Skin endocrine function and systemic aging
Introduction: Redefining Cutaneous Biology in the Context of Gerontology
For decades, the predominant paradigm within both clinical dermatology and broader gerontology has relegated the phenomenon of skin aging to the domain of cosmetic deterioration. It was widely and erroneously conceptualized as an inert boundary layer slowly succumbing to the mechanical and environmental wear of chronological time, manifesting primarily in superficial rhytides, elastosis, and dyschromia. However, this anatomically localized framework has been entirely overturned by recent, highly robust advances in molecular biology, transcriptomics, and endocrinology. The human skin is not merely a passive envelope; it is the largest, most functionally diverse, and biochemically active peripheral endocrine organ in the human body 112. It possesses absolute enzymatic autonomy to synthesize, metabolize, activate, and deploy a vast array of hormones, neurotransmitters, and biologically active peptides that profoundly influence not only local tissue health but global systemic homeostasis 2345.
Furthermore, as the primary biological interface between the internal physiological milieu and the external "exposome" - a collective term encompassing ultraviolet (UV) radiation, particulate matter (PM2.5), airborne pollutants, and temperature fluctuations - the skin sustains repeated, chronic genotoxic stress throughout the duration of a human lifespan 567. This persistent environmental bombardment, coupled with intrinsic metabolic decline, dramatically accelerates the accumulation of senescent cells within both the epidermal and dermal compartments 8910. Rather than undergoing silent apoptosis, these senescent cells experience radical chromatin reorganization and metabolic reprogramming, adopting what is known as the senescence-associated secretory phenotype (SASP) 11121413. Through the continuous, unregulated secretion of pro-inflammatory cytokines, chemokines, and matrix-degrading proteases, the aging cutaneous organ actively drives a chronic, low-grade systemic inflammatory state known as "inflammaging" 9141316.
This exhaustive research report investigates the skin as a central endocrine organ and a primary, active driver of systemic biological aging. It rigorously catalogs the specific hormonal pathways operating within the cutaneous microenvironment, maps the systemic repercussions of the inflammaging hypothesis, analyzes the complex dual-edged biological role of cellular senescence, and meticulously evaluates the translational realities of emerging senolytic therapeutics. In doing so, it prioritizes high-impact clinical data, geographically diverse patient profiling, and rigorously filters out the hyperbolic claims often perpetuated by the cosmetic marketing industry.
The Cutaneous Endocrine System: Synthesizing Hormones and Orchestrating Systemic Homeostasis
The traditional physiological view of endocrinology relies on a centralized model, wherein central glands (such as the pituitary, thyroid, and adrenals) secrete hormones into the systemic circulation to act on distant target tissues. The skin subverts this classical model by operating through highly localized paracrine, autocrine, and intracrine mechanisms, while simultaneously releasing active metabolites back into the systemic circulation, thereby modulating whole-body homeostasis and immune regulation 24514.
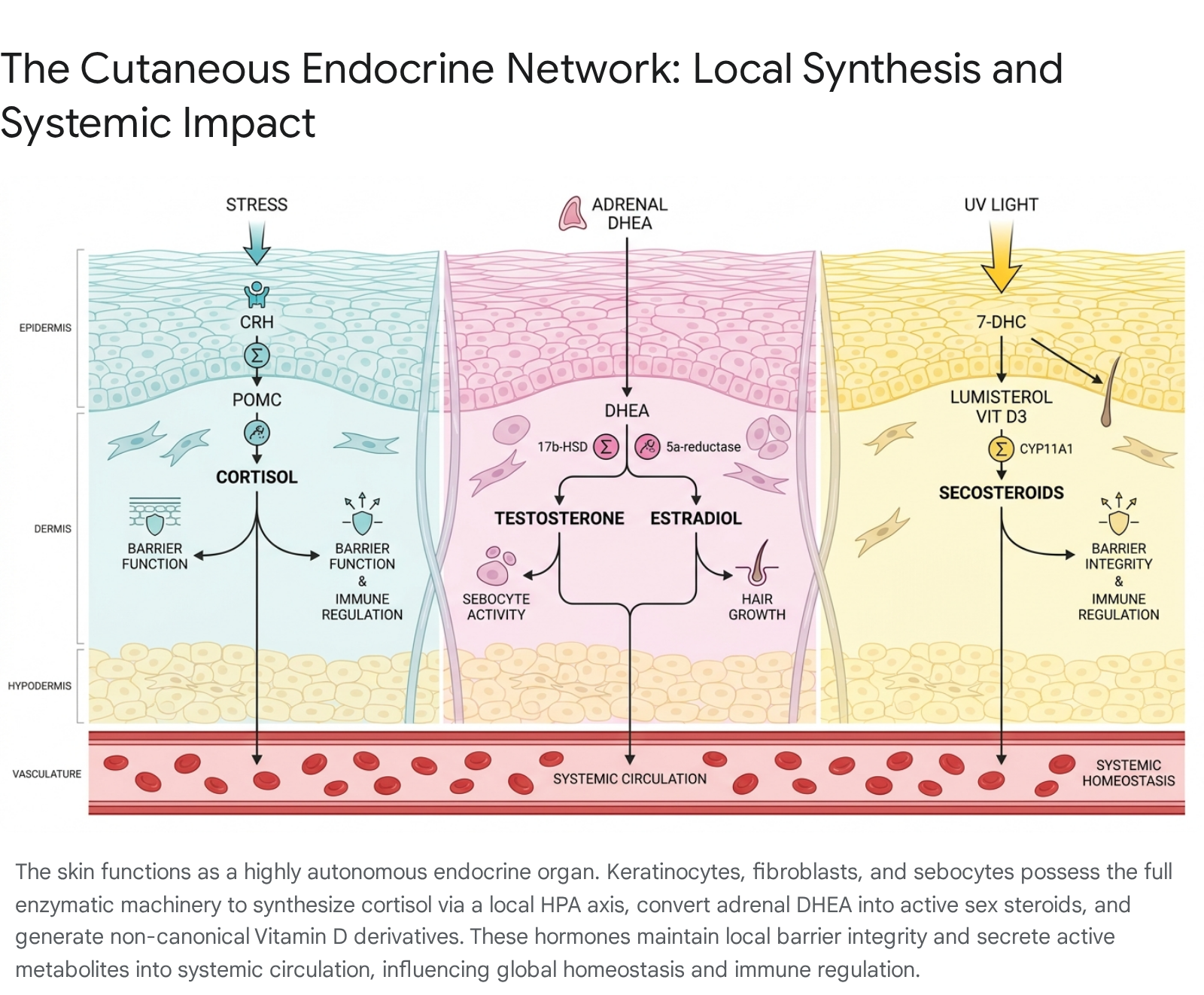
The Cutaneous Hypothalamic-Pituitary-Adrenal (HPA) Axis and Local Cortisol Synthesis
It is now a well-established physiological fact that human skin expresses the complete enzymatic machinery necessary to mimic the central Hypothalamic-Pituitary-Adrenal (HPA) axis. In response to localized external stressors - most notably ultraviolet B (UVB) and ultraviolet C (UVC) radiation, as well as pro-inflammatory cytokines - epidermal keratinocytes and melanocytes actively synthesize corticotropin-releasing hormone (CRH) 141516. The release of CRH locally stimulates the production of proopiomelanocortin (POMC), a complex precursor polypeptide 3141516. POMC is subsequently cleaved by specific convertases into active neuropeptides, including adrenocorticotropic hormone (ACTH), $\alpha$-melanocyte-stimulating hormone ($\alpha$-MSH), and $\beta$-endorphin 3141516.
Crucially, the skin does not rely solely on systemically delivered glucocorticoids originating from the adrenal cortex. It possesses the remarkable capability for de novo glucocorticoid synthesis starting directly from basic cholesterol precursors 1314. Dermal fibroblasts, epidermal keratinocytes, and resident melanocytes express the cholesterol side-chain cleavage enzyme CYP11A1, alongside a full suite of steroidogenic enzymes including 3$\beta$-HSD, CYP17A1, CYP21A2, and CYP11B1 314. Through these meticulously coordinated pathways, the skin actively synthesizes corticosterone and cortisol 314. Furthermore, the local concentration of active cortisol is tightly regulated at the cellular level by the isoenzymes 11$\beta$-hydroxysteroid dehydrogenase type 1 and type 2 (11$\beta$-HSD1 and 11$\beta$-HSD2), which catalyze the bidirectional interconversion of biologically active cortisol and inactive cortisone 31417.
The systemic implications of this highly localized HPA axis are profound. Under conditions of acute stress, localized cortisol release acts to buffer the skin from excessive inflammatory damage, regulating localized leukocyte trafficking and preventing immune hyperactivation. However, in the context of chronic aging, persistent UV exposure, and sustained psychosocial stress, the dysregulation of this cutaneous HPA axis contributes to systemic glucocorticoid resistance, significantly impaired wound healing, epidermal atrophy, and a shift toward a pro-inflammatory systemic state 1141518. By producing excessive quantities of cytokines such as IL-1 and TNF-$\alpha$ in response to sustained stress, the skin's neuroendocrine output dynamically interacts with the central nervous system and the draining lymph nodes, demonstrating that cutaneous distress directly translates to profound immunological and neuroendocrine strain throughout the entire organism 31518.
Intracrine Synthesis and Regulation of Sex Steroids
The structural integrity, elasticity, and hydration of youthful skin are heavily reliant on the presence of circulating sex hormones, which dictate fibroblast proliferation, extracellular matrix (ECM) protein synthesis (such as collagen and elastin), and lipid-rich sebum production. During the biological aging process - particularly during the abrupt hormonal shifts of post-menopause in women and the gradual hormonal decline of andropause in men - central gonadal synthesis of sex steroids sharply declines 2419. In the wake of this systemic deficit, peripheral synthesis within the skin becomes the primary, and often sole, source of these vital regulatory hormones 4.
The skin circumvents central gonadal failure by utilizing circulating, inactive adrenal precursor molecules, namely dehydroepiandrosterone (DHEA), its sulfated form (DHEAS), and androstenedione 34. Specialized skin cells, particularly sebocytes in the sebaceous glands and specialized fibroblasts within the dermal papilla of hair follicles, express high levels of 5$\alpha$-reductase, aromatase, and various isotypes of 17$\beta$-hydroxysteroid dehydrogenase (17$\beta$-HSD) 234. This robust enzymatic profile allows the skin to independently convert circulating inactive precursors into highly potent, biologically active sex steroids, including testosterone, 5$\alpha$-dihydrotestosterone (DHT), and 17$\beta$-estradiol 234.
Because these highly specific enzymatic reactions occur intracellularly, and the active hormones are utilized locally by the very cells that synthesized them (or their immediate neighbors) before being rapidly inactivated and released, this mechanism is termed "intracrinology" 24. This intracrine mechanism allows the cutaneous organ to regulate its own microenvironmental homeostasis completely independently of systemic gonadal function 24. The eventual age-related failure or exhaustion of these local intracrine conversion pathways directly precipitates the most severe clinical signs of intrinsic skin aging, including profound xerosis (dryness), critical loss of dermal tensile strength, and significantly impaired barrier function.
Vitamin D Derivatives and the Non-Canonical Regulatory Repertoire
While the skin's foundational role in synthesizing cholecalciferol (Vitamin D3) upon exposure to UVB radiation has been recognized for over a century, recent high-impact biochemical research demonstrates that the skin's Vitamin D metabolism extends exponentially beyond simple calcium and phosphate homeostasis 202122. The classical, systemic pathway involves the transport of cutaneous Vitamin D3 to the liver and kidneys, where it undergoes sequential hydroxylation to produce the systemically active hormone 1$\alpha$,25-dihydroxyvitamin D3 ($1\alpha,25(OH)_2D_3$). However, within the highly autonomous environment of the skin itself, the enzyme CYP11A1 acts directly on both Vitamin D3 and its isomer, lumisterol, to generate a massive, novel array of non-canonical secosteroids 2021.
These unique cutaneous Vitamin D and lumisterol derivatives do not merely interact with the classical Vitamin D Receptor (VDR). They exhibit immense pleiotropy, functioning as inverse agonists for retinoic acid receptor-related orphan receptors (such as ROR$\alpha$ and ROR$\gamma$) and acting as highly potent agonists for the aryl hydrocarbon receptor (AHR) and Liver X Receptor (LXR) 2021. By engaging this exceptionally complex nuclear receptor network, skin-derived secosteroids actively upregulate the NRF2 antioxidant pathway, profoundly suppress NF-$\kappa$B and IL-17-mediated inflammatory signaling cascades, and heavily stimulate inherent DNA repair mechanisms 202122.
The age-related decline in cutaneous CYP11A1 enzymatic activity, coupled with a reduced baseline availability of the 7-dehydrocholesterol precursor molecule in older skin, critically compromises this robust protective network. This failure exposes the aging cutaneous organ to significantly higher levels of oxidative stress, impairs the epidermal barrier, and actively facilitates the transition of damaged keratinocytes and fibroblasts into a permanent senescent phenotype, thereby accelerating the overall aging process 202122.
Intrinsic vs. Extrinsic Skin Aging Mechanisms: A Comparative Molecular Analysis
Skin aging is classically and clinically bifurcated into intrinsic (chronological) and extrinsic (environmental) processes. While they ultimately converge on shared pathological endpoints - most notably the induction of cellular senescence, severe extracellular matrix degradation, and macroscopic structural collapse - their initiation pathways, temporal progression, and specific biochemical drivers are highly distinct.
Table 1: Comparative Molecular Mechanisms of Intrinsic vs. Extrinsic Skin Aging
| Feature | Intrinsic (Chronological) Aging | Extrinsic Aging (The Exposome) |
|---|---|---|
| Primary Pathophysiological Drivers | Progressive telomere attrition during cellular replication, spontaneous genetic and epigenetic alterations, and severe endocrine decline (specifically the loss of local intracrine DHEA and sex steroid conversions) 5671121. | Chronic exposure to the "exposome": Ultraviolet radiation (broad-spectrum UVA and UVB), atmospheric particulate matter (PM2.5), airborne polycyclic aromatic hydrocarbons (PAHs), and tobacco smoke 5671021. |
| Macroscopic Clinical Presentation | Epidermal thinning (atrophy), fine and superficial rhytides, severe xerosis due to lipid depletion, profound loss of subcutaneous fat volume, and general tissue laxity 81023. | Coarse, deep, and heavily furrowed wrinkles, severe solar elastosis (abnormal, dysfunctional elastin clumping in the dermis), prominent hyperpigmentation, lentigines, and telangiectasia 82324. |
| Primary Cellular Senescence Triggers | Replicative exhaustion reaching the Hayflick limit, coupled with a slow, progressive accumulation of mitochondrial dysfunction and decreased ATP production over decades 7252627. | Acute and chronic genotoxic stress directly causing DNA double-strand breaks, alongside severe proteostatic collapse triggered by overwhelming environmental free radical exposure 91128. |
| Overlapping Biochemical Mediators | Constant, low-level production of endogenous Reactive Oxygen Species (ROS) resulting from inefficient mitochondrial oxidative phosphorylation; steady activation of tumor suppressor pathways p53/p21 and p16INK4a 525262930. | Massive, immediate exogenous ROS generation; robust and sustained activation of the NF-$\kappa$B inflammatory pathway; pronounced and immediate induction of specific Matrix Metalloproteinases (MMP-1, MMP-3, MMP-9) 59212531. |
| Characteristics of the SASP Profile | A chronic, low-level baseline secretion of interleukins leading to a slow, almost imperceptible decline in extracellular matrix integrity over the lifespan 132530. | An aggressive, hyperactive "photoaging" SASP; marked by the dense infiltration of pro-inflammatory macrophages and prolonged, highly destructive cytokine storms (specifically driven by IL-1$\alpha$, IL-6, and IL-8) 91014. |
The Inflammaging Hypothesis: The Cutaneous Organ as an Active Driver of Systemic Decline
To characterize aged skin as merely a passive victim of chronological time is to fundamentally ignore its massive biological volume and profound systemic interconnectivity. Covering nearly 2 square meters and weighing approximately 16% of total adult body mass, the skin is arguably the largest single repository of senescent cells in the aging human body 891011.
The pioneering concept of "inflammaging" - first articulated by Italian immunologist Claudio Franceschi in 2000 - describes a chronic, low-grade, sterile systemic inflammation that accelerates biological aging and serves as the primary pathogenic driver for a multitude of age-related morbidities, including severe cardiovascular disease, Alzheimer's disease, osteoarthritis, and metabolic syndrome 131635. Modern gerodermatology has radically updated this framework, firmly positioning the aging skin not just as a participant, but as a massive primary origin point for this systemic pathology.
When cutaneous fibroblasts, keratinocytes, and melanocytes sustain sufficient DNA damage to undergo senescence, they cease cellular division to prevent malignant transformation. However, they do not remain metabolically dormant; rather, they become metabolically hyperactive, undergoing profound chromatin restructuring to develop the Senescence-Associated Secretory Phenotype (SASP) 142530. The cutaneous SASP secretes a highly toxic, relentless milieu of pro-inflammatory cytokines (predominantly IL-1$\beta$, IL-6, and TNF-$\alpha$), chemotactic chemokines (such as IL-8/CXCL8 and CCL2), and destructive matrix metalloproteinases 10141325.
Because the underlying dermis is an intensely vascularized tissue layer, these potent SASP factors do not remain safely localized within the skin. They diffuse rapidly into the systemic circulation, elevating acute-phase proteins like high-sensitivity C-reactive protein (hs-CRP) globally and altering the entire organism's inflammatory baseline 141316. This continuous systemic drip of inflammatory mediators from the skin creates a highly destructive, vicious physiological cycle. The peripherally generated cutaneous SASP promotes insulin resistance in distant metabolic tissues, impairs central immune surveillance (contributing heavily to immunosenescence), and directly damages vascular endothelial cells, thereby facilitating atherogenesis and hypertension 14133032.
Furthermore, recent discoveries regarding mitochondrial dysfunction have illuminated exactly how this inflammation is sustained. Under extreme oxidative stress, senescent skin cells experience mitochondrial pore opening, leaking fragments of mitochondrial DNA (mtDNA) directly into the cell cytosol 2633. This misplaced cytosolic DNA acts as a highly immunogenic Damage-Associated Molecular Pattern (DAMP), triggering the cGAS-STING (Stimulator of Interferon Genes) pathway 2633. The STING pathway falsely signals the cell that it is under viral attack, locking the senescent cell into a permanent state of aggressive cytokine production that constantly feeds the systemic inflammaging loop 33. Consequently, severe photoaging is increasingly viewed by modern clinicians not just as a historical marker of cumulative UV exposure, but as a highly accurate clinical biomarker for internal, systemic biological age, and a mathematically independent risk factor for systemic frailty and mortality 91416.
The Dual Biological Role of Cellular Senescence: Acute Utility vs. Chronic Pathology
While the massive accumulation of senescent cells is undeniably deleterious in the context of chronic aging and inflammaging, senescence itself is an evolutionarily conserved, highly programmed cellular state that plays an absolutely critical, beneficial role in acute tissue repair and embryonic development. Understanding this Janus-faced duality is paramount when conceptualizing therapeutic interventions, as indiscriminate eradication of senescent cells can severely compromise physiological survival 8113334.
The Beneficial Role: Orchestrating Acute Wound Healing
In the immediate aftermath of acute cutaneous trauma - such as a surgical incision, laceration, or acute thermal burn - a rapid, highly transient wave of cellular senescence is absolutely necessary to orchestrate optimal wound healing. Shortly after the initial hemostatic phase, a specific subset of dermal fibroblasts and endothelial cells located strictly at the wound margins undergo stress-induced senescence 27333435.
In this specific, acute setting, the SASP is highly beneficial and tightly controlled. These acute senescent cells secrete high concentrations of Platelet-Derived Growth Factor AA (PDGF-AA), which forcefully stimulates the differentiation of surrounding healthy fibroblasts into highly contractile myofibroblasts, ensuring rapid wound contraction and physical closure of the defect 82733. Furthermore, the transient inflammatory cytokines (such as IL-6 and IL-8) secreted by these cells act as a powerful homing signal, recruiting immune cells - primarily neutrophils and M1 macrophages - to the wound bed to clear necrotic debris and prevent bacterial colonization 3334. Once their job is complete, the recruited immune cells undergo a phenotypic switch and efficiently phagocytize and clear the senescent cells, resolving the inflammation and allowing the tissue to return to a state of quiescent homeostasis 3334.
The Detrimental Role: Chronic Accumulation and Tissue Paralysis
The entire physiological system breaks down drastically with advancing chronological age, or when compounded by pathological metabolic states such as severe Diabetes Mellitus. In chronic conditions such as Diabetic Foot Ulcers (DFUs) and venous stasis ulcers, the initial senescent response occurs as programmed, but the immune system - which is itself suffering from systemic immunosenescence and impaired chemotaxis - completely fails to clear the acute senescent cells 273435.
These senescent cells persist indefinitely and accumulate in massive numbers, forcibly shifting the wound microenvironment from a regenerative state to a highly chronic, proteolytic, and inflammatory state 2735. The persistent SASP continuously secretes elevated levels of MMPs that aggressively degrade nascent collagen matrices faster than they can be synthesized, while simultaneously producing excessive IL-6 that halts the necessary proliferation of adjacent healthy keratinocyte stem cells 81427.
Furthermore, the continuous, high-dose exposure to SASP factors actively induces "paracrine senescence" (often termed the "bystander effect") in surrounding, previously healthy cells 81433. Most detrimentally, this includes inducing senescence in the tissue-resident macrophages themselves. The secretion of high levels of CXCL1 and CXCL2 by senescent fibroblasts paralyzes the phagocytic capabilities of local macrophages, locking the tissue in a non-healing, highly inflammatory state that often leads to severe necrosis, systemic infection, and eventual amputation 83334.
Foundational Context: Hamilton et al. and the Evolution of Modern Aging Biology
To fully appreciate the current trajectory of advanced senolytic therapies and SASP modulators, it is essential to contextualize the foundational scientific literature that shaped modern biogerontology. The conceptual understanding of why cells age, and how they transition to a senescent state, underwent a massive paradigm shift in the early 21st century, moving away from simplistic models of damage toward complex models of epigenetic and metabolic regulation.
For much of the late 20th century, the Free Radical Theory of Aging - which posited that the accumulated, random damage from endogenous Reactive Oxygen Species (ROS) was the primary, unilateral driver of chronological aging - dominated the scientific consensus. Foundational research papers, such as the landmark in vivo studies published by Hamilton et al. (2001, 2003), rigorously tested this long-standing hypothesis using highly controlled, genetically modified murine models 2936373839. By creating heterozygous mutant mice with a lifelong, systemic reduction in Manganese Superoxide Dismutase (MnSOD, or Sod2+/-) - a critical mitochondrial antioxidant enzyme - researchers anticipated witnessing a dramatic, undeniable acceleration of the aging phenotype 39.
While the Sod2+/- mutant mice did indeed exhibit significantly elevated, lifetime levels of severe oxidative DNA damage (measured precisely via high-pressure liquid chromatography as 8-oxodG) and suffered from notably higher incidences of spontaneous cancer, their maximum and mean lifespans were remarkably indistinguishable from wild-type, healthy controls 3739.
This revelation was entirely paradigm-shifting. It provided definitive, in vivo proof that while oxidative stress is a critical mechanism of macromolecular damage and specific disease pathogenesis (particularly oncogenesis), it does not function as the singular, unilateral clock that dictates chronological aging or absolute lifespan 39. This unexpected finding forced researchers to look deeper into the complex intracellular signaling cascades that are triggered by ROS, rather than focusing solely on the physical damage itself.
Subsequent, highly sophisticated research by Hamilton and colleagues (2005) and other leading geneticists expanded the focus away from basic oxidation and deeply into the epigenome 111240. They identified key histone methyltransferases - such as SET-2 and MET-1 in model organisms, which regulate the deposition of H3K36me3 along the gene body - that are absolutely vital for preserving transcriptional fidelity and suppressing spurious, chaotic gene expression 111240. In the aging skin, the gradual breakdown of these critical epigenetic regulators leads to the formation of Senescence-Associated Heterochromatin Foci (SAHF) 1112. SAHFs permanently silence vital structural genes (like those coding for collagens) while simultaneously leaving the gene loci for SASP cytokines permanently open and hyperactive 1112.
More recently, intense investigations into cellular metabolism have mapped exactly how alterations in branched-chain amino acid (BCAA) metabolism and the dysregulation of bromodomain proteins (targeted by inhibitors like AZD5153) act upstream of the mTORC1 complex to lock damaged skin cells permanently into the senescent state 4541. This profound intellectual continuum - from recognizing the stark limitations of the free radical theory to precisely mapping the epigenetic and metabolic drivers of the SASP - laid the essential theoretical groundwork that makes the targeted pharmacological clearance of senescent cells possible today.
SASP Profiling Across Diverse Populations: The Fitzpatrick Scale Imperative
A glaring, systemic deficiency in historical dermatological, pharmacological, and gerontological research has been the intense, almost exclusive overrepresentation of individuals with Fitzpatrick Skin Types (FST) I-III (lighter skin tones) in clinical trials and genomic databases 42434445. This historic bias has led to a highly skewed, incomplete understanding of global skin aging mechanisms and the expression variations of the SASP across diverse human populations.
Recent high-impact literature (2023 - 2025) underscores the critical reality that photoaging trajectories and SASP manifestations are profoundly dependent on an individual's melanocompetence and ethnic genetic background 23284546. While individuals with FST IV-VI (darker skin tones) possess higher levels of basal melanin - which offers superior physical protection against UV-induced DNA double-strand breaks, thereby delaying the onset of coarse rhytides and structural collapse - these skin types possess a much more reactive immune baseline and mount a far more robust inflammatory response to environmental stress 2328.
In highly pigmented skin, the cutaneous SASP is heavily driven by elevated levels of IL-6 and specialized chemokines that interact deeply with the local melanocyte population 28. Consequently, whereas chronological and photo-aging in FST I-II is characterized by severe elastosis, volume loss, and dermal thinning, the aging process in FST IV-VI is primarily characterized by highly persistent Post-Inflammatory Hyperpigmentation (PIH), intractable melasma, and significant variations in skin texture and pore architecture 284244.
The historical reliance on simple visual categorization via the traditional Fitzpatrick scale has also proven entirely inadequate for modern molecular research. The scale lacks objective resolution, frequently struggles to differentiate between subtle undertones in mixed-race individuals, and fails to capture the true breadth of ethnic diversity required for precise transcriptomic profiling 244347. To accurately profile SASP and aging markers on a global scale, modern dermatological AI systems and high-tier clinical trials are rapidly adopting hardware like broadband optical sensors (measuring reflected light across 410 - 940 nm) coupled with K-means unsupervised algorithms 4345. These technologies quantify tissue health, oxygenation, and melanin density without subjective human bias, ensuring that emerging anti-aging interventions and senolytic dosing protocols are safe and highly efficacious across the entire global demographic spectrum 4345.
Translational Limitations of Senolytics: The Case of ABT-263 (Navitoclax)
The advent of "senolytics" - pharmacological compounds explicitly designed to selectively induce apoptosis in persistent senescent cells while leaving healthy cells unharmed - represents the most aggressive and highly anticipated front in modern anti-aging medicine. Navitoclax (ABT-263), a highly potent BH3 mimetic originally developed as a targeted oncological agent, functions by chemically inhibiting the anti-apoptotic proteins Bcl-2, Bcl-xL, and Bcl-w. Senescent cells heavily upregulate these specific survival proteins to prevent themselves from dying in the highly toxic, ROS-rich internal environment they create 454849.
Recent, highly detailed in vivo murine studies published between 2024 and 2025 demonstrate that the topical application of ABT-263 directly to aged skin (in 24-month-old mice) drastically reduces the transcriptomic expression of classical senescence markers (specifically p16INK4a, p21CIP1, and SA-$\beta$-gal) 555057. Furthermore, by successfully clearing these senescent fibroblasts and temporarily inducing a localized macrophage-driven repair response, ABT-263 remarkably accelerates the closure of subsequent acute wounds by reviving dormant extracellular matrix remodeling pathways and stimulating angiogenesis 555057.
However, the aggressive push to translate the efficacy of ABT-263 - and similar broad-spectrum, first-generation senolytics - from highly controlled mouse models to widespread human clinical outcomes is heavily obstructed by several profound biological toxicities and pharmacological limitations:
- Severe On-Target Toxicity in High-Turnover Tissues: The anti-apoptotic survival pathways hijacked by senescent cells (particularly those involving Bcl-xL) are also absolutely critical for the survival and maintenance of essential healthy cells. The systemic administration of ABT-263 routinely causes severe, dose-limiting thrombocytopenia in clinical trials, because human blood platelets fundamentally rely on Bcl-xL for their daily survival in circulation 454849.
- Collateral Damage to Vascular Infrastructure: Recent, rigorous tissue evaluations reveal a darker side to the drug's mechanism; ABT-263 does not exclusively target senescent cells. At the concentrations required to achieve senolysis, it induces massive, indiscriminate apoptosis in non-senescent, healthy human smooth muscle cells and delicate vascular endothelial cells, raising severe, potentially fatal safety concerns regarding long-term vascular integrity and the risk of catastrophic hemorrhage 51.
- Profound Sexual Dimorphism in Drug Response: A landmark 2025 study examining atherosclerotic LDLr-/-;hApoB100+/+ (ATX) mice highlighted a highly dangerous divergence in drug response based entirely on biological sex. While ABT-263 administration successfully improved spatial memory, reduced glial activation, and increased endothelial density in aged male mice, the exact same dosing regimen in young and middle-aged female ATX mice was disastrous. It exacerbated atherosclerotic plaque formation, severely decreased vital endothelial cells, and paradoxically triggered a massive, systemic surge in inflammaging markers 52.
- Accelerated Ovarian Aging and Infertility: Parallel, high-impact studies in 2024 demonstrated that ABT-263 actively accelerates ovarian aging when administered to older female mice. The drug indiscriminately destroyed senescent-like supporting structures within the ovary, leading to a profound, irreversible reduction in the number of primordial follicles, gross ovarian shrinkage, and structural hemorrhage, effectively inducing premature and permanent infertility 60.
These severe, life-altering limitations indicate that the "hit-and-run" or broad-spectrum systemic application of early-generation senolytics is currently untenable for human longevity therapies. In response to these catastrophic toxicities, the gerontology field is currently pivoting rapidly toward "senomorphics" (compounds that gently modulate the SASP without killing the cell, for example, via precise mTOR inhibition) and investing heavily in targeted, antibody-conjugated nanocarrier delivery systems designed to localize cytotoxicity purely to pathological, fibrotic tissues, sparing healthy organs 454961.
Evaluating Clinical Evidence: Retinoids vs. Emerging Anti-Aging Interventions
As molecular research advances at an unprecedented pace, the commercial skincare and clinical dermatological markets have been completely flooded with novel interventions claiming to reverse cellular senescence, rebuild shattered DNA, and massively stimulate neocollagenesis. However, the actual strength of the peer-reviewed evidence supporting these claims varies drastically across modalities.
Table 2: RCT Evidence Strength - Retinoids vs. Emerging Anti-Aging Modalities (2024-2025)
| Modality | Mechanism of Action | Strength of Clinical Evidence (RCTs) | Clinical Realities, Limitations, and Marketing Context |
|---|---|---|---|
| Topical Retinoids (Tretinoin, Retinol, Retinaldehyde) | Lipophilic molecules that penetrate the cell membrane, bind directly to nuclear retinoic acid receptors (RAR/RXR), and physically reprogram gene expression to accelerate keratinocyte turnover, synthesize Type I/III collagen, and aggressively inhibit MMPs 625364. | Gold Standard (Unmatched/Robust). Supported by over four decades of large-scale, double-blind Randomized Controlled Trials (RCTs) proving definitive histological reversal of photoaging, massive reduction in fine lines, and dyschromia correction 625364. | The primary limitation remains tolerability. Can induce severe retinoid dermatitis (erythema, desquamation, barrier impairment). Requires consistent application for weeks to months to manifest visible structural changes, leading to frequent patient non-compliance 536465. |
| Exosomes (MSC, Plant, and Platelet-Derived Vesicles) | Nano-sized extracellular vesicles delivering a highly complex payload of microRNAs, mRNAs, and regenerative growth factors directly into target cells, modulating intercellular communication and promoting rapid fibroblast proliferation 62665455. | Promising but Highly Preliminary. Early, small-scale trials show a rapid reduction in inflammation and massively accelerated healing, especially when combined with microneedling to bypass the stratum corneum 62665670. | Lacks long-term, standardized Phase III RCTs. There is massive product heterogeneity (e.g., bovine colostrum vs. plant vs. human platelet sources vary wildly in efficacy). Extreme risk of cosmetic marketing outstripping rigorous peer-reviewed validation (e.g., claims of "7.2x better wrinkle reduction" are often based on isolated, unverified manufacturer data) 62646656. |
| Peptides & Synthetic Growth Factors | Short chains of specific amino acids or large, complex proteins designed to signal fibroblasts to upregulate structural proteins (e.g., palmitoyl tripeptide-1) or mimic the behavior of natural regenerative cytokines 626457. | Moderate/Highly Variable. In vitro laboratory data is exceptionally strong. However, in vivo human RCTs show only modest, superficial improvements in elasticity, barrier hydration, and slight wrinkle reduction 6457. | Peptides and large recombinant growth factors struggle massively with transdermal penetration due to their high molecular weight (the 500 Dalton rule). Their clinical effects are generally supportive and transient, rather than fundamentally transformative 6264. |
| Senomorphics and Topical Senolytics | Small molecule drugs (e.g., Rapamycin, Fisetin) that chemically suppress the mTOR pathway, dampening the SASP without inducing apoptosis; or targeted senolytics designed to clear senescent cells locally in the dermis 4961. | Emerging (Preclinical/Phase I). Possesses strong ex vivo human skin data and highly promising murine data showing significantly reduced p16/SASP markers and objectively increased collagen density 49555061. | Formulations optimized for human topical use are still highly experimental. The primary pharmacological challenge remains achieving optimal dermal delivery to the fibroblasts without triggering local tissue toxicity, systemic absorption, or initiating severe inflammation 343549. |
Conclusion: The Future Trajectory of Gerodermatology
The foundational, biological conceptualization of the skin has irreversibly shifted over the last decade. It can no longer be viewed through the narrow lens of cosmetics; it must be recognized as a highly active, totally autonomous endocrine organ capable of profound systemic metabolic orchestration. The inflammaging hypothesis dictates that the cosmetic deterioration of the skin - driven internally by intrinsic cellular exhaustion and externally by the relentless assault of the exposome - is not a localized, isolated aesthetic event. Rather, the massive accumulation of cutaneous senescent cells, and their relentless, lifelong emission of toxic SASP factors, acts as a primary physiological engine for systemic, age-related inflammatory diseases that dictate human morbidity and mortality.
While the pharmacological eradication of these highly damaging cells via early-generation senolytics like ABT-263 presents a tantalizing theoretical cure, the severe translational limitations - ranging from catastrophic vascular toxicity and immune suppression to profound sexual dimorphism and the induction of premature infertility - demand extreme scientific caution. The complex, dual nature of cellular senescence, being absolutely essential for acute wound healing and survival but lethal in chronic, unmanaged accumulation, means that therapeutic interventions cannot be administered as blunt instruments.
Future dermatological and systemic anti-aging interventions will undoubtedly require highly localized, transient application of modified senolytics utilizing advanced nanocarrier delivery systems, or the widespread utilization of gentle senomorphics that safely silence the SASP while completely preserving the structural and cellular integrity of the tissue. Until those advanced technologies fully mature through rigorous, geographically diverse, and highly standardized long-term randomized controlled trials, foundational neuroendocrine modulators and classical, evidence-based receptor-binding agents like topical retinoids remain the absolute zenith of anti-aging intervention. The ultimate, overarching objective of modern gerodermatology is no longer merely preserving the aesthetic surface of the human body, but actively and pharmacologically severing the biological link between cutaneous deterioration and systemic physiological decline.