Targeting Aging with Metformin trial
The Geroscience Paradigm
For the past century, the standard medical and regulatory paradigm has approached age-related diseases as distinct, isolated pathologies. Under this framework, cardiovascular disease, oncology, neurology, and endocrinology operate as disparate medical specialties utilizing highly targeted therapeutics. However, the emerging field of geroscience posits an alternative model: that biological aging itself is the primary, shared risk factor driving the onset of these chronic conditions 12. By targeting the fundamental molecular hallmarks of aging - such as genomic instability, loss of proteostasis, mitochondrial dysfunction, cellular senescence, and stem cell exhaustion - geroscience suggests that the progression of multiple diseases could be delayed simultaneously 34. This approach shifts the medical objective from incrementally extending lifespan by curing individual diseases to extending "healthspan," defined as the period of life spent free from chronic disease and disability 15.
Translating the geroscience hypothesis from successful laboratory animal models to human clinical application faces a severe administrative bottleneck. Major regulatory bodies, including the United States Food and Drug Administration (FDA) and the European Medicines Agency (EMA), do not classify aging as a treatable disease 667. Because regulatory agencies only approve therapeutics for specific, recognized disease indications, there is no standardized pathway to run a clinical trial for an aging intervention. Pharmaceutical developers are thus forced to test potential geroprotectors against single diseases, which severely limits the scope of the data collected and dampens the commercial viability of broad, preventative anti-aging therapeutics 79.
Conceived to break this regulatory deadlock, the Targeting Aging with Metformin (TAME) trial was proposed by Dr. Nir Barzilai and colleagues at the Albert Einstein College of Medicine, operating in collaboration with the American Federation for Aging Research (AFAR) 89. TAME is designed not merely as a test of a specific molecule, but as a proof-of-concept regulatory template. Its primary objective is to convince the FDA to accept a composite clinical endpoint that serves as a proxy for biological aging, thereby creating a pathway for future therapeutic approvals 7910.
Pharmacological Profile of Metformin
Metformin (dimethylbiguanide) is a first-line oral antihyperglycemic agent that has been used successfully to treat type 2 diabetes mellitus for over six decades 1311. Originally derived from compounds found in the French lilac (Galega officinalis), it is an inexpensive, off-patent generic drug with an extensively documented and highly favorable safety profile 5713. The decision to utilize metformin for the landmark TAME trial was not necessarily because biogerontologists believe it to be the most potent geroprotector known to science, but because its unparalleled safety record presented the path of least regulatory resistance for an unprecedented trial design 7.
Interest in metformin as a longevity agent was catalyzed heavily by large-scale retrospective observational studies. Most notably, a 2014 study involving over 78,000 diabetic patients in the United Kingdom indicated that type 2 diabetics prescribed metformin exhibited lower all-cause mortality rates than age-matched, non-diabetic controls who were not taking the drug 151617. While subsequent researchers have pointed out potential survival biases and methodological artifacts in these early observational studies, the data provided a compelling initial justification for prospective clinical trials 1017. Furthermore, metformin use in diabetic populations has been continuously associated with a reduced incidence of certain cancers, cardiovascular events, and cognitive decline 121314.
Metformin exerts complex, multi-systemic effects that overlap significantly with the recognized hallmarks of aging 4. Its primary identified mechanism involves the mild, transient inhibition of complex I of the mitochondrial electron transport chain 1715. This partial inhibition reduces intracellular ATP production, leading to an increase in the AMP-to-ATP ratio. This metabolic shift activates AMP-activated protein kinase (AMPK), a highly conserved cellular energy sensor 1515.
Once activated, AMPK downregulates the mechanistic target of rapamycin (mTOR) signaling pathway, specifically mTORC1. The suppression of mTOR and the simultaneous activation of AMPK effectively mimic the physiological state of caloric restriction, shifting the cell away from active growth and protein synthesis and toward stress resistance, autophagy, and efficient lipid metabolism 1516. Additionally, metformin administration has been shown to reduce the secretion of pro-inflammatory cytokines, mitigating the chronic, low-grade systemic inflammation often termed "inflammaging," while also exerting favorable modulations on the gut microbiome and reducing oxidative stress 121516. By lowering circulating insulin and insulin-like growth factor 1 (IGF-1) levels, metformin influences nutrient-sensing pathways that have been evolutionarily conserved to regulate lifespan in various model organisms, including nematodes and rodents 1517.
The TAME Trial Architecture
The TAME trial represents the first explicitly designed geroscience-guided aging outcomes trial in humans 18. The architecture of the trial was negotiated directly with the FDA over several years to ensure that, if successful, its results would be sufficient to grant an official medical indication for treating aging-related morbidity 119.
The study protocol outlines the recruitment of approximately 3,000 ethnically diverse, non-diabetic men and women between the ages of 65 and 79 820. Participants are to be randomized in a double-blind, placebo-controlled format to receive either 1,500 mg of metformin daily or a placebo over a period of up to six years 1820. The trial is designed to be distributed across 14 leading clinical research institutions in the United States, with the Wake Forest University School of Medicine serving as the primary coordinating center 8.
The Composite Primary Endpoint
The central, paradigm-shifting innovation of the TAME trial is its endpoint selection. Because measuring biological aging directly via molecular clocks remains highly debated and lacks standardized regulatory consensus, TAME utilizes a composite primary endpoint based on clinical disease incidence 1821. Rather than measuring the cure, reversal, or prevention of a single specific disease, the trial measures the time to the first incidence of any one of a cluster of major age-related chronic diseases 122.
This composite endpoint includes myocardial infarction, congestive heart failure, stroke, most forms of cancer, mild cognitive impairment (MCI) or dementia, and all-cause mortality 2021.
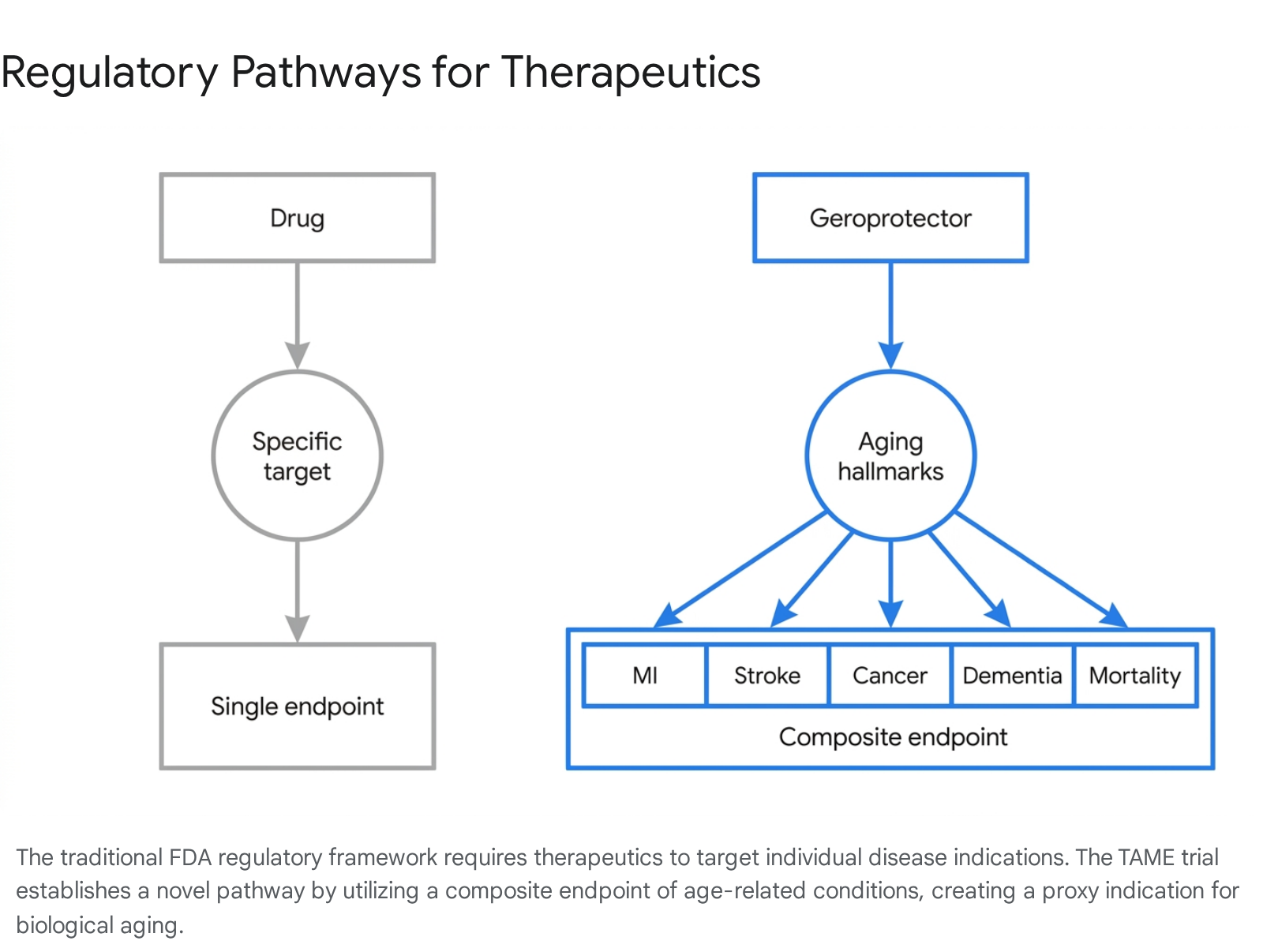
Participants trigger the endpoint if they develop any of these conditions during the observation period. The underlying statistical hypothesis is that targeting the fundamental biological aging process will delay the onset of the entire cluster of diseases. The trial is powered at 90% to detect a 22.5% reduction in the incidence of this composite endpoint 20.
Biomarker Analysis and TAME BIO
In addition to tracking macroscopic clinical events, the trial protocol includes the establishment of a biologic specimen repository. An ancillary project, referred to as TAME BIO, aims to collect blood, plasma, urine, stool, and genetic material to track how metformin impacts specific molecular biomarkers of aging over the six-year period 23. Candidate blood-based biomarkers proposed by the TAME Biomarkers Workgroup include Interleukin-6 (IL-6), Insulin-like Growth Factor 1 (IGF-1), Cystatin C, Growth Differentiation Factor 15 (GDF15), and High-Sensitivity C-Reactive Protein (hsCRP) 24. Monitoring these markers provides a dual benefit: it elucidates the molecular mechanisms of the intervention in healthy humans and helps validate surrogate endpoints for future, shorter-duration clinical trials testing next-generation geroprotectors.
| Trial Component | Specification |
|---|---|
| Study Design | Double-blind, placebo-controlled, multi-center randomized trial 1820 |
| Target Population | ~3,000 non-diabetic men and women, ages 65 - 79 820 |
| Intervention | Metformin (1,500 mg/day) vs. Placebo 1220 |
| Duration | Up to 6 years of follow-up 818 |
| Coordinating Center | Wake Forest University School of Medicine (across 14 U.S. sites) 8 |
| Composite Primary Endpoint | Incidence of myocardial infarction, congestive heart failure, stroke, cancer, MCI/dementia, or death 2021 |
| Statistical Power | 90% power to detect a 22.5% reduction in the primary endpoint 20 |
| Key Biomarkers Tracked | IL-6, IGF-1, Cystatin C, GDF15, HbA1c, hsCRP (via TAME BIO) 2324 |
Regulatory Implications and the ICD-11
The greatest hurdle in the longevity biotechnology sector is not merely discovering effective compounds, but navigating a regulatory framework that was designed exclusively for infectious and acute diseases. The FDA's steadfast refusal to classify aging as a disease means that without the TAME trial's composite endpoint template, it is effectively impossible to bring a drug to market for the explicit purpose of extending healthspan 79.
While the FDA has yet to officially formalize a blanket regulatory pathway for aging therapeutics outside of the specific TAME negotiations, international health frameworks are evolving more rapidly. The World Health Organization (WHO) implemented the 11th Revision of the International Classification of Diseases (ICD-11), which introduced significant changes to the categorization of age-related physical decline 2526.
Advocacy by organizations such as the International Longevity Alliance resulted in the inclusion of the extension code XT9T, officially designated as "Ageing-related" 2527. This code acts as an etiology modifier that can be attached to other pathologies, explicitly recognizing biological aging as a causal factor for disease. Furthermore, the stem code MG2A, originally designated with the nebulous term "Old age," was refined to "Aging-associated decline in intrinsic capacity," providing a far more measurable, symptom-based framework for clinical intervention 282930.
These ICD-11 classifications bridge a vital gap between traditional clinical medicine and geroscience. By providing specific billing, diagnostic, and causal codes for aging-related decline, the WHO has enabled researchers and clinicians outside the United States to register trials and target aging parameters directly, without waiting for the FDA to formally alter its definition of a disease 2937. For the TAME trial, achieving a formal indication from the FDA remains the ultimate goal, as an FDA indication unlocks insurance reimbursement models, incentivizes private pharmaceutical investment, and validates the geroscience paradigm on the global stage 91331.
| Regulatory Body | Current Stance on Biological Aging | Coding / Pathway Mechanism | Implications for Longevity Clinical Trials |
|---|---|---|---|
| U.S. FDA | Aging is a natural process, not a disease indication. | Single-disease endpoints (Traditional) vs. Composite Endpoint (TAME exception). | Trials must focus on disease clusters; anti-aging therapeutics face high friction. 67 |
| WHO (ICD-11) | Aging is a major disease risk factor and etiology. | Extension Code XT9T ("Ageing-related") & Stem Code MG2A. | Allows international researchers to target aging directly as a medical condition. 252629 |
Critiques and the Rapamycin Comparison
While the regulatory design of the TAME trial is universally lauded as a necessary evolution by the geroscience community, the choice of metformin as the primary therapeutic agent is highly debated among biogerontologists. As longitudinal data and meta-analyses accumulate into 2026, a growing contingent of researchers argue that metformin's effects in healthy, non-diabetic populations may be negligible, overstated, or even detrimental in specific contexts 71117.
A central critique revolves around the early retrospective studies that catapulted metformin into the longevity spotlight. Critics point out that these studies suffered from significant methodological artifacts, most notably survival bias and immortal time bias 10. The seminal Bannister study, for instance, unintentionally selected the healthiest diabetic patients - those who successfully managed their condition with first-line metformin - and compared them against patients whose diabetes was harder to control and required second-line medications. When patients' diabetes worsened and they were taken off metformin, they were systematically dropped from the metformin cohort's data, creating an illusion that metformin users lived longer than the general population solely because the study only counted users who remained healthy enough to stay on the drug 10.
Compared to other candidate geroprotectors, metformin's performance in rigorous animal longevity models is also inconsistent. In the National Institute on Aging's Interventions Testing Program (ITP) - considered the gold standard for assessing lifespan extension in genetically heterogeneous mice across multiple independent laboratories - metformin showed weak or negligible effects on maximum lifespan 732.
In sharp contrast, rapamycin (and its analogues, rapalogues), an immunosuppressive drug that acts as a direct, allosteric inhibitor of the mTOR complex, has consistently demonstrated robust lifespan extension across multiple species, from yeast and nematodes to mice and rhesus monkeys 3233. A 2025 meta-analysis published in Aging Cell confirmed that rapamycin's lifespan-boosting effects rival those of strict dietary restriction, whereas metformin showed no reliable uptick in longevity across non-human vertebrates 323334. Rapamycin has also demonstrated profound effects on immune rejuvenation in older adults, improving vaccine responses and antiviral gene signatures in human trials 33.
| Feature / Mechanism | Metformin | Rapamycin |
|---|---|---|
| Primary Target | Mitochondrial Complex I; Indirect AMPK activation 1517 | Direct allosteric inhibition of mTORC1 3342 |
| Lifespan Extension (Animal Models) | Inconsistent; minimal to zero gains in robust, multi-site trials (e.g., NIA ITP) 732 | Highly consistent; double-digit percentage lifespan extension in mice 3242 |
| Human Clinical Use History | Approved for Type 2 Diabetes; >60 years of widespread use 1311 | Approved as an immunosuppressant for organ transplants (e.g., kidney) 3242 |
| Safety Profile | Extremely high; low risk of hypoglycemia or lactic acidosis 1122 | High continuous doses cause immunosuppression, mouth ulcers, glucose dysregulation, and lipidemia 344235 |
| Suitability for TAME Trial | Selected primarily due to pristine safety profile and low regulatory friction 7 | Rejected for massive primary prevention trials due to side-effect profile and perceived FDA hurdles 7 |
Despite rapamycin's superior biological efficacy in model organisms, its side-effect profile made it an unviable candidate for a massive, preventative clinical trial in healthy seniors. Continuous dosing of rapamycin carries risks of hyperlipidemia, hyperglycemia, recurrent mouth ulcers, and potentially dangerous immunosuppression, rendering it unacceptable for the TAME trial's objectives under current FDA risk-tolerance paradigms 7344235. Consequently, longevity researchers are forced to test the less effective but immensely safer metformin to establish the regulatory precedent.
The Exercise Blunting Paradox
Beyond debates regarding metformin's baseline efficacy, one of the most critical concerns regarding its use as a geroprotector is its interaction with physical exercise. Physical exercise - specifically progressive resistance training and aerobic conditioning - remains the most robust, evidence-based intervention for extending human healthspan, preserving metabolic function, and preventing sarcopenia 353637. Because metformin inhibits mitochondrial respiration and fundamentally alters cellular energy sensing, it can actively interfere with the physiological adaptations normally induced by physical exertion 38.
The MASTERS (Metformin to Augment Strength Training Effective Response in Seniors) trial provided compelling, randomized, double-blind evidence of this phenomenon. In this trial, healthy adults aged 65 and older underwent 14 weeks of supervised progressive resistance training (PRT). Participants taking 1,700 mg of metformin daily exhibited significantly blunted gains in lean muscle mass, thigh muscle volume, and cross-sectional area compared to the placebo group 3940. Transcriptomic profiling of muscle biopsies revealed that metformin suppressed mTORC1 signaling - the primary driver of muscle protein synthesis - and attenuated the activation of core anabolic and mitochondrial biogenesis pathways 3941.
Similar studies evaluating aerobic exercise training (AET) have shown that metformin attenuates improvements in cardiorespiratory fitness (measured by VO2 peak) and blunts exercise-induced enhancements in whole-body insulin sensitivity 3842. A 2026 meta-analysis confirmed that metformin use was associated with significantly smaller improvements in aerobic capacity and attenuated reductions in both systolic and diastolic blood pressure compared to exercise alone 42.
This presents a profound clinical paradox: a drug intended to delay biological aging may actively inhibit the hypertrophic and metabolic benefits of exercise, potentially accelerating age-related muscle loss and frailty in active older adults 3741. The mechanism underlying this paradox likely involves metformin's reduction of reactive oxygen species (ROS) generation; while reducing ROS lowers oxidative stress at rest, it also dampens the necessary redox-sensitive signals required to trigger optimal training adaptations in skeletal muscle 42. For patients reliant on strength training to maintain mobility and independence, the adjunctive use of metformin remains highly controversial.
| Exercise Modality | Impact of Metformin Addition | Physiological Mechanism of Interference |
|---|---|---|
| Progressive Resistance Training (PRT) | Blunts gains in lean body mass, thigh muscle area, and density 39 | Suppresses mTORC1 signaling; inhibits type I fiber frequency expansion 3941 |
| Aerobic Exercise Training (AET) | Attenuates improvements in VO2 peak and insulin sensitivity 3842 | Direct inhibition of mitochondrial complex I; dampening of redox-sensitive (ROS) signals 3842 |
| Cardiovascular Response | Attenuates exercise-induced reductions in systolic and diastolic blood pressure 42 | Interaction with core transcription factors involved in angiogenesis and oxidative capacity 3842 |
Trial Funding and Current Status
Despite receiving unprecedented conceptual approval for its composite study design from the FDA in 2015, the TAME trial has been persistently plagued by funding shortfalls. As of mid-2026, the trial has struggled to initiate full-scale participant enrollment and dosing across its intended 14 centers 921.
The fundamental obstacle facing TAME is the economic structure of traditional pharmaceutical development. The trial requires an estimated $45 million to $75 million to execute fully over its six-year duration 7943. Because metformin is a widely available, off-patent generic drug that costs pennies per pill, no pharmaceutical company stands to gain exclusive market rights or recoup the massive costs of the clinical trial 943. Consequently, traditional industry funding is non-existent.
The trial relies entirely on public grants and philanthropic donations. The National Institute on Aging (NIA) contributed initial grant funding (approximately $5 million to $9 million) primarily directed toward biomarker development, but government organizations have historically allocated minimal budgets to non-disease-specific basic geroscience 79. While a $40 million commitment from a private donor helped close a significant portion of the budget gap in 2019, the logistical and financial complexities of maintaining a 3,000-person, multi-year clinical trial have led to ongoing delays, with some academic reviews explicitly noting that the trial is stalled solely due to capital constraints 9944.
The TAME trial highlights a systemic market failure in global healthcare: billions of dollars in private venture capital flow readily into proprietary longevity biotech startups developing patented molecules, while a comparatively inexpensive public trial that could unlock an entirely new regulatory framework languishes for lack of an immediate profit incentive 9. U.S. healthcare spending allocates trillions of dollars to treating the downstream consequences of aging, but only a fraction of a percent to researching the biological processes that cause them 9.
Due to these systemic delays, the pharmaceutical industry is increasingly looking to adapt the TAME regulatory template for highly profitable, patented compounds. Reports in 2026 indicate that major pharmaceutical entities, such as Eli Lilly, are negotiating with the FDA to run "TAME-like" trials using proprietary drugs - specifically GLP-1 receptor agonists - which solve the funding equation that an off-patent drug like metformin could not 9.
Conclusion
The true legacy of the Targeting Aging with Metformin trial may ultimately be administrative and regulatory rather than pharmacological. The scientific community remains divided on whether metformin will prove to be a potent geroprotector in healthy, non-diabetic humans. Given its inconsistent performance in robust animal lifespan models and its documented tendency to blunt the vital physiological adaptations of exercise, metformin may not be the elusive "longevity pill" that early epidemiological studies suggested 5739.
However, whether metformin extends human healthspan by a significant margin, or whether its dampening of exercise adaptations renders it a net-neutral intervention in active populations, the TAME trial has already achieved a historic milestone 579. By negotiating a composite endpoint of age-related morbidities with the FDA, Dr. Nir Barzilai, AFAR, and their colleagues have successfully charted a navigable regulatory pathway where none previously existed 19.
If fully executed and successful, TAME will prove that the incidence of multiple chronic diseases can be delayed simultaneously by a single therapeutic agent. This proof-of-concept will force the global healthcare system to transition from a reactive, disease-specific model to a proactive, healthspan-extending paradigm 1619. Even if the trial ultimately serves solely as a bureaucratic template for newer, more potent, and commercially viable geroprotectors like rapalogues or GLP-1 agonists, TAME represents the vital first step toward classifying and treating biological aging as a modifiable medical condition.