents a significant biological anomaly. Female chimpanzees experience ovarian aging at rates similar to humans and undergo menopause around age 50; however, they rarely survive more than a few years post-fertility 1219. Human females, conversely, routinely experience decades of vigorous post-reproductive life 2320.
The Grandmother Hypothesis
The Grandmother Hypothesis, an adaptationist model rooted in kin selection, suggests that the extended human lifespan evolved because post-menopausal women could significantly increase their inclusive fitness by provisioning their grandchildren 19202122. By reallocating labor from direct reproduction, which becomes increasingly dangerous with age, to alloparental care, older females subsidized the energetic costs of their adult daughters 1923. This subsidy allowed mothers to shorten interbirth intervals and overlap multiple dependent offspring 192024.
Evidence from historical and modern foraging populations provides empirical backing for this hypothesis. Cross-cultural studies, including analyses of the Hadza and rural Gambian families, indicate that the presence of an active maternal grandmother can halve toddler mortality rates and significantly increase the number of a daughter's surviving offspring 121. Studies analyzing 17th and 18th-century French settlers in Quebec found that daughters who began reproducing while their mother was alive birthed an average of 2.08 more offspring across their lifespan, a fitness benefit that declined rapidly with geographic distance 1. The evolutionary logic suggests that the inclusive fitness benefits generated by grandmaternal care were sufficient to select for alleles promoting somatic durability and resistance to cognitive decline well into advanced age 219.
Demographic Transitions and Evidentiary Limitations
Despite its theoretical strength, the Grandmother Hypothesis faces challenges concerning the limits of post-reproductive care. Studies utilizing historical pre-industrial register data from Finland reveal that the evolutionary benefits of grandmothering are distinctly age-dependent 12125. The positive effect of grandmaternal presence on grandchild survival peaks when the grandmother is in her early 60s and precipitously declines by age 75 12125.
Furthermore, the data reveal complex dynamics regarding lineage. While maternal grandmothers consistently improve offspring survival, paternal grandmothers over the age of 75 can inflict a net cost on grandchild survivorship. This is likely due to intergenerational resource competition, as the aging matriarch becomes frail and requires care herself from the extended family 12123. Additionally, the modern demographic transition, characterized by plummeting infant mortality rates due to better nutrition and medicine, has largely decoupled grandmothering from its historical evolutionary imperative. As child mortality drops, the critical life-preserving function of the grandmother becomes biologically redundant, even though the genetic architecture supporting post-menopausal longevity remains intact 25.
Parity and Late-Life Mortality Risk
To isolate the physiological toll of reproduction from broad evolutionary theory, researchers utilize large-scale epidemiological meta-analyses assessing the correlation between parity and late-life mortality. The data reveal that the relationship between reproduction and longevity is not a linear tradeoff, but rather exhibits a distinct dose-response curve.
All-Cause Mortality and the U-Shaped Risk Curve
A dose-response meta-analysis of cohort studies encompassing over 2.8 million participants provides a definitive quantification of the parity-mortality relationship 2627. The data demonstrate a statistically significant non-linear, U-shaped association between the number of live births and the relative risk of all-cause mortality 2627.
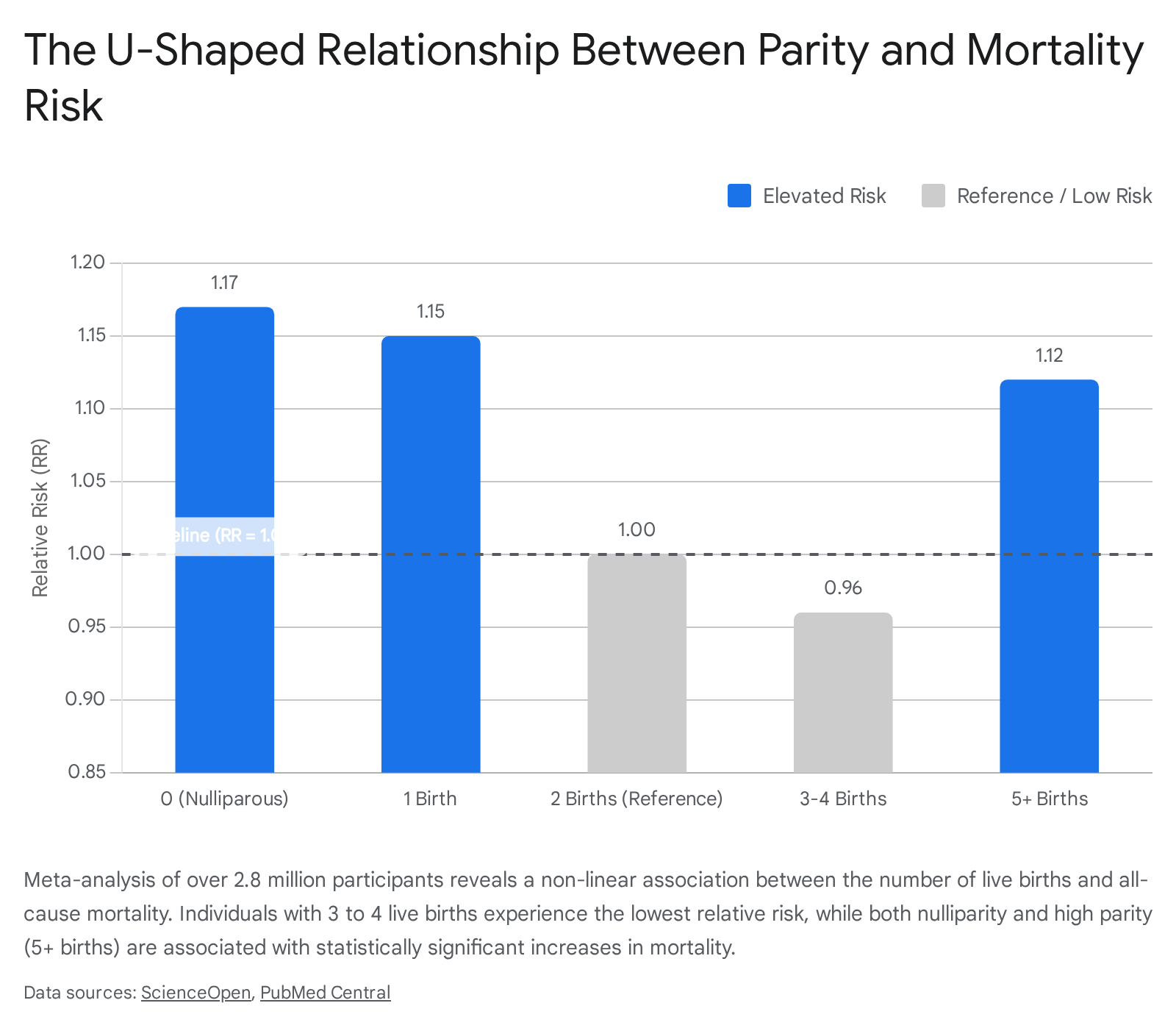
| Parity Bracket (Number of Live Births) | Relative Risk (RR) of All-Cause Mortality | 95% Confidence Interval | Clinical and Demographic Observations |
|---|---|---|---|
| 0 (Nulliparous) | 1.17 to 1.19 | 1.03 - 1.38 | Highest risk category compared to women with live births. Often confounded by underlying biological infertility or adverse early-life health factors. |
| 1 Birth | 1.15 | 1.09 - 1.20 | Elevated risk relative to moderate parity. Associated with increased risk of accidental deaths in specific historical cohorts. |
| 2 Births | 1.00 (Reference) | N/A | Baseline mortality risk utilized for comparative meta-analysis. |
| 3 to 4 Births | 0.96 | 0.94 - 0.98 | Lowest risk of all-cause mortality. Represents the optimal balance between reproductive health, socio-behavioral stability, and physiological cost. |
| 5+ Births (High Parity) | 1.12 | 1.03 - 1.21 | Significantly elevated risk. Mortality is heavily driven by vascular complications, hemorrhagic stroke, and systemic metabolic exhaustion. |
Table 1: Meta-analytical relationship between parity and all-cause mortality risk, utilizing a reference baseline of 2 live births. Data synthesized from extensive multi-national cohort analyses 26272829.
Nulliparous individuals face a significantly elevated risk of all-cause mortality (RR = 1.17 to 1.19) compared to those with live births 2627. This elevation is likely partially attributed to underlying health conditions that cause involuntary infertility, which may simultaneously increase mortality risk. Conversely, the lowest risk of all-cause mortality is observed in individuals with three to four live births (RR = 0.96) 2729.
When parity exceeds five live births, the mortality risk climbs substantially (RR = 1.12) 2729. This high-parity mortality penalty is heavily driven by vascular complications. Cohort data from isolated populations, such as Northern Finland, show that women with 10 or more births face up to a fourfold increase in mortality from hemorrhagic stroke, underscoring the profound cardiovascular stress of repeated gestations 28.
Maternal Age and Demographic Modulators
The timing of reproductive events also drastically alters the mortality landscape. Epidemiological data indicates that both extreme youth and advanced age at parturition exacerbate mortality risks. Nulliparous women under the age of 18 face the highest odds of adverse neonatal outcomes and long-term health detriments 30. Conversely, maternal mortality data from the United States spanning 2000 to 2019 demonstrates that the acute risk of death during or immediately following pregnancy rises exponentially with age. Compared to women aged 25 - 29, the odds ratio for maternal mortality is 28.49 for women aged 45 - 49, and reaches an alarming 343.50 for women aged 50 - 54 31. Demographic factors, such as marital status, further modulate baseline risks; single and divorced individuals demonstrate significantly higher all-cause and cardiovascular mortality compared to married counterparts, emphasizing the protective effect of social and economic stability 32.
Molecular Biomarkers of Reproductive Costs
The mechanisms underlying the high-parity mortality penalty can be traced to cumulative systemic damage, notably through oxidative stress and cellular senescence.
Oxidative Stress and Lipid Peroxidation
The peroxidation of lipids and oxidative damage to DNA are central components of cellular aging and metabolic dysfunction 3334. Specific biomarkers reliably quantify this physiological toll. F2-isoprostanes, prostaglandin-like compounds formed non-enzymatically by free radical attack on arachidonic acid, are widely considered the gold standard for measuring in vivo lipid peroxidation 333435. Similarly, 8-hydroxy-2′-deoxyguanosine (8-OHdG) serves as a stable marker of cumulative oxidative DNA damage 3336.
High maternal parity has been correlated with sustained elevations in these systemic oxidative stress markers, demonstrating that repeated allocation of energetic resources toward fetal development results in a measurable failure of somatic antioxidant defenses 143536. The sustained elevation of these markers mirrors the molecular signatures seen in advanced age-related pathologies, underscoring the severe metabolic taxation of the reproductive process 3337.
Tissue-Specific Cellular Senescence
At the cellular level, aging is driven by the accumulation of senescent cells - cells that have undergone irreversible growth arrest but remain metabolically active, secreting a toxic cocktail of pro-inflammatory cytokines known as the Senescence-Associated Secretory Phenotype (SASP) 384039. The primary markers for these cells are the cyclin-dependent kinase inhibitors p16Ink4a and p21Cip1 3840.
Recent single-cell RNA sequencing analyses of human and murine tissues reveal a startling complexity: p16-positive and p21-positive senescent cells are distinct, non-overlapping populations that possess entirely different SASP profiles and trajectory dynamics 383941. Furthermore, their accumulation is highly tissue-specific. While the skin, pancreas, and kidneys show age-related increases in both p16 and p21 expressing cells, the lungs show no such increase, and skeletal muscle appears entirely devoid of them 4040.
This tissue-specific heterogeneity provides a critical mechanism for the reproductive-longevity tradeoff. Because the physiological demands of reproduction stress various organ systems asymmetrically, the accumulation of senescent cells and the subsequent exhaustion of stem cell pools occur unevenly across the body. The maternal mortality risk from cardiovascular complications at high parities is likely a direct result of localized, stress-induced cellular senescence in the vascular endothelium that bypasses systemic resilience 2838.
Genetic and Epigenetic Mediators
The relationship between reproductive capacity and extreme longevity is not purely detrimental; in certain genetic profiles, late-life fertility is highly correlated with exceptional lifespan. Epidemiological data on female centenarians reveal that over 20% bore children after the age of 40, compared to less than 5% in the general population 42. This suggests that an extended reproductive span may be a phenotypic marker for a fundamentally slower rate of biological aging.
Genome-Wide Association Studies
Attempts to identify the genetic architecture of human longevity have proven difficult. Despite the heritability of lifespan being estimated between 15% and 25%, massive Genome-Wide Association Studies (GWAS) - such as those conducted by the CHARGE consortium utilizing over 6,000 longevity cases - have yielded surprisingly few universally replicable loci 43444546.
To date, only two gene loci have been consistently replicated across independent global cohorts: Apolipoprotein E (APOE) and Forkhead box O3 (FOXO3) 43444547. APOE variants are heavily implicated in lipid transport and cardiovascular disease risk, while FOXO3 is a critical transcription factor in the insulin/IGF-1 signaling pathway, directly regulating stress resistance, apoptosis, and cellular maintenance 4348. Other loci, such as CADM2 and GRIK2, show suggestive but less robust associations 4344. The scarcity of definitive "longevity genes" suggests that extreme lifespan is a highly polygenic trait influenced by thousands of variants with minute effect sizes, or that it relies on rare protective variants not easily captured by standard imputation 4445. The ability to reproduce late in life and survive past a century is driven by a systemic genetic resistance to metabolic decline.
Epigenetic Clocks and Biological Age
While fixed genomic variants provide the baseline blueprint, the actual rate of biological aging is highly sensitive to environmental and reproductive events, a dynamic tracked via epigenetics. Epigenetic modifications, particularly DNA methylation (DNAm) at specific CpG sites, allow for the calculation of an individual's biological age relative to their chronological age 495051. Highly accurate algorithms, such as the Horvath, Hannum, Levine, and GrimAge clocks, measure this biological degradation 5052. Recent breakthroughs have even allowed for the development of methylation-based models that accurately predict maximum lifespan, gestational time, and age at sexual maturity across diverse mammalian species 51.
Research indicates that parity and the timing of reproductive events directly modulate epigenetic aging rates. Adolescents experiencing early environmental perturbations and early childbirth exhibit accelerated epigenetic aging, aligning with the Antagonistic Pleiotropy framework 44955. Conversely, women who undergo late-onset menopause (after age 55) exhibit significantly younger biological ages based on epigenetic clocks 42. The physiological stress of reproduction, when initiated prematurely or executed at very high frequencies, disrupts cellular homeostasis and leaves permanent, accelerating marks on the epigenome.
Environmental Context and Phenotypic Plasticity
The evolutionary tradeoffs between reproduction and longevity are not static physical laws; they are highly plastic variables mediated by the abundance of resources and the severity of the external environment 856.
Baseline Mortality in Foraging Populations
To accurately gauge the evolutionary baseline of the reproductive-longevity tradeoff, biogerontologists analyze mortality data from ethnographically observed hunter-gatherer populations, such as the !Kung and Aché. In these populations, which approximate the conditions under which human life history evolved, the mean life expectancy at birth is roughly 31 years, driven by staggering early-life mortality exceeding 35% before age 15 575354.
However, this low average conceals a remarkable capacity for adult longevity. Conditional on surviving the intense selective pressures of childhood, hunter-gatherers routinely live into their sixth and seventh decades, allowing them to fulfill the evolutionary mandate of the Grandmother Hypothesis 5354. Comparative biodemography shows that the mortality improvements achieved in modernized societies over the last century represent a phenotypic plasticity that is virtually unprecedented in the animal kingdom 5360. For context, a 15-year-old wild chimpanzee experiences an annual probability of death of 4.7%; an average hunter-gatherer does not reach this probability until age 63, and modern populations in developed nations do not reach it until roughly age 69 53.
Socioeconomic Status and the Wealth Effect
In historical pre-industrial populations, the strict negative correlation between high parity and longevity only applied to the lowest socioeconomic tiers. Data from the historical Krummhörn population in Germany (1720 - 1870) and pre-industrial Swedish registers reveal that the mortality penalty of high fertility was exclusively borne by landless households 8. In higher socioeconomic classes, the parity-mortality tradeoff disappeared entirely, and in some cases, high fertility correlated with increased lifespan 8.
This phenomenon, termed the "wealth effect," indicates that when metabolic resources are abundant, the competition between somatic maintenance and reproductive investment described by the Disposable Soma Theory is alleviated 56. An organism does not have to choose between repairing DNA and nourishing offspring if caloric and structural resources are limitless. Consequently, the modern demographic landscape, characterized by advanced healthcare and low extrinsic mortality, obscures the harsh biological trade-offs that forged human evolution, allowing humans to achieve both high reproductive success and extended longevity if socio-economic conditions permit 25565355.