Spermidine biology and longevity research on autophagy
Introduction to Polyamine Metabolism
Spermidine (N-(3-aminopropyl)butane-1,4-diamine) is a ubiquitous, naturally occurring aliphatic polyamine present in the cells of all eukaryotic organisms 123. First isolated from human semen, spermidine belongs to a critical family of polycationic compounds that includes its precursor, putrescine, and its derivative, spermine 345. Because these molecules are highly charged at physiological pH, they interact closely with negatively charged cellular macromolecules, including DNA, RNA, and proteins, making them essential for fundamental cellular operations such as gene expression, cellular proliferation, differentiation, and apoptosis 136.
Polyamine concentrations within the mammalian body are tightly regulated by endogenous biosynthesis, cellular transport, and catabolism, as well as exogenous contributions from dietary intake and microbial synthesis in the gastrointestinal tract 378. Within the cellular environment, putrescine is synthesized from ornithine, which is subsequently converted into spermidine, and then into spermine through specific enzymatic pathways 34. However, intracellular concentrations of spermidine and other polyamines decline significantly as organisms chronologically age, a biological phenomenon observed across various tissues in both animal models and humans 91011.
This progressive decline in spermidine levels is strongly correlated with the manifestation of age-related pathologies, including neurodegeneration, metabolic dysfunction, and cardiovascular disease 1213. Conversely, restoring polyamine levels through external supplementation - either via diet or purified compounds - induces biochemical changes that mimic the physiological effects of caloric restriction 314. Consequently, spermidine is classified functionally as a "caloric restriction mimetic," capable of triggering cellular stress-response pathways that promote longevity without necessitating actual nutrient deprivation 31415.
Mechanisms of Autophagy and Cellular Rejuvenation
The primary geroprotective value of spermidine stems from its potent ability to induce autophagy, the strictly regulated cellular process responsible for degrading and recycling damaged organelles, misfolded proteins, and dysfunctional intracellular components 21316. The efficiency of autophagic flux naturally deteriorates with advancing age, leading to the toxic accumulation of cellular debris 313.
Epigenetic Regulation and Protein Deacetylation
Spermidine triggers autophagic pathways through direct epigenetic modulation. The polyamine operates as a competitive inhibitor of the acetyltransferase enzyme EP300 (E1A-associated protein p300) 4. The competitive inhibition of EP300 results in the widespread deacetylation of multiple autophagy-related (ATG) proteins, as well as the deacetylation of histones and structural proteins, which collectively initiates the autophagic cascade 4. Clinical investigations involving human pilot trials confirm that daily supplementation with highly concentrated spermidine extracts significantly elevates systemic biomarkers of autophagy, notably Beclin-1 and Unc-51-like kinase 1 (ULK1) 17.
The eIF5A1 Translation Factor
Beyond epigenetic modifications, spermidine is structurally required for the activation of a specific protein translation factor: eukaryotic translation initiation factor 5A (eIF5A) 12. Spermidine serves as the exclusive substrate for the hypusination (a unique post-translational modification) of eIF5A1 1218. In healthy, non-malignant tissues, spermidine-activated eIF5A1 regulates the synthesis of specific proteins necessary for preserving mitochondrial homeostasis and executing autophagy 12. By supporting mitochondrial renewal (mitophagy), spermidine mitigates the accumulation of reactive oxygen species and sustains cellular energy output 1920.
Mitochondrial Trifunctional Protein Activation
Spermidine also influences cellular metabolism through direct enzymatic activation. Research indicates that spermidine allosterically activates the mitochondrial trifunctional protein (MTP), a bipartite protein complex localized to the inner mitochondrial membrane 2122. The activation of MTP enhances enzymatic fatty acid oxidation (FAO) and significantly increases the production of adenosine triphosphate (ATP), reversing the metabolic exhaustion frequently observed in aged cells 2223. This metabolic rejuvenation appears critical for maintaining the function of highly energetic cell types, such as cardiac myocytes and circulating immune cells 2122.
The Polyamine Paradox in Malignancy
While spermidine's ability to induce autophagy positions it as a powerful geroprotector, polyamines simultaneously occupy a complex and somewhat contradictory role in oncology, an intersection referred to as the "polyamine paradox" 12. Polyamines are inherently necessary for cell proliferation, meaning that highly active, dividing cells require vast quantities of them 721. Consequently, elevated polyamine levels are consistently documented within various cancer types, raising valid questions regarding the safety of polyamine supplementation 12.
eIF5A2 and Aerobic Glycolysis
The distinction between spermidine's anti-aging effects and its role in tumor progression relies heavily on the biological context of the tissue and the specific cellular machinery it interacts with 124. Proteomic studies evaluating over 6,700 proteins in human cancer cell lines reveal that in malignant or pre-malignant tissues, polyamines interact with a variant protein: eIF5A2 124.
While polyamines activate eIF5A1 in healthy cells to promote mitochondrial respiration, they function in malignant cells to suppress an RNA molecule (miR-6514-5p) that normally acts as a natural brake on eIF5A2 production 118.
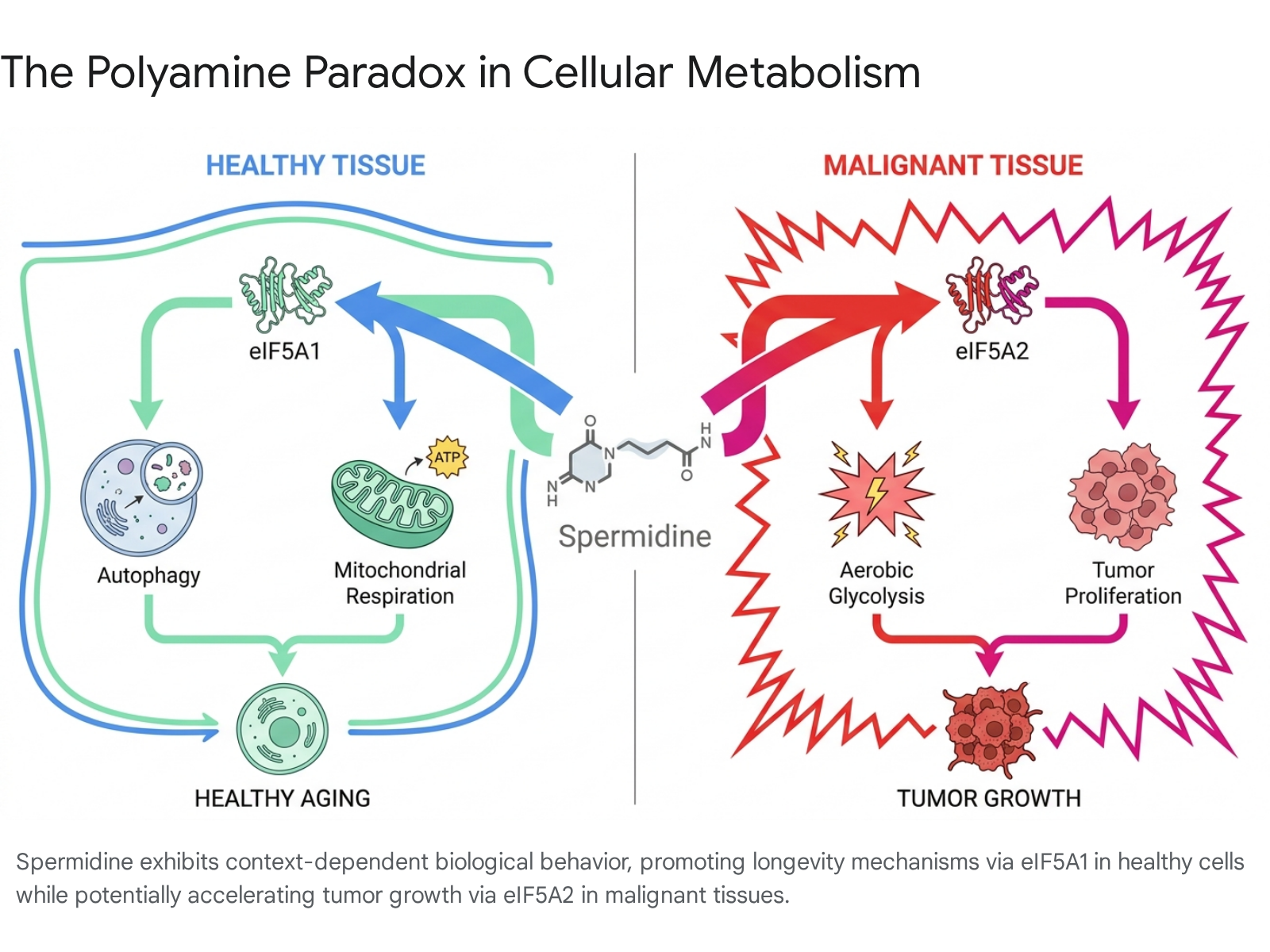
Unfettered eIF5A2 expression fundamentally rewires the cell's metabolism, forcefully driving the cell away from healthy mitochondrial respiration and toward aerobic glycolysis 21218. This metabolic shift - rapidly converting glucose to usable energy regardless of oxygen availability - provides the necessary fuel for aggressive tumor proliferation 12. Furthermore, polyamine abundance in these environments upregulates the synthesis of five specific ribosomal proteins (including RPS27A, RPL36AL, and RPL22L1) tightly associated with cancer severity and aggressive cellular expansion 1224.
Anti-Tumor Immunosurveillance
Despite its potential to fuel established tumors, exogenous spermidine does not appear to act as a primary carcinogen 1821. In fact, lifelong supplementation of spermidine in wild-type mice did not increase the incidence of spontaneous tumors 31021. Instead, recent immunological research has uncovered a counterbalancing mechanism: spermidine is critically necessary for anti-tumor immune surveillance 622.
CD8+ T cells, the primary immune effectors responsible for identifying and destroying malignant cells, suffer from age-related exhaustion 2223. A study published in Science demonstrated that spermidine concentrations in CD8+ T cells decline by approximately 50% in aged mice compared to young subjects 23. This intracellular deficiency directly impairs mitochondrial ATP production, effectively neutralizing the cells' capacity to combat pathogens and tumors 2223.
Supplementing aged immune systems with spermidine restores mitochondrial fatty acid oxidation and rejuvenates CD8+ T cells 22. When combined with cancer immunotherapies - specifically monoclonal antibodies targeting the PD-1/PD-L1 checkpoint pathways - spermidine robustly stimulated T cell proliferation and cytokine production 2223. The combination therapy impeded tumor growth significantly more than the antibody therapy alone, indicating that while tumors rely on polyamines, the immune system equally relies on them to sustain the metabolic firepower required to eliminate malignancies 2123.
Clinical Considerations in Oncology
Due to the polyamine paradox, oncological guidelines approach dietary polyamines with caution. A class of chemotherapeutic agents utilizes difluoromethylornithine (DFMO, eflornithine) to actively blockade the body's synthesis of endogenous polyamines, intentionally starving cancer cells of the molecules required for proliferation 2125. In these clinical contexts, patients undergoing polyamine-lowering therapies must strictly avoid spermidine supplementation, as external intake directly interferes with the drug's mechanism of action 25. While observational data suggests that naturally occurring dietary spermidine correlates with lower cancer mortality in the general population, the introduction of high-dose supplements into patients with established, active malignancies requires explicit supervision by an oncology team 3425.
Preclinical Evidence in Model Organisms
Spermidine's ability to extend lifespan has been extensively documented across evolutionary diverse model organisms. Interventions supplying external spermidine to simple organisms such as yeast, nematodes, and fruit flies reliably demonstrate longevity extensions 313. In wild-type yeast strains, specific doses of spermidine not only slowed chronological aging but actively rejuvenated older replicative cells while enhancing their resistance to oxidative and thermal stress 19.
In mammalian models, the results are equally substantive. When administered continuously in the drinking water of Mus musculus (laboratory mice), spermidine supplementation prolonged median lifespan by approximately 10% 316. Autopsies performed on the aged murine subjects indicated that this extension was primarily driven by the suppression of age-related cardiovascular decline, with no corresponding increase in neoplastic diseases or fibrotic events 310.
Mitigation of Reproductive Aging
In addition to systemic lifespan extension, spermidine appears to counter tissue-specific senescence. Mammalian female reproductive systems exhibit rapid functional declines with age, largely driven by mitochondrial dysfunction and reactive oxygen species (ROS) damage to oocytes 20. Preclinical studies utilizing aging female mice revealed that ovarian spermidine levels correlate inversely with chronological age 20.
Supplementation of spermidine in the drinking water of aged mice enhanced the systemic clearance of damaged mitochondria via mitophagy, actively improving mitochondrial function in the ovaries 20. This intervention resulted in a quantifiable restoration of oocyte quality and an improvement in overall fertility potential 20. These findings were subsequently replicated ex vivo using porcine oocytes subjected to oxidative stress, suggesting that the geroprotective mechanisms of polyamines in reproductive tissues are highly conserved across mammalian species 20.
Epidemiological Associations with Human Mortality
While animal models provide mechanistic proof-of-concept, translating longevity data directly to humans is complex due to genetic diversity, environmental variables, and the redundancy of metabolic networks in higher-order species 19. Nonetheless, extensive prospective cohort studies evaluating human dietary patterns strongly suggest that regular polyamine intake imparts significant survival benefits 34.
A seminal 20-year prospective investigation evaluating 829 adults provided robust epidemiological evidence connecting dietary spermidine to reduced human mortality 26. Dietary assessments revealed that spermidine constitutes approximately 26% of human dietary polyamine consumption 26. The cohort demonstrated a strictly linear, dose-response relationship: higher intakes of spermidine-rich foods correlated with profound reductions in all-cause mortality 2626.
Participants ranked in the highest tertile of daily spermidine consumption (>79.8 μmol/day) exhibited a 39% lower mortality risk compared to those in the bottom tertile 26. When fully adjusted for confounding variables - such as age, sex, BMI, and socioeconomic status - each one-standard-deviation increase in dietary spermidine reduced all-cause mortality risk by 24% (HR: 0.76; 95% CI: 0.67-0.86) 26. Demographically, this risk reduction equated to a biological advantage of being 5.7 years younger chronologically 26.
Further analysis of the UK Biobank and the Women's Health Initiative Observational Study (evaluating over 87,000 postmenopausal women) found that while extreme overconsumption of polyamines did not necessarily yield linear benefits, moderate to high dietary polyamine intake was associated with a reduced risk of colorectal cancer and cardiovascular events, independent of overall adherence to traditional healthy eating indexes 4725.
Dietary Sources and Biochemical Variability
The physiological benefits of spermidine observed in epidemiological cohorts are derived entirely from whole-food sources. While polyamines are ubiquitous in organic tissue, their concentrations vary wildly across food categories, influenced by genetics, soil composition, and preparation methods 2728.
High-Concentration Whole Foods
Cereal grains and legumes represent the highest potential sources of dietary polyamines 29. Wheat germ - the reproductive embryo of the wheat kernel - contains the highest recorded spermidine density among common foods, ranging from 24 to 35 mg per 100 grams (approximately 243 - 2,437 nmol/g depending on the agricultural dataset) 293031.
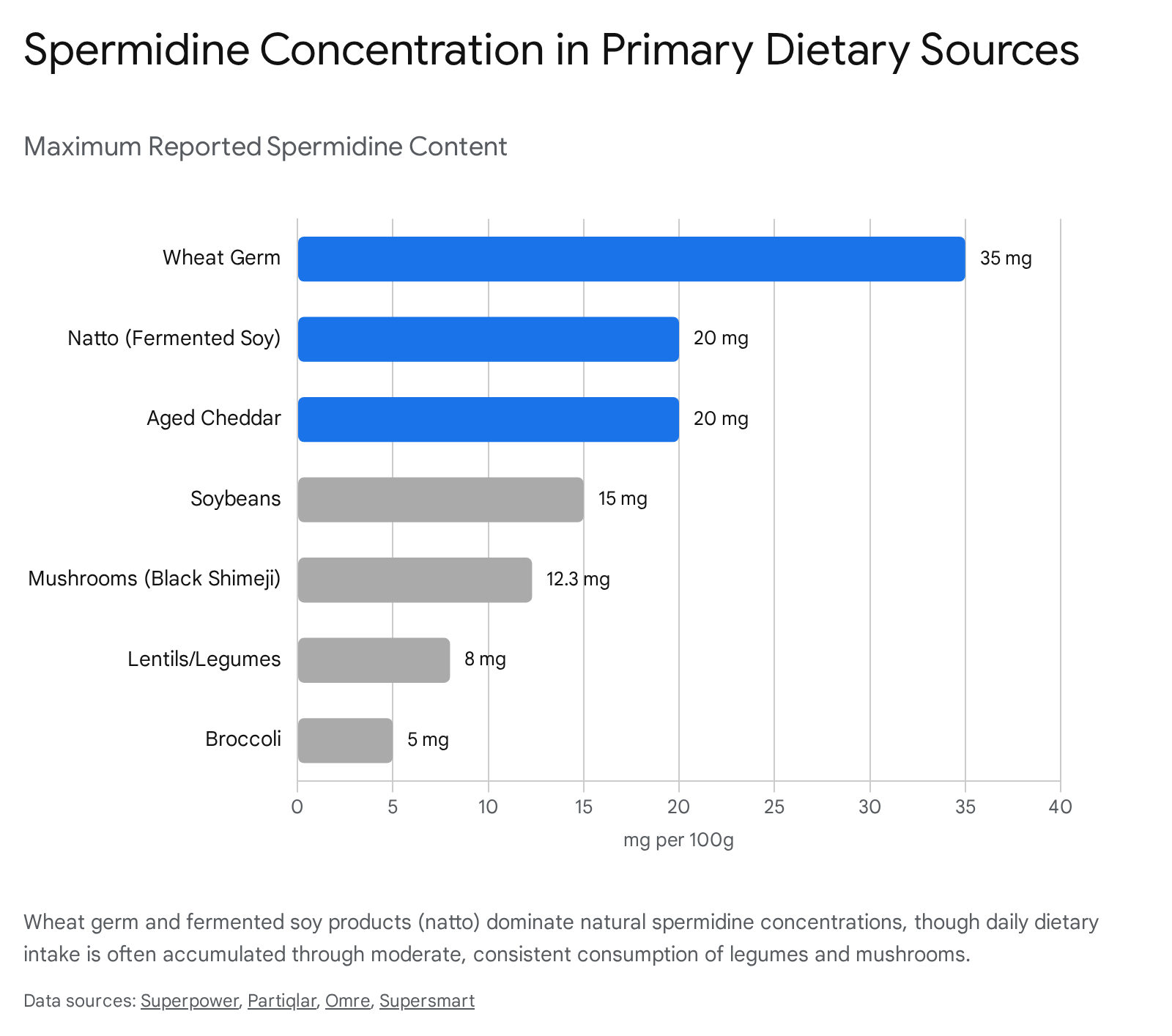
Due to its density, just a single tablespoon of wheat germ delivers roughly 2.0 to 2.5 mg of spermidine, providing an efficient dietary intervention without the need for supplementation 3031.
Soybeans (Glycine max) and their derivatives rank second, providing an average of 6.0 to 15.0 mg of spermidine per 100 grams in their cooked forms 930. Beyond cereals and legumes, certain culinary mushrooms (e.g., black shimeji and oyster mushrooms) contain elevated levels ranging from 5.0 to 12.3 mg per 100 grams 91130. Moderate amounts are widely distributed across cruciferous vegetables (broccoli, 2.5 mg/100g), aged hard cheeses (4.0 - 20.0 mg/100g), and tree nuts 1130.
The Impact of Fermentation and Processing
Fermentation significantly alters the polyamine profile of agricultural products, primarily due to the metabolic actions of the inoculating bacteria. Natto, a Japanese condiment produced via the fermentation of soybeans with Bacillus subtilis, achieves spermidine concentrations of 11.0 to 20.0 mg per 100 grams, noticeably higher than unfermented tofu 93032.
Beyond East Asian cuisine, traditional African fermentation practices yield similarly dense polyamine profiles. Dawadawa (also known as sumbala) is a pungently flavored condiment created by fermenting African locust beans (Parkia biglobosa) across West Africa 3334. 16S rRNA sequencing of dawadawa samples from diverse communities in Northern Ghana and Nigeria reveals that Bacillus subtilis is the dominant bacterial agent responsible for the fermentation 3335. The microbial processing of the locust bean not only generates variable quantities of polyamines but also substantially increases the bioavailability of crude protein (ranging from 35.2% to 50.0% depending on the region) and essential minerals like calcium, iron, and zinc 333536. The high presence of free amino acids - combined with robust antioxidant capacities measured via DPPH and ABTS radical scavenging assays - positions dawadawa as a highly functional, geroprotective dietary staple 3637. However, improper fermentation that allows excessive accumulation of other biogenic amines (such as cadaverine and tyramine) can lead to putrefaction and toxicity, highlighting the necessity of precise microbial control 3839.
Regional Varietals: The Common Bean
In Latin America, the common bean (Phaseolus vulgaris) forms a critical cornerstone of human nutrition 4041. Advanced analytical techniques, including Direct Analysis in Real Time Mass Spectrometry (DART-MS) and Ultra-High Performance Liquid Chromatography (UHPLC), have been deployed to assess the chemical composition of Mexican and South American landraces 4243.
Testing of 12 common bean cultivars revealed that spermidine content varies significantly based on genetic lines and soil fertility, ranging from 2.01 mg/kg to 12.08 mg/kg 42. While absolute polyamine concentrations in the common bean are lower than those found in wheat germ or soybeans, specific regional varieties - such as the black bean cultivars "Negro San Luis," "Negro 8025," and "Negro Jamapa" - compensate through extraordinarily high concentrations of synergistic bioactive compounds 4043. The seed coats of these black beans are dense with condensed tannins, anthocyanins, quercetin, and genistein (a potent isoflavone that increases during germination) 404143. When consumed regularly, the moderate spermidine content of these legumes acts in concert with these potent polyphenols to deliver broad-spectrum anti-inflammatory and antioxidant health benefits 40.
| Food Category | Specific Food Item | Spermidine Content Range | Notable Co-factors & Context |
|---|---|---|---|
| Cereals | Wheat Germ | 240 - 350 mg/kg | Highest density; absent in highly refined flours; contains gluten. 2930 |
| Fermented Soy | Natto | 110 - 200 mg/kg | Enhanced by Bacillus subtilis; strong flavor profile. 3032 |
| Legumes | Soybeans (Whole) | 60 - 150 mg/kg | High protein; closely correlated with putrescine levels. 2830 |
| Fungi | Culinary Mushrooms | 50 - 123 mg/kg | Black shimeji and oyster varieties contain the highest yields. 930 |
| Fermented Seed | Dawadawa (Parkia biglobosa) | Variable (Processing dependent) | Fermented African locust bean; rich in bioavailable iron and zinc. 3336 |
| Common Bean | Black Beans (Phaseolus vulgaris) | 2.0 - 12.0 mg/kg | Lower spermidine but dense in condensed tannins and genistein. 4042 |
Human Clinical Trials on Cognitive Decline and Safety
Translating the longevity benefits of spermidine from model organisms to humans currently relies on Phase II randomized controlled trials (RCTs). Because aging itself is difficult to measure over short clinical windows, researchers frequently utilize cognitive decline - specifically Subjective Cognitive Decline (SCD) in older adults - as a primary clinical endpoint 104445.
Trial Outcomes in Cognitive Aging
Interventional studies investigating spermidine's efficacy in mitigating memory loss have produced highly mixed results, largely dependent on the dosage administered and the specific cognitive assessments utilized 4546.
An early randomized, double-blind Phase II trial by Wirth et al. (2018) evaluated 30 older adults with SCD. Participants received a daily dose of 1.2 mg of spermidine (via a plant extract) for three months 1045. The study reported high compliance (>85%) with no adverse effects, and subjects demonstrated moderate improvements in memory performance as measured by the Mnemonic Similarity Task (MST) 1045. A subsequent trial by Pekar et al. (2021) involving 85 participants tested a higher dosage (3.3 mg/day versus a 1.9 mg/day control) over three months. The higher-dose group exhibited a statistically significant improvement in the CERAD-Plus cognitive test scores (a 6.25 point increase compared to a 4.00 point increase in the low-dose group), indicating a dose-dependent cognitive benefit 45.
However, the largest and most robust longitudinal study to date - the SmartAge trial (Schwarz et al., 2022) - failed to replicate these cognitive benefits 4547. The SmartAge trial was a 12-month, randomized, double-blind Phase IIb study involving 100 participants aged 60 to 90 with SCD 47. Participants were administered 0.9 mg/day of spermidine extracted from wheat germ. After 12 months, researchers found absolutely no significant changes in mnemonic discrimination performance (between-group difference: - 0.03; 95% CI: - 0.11 to 0.05; P = .47) 444647. While the intervention was safe, the lack of cognitive improvement suggests that dosages below 1.0 mg/day may be clinically inert, failing to cross the therapeutic threshold required to alter neurobiology 4448.
Systemic Safety and Autophagic Biomarkers
Despite the ambiguous cognitive outcomes, the safety profile of spermidine supplementation is overwhelmingly positive. A pilot interventional trial by Bruno et al. (2025) evaluated the systemic biomarkers of 12 healthy adults administered either 1.5 mg or 3.3 mg of spermidine from rice germ extract over 56 days 17. The 3.3 mg dose successfully altered circulating biomarkers, resulting in a 7.3% increase in Beclin-1 and a 13.4% increase in ULK-1 (both markers of systemic autophagy), alongside a 12.1% increase in brain-derived neurotrophic factor (BDNF) 17. Furthermore, the intervention reduced high-sensitivity C-reactive protein (hs-CRP) by 20.8%, indicating a mitigation of systemic inflammation 17.
| Clinical Trial | Study Design & Duration | Intervention Dosage | Key Outcomes | Source |
|---|---|---|---|---|
| Wirth et al. (2018) | RCT; n=30; 3 months | 1.2 mg/day (Wheat germ) | Safe; moderate improvement in mnemonic memory. | 1045 |
| Pekar et al. (2021) | RCT; n=85; 3 months | 3.3 mg vs 1.9 mg/day | Higher dose correlated with improved CERAD-Plus scores. | 45 |
| SmartAge (2022) | Phase IIb RCT; n=100; 12 months | 0.9 mg/day (Wheat germ) | No significant cognitive improvement; possible reduction in inflammation. | 47 |
| Bruno et al. (2025) | Pilot RCT; n=12; 56 days | 1.5 mg vs 3.3 mg/day (Rice germ) | 3.3 mg increased autophagy markers (Beclin-1) and decreased hs-CRP. | 17 |
Pharmacokinetics and Supplement Formulation
The ambiguity surrounding low-dose efficacy has driven intense research into the pharmacokinetics of oral spermidine and the optimal formulation of longevity supplements.
The Spermine Conversion Pathway
A fundamental challenge in evaluating spermidine trials is the molecule's unique presystemic metabolism. A 2023 randomized, triple-blinded pharmacokinetic crossover trial administered 15 mg/day of spermidine to healthy volunteers for five days 49. Subsequent analysis of blood and saliva using liquid chromatography-mass spectrometry (LC-MS/MS) and nuclear magnetic resonance (NMR) metabolomics yielded a highly counterintuitive result: oral spermidine supplementation did not increase the concentration of spermidine in the blood plasma 4849.
Instead, the supplementation triggered a significant increase in systemic spermine levels 49. This indicates that dietary or supplemental spermidine undergoes rapid presystemic conversion - likely in the gastrointestinal tract or the liver - into spermine before entering systemic circulation 1449. Consequently, researchers hypothesize that the clinical, in vivo geroprotective effects attributed to spermidine may actually be executed, at least in part, by its downstream metabolite, spermine 144849. Furthermore, pharmacokinetic data suggests that due to this rapid conversion, oral doses below 15 mg/day are unlikely to exert immediate, measurable short-term effects on systemic polyamine pools 154849.
Synthetic Versus Food-Derived Formulations
The push for higher, more precise dosing has led to the development of synthetic, high-purity (98%) spermidine trihydrochloride (hpSPD) formulations 515253.
A recent exploratory double-blind RCT conducted by Chrysea Labs administered 40 mg/day of synthetic hpSPD to 37 older men for 28 days 485051. The massive dose proved highly safe and well-tolerated, producing no adverse effects on lipid profiles, clinical chemistry, or hematology 5051. Remarkably, even at 40 mg/day, there were no substantial alterations to serum and urine polyamine concentrations, demonstrating that the human body maintains extraordinarily tight homeostatic control over polyamine levels without metabolic adaptation 4851.
However, the longevity community remains divided on the superiority of synthetic isolates versus whole-food extracts. * Synthetic Spermidine: Offers precise dosing free from agricultural contaminants and allergens (such as gluten from wheat germ or soy proteins). It permits clinical testing at much higher ranges (up to 40 mg) safely 5352. * Food-Derived Spermidine: Proponents argue that plant extracts deliver a synergistic "recycling loop." Natural extracts contain co-factors like putrescine and spermine, alongside prebiotic resistant starches that selectively nourish the gut microbiome, encouraging endogenous bacterial synthesis of polyamines 85253. The European Food Safety Authority (EFSA) explicitly recognizes food-derived spermidine, establishing a safe upper intake limit of 6.0 mg per day, whereas synthetic molecules lack historical dietary precedent and widespread regulatory consensus 155253.
Methodological Limitations and Conflicts of Interest
The current body of clinical evidence regarding spermidine is encumbered by significant methodological limitations. The majority of human trials rely on severely constrained sample sizes (frequently under 40 participants) and brief observational windows (1 to 3 months) that are inadequate for measuring true anti-aging endpoints 104649. Furthermore, inconsistencies in biological measurement - ranging from assessing polyamines in whole blood to saliva to specific cellular compartments - prevent standardized comparisons across studies 46.
Compounding these structural weaknesses is the pervasive influence of industry funding and potential conflicts of interest (COI) in longevity research 5455. A significant number of the flagship clinical trials demonstrating the efficacy of spermidine have been funded by, or co-authored by, individuals with direct financial ties to supplement manufacturers 4456. For example, studies evaluating memory improvements frequently disclose authors who hold board positions, equity, or patents associated with commercial entities like Longevity Labs (which markets wheat germ extract) or Chrysea Labs (which develops synthetic spermidine) 4451.
While industry funding does not invalidate empirical data, extensive meta-analyses of biomedical publishing demonstrate that industry-sponsored trials are statistically far more likely to report results favorable to the sponsor's product 5457. Compounding this issue, advocacy organizations such as the Center for Science in the Public Interest (CSPI) have highlighted that databases like PubMed do not universally require the explicit, upfront disclosure of funding sources and COIs in study abstracts, potentially misleading physicians and consumers who do not access the full journal text 57. To establish definitive clinical consensus, the spermidine field urgently requires large-scale, independently funded, multi-year trials that track hard biological endpoints rather than subjective cognitive questionnaires.
Animal-to-Human Dose Extrapolation Methodology
Because the most compelling longevity data for spermidine originates in murine models (e.g., life-long administration yielding a 10% lifespan extension), establishing safe and effective human dosages relies on complex pharmacological extrapolation 358.
Extrapolating a dose from a mouse to a human cannot be accomplished through a simple 1:1 body weight (mg/kg) conversion. Larger mammals, including humans, possess significantly slower basal metabolic rates and altered pharmacokinetic clearance speeds compared to rodents 5859. To account for these physiological disparities, pharmacologists and regulatory bodies (such as the FDA) utilize an Allometric Scaling method based on Body Surface Area (BSA) normalization 5860.
The $K_m$ Factor and Allometric Scaling
The conversion relies on a constant known as the $K_m$ factor, which represents the mathematical relationship between an organism's body weight (kg) and its body surface area ($m^2$) 5859.
| Species | Reference Body Weight (kg) | $K_m$ Factor | Factor to Convert Animal mg/kg to Human Equivalent Dose (HED) |
|---|---|---|---|
| Human (Adult) | 60.0 | 37 | N/A |
| Mouse | 0.02 | 3 | Divide animal dose by 12.33 (or multiply by 0.081) 58 |
| Rat | 0.15 | 6 | Divide animal dose by 6.17 (or multiply by 0.162) 5861 |
| Guinea Pig | 0.40 | 8 | Divide animal dose by 4.63 (or multiply by 0.216) 5861 |
| Dog | 10.0 | 20 | Divide animal dose by 1.85 (or multiply by 0.541) 5859 |
To calculate the Human Equivalent Dose (HED) from a preclinical animal trial, researchers use the following standard equation:
$$HED (mg/kg) = Animal Dose (mg/kg) \times \left( \frac{Animal K_m}{Human K_m} \right)$$
For example, if a longevity study establishes that a highly effective and safe dose of spermidine in a laboratory mouse is 100 mg/kg, the translation to humans uses the mouse $K_m$ (3) and the human $K_m$ (37) 5861:
$$HED (mg/kg) = 100 \times \left( \frac{3}{37} \right) = 100 \times 0.081 = 8.1 \text{ mg/kg}$$
For an average adult human weighing 60 kg, the biologically equivalent systemic dose is approximately 486 mg per day 61.
However, in clinical drug development, the calculated HED is not immediately administered to human subjects. To ensure utmost safety during Phase 1 "first-in-human" trials, the HED is conventionally divided by a safety factor, historically set at 10, to account for unforeseen interspecies biochemical differences 586061. Applying this safety factor to the 486 mg equivalent yields a starting test dose of roughly 48.6 mg/day. This pharmacological calculation perfectly aligns with the current clinical trajectory of synthetic spermidine, where researchers are actively executing safety protocols at a maximum dose of 40 mg/day 485158.
Conclusion
Spermidine occupies a uniquely promising yet complex position within the landscape of longevity research. Biologically, its capacity to induce autophagy through the epigenetic deacetylation of ATG proteins and the activation of eIF5A1 translation establishes a highly plausible mechanism for cellular rejuvenation. This is robustly supported by life-extension data in diverse model organisms and extensive epidemiological cohorts demonstrating that populations with high dietary polyamine intake experience significantly reduced all-cause mortality.
However, translating these biological realities into supplemental therapies reveals profound nuances. The "polyamine paradox" dictates that while spermidine preserves mitochondrial function in healthy cells and rejuvenates the anti-tumor capabilities of CD8+ T cells, it can simultaneously be hijacked by malignant tissues to fuel rapid aerobic glycolysis via eIF5A2. Consequently, uncontrolled, high-dose supplementation poses theoretical risks for individuals with active oncological profiles.
Furthermore, human interventional trials targeting cognitive decline have yielded ambiguous and often dose-dependent results, hindered by small sample sizes, short durations, and the metabolic reality that oral spermidine converts rapidly to spermine before systemic circulation. Until large-scale, independently funded longitudinal trials determine the absolute efficacy of high-dose synthetic spermidine, the most scientifically validated approach to polyamine-mediated geroprotection remains the consistent dietary consumption of concentrated whole foods - such as wheat germ, fermented soy, and diverse legumes - which safely deliver spermidine within a synergistic matrix of complementary phytonutrients.