Sunlight and circadian mood regulation
Neuroanatomical Pathways of Light Reception
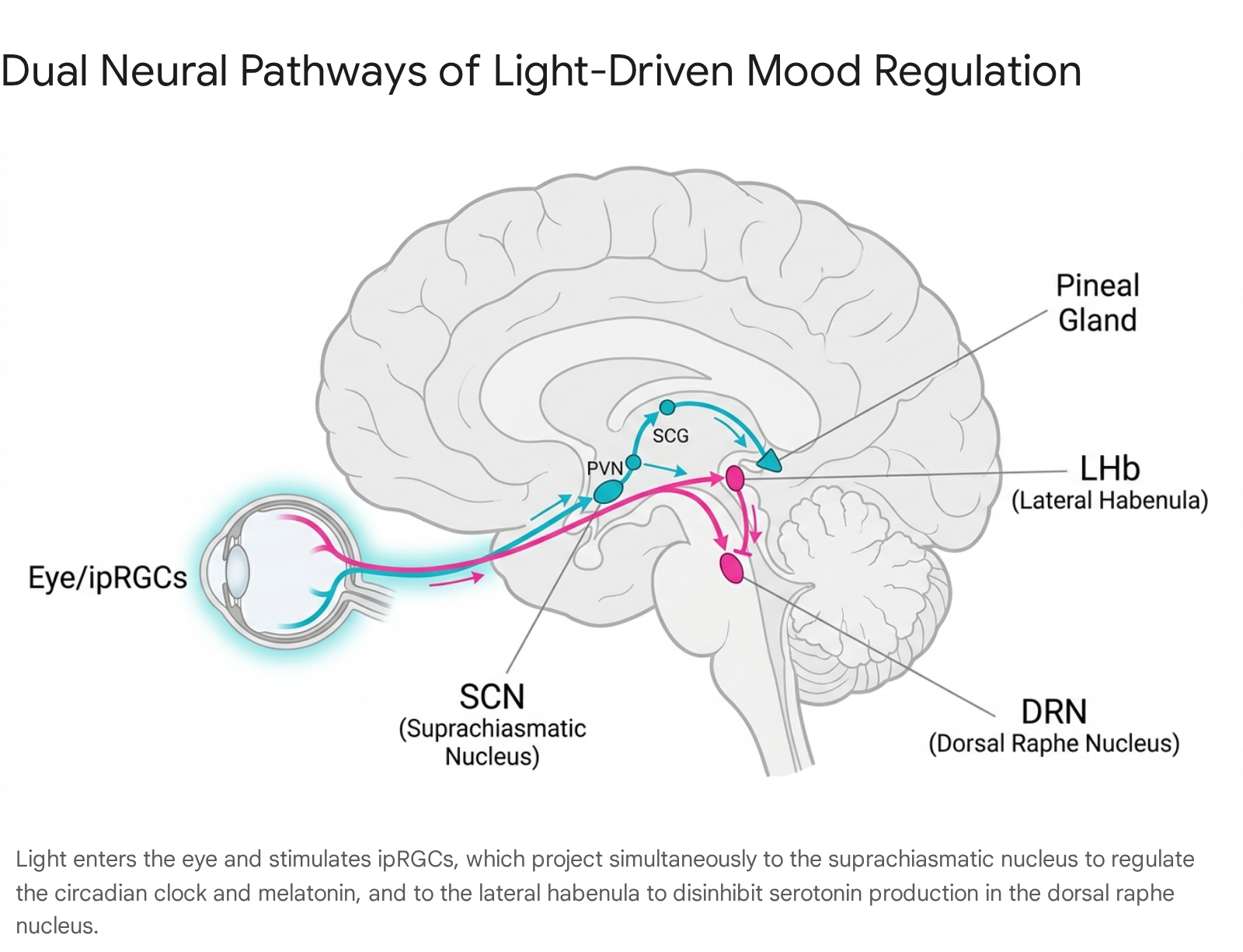
The biological impact of light on human physiology begins within the retina, but it extends far beyond the visual cortex. A specialized subset of retinal neurons, known as intrinsically photosensitive retinal ganglion cells (ipRGCs), serves as the primary conduit for environmental light data 1. Unlike rods and cones, which are responsible for image formation, ipRGCs - specifically the M1 subtype - express melanopsin, a photopigment highly sensitive to short-wavelength blue light within the 460 - 480 nanometer (nm) range 12. These cells form the afferent projections of the retinohypothalamic tract (RHT), a monosynaptic neural pathway that communicates ambient light levels directly to the brain 1.
The Retinohypothalamic Tract and Suprachiasmatic Nucleus
The retinohypothalamic tract terminates in the ventrolateral core region of the suprachiasmatic nucleus (SCN), a structure located in the hypothalamus that functions as the body's master circadian pacemaker 13. At the RHT terminals within the SCN, the principal neurotransmitters released are glutamate and pituitary adenylate cyclase-activating polypeptide (PACAP) 1. Glutamate binds to N-methyl-D-aspartate (NMDA) receptors on SCN neurons, triggering an intracellular signaling cascade that synchronizes the molecular clock to the external solar day 1. PACAP acts as a modulator, adjusting the sensitivity of the SCN and ensuring entrainment stability 1. The SCN subsequently orchestrates the alignment of peripheral cellular clocks across all organs and tissues, establishing the physiological homeostasis required for optimal metabolic and neurological function 4.
Multisynaptic Pathways for Melatonin Regulation
The downstream effects of SCN activation bifurcate into two primary chemical cascades that regulate sleep and mood: the synthesis of melatonin and the modulation of serotonin. The regulation of melatonin is orchestrated by the SCN via a complex multisynaptic circuit. During periods of light exposure, SCN activity inhibits this circuit 1. In the absence of light, SCN activity wanes, and signals are projected to the paraventricular nucleus (PVN) of the hypothalamus 1. The PVN then signals preganglionic sympathetic neurons located in the intermediolateral cell column (IML) of the spinal cord 1. These neurons drive postganglionic fibers from the superior cervical ganglion (SCG), which ultimately innervate the pineal gland, the primary site of melatonin synthesis 1.
Direct Neural Projections for Serotonin Modulation
While the SCN pathway manages the timing of sleep through melatonin, light actively facilitates serotonin transmission through a secondary, non-circadian neural circuit. Certain ipRGC subtypes bypass the SCN entirely and project directly to the lateral habenula (LHb) 1. The LHb exerts inhibitory control over the midbrain's monoaminergic systems, specifically the dorsal raphe nucleus (DRN), which houses the brain's primary serotonergic neurons 1. Under dark conditions, the LHb suppresses the DRN. However, acute light exposure suppresses the firing of LHb neurons, effectively disinhibiting the DRN 1. This disinhibition facilitates an immediate release of serotonin, thereby elevating mood, motivation, and alertness independently of the circadian phase 1.
Biochemical Mechanisms of Hormone Synthesis
Serotonin N-Acetyltransferase and Pineal Regulation
Within the pineal gland, melatonin is synthesized from serotonin through a highly regulated biochemical conversion. This process requires two rate-limiting enzymes: serotonin N-acetyltransferase (SNAT, also referred to as arylalkylamine N-acetyltransferase, or AANAT) and hydroxyindole O-methyltransferase (HIOMT) 567. SNAT catalyzes the conversion of serotonin to N-acetylserotonin, which HIOMT subsequently methylates into melatonin 58.
This synthesis is entirely dependent on the light-dark cycle. During dark hours, intracellular calcium levels increase, potentiating protein kinase C (PKC) and calcium-calmodulin protein kinase (CaM kinase) 5. This leads to an increase in cyclic AMP (cAMP) and the phosphorylation of SNAT by protein kinase A (PKA) 59. Phosphorylated SNAT binds to 14-3-3 proteins, forming a complex that protects the enzyme from proteolytic destruction, resulting in a dramatic 7- to 150-fold increase in enzyme activity during the night 69. Environmental light exerts a powerful suppressive effect on this process; acute light exposure detected by ipRGCs causes an immediate degradation of SNAT, halting melatonin synthesis and inducing wakefulness 16.
Tryptophan Metabolism and Serotonergic Tone
The availability of serotonin itself is subject to seasonal and photic fluctuations. Serotonin synthesis relies on the availability of its precursor, the essential amino acid tryptophan, and the rate-limiting enzyme tryptophan hydroxylase (TPH) 79. Brain serotonin levels exhibit significant seasonal variations, dropping precipitously during the winter months 10. Positron Emission Tomography (PET) studies have confirmed that serotonin transporter binding potential in the brain is significantly higher during fall and winter compared to spring and summer 10. This heightened transporter activity aggressively clears serotonin from the synaptic cleft, effectively reducing serotonergic tone and elevating the risk of depressive symptoms during months with reduced photoperiods 10.
Physics of Light Exposure and Environmental Constraints
Illuminance Metrics and Melanopic Efficacy
The biological efficacy of light depends heavily on its intensity, which is quantified in lux (lumens per square meter), alongside its spectral power distribution 11. The human visual system adapts seamlessly to dramatic variations in luminosity, which masks the vast discrepancy between natural outdoor light and artificial indoor environments 11.
Natural daylight provides an illuminance stimulus that standard indoor environments cannot replicate. Direct sunlight generates between 32,000 and 130,000 lux, while an overcast sky yields 1,000 to 10,000 lux 12. In stark contrast, standard indoor lighting is calibrated primarily for visual acuity, typically providing only 300 to 500 lux at the work plane 13. To quantify the biological impact of these light levels on the ipRGC melanopsin system, researchers utilize the melanopic equivalent daylight illuminance (mel-EDI) metric 141715. The Commission Internationale de l'Eclairage (CIE) recommends a minimum mel-EDI of 250 lux measured vertically at eye level throughout the daytime to promote optimal physical and mental health 1716.
| Lighting Environment | Typical Illuminance (Lux) | Biological Impact / Melanopic Efficacy |
|---|---|---|
| Direct Summer Sunlight | 32,000 - 130,000 | Rapid circadian entrainment; robust acute serotonin release 1112. |
| Overcast Daylight | 1,000 - 10,000 | Sufficient for circadian maintenance; typical benchmark for light therapy devices 13. |
| Well-Lit Office | 300 - 500 | Visually adequate; frequently insufficient for optimal melanopsin activation (mel-EDI) 13. |
| Residential Living Room | 50 - 100 | Promotes twilight physiology; initiates early melatonin synthesis stages 12. |
| Civil Twilight | 1 - 10 | Minimal visual acuity; threshold for deep circadian resting phase 1213. |
These vast disparities explain why modern populations, spending upwards of 90% of their time indoors, experience chronic, low-grade circadian disruption. The visual system can read a monitor at 300 lux, but the ipRGC network registers this environment as biological twilight 17.
Spectral Filtration by Architectural Glazing
The barrier between natural outdoor light and the indoor environment is mediated by window glazing, which profoundly alters both the intensity and spectral composition of incoming light. Recent analyses evaluating the melanopic action factor of glazing ($MAF_{Glazing}$) demonstrate that standard architectural glass severely limits circadian light transmission 14.
Modern energy-efficient buildings heavily utilize three-layered low-emissivity (Low-E) glass to control thermal transfer. Spectroradiometric laboratory comparisons reveal that this three-layered glass reduces the active spectral range required to stimulate ipRGCs (460 - 480 nm) by approximately 20% when compared to two-layered low-iron glass 1819. Furthermore, these heavy glazings entirely block UV-B transmission (280 - 315 nm), eliminating natural sunlight's capacity to drive vitamin D synthesis indoors and mitigating potential germicidal effects on airborne pathogens 18.
Because of light scattering, spectral filtration, and structural shading, researchers conclude that viewing sunlight through a standard residential or automotive window is up to 50 times less biologically effective for circadian entrainment than direct, unmediated outdoor exposure 202122. Light must strike the retina unhindered at a sufficient intensity to trigger the SCN. Therefore, even highly illuminated indoor spaces adjacent to windows frequently fail to meet the biological requirements for circadian maintenance 1523.
Chronodisruption and Psychiatric Vulnerability
Sleep Disturbance as a Transdiagnostic Risk Factor
The desynchronization of the internal biological clock from the external solar day - a state defined as chronodisruption - is increasingly viewed as a foundational etiology in psychiatric disorders 42425. A robust body of evidence links sleep timing variability and poor circadian amplitude to major depressive disorder (MDD), anxiety, and schizophrenia 252627.
Individuals displaying an evening chronotype (a behavioral predisposition to late sleep-wake cycles) endure significantly higher rates of severe psychiatric symptoms and metabolic comorbidities 28. This vulnerability is compounded by "social jetlag," the chronic misalignment between a person's endogenous circadian preference and the strict schedules demanded by work or education 428. Meta-analyses indicate that shift workers subjected to regular circadian inversion are 25% to 40% more likely to develop depression and anxiety 28. Furthermore, a lack of consolidated sleep actively alters cognition; sleep-deprived brains exhibit a strong negative salience bias, retaining memories of negative experiences while failing to consolidate positive ones 29.
Intervention studies confirm a causal direction between circadian sleep health and mood. A comprehensive meta-analysis of 65 randomized controlled trials (N = 8,608) assessing the psychiatric impact of sleep interventions demonstrated significant improvements across multiple domains 30. Improving sleep quality yielded a medium-sized effect on composite mental health (g = -0.53), depression (g = -0.63), anxiety (g = -0.51), and rumination (g = -0.49), alongside small-to-medium effects on stress (g = -0.42) and positive psychosis symptoms (g = -0.26) 30. This dose-response relationship suggests that erratic circadian rhythms actively drive pathology rather than merely presenting as secondary symptoms of mental illness 3031.
Bipolar Disorder and Mood Seasonality
The influence of seasonality and photoperiod shifts is particularly acute in bipolar disorder (BD). Longitudinal data from the Prechter Longitudinal Study of Bipolar Disorder reveals profound seasonal patterns in both BD Type I and BD Type II 3233. Across monitored patients, depressive symptoms consistently peak during the autumn and winter months, correlating inversely with daylight availability 33. Conversely, episodes of mania and hypomania surge during the spring and summer, tracking with the lengthening of the photoperiod 33.
Individuals with bipolar disorder exhibit significantly higher scores on the Global Seasonality Score (GSS) than healthy controls, and high seasonality correlates strongly with a greater variance in overall mood instability 32. The circadian pacemaker in bipolar patients appears highly fragile. During mixed manic episodes, patients frequently display circadian phase delays of 6 to 7 hours, whereas depressive episodes show delays of 4 to 5 hours, underscoring the severe chronobiological dysfunction inherent to the disorder 27. Because of this hyper-sensitivity, targeted circadian interventions are increasingly recognized as necessary tools for relapse prevention in bipolar populations 33.
Genetic Susceptibility and Population Variations
Clock Gene Variants and Polygenic Risk
While reduced photoperiod is the primary environmental trigger for seasonal mood changes, susceptibility to these changes is highly individualized and deeply rooted in genetics. Molecular pathways governing mood are intrinsically linked to core circadian clock gene variants 1034.
Machine learning models utilizing deeply phenotyped population samples have identified sex-specific polygenic risk factors for seasonality and Seasonal Affective Disorder (SAD) 34. In males, genotypic combinations involving variants of CLOCK, ZBTB20, and PER2 serve as significant genetic risk factors. In females, risk is more heavily mediated by variants in CRY2 and PER3 34. Conversely, specific variants, such as CRY1-GG, provide protective effects against SAD symptoms 34. The transcription factor ZBTB20 is particularly notable, as it is involved in synaptic transmission impacting glucose homeostasis, glucocorticoid stress pathways, and monoaminergic depression pathways, suggesting a direct mechanistic link between cellular clock function and systemic mood regulation 34.
Polygenic risk scores analyzing broader psychiatric vulnerability confirm that genetic predispositions for MDD, BD, anxiety disorders, and low vitamin D levels are positively associated with a continuous measure of seasonality, the general seasonality score (GSS) 35. This indicates a shared genetic architecture between biological rhythm sensitivity and affective disorders 35.
Latitudinal Anomalies and Cultural Isolation
Epidemiological meta-regressions confirm a significant positive association between latitude and the prevalence of SAD and subsyndromal SAD (SSAD) 3640. As distance from the equator increases, the winter photoperiod shrinks, triggering melatonin overproduction and serotonin deficits in susceptible individuals 4037. For instance, SAD prevalence is approximately 1% in Florida but scales to nearly 9% in Alaska 40.
| Geographic Region | Latitude | SAD Prevalence Estimate | Contextual Note |
|---|---|---|---|
| Florida, USA | ~27° N | 1.0% | High winter solar insolation; low baseline risk 40. |
| Maryland, USA | 39° N | 4.3% | Mid-latitude baseline 40. |
| New York, USA | 40° N | 4.7% | Mid-latitude baseline 40. |
| Alaska, USA | ~60° N+ | 9.0% | Extreme photoperiod reduction; high expected prevalence 40. |
| Iceland | 64 - 67° N | 3.6% | Latitudinal anomaly; unexpectedly low prevalence despite extreme darkness 3839. |
| Manitoba, Canada (Non-Icelandic descent) | 50° N | 9.1% | Control population matching expected latitudinal prevalence 3839. |
| Manitoba, Canada (Icelandic descent) | 50° N | 4.8% | Demonstrates retained genetic resilience independent of latitude 3839. |
The latitudinal hypothesis presents notable anomalies that underscore the importance of genetic isolation and environmental adaptation. The most prominent outlier is Iceland. Despite being situated at high latitudes with extreme winter darkness, the native Icelandic population exhibits surprisingly low rates of SAD (approximately 3.6%) 3839.
To isolate whether this resilience is cultural or genetic, researchers examined populations of wholly Icelandic descent living in Manitoba, Canada, comparing them to non-Icelandic populations in the same geographic area 3839. The Canadian-Icelandic cohort demonstrated a SAD prevalence of only 4.8%, significantly lower than the 9.1% rate seen in the non-Icelandic local population 3839. This discrepancy suggests that generations of exposure to extreme photoperiods have selected for unique genetic adaptations in the Icelandic genome, conferring biological resilience against dark-induced mood degradation 4038.
Behavioral Interventions and Non-Photic Zeitgebers
Time-Restricted Eating and Metabolic Clocks
While light remains the primary zeitgeber for the central suprachiasmatic nucleus, peripheral clocks located in the liver, gut, and muscle tissues are heavily entrained by non-photic cues, most notably food intake 3440. Extensive research by chronobiologists into feeding-fasting cycles indicates that erratic eating patterns decouple peripheral metabolic clocks from the central SCN pacemaker, contributing to systemic chronodisruption and psychiatric metabolic comorbidities 22840.
Time-restricted eating (TRE), which confines caloric intake to a consistent 8- to 12-hour diurnal window, aligns digestive processes with optimal metabolic phases 345. Observational data utilizing smartphone-based food logs from over 20,000 adults reveals significant variability in modern food timing, often extending late into the night 40. Aligning meal timing prevents circadian misalignment, supports diurnal rhythmicity in the gut microbiota, and increases systemic resilience against both metabolic and mental health disorders 284045.
Nocturnal Light Avoidance
The intrusion of artificial light at night (ALAN) heavily exacerbates chronodisruption. Exposure to bright, short-wavelength light between 10:00 PM and 4:00 AM profoundly suppresses nocturnal melatonin synthesis and alters the normal circadian phase 4142. Furthermore, nocturnal exposure to ultraviolet and blue light is shown to decrease dopamine output in the brain, directly worsening feelings of anxiety and depressive anhedonia 4248.
To mitigate this, specialized behavioral protocols utilizing long-wavelength light at night are recommended. Light in the red spectrum (longwave) penetrates tissue but does not activate the melanopsin receptors in ipRGCs 4248. Consequently, utilizing dim red or amber lighting during nocturnal awakenings preserves cortisol nadirs and leaves melatonin synthesis undisturbed, protecting the neurochemical environment required for restorative sleep and emotional regulation 4248.
Clinical Light Therapy Protocols
Efficacy in Seasonal Affective Disorder
Bright Light Therapy (BLT) remains the gold standard, first-line intervention for Seasonal Affective Disorder. By artificially extending the photoperiod and providing a high-intensity circadian anchor, BLT corrects phase-delayed circadian rhythms and normalizes neurotransmitter metabolism 4344.
The clinically accepted standard protocol involves exposure to a 10,000-lux, cool-white fluorescent or LED light box, administered for 30 to 60 minutes immediately upon waking 17434546. The light must strike the retina at an angle of 30 to 60 degrees to prevent glare and retinal damage, requiring patients to sit roughly 60 to 80 centimeters from the device with their eyes open 47. Efficacy is highly dependent on timing. Morning administration yields remission rates exceeding 53%, compared to significantly lower efficacies when administered at mid-day (32%) or evening (38%) 1745. When matched against pharmacological interventions, trials indicate that 6 weeks of BLT is as effective as cognitive behavioral therapy or fluoxetine monotherapy, with patients often exhibiting a faster initial response 4547.
Recent network meta-analyses comparing the spectral wavelength (color) of light therapies specifically for SAD reveal that full-spectrum white light remains the most effective intervention 48. While narrow-band blue light efficiently targets the melanopsin peak, clinical data indicates that white light provides superior overall alleviation of seasonal mood dysregulation, followed sequentially by green, blue, and red light 48. Red light serves primarily as a placebo control in these specific SAD phototherapy contexts 48.
Efficacy in Non-Seasonal Depressive Disorders
Historically relegated exclusively to seasonal pathologies, BLT is now recognized as a potent intervention for non-seasonal major depressive disorder. A landmark 2024 systematic review and meta-analysis published in JAMA Psychiatry analyzed 11 randomized clinical trials comprising 858 patients 465649. The study evaluated the efficacy of BLT (10,000 lux for 30 - 60 minutes) used either as monotherapy or as an adjunctive treatment to standard antidepressants, compared against controls utilizing dim red light or placebo pills.
The findings conclusively demonstrate that BLT is an effective adjunctive treatment for non-seasonal depression. Patients receiving BLT exhibited a statistically significant remission rate of 40.7%, outperforming the 23.5% remission rate in the control group 56. Furthermore, the overall clinical response rate - defined as a clinically significant reduction in depressive symptoms - was 60.4% in the BLT cohort versus 38.6% in the control cohort 56.
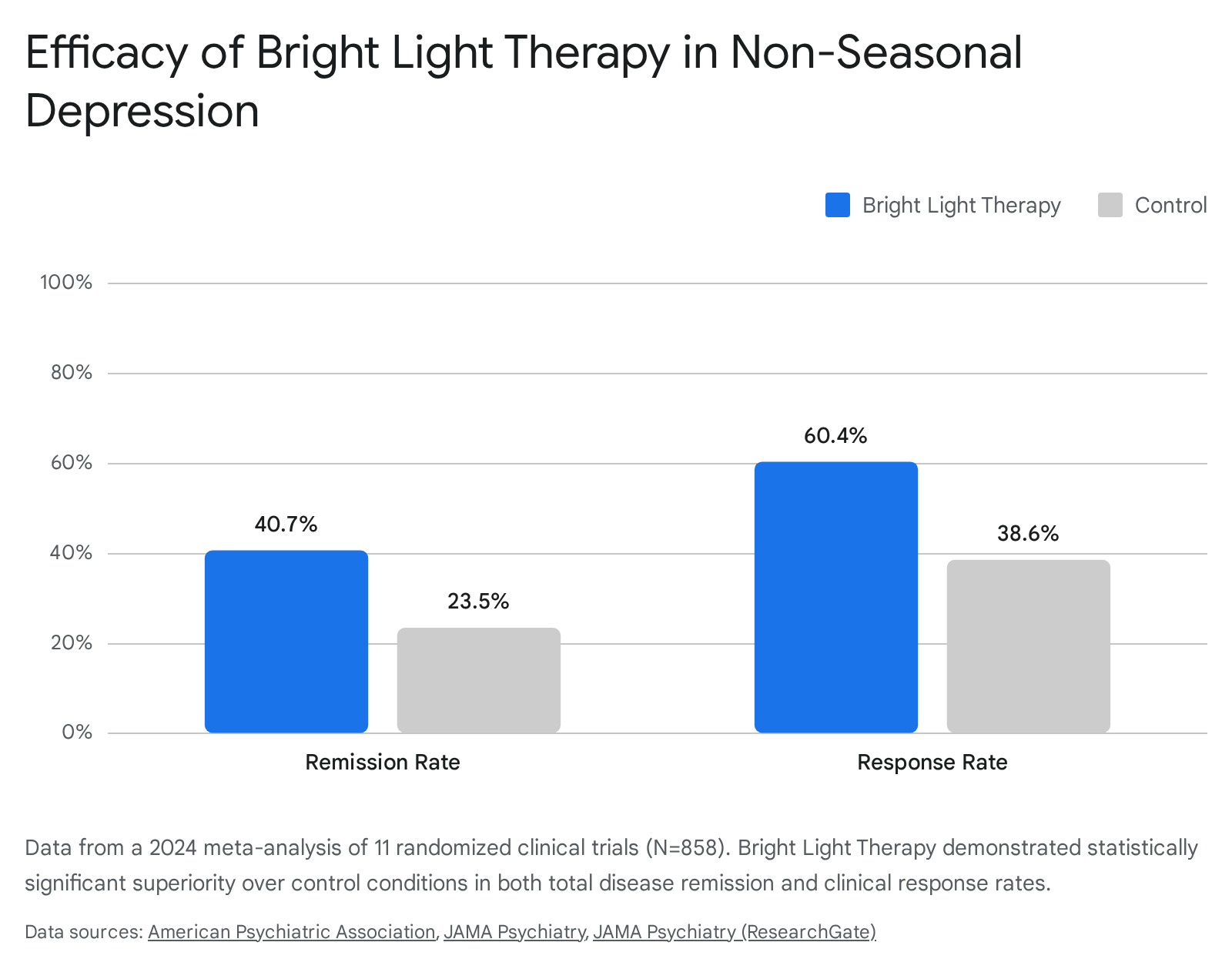
Subgroup analyses indicated that BLT accelerated the response time to initial antidepressant treatments, with prominent therapeutic effects emerging in less than four weeks 56.
These findings indicate that the modulation of circadian rhythms via intense optical light is a fundamental biological lever that transcends seasonal mood fluctuations. As modern lifestyles increasingly divorce human populations from the solar cycle, clinical alignment of the circadian pacemaker offers a low-risk, highly efficacious protocol for stabilizing broad-spectrum psychiatric health.