Thymic involution and regeneration
Pathophysiology and Structural Dynamics of Thymic Involution
Anatomical Compartmentalization and Adipogenic Transition
The thymus is a primary lymphoid organ situated in the superior and inferior mediastinum, operating as the central site for the maturation, differentiation, and positive and negative selection of T lymphocytes. Organogenesis begins during early fetal development, specifically between the seventh and fourteenth gestation weeks, originating from the third pharyngeal arch 1. While the organ is fully developed and highly active before birth, it undergoes a profound and continuous lifelong structural transformation known as thymic involution 12.
Histologically, the human thymus is divided into two structurally and functionally distinct compartments: the true thymic epithelial space (TES) and the perivascular space (PVS) 23. The TES forms the epithelial framework necessary for thymopoiesis and is further subdivided into the cortex and the medulla. The thymic cortex contains densely packed, immature thymocyte precursors undergoing rapid division and positive selection. The medulla is characterized by a looser arrangement of thymocytes and the presence of terminally differentiated epithelial structures known as Hassall's corpuscles 34.
During the aging process, the TES undergoes severe and progressive atrophy, systematically losing functional thymic epithelial cells (TECs) and developing thymocytes. Conversely, the PVS increases in volume. This expanding PVS fills with adipose tissue and a diverse array of mature peripheral immune cells, including T cells, B cells, monocytes, and eosinophils 3. This adipogenic transformation is the defining histological hallmark of involution. As the TES shrinks, the structural integrity of the stroma deteriorates, culminating in an organ primarily composed of fat and fibrotic tissue by middle age 56. Despite this dramatic reduction in the functional epithelial space, localized thymopoiesis and active T-cell receptor (TCR) gene rearrangement continue within the microscopic cellular remnants of the TES well into late life 3.
Chronological Modulations in Thymic Mass, Volume, and Morphology
The macroscopic mass and volume of the thymus fluctuate significantly across the human lifespan. Historically, clinical dogma suggested that the thymus grew steadily until puberty, at which point the surge of sex hormones triggered a rapid decline. However, precise morphometric analyses utilizing silver impregnation techniques and stereological methods indicate that the involution of the functional TES begins much earlier - as early as the first year of life - and progresses independent of pubertal hormonal changes 24.
The gross weight of the thymus grows rapidly from approximately 10 to 13 grams at birth to reach its maximum physical dimensions - typically between 30 and 50 grams, though individual maximums can reach up to 70 grams - during childhood or early adolescence 1478. Following this peak, the rate of true epithelial decline is estimated at roughly 3% per year until middle age (approximately 35 to 45 years), after which the rate of loss decelerates to approximately 1% per year until death 27.
Radiological evaluations using multidetector computed tomography (MDCT) provide critical quantitative insights into the organ's changing morphology in vivo. The thymus typically transitions from a quadrilateral, square, or mass-like shape in infancy to a distinct arrowhead shape in adulthood 18. Furthermore, age influences its precise anatomical position; the thymus tends to migrate inferiorly with age, shifting from the T4 - T5 vertebral level in youth down to the T6 - T7 vertebral level in older adults 111.
| Age Group (Years) | Mean Thymus Weight (g) | Mean Total Volume (cm3) | Morphological Shape | Predominant Tissue Composition |
|---|---|---|---|---|
| 0 - 2 | 9.72 - 13.00 | 10.04 | Quadrilateral / Square | Dense Thymic Epithelial Space (TES) |
| 2 - 8 | 20.17 | 12.48 | Arrowhead | Peak TES expansion, enlarging PVS |
| 8 - 16 | 30.00 - 40.00 | 12.78 | Arrowhead / Bilobal | Peak gross mass, early adiposity |
| 16 - 40 | 18.90 - 19.39 | 18.96 | Arrowhead | Accelerating adipose replacement |
| 40 - 60 | 13.75 - 17.52 | 11.88 | Arrowhead / Indistinct | >50% Adipose, shrinking TES |
| > 60 | 5.70 - 7.31 | < 10.00 | Indistinct from fat | Fibrotic and fatty remnants |
| Table 1: Age-related dynamics of human thymic mass, volume, and composition. Data is aggregated from human cadaveric morphometry and radiological MDCT imaging studies 17811129. Note: Volumetric measurements via CT often capture the total mediastinal shadow, including newly deposited adipose tissue within the PVS, resulting in volumetric stabilization even as the functional epithelial mass rapidly declines. |
Cellular Mechanisms Driving Epithelial Atrophy
The precise molecular drivers of thymic involution involve a complex interplay of systemic hormones and intrinsic stromal failure. While early hypotheses focused heavily on the inhibitory effects of sex steroids (testosterone and estrogen), recent evidence points toward the degeneration of TECs as the primary catalyst 710. The maintenance of the thymic microenvironment relies heavily on the transcription factor Forkhead-box N1 (FOXN1), a master regulator of TEC lineage specification 11. During aging, the continuous expression of FOXN1 declines, which fundamentally impairs the ability of bipotent progenitor cells to differentiate into functional cortical and medullary TECs 1112.
Additionally, the RANK-RANKL signaling axis plays a pivotal role. The administration of exogenous receptor activator of nuclear factor kappa-B ligand (RANKL) has been shown to stimulate endothelial and epithelial cells within human thymic organocultures, suggesting that deficits in specific trophic signaling pathways contribute to the age-related collapse of the thymic stroma 17.
Immunological Consequences of Thymic Atrophy
Quantitative Shifts in T-Cell Subsets and Reference Ranges
The most profound physiological consequence of a shrinking thymic epithelial space is a severe reduction in the output of newly generated naive T cells (Tn). Because the human peripheral T-cell pool requires the continuous generation of cells to maintain homeostasis and replace those lost to apoptosis or differentiation, the decline in thymic export forces the immune system to rely heavily on the peripheral proliferation of existing naive and memory T cells 51713.
Quantifying these cellular shifts requires high-resolution, multi-color flow cytometry. Using specific cluster of differentiation (CD) surface markers, researchers classify peripheral T cells into distinct functional subsets: naive T cells (Tn; CD45RA+CD45RO-CD62L+CD95-), stem cell memory T cells (Tscm; CD95+CD62L+), central memory T cells (Tcm; CD45RO+CD95+CD62L+), effector memory T cells (Tem; CD45RO+CD95+CD62L-), and terminal effector T cells (Tte) 141516.
Cross-sectional studies, including extensive single-platform analyses of hundreds of healthy volunteers, demonstrate that the absolute counts of Tn and Tscm cells decrease linearly with advancing age 1422. Conversely, Tcm and Tem populations expand significantly between the ages of 18 and 64, plateauing in late life. The overall lymphocyte compartment transitions from a highly diverse, naive-dominant profile in youth to an oligoclonal, memory-dominant profile in older adulthood 162217.
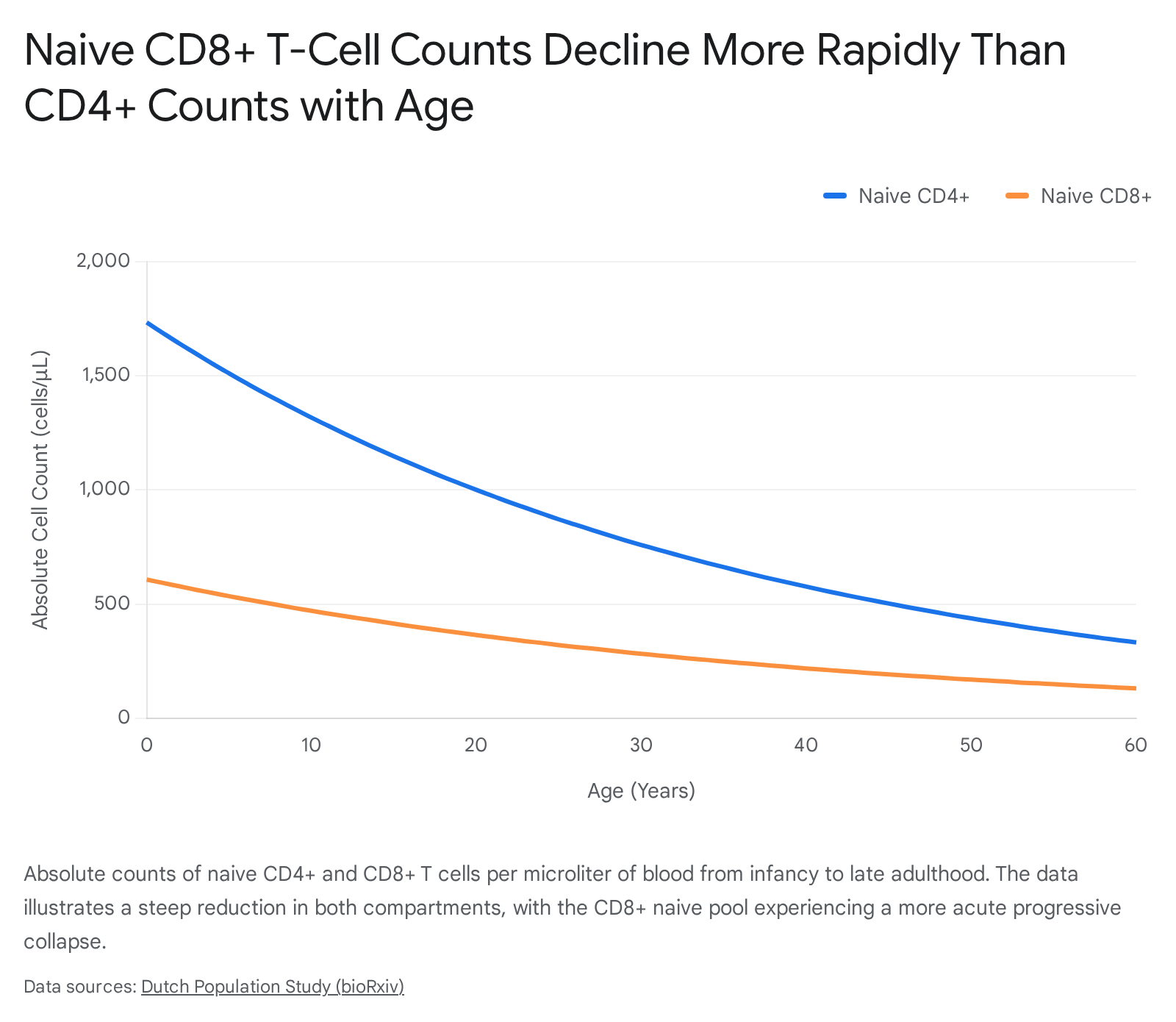
Crucially, the decline in naive cells is highly asymmetrical across T-cell lineages. While CD4+ naive T cells exhibit a gradual decline, the drop in CD8+ naive T cells is highly pronounced. Over a 50-year span, naive CD4+ counts decline approximately four-fold (e.g., from 1,733 to 438 cells/μL), whereas naive CD8+ counts experience an even steeper relative drop, falling from 608 to 170 cells/μL 24.
The measurement of T-cell receptor excision circles (TRECs) - circular DNA fragments formed during TCR gene rearrangement that serve as stable markers of recent thymic emigrants (RTEs) - provides insight into the rate of this decline. Studies indicate that the concentration of TRECs per naive T cell remains relatively stable during the first two decades of life. This suggests that the early maintenance of the naive pool is heavily dependent on the peripheral division of existing naive cells rather than relying purely on direct thymic export 13. However, as thymic output diminishes further in later life, the intervals between peripheral divisions lengthen, making the naive population progressively less dynamic and increasingly senescent 13.
T-Cell Receptor Repertoire Collapse and Cellular Exhaustion
The failure of the aged thymus to generate a diverse repertoire of naive T cells precipitates a clinical condition known as immunosenescence 618. Without a continuous influx of newly generated, unique T-cell receptors, the peripheral immune system's capacity to recognize novel, previously unencountered antigens is drastically reduced 19. Current models indicate that humans experience a profound and irreversible collapse in TCR repertoire diversity around 63 years of age 20.
Simultaneously, the existing peripheral T-cell pool becomes exhausted due to a lifetime of continuous antigenic exposure, chronic viral infections (such as Cytomegalovirus), and relentless homeostatic proliferation. This exhaustion is phenotypically characterized by the upregulation of inhibitory surface receptors, such as PD-1 and Tim-3, which are found predominantly on CD8+ T cells in older adults 1820.
Clinical Manifestations of Immunosenescence
The downstream effects of thymic involution extend far beyond microscopic cellular shifts; they are directly implicated in the increased morbidity and all-cause mortality observed in the elderly. The exhaustion of the CD8+ cytotoxic T-cell compartment impairs the body's immunosurveillance mechanisms, driving elevated susceptibilities to viral and bacterial infections, and facilitating increased cancer incidence as malignant cells escape immune detection 21020. Furthermore, the lack of robust naive T cells results in notoriously poor responses to novel vaccines 10. Finally, regulatory failure within the aging immune system leads to a rise in subclinical chronic inflammation ("inflammaging") and an increased prevalence of autoimmune conditions 21020.
Pharmacological Interventions and the Debate on Epigenetic Reversal
Recombinant Human Growth Hormone and the TRIIM Trials
Given the central role of thymic involution in age-related physical and immunological decline, therapeutic interventions aiming to regenerate the thymus have garnered significant clinical and academic attention. The most notable human in vivo effort to date is the Thymus Regeneration, Immunorestoration, and Insulin Mitigation (TRIIM) trial, conducted between 2015 and 2017.
The TRIIM trial investigated whether the administration of recombinant human growth hormone (rhGH) could physically regenerate the thymus and reverse signs of immunosenescence in a cohort of putatively healthy men aged 51 to 65. Previous animal models and studies on HIV patients had demonstrated that growth hormone possesses thymotrophic properties. However, because rhGH can induce hyperinsulinemia and possesses distinct diabetogenic properties, the TRIIM protocol combined rhGH with dehydroepiandrosterone (DHEA) and metformin, aiming to mitigate insulin resistance and counterbalance hormonal drift 182129.
Over the 12-month treatment period, high-resolution magnetic resonance imaging (MRI) revealed a highly significant increase in the thymic fat-free fraction (TFFF) among the participants. This metric indicated a physical reduction of adipose tissue within the mediastinum and the localized restoration of functional thymic mass 1820. Immunologically, the treatment induced a robust decline in total and CD38-positive monocytes, resulting in a highly favorable lymphocyte-to-monocyte ratio (LMR). The protocol also triggered a statistically significant increase in the absolute counts of naive CD4+ and CD8+ T cells, alongside a reduction in exhausted PD-1+ CD8+ T cells, suggesting genuine functional immune rejuvenation 1820.
Building on these preliminary but highly promising findings, an expanded follow-up study, the TRIIM-X trial (2020 - 2025), enrolled a larger, more diverse cohort - including both men and women aged 40 to 80 - to evaluate personalized, adaptive dosing regimens 222324. Interim data from TRIIM-X suggests that beyond simply improving immune markers, participants experienced improvements in functional physical metrics, including increased VO2 max and lower-limb strength. These findings hint that restoring immune tolerance and reversing thymic involution may have systemic, pleiotropic benefits on physical frailty and the broader aging process 212925.
Epigenetic Clocks and the Complexity of T-Cell Differentiation
The most widely publicized and heavily debated outcome of the TRIIM trial was its reported effect on epigenetic age. Using advanced DNA methylation-based algorithms - specifically the Horvath clock, Hannum clock, PhenoAge, and GrimAge - researchers calculated the biological age of the participants' peripheral blood mononuclear cells. The TRIIM cohort demonstrated a mean epigenetic age reduction of 1.5 to 2.5 years after the 12-month intervention, meaning their cellular DNA appeared biologically younger than their chronological age 182026. The rate of epigenetic aging reversal even appeared to accelerate during the final months of the trial, and improvements in the GrimAge predictor - a highly accurate surrogate for mortality risk - persisted six months after the treatment was discontinued 1820.
However, the interpretation of these epigenetic results is subject to intense, rigorous scientific debate. Epigenetic clocks measure methylation patterns at specific cytosine-phosphate-guanine (CpG) sites across the genome 26. Critics within the field of computational biology note that T-cell differentiation states fundamentally alter these precise methylation patterns. Specifically, naive T cells possess distinct chromatin structures, maintaining stemness-associated transcription factors (such as FOXO1, KLF2, LEF1, and TCF7) with very low DNA methylation and specific activating histone modifications (e.g., H3K4me3) 3527. As a result of this quiescent, stem-like state, naive T cells generate an intrinsically "younger" epigenetic signature compared to terminally differentiated memory or effector T cells, which accumulate suppressive methylation marks 3527.
Because the TRIIM intervention successfully increased the proportion of naive T cells and decreased the number of exhausted memory cells in the bloodstream, the aggregate DNA sample drawn from the participants naturally analyzed as biologically younger. The unresolved critique is whether the rhGH-based treatment genuinely reversed the intrinsic biological aging of individual cells (representing true intracellular rejuvenation), or merely shifted the macroscopic cellular composition of the blood toward a higher ratio of naive cells 2935. Furthermore, the long-term safety of sustained rhGH therapy regarding cancer promotion remains a persistent concern, making isolated analysis of the intervention's individual components difficult in the absence of a large-scale, placebo-controlled clinical trial 26.
Molecular Therapies and Endogenous Repair Mechanisms
Transient Hepatic Reprogramming via mRNA Lipid Nanoparticles
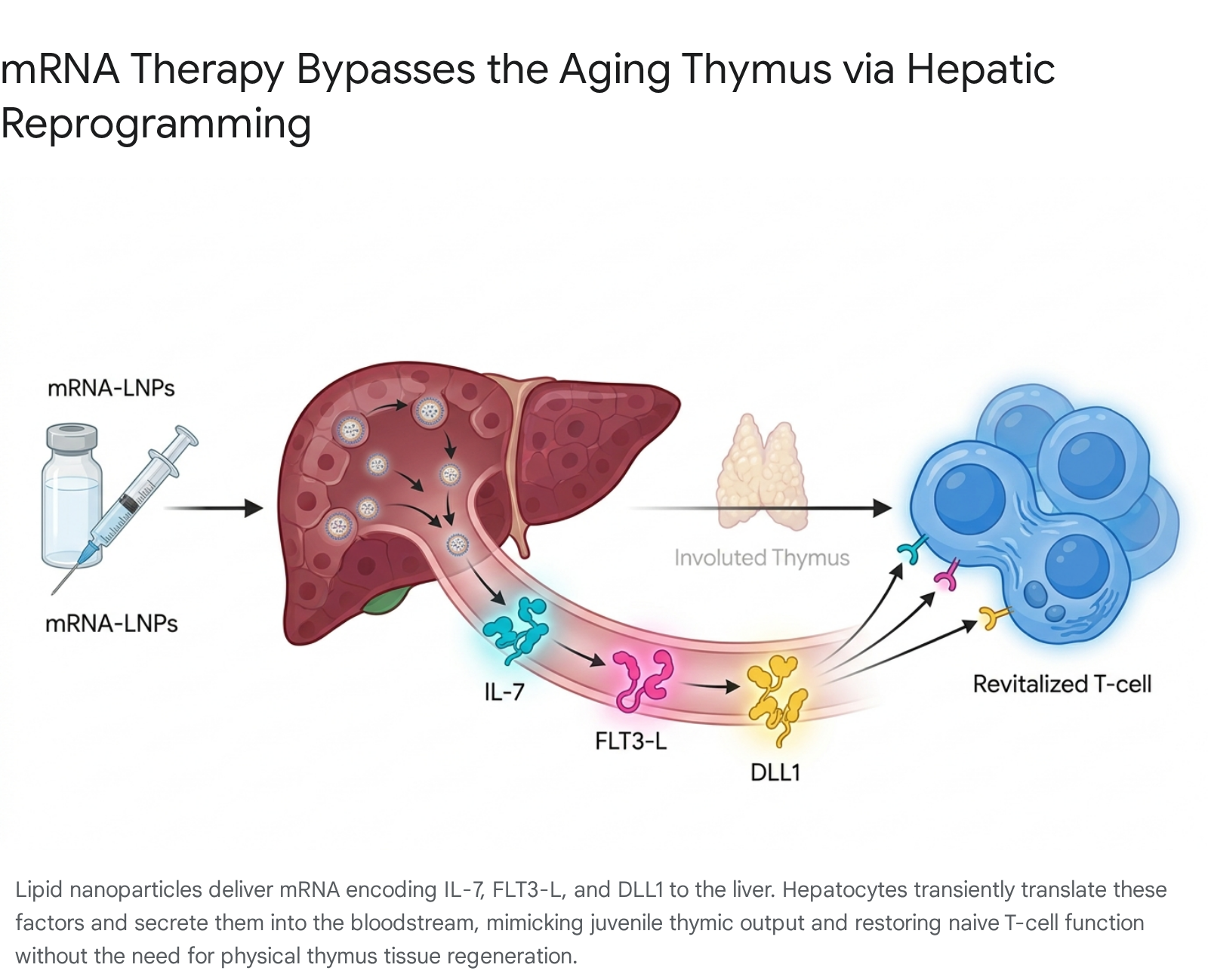
As an alternative to hormone-mediated thymic regrowth, emerging strategies seek to bypass the physical constraints of the degenerated thymus entirely by engineering other organs to fulfill its molecular and immunological duties. In a breakthrough 2025 study published in Nature by Friedrich, Zhang, et al. at the Massachusetts Institute of Technology and the Broad Institute, researchers utilized mRNA technology to reprogram the liver into a temporary "thymus factory" 28293031.
Through highly complex single-cell transcriptional analyses, the researchers identified that the aged immune system specifically lacks three critical trophic and maturation signals normally provided by the juvenile thymus: Interleukin-7 (IL-7), FMS-like tyrosine kinase 3 ligand (FLT3-L), and Delta-like canonical Notch ligand 1 (DLL1) 2830. IL-7 is vital for T-cell survival, FLT3-L promotes the expansion of dendritic cells and hematopoietic progenitors, and DLL1 is essential for driving Notch signaling, which commits progenitors to the T-cell lineage.
The research team encapsulated synthetically engineered mRNA encoding these three trophic factors into lipid nanoparticles (LNPs) and administered them intravenously to 18-month-old mice (biologically equivalent to humans in their late 50s). The LNPs specifically targeted hepatocytes due to the liver's exceptionally robust protein synthesis capacity and its high blood flow, which ensures the efficient systemic distribution of the secreted immune factors 2832.
Because mRNA translation is inherently transient, the liver produced the thymic signals only for a short, tightly controlled period. This temporal limitation is highly advantageous from a safety perspective, allowing for fine-tuned dosing while effectively avoiding the severe systemic toxicity, inflammation, and potential autoimmunity traditionally associated with the continuous administration of recombinant protein therapies 3233.
Synergistic Effects on Antitumor Immunity and Vaccine Efficacy
The immunological outcomes of this synthetic hepatic reprogramming were profound and rapid. Mice receiving repeated doses of the mRNA-LNP cocktail over four weeks exhibited a substantial increase in both the absolute size and functional diversity of their T-cell populations 2831.
When challenged with an ovalbumin vaccine - a standard test for measuring de novo immune responses - the treated aged mice produced double the number of antigen-specific cytotoxic T cells compared to untreated aged controls 2831. Furthermore, the intervention synergized powerfully with established cancer immunotherapies. In aggressive melanoma models treated with PD-L1 checkpoint inhibitors (drugs designed to release the "brakes" on the immune system), mice that received the mRNA pretreatment demonstrated significantly higher survival rates and prolonged lifespans, successfully suppressing tumor growth that typically overwhelms the weakened aged immune system 3132. These results present a paradigm shift: rather than attempting the difficult task of physically rebuilding the complex stromal architecture of the aged thymus, researchers can achieve robust, functional immune rejuvenation by synthetically replacing its missing molecular output via the liver.
Amphiregulin-Mediated Regeneration via Regulatory T Cells
Endogenous mechanisms of thymic repair also offer novel therapeutic targets that do not require exogenous organ reprogramming. A 2025 study published in Immunity by Lemarquis et al. identified a highly specific, previously undescribed population of recirculating regulatory T cells (Tregs) that actively mediate thymic regeneration following acute damage, such as from radiation or severe stress 43.
These specialized Tregs migrate back from the peripheral bloodstream into the thymus, where they accumulate and secrete a potent growth factor known as amphiregulin. Amphiregulin acts directly on the thymic stroma to promote rapid tissue repair and stimulate the de novo generation of T cells. When researchers isolated these specific Tregs and administered them intravenously in murine models, the cells homed exclusively to the thymus and significantly accelerated its overall regeneration 43. Exploiting this native pathway - potentially by synthetically engineering a patient's own T cells to overproduce amphiregulin - offers a highly targeted, non-hormonal avenue for ameliorating immune fatigue in aging populations and in patients recovering from cytotoxic cancer therapies 43.
FOXN1 Upregulation and Cellular Reprogramming
For researchers remaining focused on true, structural organ regeneration, the Forkhead-box N1 (FOXN1) transcription factor remains the primary molecular target. As the master regulator of thymic epithelial cell specification, its presence is mandatory for the survival of the TES 11.
Experimental gene therapies utilizing transgenic mouse models (such as Rosa26 Cre-driven models) have demonstrated that forced upregulation of FOXN1 in fully involuted, aged thymi is sufficient to drive the rapid proliferation of dormant progenitor TECs. In these models, conditional overexpression of FOXN1 completely reversed the hallmarks of age-related involution, restoring the thymic architecture, transcriptional gene expression profile, and functional T-cell output to a state closely resembling the juvenile organ 12. Furthermore, studies have shown that embryonic fibroblasts can be directly reprogrammed into induced thymic epithelial cells (iTECs) via forced FOXN1 overexpression 3445. This raises the highly promising possibility of autologous cell therapies, wherein a patient's own skin fibroblasts are extracted, genetically altered ex vivo to express FOXN1, and transplanted back into the mediastinum to successfully rebuild the thymic stroma without the risk of immune rejection 34.
Bioengineering, Synthetic Scaffolds, and Thymic Organoids
Differentiation of Human Pluripotent Stem Cells
The ultimate frontier in thymic regeneration and the broader field of regenerative medicine is the complete ex vivo biofabrication of transplantable thymic organoids. Given the limited availability of primary donor tissue and the risks of allogeneic rejection, regenerative tissue engineering has turned heavily to human pluripotent stem cells (hPSCs).
Through the precise, sequential modulation of developmental signaling pathways - utilizing factors such as Activin A, bone morphogenetic proteins (BMP), Wnt, and retinoic acid - hPSCs can be successfully directed to differentiate into definitive endoderm, and subsequently into thymic epithelial progenitors (TEPs) 4535. Recently, researchers have successfully engineered isogenic stem cell-derived thymic organoids (sTOs) by co-culturing these pure TEPs with mesenchymal cells and hematopoietic progenitor cells derived entirely from the same hPSC line. In vitro, these sTOs accurately replicate the complex thymic niche. They express the crucial autoimmune regulator (AIRE) protein necessary for the negative selection of autoreactive cells, and they successfully support the development of functional, self-tolerant CD4+ and CD8+ T cells 3637.
Natural Hydrogels and 3D Bioprinting Techniques
To scale these microscopic cellular aggregates into functional, macroscopic organs suitable for transplantation, advanced 3D bioprinting technologies are utilized. Bioprinting allows for the precise, computer-aided spatial distribution of "bioinks" laden with TECs, mesenchymal support cells, and necessary growth factors 383940.
Natural polymers, such as collagen, gelatin methacryloyl (GelMA), alginate, and decellularized extracellular matrix (dECM) from donor thymi, are highly favored for their excellent biomimetic properties. Collagen scaffolds, in particular, can closely reproduce the endogenous thymic microenvironment, successfully supporting TEC adhesion, migration, and proliferation while preventing apoptosis during the extrusion phase of printing 104041. However, natural hydrogels face significant biomechanical challenges; they are often too soft and lack the necessary structural rigidity required for long-term implantation and vascularization in a living host. Overcoming these limitations often requires rapid pH changes for self-assembly, photocurable cross-linking, or hybridization with tougher synthetic materials 1040.
Advanced Synthetic Scaffolds and Porous Tantalum Matrices
To overcome the inherent structural instability of natural hydrogels, biomedical engineers are actively investigating advanced synthetic biomaterials to serve as the physical backbone for regenerated thymi. Highly porous metal scaffolds, such as those made of 3D-printed tantalum (Ta), are traditionally utilized in load-bearing orthopedics due to their high compressive strength (10 - 200 MPa) and low elastic modulus (1 - 30 GPa), which closely mimics the physical properties of cancellous bone, thus preventing the "stress shielding" effect 4243.
In the specific context of soft tissue and immune organ engineering, tantalum's exceptional biocompatibility, high surface energy, excellent corrosion resistance, and highly interconnected biomimetic pore structure (ranging from 100 to 400 μm) provide an ideal, unyielding architectural foundation for delicate epithelial cells 4244. Experimental studies have demonstrated that tantalum-coated carbon scaffolds can successfully encapsulate fragile TECs alongside hematopoietic stem cells. When implanted, these rigid synthetic scaffolds actively prevent the physical collapse, degradation, and fragmentation that frequently plague soft hydrogel grafts in vivo. They successfully support the long-term progression of naive to mature T-cell development, effectively acting as an indestructible artificial stroma 10.
As the field of tissue engineering matures over the next decade, the integration of artificial intelligence for optimal porous scaffold design, automated closed-system bioreactors for rapid TEC expansion, and highly precise synthetic bio-inks will likely transition thymic organoids from theoretical laboratory models into viable, life-extending clinical therapies 3956.