Sharp-wave ripples and sleep spindles in memory consolidation
Introduction to Memory Consolidation
The transformation of transient, labile experiences into stable, long-term memory representations is a foundational mechanism of mammalian cognition. This time-dependent stabilization process, termed memory consolidation, encompasses two distinct but deeply interrelated dimensions: synaptic consolidation and systems consolidation. Synaptic consolidation refers to the localized stabilization of a memory trace at the cellular level, driven by molecular mechanisms such as long-term potentiation (LTP) and the structural remodeling of dendritic spines, which occurs in the hours immediately following encoding. Conversely, systems consolidation describes the large-scale reorganization of memory engrams across distributed brain networks over days, months, or even years 1234.
At the systems level, the mammalian brain relies on a division of labor between the hippocampus and the neocortex. During wakefulness, the hippocampus operates as a fast-learning network, rapidly binding the disparate sensory, spatial, and temporal elements of a novel experience into a cohesive episodic representation. However, this hippocampal trace is highly susceptible to interference and decay. For permanent storage and integration into existing knowledge frameworks (schemas), the representation must be gradually redistributed to the neocortex, a slow-learning network that extracts statistical regularities and generalized concepts 2567.
The offline state of sleep provides an optimal neurophysiological and neurochemical environment for this hippocampal-neocortical dialogue. Divorced from the continuous influx of external sensory stimuli, the sleeping brain actively processes, sorts, and solidifies newly acquired information. This systems-level reorganization is driven by the spontaneous reactivation or "replay" of waking neural activity patterns, a process heavily orchestrated by specific macroscopic brain oscillations - most notably cortical slow oscillations, thalamocortical sleep spindles, and hippocampal sharp-wave ripples 158910.
Theoretical Models of Consolidation
The mechanisms governing memory processing during sleep have been formalized primarily through two dominant theoretical frameworks, which highlight complementary aspects of neuroplasticity: Active Systems Consolidation and the Synaptic Homeostasis Hypothesis.
Active Systems Consolidation
The Active Systems Consolidation (ASC) model posits that memory formation during sleep relies on the selective, repeated reactivation of specific hippocampal engrams formed during prior wakefulness. Rather than passively protecting memories from interference, sleep involves a highly organized replay of neural sequences, functioning as an internal training signal for the neocortex 56.
During non-rapid eye movement (NREM) sleep, specifically the deep stages of slow-wave sleep (SWS), the brain orchestrates a complex dialogue between structures. The hippocampus relays compressed packages of memory information to the neocortex. Over successive sleep cycles, the persistent replay strengthens the distributed neocortical synapses connecting the components of the memory trace. Eventually, the memory becomes independent of the hippocampus, residing entirely within distributed neocortical networks - a process known as corticalization or memory abstraction 6.
Crucially, ASC dictates that this process is highly selective. The brain does not consolidate every waking sensory input equally; it preferentially strengthens memories that hold future relevance, emotional salience, or associative reward 5611.
Synaptic Homeostasis Hypothesis
The Synaptic Homeostasis Hypothesis (SHY), formulated by Giulio Tononi and Chiara Cirelli, approaches sleep through a neuroenergetic and computational lens. According to SHY, wakefulness is inherently associated with learning and continuous synaptic potentiation. As an organism interacts with its environment, synapses across the brain strengthen to encode new information. However, this net increase in synaptic weight is biologically unsustainable. Potentiated synapses demand high metabolic energy, occupy physical space, decrease the overall signal-to-noise ratio in neural circuits, and push the brain toward a saturation point where new learning becomes impossible 12131415.
SHY posits that the primary function of sleep is to restore synaptic homeostasis through a process of global synaptic downscaling. During the slow, synchronized neuronal firing characteristics of SWS, all synapses undergo a proportional weakening. This downselection process reduces the absolute strength of synaptic connections while preserving their relative weight differences 121416. By uniformly downscaling connections, the brain conserves energy, restores its capacity for novel encoding the following day, and prunes weak, noisy, or irrelevant synaptic traces 131517.
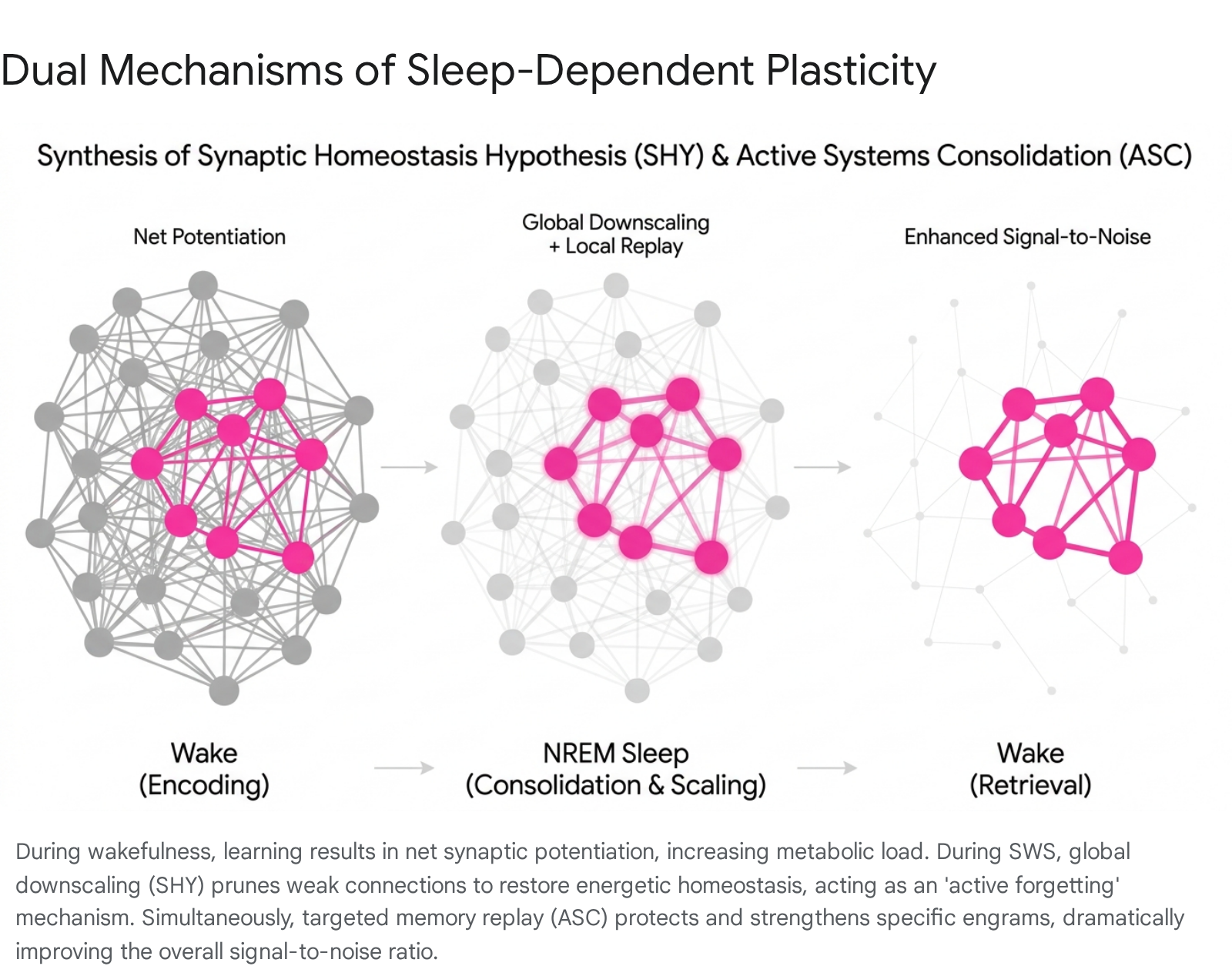
Synthesis of Consolidation and Homeostasis
Initially perceived as contradictory - with ASC emphasizing synaptic strengthening and SHY emphasizing synaptic weakening - recent neuroscientific consensus views these mechanisms as operating in parallel to optimize memory networks.
The global downscaling proposed by SHY functions as a mechanism for "active forgetting," an adaptive computational process that prevents catastrophic interference and clears associative clutter from neural networks. Concurrently, the targeted memory replay central to the ASC model protects specific, high-value engrams from this global downscaling 6111317. In specific highly active circuits, sleep may even drive localized upscaling and the formation of new dendritic spines. Consequently, the simultaneous execution of global downscaling and targeted replay dramatically increases the signal-to-noise ratio of relevant memories, allowing them to stand out against a quieted cortical background 131516.
Neurophysiology of Non-Rapid Eye Movement Sleep
The communication of memory representations between the hippocampus and neocortex relies on the precise temporal coordination of specific neurophysiological oscillations. During NREM sleep, three cardinal waveforms dominate the electroencephalographic landscape.
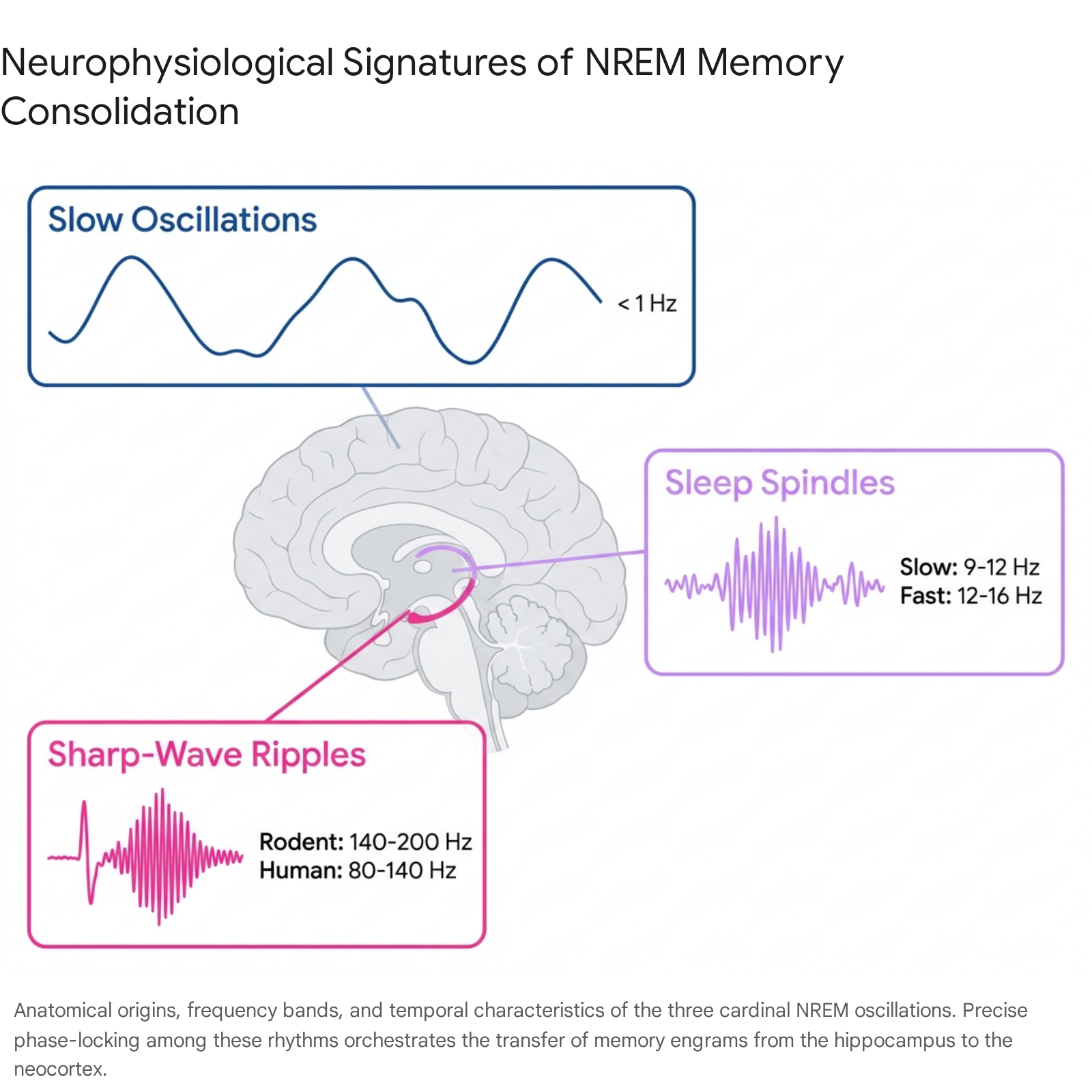
Cortical Slow Oscillations
Slow oscillations (SOs) are large-amplitude, low-frequency waves occurring at less than 1 Hz, with a peak frequency around 0.75 Hz in human subjects. These oscillations are generated within widespread neocortical networks, originating predominantly in the prefrontal cortex before propagating across the anterior-to-posterior axis 12917.
The defining characteristic of the SO is the bistability of cortical neuronal membrane potentials. Neurons toggle synchronously between two distinct states: a highly depolarized "UP state" characterized by intense, sustained neuronal firing, and a hyperpolarized "DOWN state" marked by widespread neuronal silence 2912. The DOWN state effectively serves as a reset, clearing the network of residual activity. The subsequent transition into the UP state acts as the master conductor for systems consolidation. It triggers top-down activation of subcortical structures, including the thalamus and the hippocampus, providing generalized windows of excitability that are conducive to synaptic plasticity 1171819.
Thalamocortical Sleep Spindles
Sleep spindles are transient, waxing-and-waning oscillatory bursts typically lasting between 0.5 and 2.0 seconds. They are generated by inhibitory interactions within the reticular nucleus of the thalamus, which in turn drive rebound excitation in thalamocortical projection neurons 9172021.
Extensive human electroencephalography (EEG) and magnetoencephalography (MEG) data categorize spindles into two functional and topographical subtypes, though exact frequency boundaries can vary between individuals.
| Characteristic | Slow Spindles | Fast Spindles |
|---|---|---|
| Frequency Range | 9 Hz - 12 Hz | 12 Hz - 16 Hz |
| Topographical Peak | Anterior / Medial Frontal Cortex | Central / Parietal Cortex |
| Phase Relationship to SO | Peaks during the transition into the SO DOWN state (waning depolarization) | Tightly phase-locked to the highly depolarized peak of the SO UP state |
| Consolidation Role | Generally not directly locked to hippocampal output; role in memory is debated | Provides the critical temporal window of excitability for hippocampal memory transfer |
Table 1: Characteristics of Thalamocortical Sleep Spindle Subtypes based on human EEG recordings 17222324.
The dichotomy between slow and fast spindles has been refined in recent years. Advanced spatial filtering methods, such as generalized eigendecomposition (GED), indicate that individualized frequency thresholds are required, as fixed group-level bands often obscure the physiological variance present in human subjects 2425. Furthermore, intracranial stereoelectroencephalographic (sEEG) data suggest that spindle frequencies exist on a spatial and temporal continuum, with the frequency of individual spindles often decelerating across their duration 26.
Methodologically, identifying these brief oscillations has evolved. Traditional spindle detection relies on static amplitude and duration thresholds. However, more resilient algorithms like the Better OSCillation (BOSC) method utilize frequency-specific power thresholds calibrated to the background 1/f noise of the individual's local field potential, significantly reducing false-positive detections and accurately capturing biologically relevant low-amplitude spindles 21. Regardless of minor classification differences, the density and amplitude of fast spindles phase-locked to SO UP states are highly predictive of next-day memory retention across declarative and procedural tasks 22728.
Hippocampal Sharp-Wave Ripples
Sharp-wave ripples (SWRs) are brief, highly synchronous local field potential events originating in the mammalian hippocampus. They represent the most synchronous physiological pattern observed in the brain, occurring primarily during SWS and periods of quiet, idle wakefulness 2930.
An SWR comprises two distinct components. The "sharp wave" is a large-amplitude deflection caused by the synchronous depolarization of the apical dendrites of CA1 pyramidal cells, driven by powerful excitatory bursts from the CA3 region 293031. Superimposed upon this sharp wave is the "ripple," a narrow-band high-frequency oscillation generated by fast-spiking GABAergic interneurons. This fast rhythm of inhibition synchronizes the firing of the active pyramidal cells, tightly restricting their action potentials to the narrow windows between inhibitory peaks 2930.
During SWRs, the sequences of place cells and other memory-encoding neurons that fired during waking exploration are reactivated. This replay occurs at a highly compressed timescale - often 10 to 20 times faster than the original waking behavior 829. SWRs thus act as the output mechanism of the hippocampus, broadcasting these compressed memory packets via the subiculum and entorhinal cortex to broad neocortical networks 1832.
Mechanisms of Neural Coupling
The individual presence of slow oscillations, spindles, and SWRs is not sufficient to guarantee successful memory consolidation. The active transfer of memory representations relies on the precise phase-amplitude coupling of all three rhythms, forming a temporal hierarchy known as "triple coupling."
The active systems consolidation theory details three coordinated steps within this hierarchy: 1. SO-Spindle Coupling: The massive cortical depolarization during the SO UP state acts as a top-down trigger, exciting the thalamic reticular nucleus. This forces the maximal amplitude of thalamocortical fast spindles to phase-lock perfectly with the peak of the SO UP state 21827. 2. Spindle-SWR Coupling: Concurrently, the descending phases of the individual cycles within the fast spindle provide optimal windows for hippocampal output. The maximal amplitude of hippocampal SWRs aligns tightly with the troughs of the spindle oscillation 21827.
By nesting extremely high-frequency hippocampal bursts (SWRs) into intermediate thalamic gating rhythms (spindles), which are themselves nested inside periods of widespread cortical depolarization (SOs), the brain ensures that reactivated memory traces arrive at neocortical synapses at the exact millisecond those synapses are most excitable.
The precision of this coupling is biologically critical. Studies spanning the human lifespan demonstrate an "age-related dispersion" of SO-spindle coupling. Older adults frequently exhibit a loss of precise temporal alignment, with spindle power increasing erroneously at the end of the SO UP state rather than at its peak. This loss of coupling precision strongly correlates with age-related declines in the retention of associative memories across sleep, underscoring that cognitive maintenance depends not just on regional brain integrity, but on the precise millisecond-level timing of neural interactions 2227.
Sub-states and Micro-structures of Sleep
Recent methodological advancements in large-scale neuronal recording and behavioral tracking have illuminated novel micro-structures within the architecture of sleep, revising earlier assumptions regarding the uniformity of SWRs and NREM sleep.
Distinct Roles of Large vs. Small Sharp-Wave Ripples
Historically treated as a monolithic class of events, research published between 2024 and 2026 demonstrates that SWRs must be functionally subdivided. High-density ensemble recordings have shown that only a specific minority subset - termed "large" sharp-wave ripples - possess the necessary amplitude, duration, and energetic power to drive the coordinated reactivation of memories in both the hippocampus and the downstream prefrontal cortex 32343334.
Smaller SWRs, while numerous, are generally not associated with significant extra-hippocampal memory reactivation. Following the learning of a novel task, the brain selectively increases the occurrence rate of large SWRs during subsequent sleep. Utilizing closed-loop optogenetics to artificially boost ripple amplitude at their peak, researchers were able to elevate standard ripples into large SWRs. This intervention enabled mice to successfully consolidate and subsequently recall brief behavioral experiences that would otherwise have been too weak to form lasting memories, confirming the causal necessity of large SWRs in the consolidation cascade 3233.
Micro-structures in Pupil Fluctuations
A persistent theoretical problem in systems consolidation is the risk of catastrophic interference: how does the brain integrate new memories into the neocortex without overwriting previously stabilized schemas?
Simultaneous pupillometry and electrophysiology in naturally sleeping rodents have identified an autonomic micro-structure within NREM sleep that resolves this conflict. Pupil size continuously fluctuates during NREM sleep, driven by locus coeruleus noradrenergic activity. Researchers discovered a profound temporal segregation of memory replay based on these pupil states: - Contracted Pupil Sub-states: Dominated by strong extrinsic excitatory inputs, SWRs occurring during contracted pupil phases preferentially reactivate neural assemblies encoding novel, recently acquired experiences 635. - Dilated Pupil Sub-states: Dominated by localized inhibition, SWRs occurring during dilated phases preferentially reactivate neural assemblies encoding older, established prior memories 635.
By compartmentalizing the processing of new information and the maintenance of old information into distinct autonomic sub-states, the brain multiplexes cognitive operations, allowing continuous learning while strictly minimizing retroactive interference 35.
Reset Mechanisms in the CA2 Region
Further refining the dynamics of the hippocampus during sleep, researchers have mapped the specific contributions of hippocampal sub-regions. While CA1 and CA3 are heavily involved in generating the ripple oscillation and the sharp wave respectively to encode time and space, the lesser-studied CA2 region appears vital for neural regulation.
During NREM sleep, parallel circuits regulated by distinct interneuron subtypes generate periods where CA1 and CA3 firing is abruptly silenced. This silencing is driven by CA2 activity and serves to "reset" the memory-encoding pyramidal neurons. This prevents neuronal exhaustion and saturation, ensuring that the same physical neural resources can be reused to encode entirely new memories the following day 36. Furthermore, specific optogenetic studies have shown that CA2 reactivation during SWRs is fundamentally essential for the consolidation of specialized domains such as social memory 37. Continuous tracking across 24-hour periods shows that these processes induce "representational drift," where waking memory traces are actively updated, pruned, and merged as they are repeatedly replayed 38.
Rapid Eye Movement Sleep and Emotional Processing
While NREM sleep facilitates the initial stabilization of memories, memory processing continues dynamically into Rapid Eye Movement (REM) sleep. REM sleep serves an essential, complementary function, specializing in the integration of newly consolidated memories into abstract schemas and the recalibration of emotional valence 123940.
The neurochemical transition from NREM to REM sleep is profound. During SWS, cholinergic (acetylcholine) tone is minimized, which is essential for permitting the uninterrupted flow of memory replay from the hippocampus to the neocortex. However, during REM sleep, subcortical cholinergic systems reactivate, pushing acetylcholine to levels equal to or exceeding those of active wakefulness. Simultaneously, aminergic systems - specifically serotonergic neurons in the Raphe nuclei and noradrenergic neurons in the locus coeruleus - are almost completely silenced 394341.
This high-acetylcholine, low-norepinephrine state completely alters network dynamics. It suppresses the recurrent feedback pathways from the hippocampus, allowing local neocortical circuits to freely recombine the elements of memories consolidated during prior NREM sleep. The dominant electrical signature shifts to continuous theta band oscillations (4 - 8 Hz). Cortical excitatory feedback loops are uninhibited, providing the computational space necessary for memory abstraction, the extraction of hidden rules, and creative integration 13940.
The Sleep to Forget, Sleep to Remember Hypothesis
REM sleep holds a privileged role in the processing of emotionally arousing memories. Functional neuroimaging demonstrates that the amygdala, hippocampus, medial prefrontal cortex, and anterior cingulate cortex are highly active during human REM sleep 3941.
The "Sleep to Forget, Sleep to Remember" (SFSR) hypothesis suggests that REM sleep serves a dual purpose for emotional experiences: it selectively strengthens the declarative core of the memory while systematically stripping away the visceral autonomic charge originally associated with the event 394342. When an individual experiences an emotional event during wakefulness, the amygdala triggers the release of noradrenaline, prompting a physical fight-or-flight response. When the brain replays this memory during REM sleep, the absolute absence of noradrenergic tone ensures that the memory is reactivated without triggering the peripheral stress response. Over successive REM cycles, this decouples the emotional memory from its physiological reactivity 4142.
When REM sleep is fragmented, or when the noradrenergic system fails to shut down during sleep, this decoupling process fails. The inability to recalibrate emotional memory via REM sleep is increasingly recognized as a foundational neurobiological mechanism underlying affective psychopathology, particularly Post-Traumatic Stress Disorder (PTSD) and severe depression 4343.
Active Forgetting via Melanin-Concentrating Hormone
Recent neurobiological investigations highlight an additional mechanism of active forgetting occurring during REM sleep. Melanin-concentrating hormone (MCH)-producing neurons in the hypothalamus densely innervate the dorsal hippocampus and are uniquely active during REM sleep. Activation of these MCH nerve terminals increases inhibitory input to hippocampal pyramidal neurons, actively suppressing their firing. Chemogenetic silencing of these neurons prevents the normative decay of novel object and conditional fear memories, strongly suggesting that REM sleep enforces the active erasure of redundant or obsolete memory traces to maintain cognitive flexibility 3.
Targeted Memory Reactivation
The growing comprehension of sleep's active role in memory has catalyzed the development of Targeted Memory Reactivation (TMR), a non-invasive behavioral intervention capable of causally manipulating memory consolidation in humans.
TMR leverages the brain's natural replay mechanisms by establishing an associative link between a learning task and an unobtrusive sensory cue - typically a sound or an odor. When the participant enters post-learning SWS, the identical cue is surreptitiously presented at a sub-arousal volume or concentration. This presentation biases the sleeping brain, forcing the selective reactivation of the specific neural ensemble tied to the cue 4445464748. Consequently, upon waking, memories subjected to TMR are retained with significantly greater accuracy than uncued control memories 49.
Modality Comparisons in Memory Reactivation
The efficacy and operational mechanics of TMR are highly contingent on the sensory modality used. The two predominant cue types - auditory and olfactory - present distinct neuroanatomical advantages and limitations.
| Feature | Olfactory TMR (Odors) | Auditory TMR (Sounds) |
|---|---|---|
| Neuroanatomy | Processing bypasses the thalamus, allowing direct access to limbic and hippocampal structures during sleep 46. | Processing is subject to thalamic gating, yet sound inputs still reliably penetrate to the cortex during SWS 46. |
| Arousal Profile | Rarely awakens sleeping subjects, making it ideal for continuous, extended presentation protocols 50. | Considerably more arousing; requires careful volume titration to prevent disrupting sleep architecture or waking the subject 50. |
| Congruency | Cues are usually incongruent and contextual (e.g., learning card locations while smelling a rose) 5051. | Allows highly congruent, item-specific cueing (e.g., pairing an image of a cat with a 'meow' sound) 475051. |
| Memory Domain Efficacy | Highly effective for declarative, spatial, and emotional memory consolidation 50. | Exceptionally versatile; demonstrates robust effects across declarative, spatial, linguistic, and procedural (motor skill) domains 4650. |
Table 2: Comparison of Olfactory and Auditory Modalities in Targeted Memory Reactivation (TMR) Protocols.
Decoding of lateralized EEG activity during auditory TMR confirms that cues not only improve behavioral recall but actively elicit learning-related neural signatures during sleep. However, TMR is not uniformly beneficial. The efficacy of the cueing is modulated by the pre-sleep strength of the memory trace. Specifically, memories that are encoded weakly or recalled with low accuracy prior to sleep benefit the most from SWS reactivation, while highly accurate, over-trained memories show negligible TMR benefits, likely because they have already achieved optimal consolidation ceilings 484952.
Furthermore, attempts to utilize TMR alongside "forgetting instructions" (cueing a sound alongside an odor previously associated with instructions to forget) have proven complex. Rather than instilling localized forgetting, multiple conflicting cues tend to evoke competing sleep-based retrieval processes that globally disrupt spatial memory reactivation, resulting in a prolonged decline in sigma power (spindle band) activity 52.
Closed-Loop Stimulation and Personalized Protocols
Initially, TMR protocols delivered cues continuously or at randomized intervals during stages N2 and N3 of NREM sleep. Modern approaches have drastically improved efficacy by integrating real-time electroencephalographic monitoring to create "closed-loop" systems.
Closed-loop algorithms detect the ascending phase of an ongoing slow oscillation and trigger the TMR auditory cue precisely as the brain approaches the highly excitable UP state. Studies directly comparing phase-timing show that UP-state cueing significantly reduces overnight forgetting, whereas cueing during the DOWN state yields behavioral results indistinguishable from uncued controls 445354. Neurophysiologically, UP-state cueing acts as an amplifier: it immediately enhances the amplitude of the ongoing slow wave and provokes a massive, subsequent increase in phase-locked fast spindle power, effectively hijacking the endogenous triple-coupling mechanism 4453.
Emerging research has begun to personalize TMR. By continuously adjusting the stimulation frequency based on a combination of the subject's baseline retrieval performance and ongoing EEG spindle features, personalized protocols can dynamically target specific, highly challenging memory circuits. Multivariate classification of post-sleep EEG data shows that personalized UP-state TMR produces distinct neural signatures indicative of deep, trace-specific system-level consolidation 55.
Translational Limitations in Sleep Research
While the high-frequency dynamics of systems consolidation are well understood in animal models, translating the neuroscience of sharp-wave ripples to human subjects involves profound anatomical and methodological challenges.
The fundamental limitation is spatial resolution. In rodents, researchers utilize microscopic, high-density silicon probes stereotactically targeted to the specific pyramidal layers of the CA1 and CA3 regions. This allows for the unambiguous recording of both the slow "sharp wave" deflection in the stratum radiatum and the high-frequency "ripple" in the pyramidal layer 8305657. Conversely, non-invasive human EEG and MEG are restricted to monitoring cortical slow oscillations and spindles; they cannot detect deep hippocampal SWRs 9.
Consequently, human SWR data relies almost entirely on intracranial stereoelectroencephalography (sEEG) sourced from patients undergoing surgical evaluation for drug-resistant epilepsy. Clinical macroelectrodes measure local field potentials spanning tens of thousands of neurons simultaneously. Due to this coarse spatial sampling, the "sharp wave" component is rarely detectable in human recordings, forcing researchers to rely heavily on simple bandpass filtering to isolate generalized high-frequency oscillations (HFOs) 857.
| Parameter | Rodent SWR Studies | Human SWR Studies |
|---|---|---|
| Frequency Range | 140 Hz - 200 Hz 293058 | 80 Hz - 140 Hz 3058 |
| Recording Instrumentation | Micro-wire arrays / Silicon probes | Clinical macroelectrodes |
| Spatial Resolution | Layer-specific (e.g., CA1 pyramidal layer isolation) 856 | Regional (broad hippocampal / subicular sampling) 57 |
| Subject Pathology | Healthy wild-type or specific genetically modified models 857 | Primarily patients with intractable epilepsy 85758 |
Table 3: Methodological and physiological disparities between rodent and human sharp-wave ripple characterization.
The reliance on epileptic populations introduces a severe confound: interictal epileptiform discharges (IEDs) and pathological ripples (p-ripples). P-ripples occur across a much broader frequency spectrum (80 - 500 Hz) and are generated by pathological, rather than physiological, synchronization. Because broad-spectrum IEDs frequently overlap with the human ripple band (80 - 140 Hz), traditional threshold-based detection methods are highly prone to false positives 85758. To circumvent this, researchers now utilize stricter morphological criteria, identifying human SWRs by searching for narrow-band frequency peaks containing multiple visible oscillation cycles on the unfiltered field potential - features that cleanly separate physiological memory replay from broad-band epileptic pathology 857.
In summary, despite these translational hurdles, the comparative data underscores a highly conserved evolutionary mechanism. The precise coordination of brain-wide oscillations during sleep remains the fundamental biological substrate for transforming the transient events of daily life into permanent architectural elements of memory.