Senolytic evidence for fisetin and quercetin
Introduction to Cellular Senescence and Senotherapeutics
Cellular senescence is a fundamental biological mechanism characterized by an essentially irreversible arrest of the cell cycle, accompanied by profound morphological, epigenetic, and metabolic changes. Initially identified in the 1961 as a tumor-suppressor mechanism designed to prevent the proliferation of damaged or precancerous cells, senescence is triggered by various intrinsic and extrinsic stressors. These include telomere attrition (replicative senescence), DNA double-strand breaks, mitochondrial dysfunction, reactive oxygen species (ROS), and oncogene activation 1234. While transient cellular senescence is beneficial for embryonic development, tissue regeneration, and wound healing, the chronic accumulation of senescent cells over a lifespan contributes directly to tissue dysfunction and the pathogenesis of a wide array of age-related diseases 566.
A defining feature of these persistent senescent cells is the senescence-associated secretory phenotype (SASP). Despite halting proliferation, senescent cells remain highly metabolically active and secrete a complex cocktail of pro-inflammatory cytokines (such as IL-6, IL-1α, and TNF-α), chemokines (such as IL-8 and MCP-1), growth factors, and matrix metalloproteinases (MMPs) 1278. The SASP creates a localized pro-inflammatory microenvironment that can degrade extracellular matrix architecture, disrupt the function of neighboring healthy progenitor cells, and induce secondary, paracrine senescence in adjacent tissues 8911. Furthermore, the chronic, low-grade systemic inflammation fueled by the SASP - often termed "inflammaging" - is recognized as a primary driver of geriatric syndromes and chronic conditions such as cardiovascular disease, neurodegeneration, osteoarthritis, pulmonary fibrosis, and metabolic dysfunction 691011.
Senescent Cell Anti-Apoptotic Pathways
To survive the highly inflammatory and pro-apoptotic environment they create, senescent cells heavily upregulate pro-survival networks, collectively termed senescent cell anti-apoptotic pathways (SCAPs) 91114. These redundant pathways include the BCL-2 family of proteins (BCL-2, BCL-xL, BCL-w), the phosphoinositide 3-kinase (PI3K)/AKT cascade, hypoxia-inducible factor 1-alpha (HIF-1α), p53/p21, and specific ephrin receptors 101213. Because senescent cells rely on these upregulated pathways to evade death, they exist in a state described as "primed for apoptosis" 61418.
Senolytics are a novel class of pharmacological agents designed to selectively eliminate senescent cells by transiently disabling these SCAP networks. By inhibiting the specific survival nodes upon which senescent cells depend, senolytics exploit the cells' pre-existing apoptotic priming without harming non-senescent, healthy cells that do not rely on these overexpressed pathways 6101415. Among the most extensively studied and promising senolytic agents are the naturally occurring flavonoid polyphenols, quercetin and fisetin.
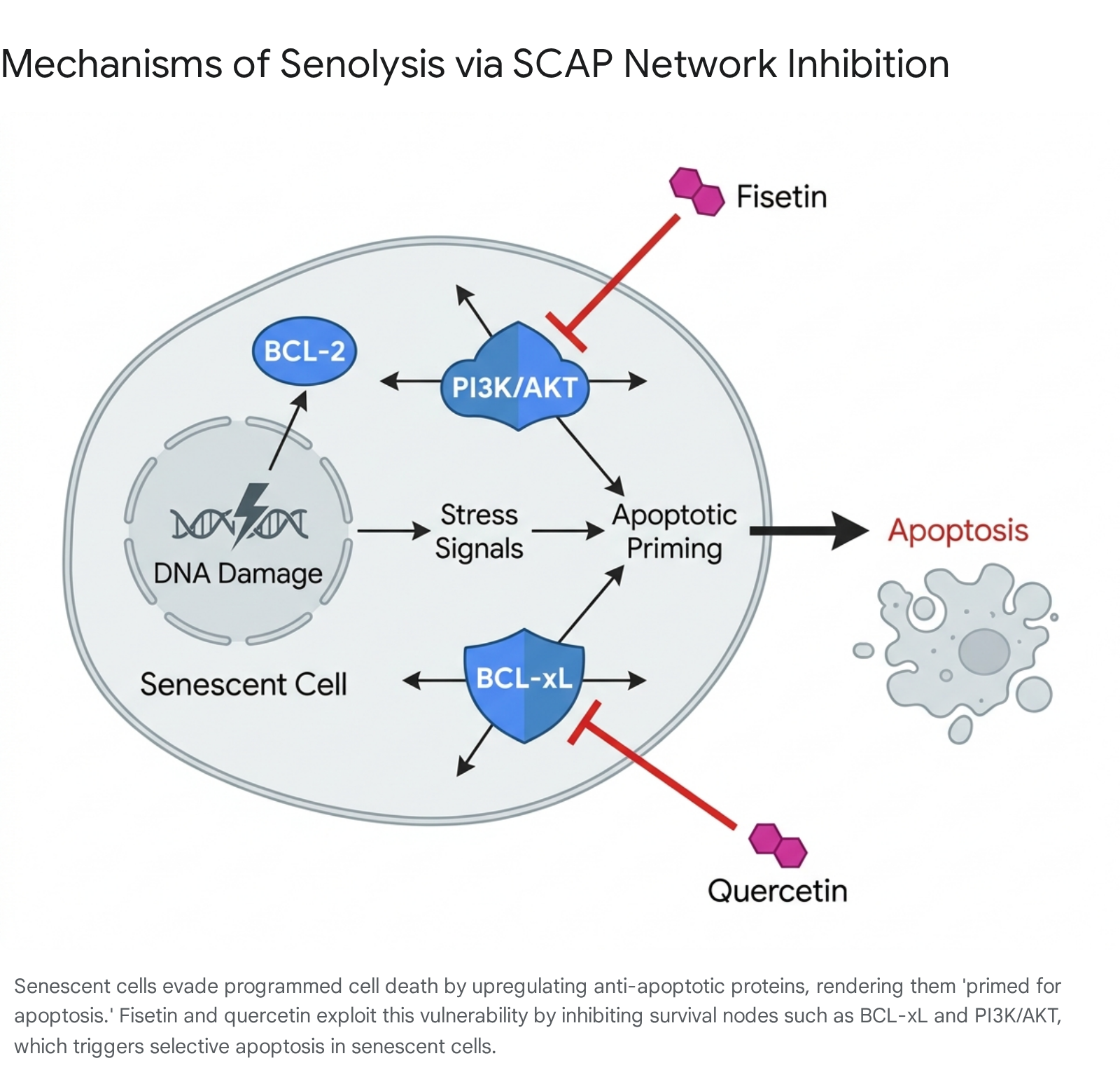
Pharmacological Mechanisms of Fisetin and Quercetin
Fisetin (3,3',4',7-tetrahydroxyflavone) and quercetin (3,3',4',5,7-pentahydroxyflavone) are closely related plant-derived flavonols found in fruits, vegetables, and teas, including strawberries, apples, onions, and persimmons 20161723. Both compounds possess antioxidant, anti-inflammatory, and neuroprotective properties. Despite their structural similarities - differing primarily by a single hydroxyl group - they exhibit distinct pharmacological profiles, tissue specificities, and molecular mechanisms within the context of senotherapeutics 162425.
Quercetin and Dasatinib Synergy
Quercetin possesses five hydroxyl groups, making it slightly more hydrophilic than fisetin 1625. Quercetin primarily exerts its senolytic effects by inhibiting the PI3K/AKT signaling cascade and interfering with the functions of BCL-2 family proteins 38918. However, quercetin is relatively selective in its target cell population. It effectively targets senescent human umbilical vein endothelial cells (HUVECs) but demonstrates weak senolytic activity against senescent fibroblasts and primary human preadipocytes when used in isolation 61427.
Due to this limited spectrum, quercetin is almost exclusively utilized in clinical and preclinical models in combination with dasatinib, an FDA-approved tyrosine kinase inhibitor originally developed for chronic myeloid leukemia 91427. The combination therapy, referred to as "D+Q," operates synergistically. Dasatinib targets senescent preadipocytes and fibroblasts by inhibiting ephrin receptors, p21, and src kinases, while quercetin broadens the therapy's reach by targeting endothelial cells via PI3K/AKT and BCL-2 family inhibition 101413. Together, the D+Q cocktail addresses a comprehensive array of redundant SCAPs, facilitating the clearance of senescent cells across multiple tissue types 1519.
Fisetin Pathway Specificity
Fisetin, conversely, demonstrates robust, broad-spectrum senolytic activity as a monotherapy. Preclinical screening of ten distinct flavonoid polyphenols (including quercetin, luteolin, rutin, and curcumin) identified fisetin as the most potent natural senolytic, capable of clearing senescent cells across a wider range of biological systems without requiring an adjunctive pharmaceutical compound 223. Fisetin operates as a dual-action molecule, simultaneously modulating several discrete survival pathways. It inhibits the PI3K/AKT/mTOR network, suppresses the expression of BCL-xL, and acts as a potent inhibitor of nuclear factor-κB (NF-κB) 91420.
The compound selectively induces apoptosis in senescent HUVECs and demonstrates strong clearance capabilities in adipose tissue, kidney, liver, and brain tissue, sometimes outperforming the D+Q combination in specific neurodegeneration models 1415. Furthermore, fisetin functions as a natural caloric restriction mimetic, modulating nutrient-sensing pathways that contribute to cellular longevity and systemic metabolic homeostasis 14.
Mechanisms of Senescence-Associated Secretory Phenotype Suppression
Beyond direct senolysis, both fisetin and quercetin exhibit potent "senomorphic" properties - the ability to suppress the detrimental SASP without inducing apoptosis in the senescent cell 481230. This dual action is crucial for mitigating the chronic tissue inflammation characteristic of aging.
The SASP is predominantly regulated by the DNA damage response, which activates downstream effectors such as p38 mitogen-activated protein kinase (MAPK) and the transcription factor GATA4 7212223. These pathways converge to activate NF-κB and C/EBPβ, the master transcriptional regulators that drive the expression of inflammatory cytokines like IL-1β, IL-6, and IL-8 2023. Quercetin directly downregulates PI3K/AKT and NF-κB signaling, dampening the secretion of SASP factors and decreasing the oxidative stress burden 8.
Fisetin suppresses the SASP through multiple overlapping mechanisms. It effectively inhibits mTOR and NF-κB pathways while activating the AMPK/SIRT1 network, which reduces overall oxidative stress 8. Fisetin has also been shown to induce dual-specificity phosphatase 1 (DUSP1), which subsequently inactivates pro-calcific p38 MAPK signaling, mitigating the progression of vascular calcification 52124. By suppressing the secretion of factors like IL-6, TNF-α, and MMPs, these flavonoids break the paracrine feedback loops that drive secondary senescence and progressive tissue deterioration 4825.
Preclinical Efficacy in Animal Models
Extensive in vitro and in vivo studies in murine models have provided the foundational evidence for the senotherapeutic potential of fisetin and quercetin. These studies have primarily utilized naturally aged mice, progeroid (accelerated aging) models, and disease-specific transgenic lines.
Adipose, Vascular, and Systemic Tissue Clearance
The D+Q combination consistently demonstrates the ability to clear senescent cells in adipose tissue and the vascular endothelium. In rodent models, D+Q administration alleviated physical dysfunction, prevented age-related bone loss, and reduced the burden of senescent cells in models of atherosclerosis 102627. D+Q has also been shown to reduce intervertebral disc degeneration, improve pulmonary function, and decrease hepatic steatosis in obese and aged mice 1028.
Fisetin's tissue penetrance and senolytic profile appear broader in these models. In a landmark study utilizing both progeroid mice carrying a p16INK4a-luciferase reporter and aged wild-type mice, acute or intermittent treatment with fisetin reduced senescence markers across multiple tissues, including adipose tissue, spleen, liver, and kidney 229. Fisetin restored tissue homeostasis, reduced the severity of age-related pathologies, and improved physiological responses analogous to a delay in biological aging 229. Furthermore, in models of lupus nephritis (MRL/lpr mice), fisetin treatment reduced the number of senescent renal tubular epithelial cells, attenuated kidney fibrosis, and decreased localized SASP expression 5.
Neurological Function and Neurodegeneration
Senescent cells accumulate significantly within the central nervous system during aging and neurodegenerative disease progression. Fisetin demonstrates notable neuroprotective efficacy across several murine models. In the APPNL-F/NL-F amyloidogenic mouse model of Alzheimer's disease, intermittent oropharyngeal administration of fisetin reduced senescent cell burden in both the brain and peripheral visceral adipose tissue 30. This clearance correlated with restored mitochondrial function, reduced neuroinflammation, and improved cognitive performance 122730.
Similar neuroprotective outcomes have been observed in SAMP8 mice (a model for sporadic Alzheimer's disease and dementia), where fisetin prevented progressive memory loss, learning disabilities, and locomotor deficits 1227. The compound restored synaptic function proteins and mitigated oxidative stress-induced nerve cell death 1231. The D+Q combination has also proven effective in neurodegenerative contexts, preventing cognitive deficits in Alzheimer's models and reducing systemic inflammation that exacerbates cognitive decline 2527.
Viral Pathogenesis and COVID-19 Models
Recent investigations have highlighted the role of cellular senescence in viral pathogenesis, particularly concerning SARS-CoV-2. Viral infections can rapidly induce a state of cellular senescence, contributing to severe acute inflammatory responses and post-viral sequelae. In advanced human brain organoid models, SARS-CoV-2 infection heavily induced senescence. Treatment with senolytics, including D+Q, navitoclax, and ABT-737, significantly reduced senescent cell burden and decreased viral load 32. D+Q specifically demonstrated the capacity to reverse the biological age of these infected organoids based on gene activity predictions 32.
In live animal studies, mice infected with SARS-CoV-2 exhibit profound neuropathy, rapid health deterioration, and abbreviated lifespans. Oral administration of fisetin or the D+Q combination every two days improved survival rates dramatically. While infected control mice survived approximately five days, treatment with either fisetin or D+Q extended the median lifespan of the infected cohorts by 60% and prevented profound neurodegeneration, establishing a clear link between senescent cell clearance and acute viral resilience 32.
Interventions Testing Program Lifespan Discrepancies
While early independent studies indicated that late-life administration of fisetin restored tissue homeostasis and extended both median and maximum lifespan in wild-type mice (by approximately 10%) 22526, highly rigorous subsequent investigations have yielded contradictory results regarding absolute lifespan extension.
The National Institute on Aging (NIA) Interventions Testing Program (ITP) - recognized globally as the gold standard for murine lifespan studies due to its rigorous, multi-site protocols - reported in 2023 that fisetin failed to significantly extend the lifespan of genetically heterogeneous UM-HET3 mice of either sex 33. The researchers posited that the discrepancy likely stemmed from the study design. The ITP study administered fisetin continuously through the diet over an extended period. This chronic, uninterrupted administration contrasts sharply with the intermittent "hit-and-run" dosing typically required for safe senolytic protocols 23334.
Continuous exposure to senolytics may induce off-target toxicities, impair normal physiological repair mechanisms, or eventually exhaust progenitor stem cell pools, thereby negating the healthspan benefits observed with pulsed, intermittent dosing 1433. Alternatively, differences in mouse strains may heavily influence senescent cell phenotypes and burden 33. Despite the failure to extend absolute maximal lifespan under continuous administration protocols, the scientific consensus remains robust that fisetin and quercetin consistently extend healthspan by delaying age-related frailty, preserving physical function, and mitigating disease progression when dosed appropriately 143536.
Pharmacokinetics and Bioavailability Formulations
A critical bottleneck in the clinical translation of flavonoid senolytics is their challenging pharmacokinetic profile. The "bioavailability paradox" dictates that while flavonoids exhibit highly potent senolytic and senomorphic effects in vitro, their physical characteristics and rapid systemic clearance severely limit therapeutic concentrations in human target tissues 4748.
The Bioavailability Paradox and Hepatic Metabolism
When ingested orally, both quercetin and fisetin are subject to rapid and extensive Phase I and Phase II metabolism in the liver and gastrointestinal tract 48. Cytochrome P450 enzymes swiftly convert these parent compounds into various conjugated forms, including glucuronides, sulfates, and methylated metabolites 1748. For fisetin, a primary active metabolite is geraldol. While geraldol retains some bioactivity, the overarching consequence of this rapid first-pass metabolism is a significant reduction in the availability of the pharmacologically potent parent molecule 1748.
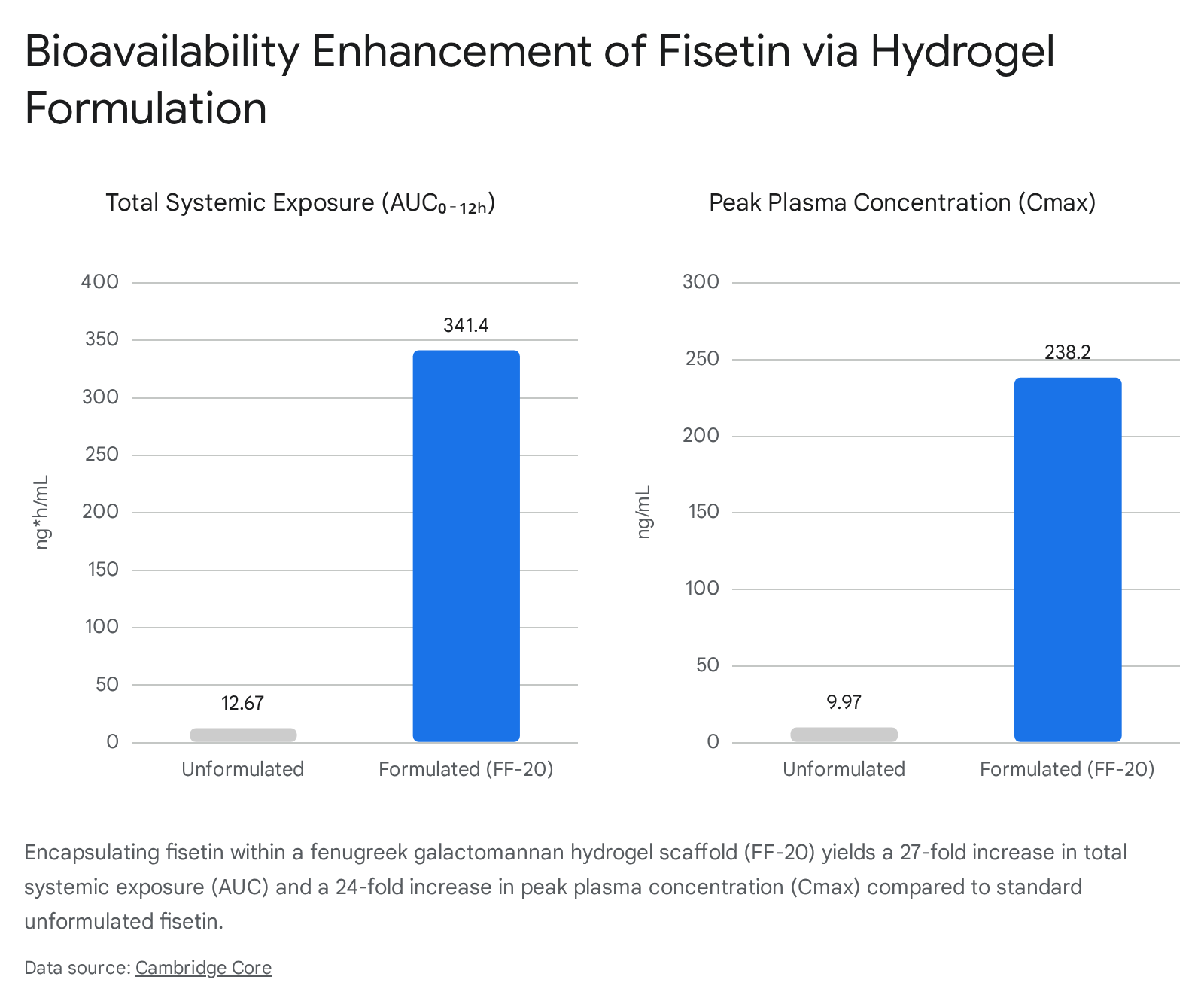
Unformulated fisetin is highly lipophilic and exhibits very poor aqueous solubility, leading to an estimated oral bioavailability of approximately 44% 48. Upon ingestion, fisetin achieves a maximum plasma concentration (Cmax) of roughly 10 ng/mL in under an hour (Tmax), and is cleared with a highly abbreviated half-life (t1/2) of merely 1.14 to 4 hours 1748. Quercetin possesses a slightly different profile; its natural glycoside forms offer a slower, more sustained release into the bloodstream, but standard unformulated quercetin extracts remain notoriously difficult for the human body to absorb efficiently 9242526.
| Pharmacokinetic Property | Unformulated Quercetin 9162425 | Unformulated Fisetin 1748 | Formulated Fisetin (FF-20) 17 |
|---|---|---|---|
| Chemical Structure | Flavonol (5 hydroxyl groups) | Flavonol (4 hydroxyl groups) | Encapsulated within Galactomannan |
| Water Solubility | Low (More hydrophilic than fisetin) | Very Low (Highly lipophilic) | Enhanced via hydrogel scaffold |
| Metabolism | High first-pass (Glucuronides) | High first-pass (Geraldol, Sulfates) | Reduced metabolic conversion |
| Half-Life (t1/2) | Rapid clearance | 1.14 hours | 1.51 hours |
| Tmax (Time to peak) | Sustained (glycosides) | 0.88 hours | 1.24 hours |
| Cmax (Peak plasma) | Poor without lipid carriers | ~9.97 ng/mL | ~238.2 ng/mL |
| Systemic Exposure (AUC) | Low | ~12.67 ng*h/mL | ~341.4 ng*h/mL |
Advanced Delivery Systems
To overcome these metabolic hurdles and achieve therapeutically relevant concentrations in deeper tissues (such as the central nervous system), the pharmaceutical and nutraceutical industries have developed advanced targeted delivery platforms.
For quercetin, researchers have widely utilized phytosome technology, which pairs the flavonoid with a phospholipid carrier (such as fenugreek galactomannans). This lipid-based encapsulation allows the molecule to bypass early metabolic degradation, improving overall bioavailability by up to 20-fold 92026. Similarly, quercetin-rosemary oil nanoemulsions are actively being explored to optimize tissue delivery for wound healing applications 16.
Fisetin absorption has also seen massive improvements through formulation technologies. A recent randomized, double-blind, human crossover study demonstrated the efficacy of a novel green technology (Hybrid-FENUMATTM), which encapsulated fisetin micelles into a fenugreek galactomannan hydrogel scaffold to create the FF-20 formulation 17. The FF-20 platform dramatically altered the compound's pharmacokinetics in human subjects. The resulting formulation increased the total systemic exposure (Area Under the Curve, AUC) of fisetin by 26.9-fold and the peak plasma concentration (Cmax) by nearly 24-fold compared to unformulated raw fisetin, while simultaneously reducing its rapid metabolic conversion into geraldol 17.
Human Clinical Trial Evidence and Outcomes
The translation of senolytics from murine models to human clinical applications is actively expanding, with several phase 1 and phase 2 trials completed, and dozens more ongoing. A central consensus in senolytic medicine dictates how these compounds must be administered to maximize efficacy while avoiding severe physiological side effects.
The Hit-and-Run Dosing Rationale
Unlike traditional pharmacological therapies in oncology or chronic disease management that require continuous daily dosing to maintain receptor saturation, senolytics are utilized via an intermittent, "hit-and-run" strategy 131949. Senescent cells do not actively proliferate; therefore, once they are eliminated by a brief, high-dose pulse of a senolytic agent, it takes several weeks or even months for new senescent cells to develop and accumulate to problematic levels 12736. Furthermore, fisetin and quercetin have short elimination half-lives, clearing the human body within roughly 24 hours 1019.
Administering a brief, intense course (e.g., 2 to 3 consecutive days separated by a two-to-four-week washout period) clears the existing senescent cell burden while allowing normal, proliferating tissue ample time to recover 273649. This intermittent approach drastically mitigates the risk of systemic toxicity, acute immunosuppression, and the potential depletion of the healthy stem cell pool that might occur with chronic daily inhibition of vital survival pathways like PI3K and BCL-2 111449.
Efficacy of Dasatinib and Quercetin Therapies
The D+Q combination is currently the most clinically advanced and robustly documented senolytic therapy in humans. Early pilot trials provided the first concrete proof-of-concept that senolytics engage their targets and decrease senescent cell burden in vivo. A phase 1 open-label trial involving patients with diabetic kidney disease demonstrated that a 3-day oral course of D+Q (100 mg dasatinib + 1000 mg quercetin) significantly reduced the abundance of p16INK4A and p21CIP1-expressing senescent cells in adipose tissue. Additionally, the therapy successfully decreased circulating SASP factors, including IL-1α, IL-6, and MMPs, measured 11 days post-treatment 1619. Another pilot study involving patients with idiopathic pulmonary fibrosis (IPF) - a fatal, progressive senescence-associated lung disease - reported measurable improvements in physical function metrics, such as gait speed, chair rise ability, and 6-minute walk distance, following intermittent D+Q administration 262728.
In 2024, the Mayo Clinic published the results of a rigorous 20-week Phase 2 randomized controlled trial evaluating D+Q for age-related osteoporosis in 60 healthy postmenopausal women 34. The study subjects received 100 mg of dasatinib and 1000 mg of quercetin for three consecutive days every 28 days. The results were highly nuanced. While the overall treatment group did not show a statistically significant reduction in bone resorption, exploratory analyses indicated that the skeletal response was entirely dependent on the baseline senescent cell burden 34. Women in the highest tertile of senescent cell burden (measured by T cell p16 mRNA levels) experienced robust increases in bone formation markers (P1NP), decreases in bone resorption markers (CTx), and an overall improvement in wrist bone mineral density 34.
This finding underscores a critical clinical reality: senolytics are highly targeted therapies that benefit individuals with a clinically significant accumulation of senescent cells, rather than serving as universal prophylactic "anti-aging" supplements for healthy individuals with low senescent burdens 34.
Fisetin Clinical Trial Landscape
Because it acts as a potent senolytic monotherapy without the need for an accompanying oncology drug like dasatinib, fisetin is the subject of intense global clinical scrutiny. While large-scale, phase 3 outcome data remains pending, several Phase 1 and Phase 2 studies are currently registered and actively recruiting across global clinical trial databases.
| Clinical Trial / Registry ID | Target Population & Condition | Fisetin Dosing Protocol | Key Primary Endpoints |
|---|---|---|---|
| NCT06133634 (Colorado) 37 | Older adults with vascular dysfunction | 2 mg/kg/day for 3 days, repeated after 2 weeks | Vascular endothelial function, aortic stiffness, circulating SASP |
| NCT05595499 (Jonsson Cancer Center) 383940 | Stage I-III breast cancer survivors (Post-chemotherapy) | 20 mg/kg/day for 3 days, every 2 weeks for 8 weeks | 6-minute walk distance, frailty phenotype, fatigue, p16 expression |
| NCT06431932 (Copenhagen) 41 | Older medical patients with multi-morbidity | 20 mg/kg/day for 2 consecutive days | Population PK modeling, safety, suPAR plasma levels |
| NCT07195318 42 | Healthy middle-aged and older adults | 100 mg daily for 7 weeks | Anti-inflammatory markers, general health metrics, biological aging |
| NCT06819254 (Wake Forest) 43 | Older adult cancer survivors with fatigue | 20 mg/kg twice daily for 2 days, repeated for 2 weeks | Efficacy of fatigue reduction, treatment adherence |
| ChiCTR265912 (China) 44 | Adults aged 45-70 with poor sleep quality | 500 mg daily for 12 weeks (alone or combined with Urolithin A) | Pittsburgh sleep quality index, physiological aging decline |
The broad range of these trials - spanning from high-dose hit-and-run protocols to address chemotherapy-induced senescence, to continuous low-dose protocols aimed at general inflammation - reflects the ongoing effort to optimize fisetin administration for specific pathological contexts.
Epigenetic Clocks and Multi-Compound Synergies
Recent clinical investigations have begun exploring the impact of senolytics on DNA methylation (DNAm) and biological "epigenetic clocks." A longitudinal pilot study (registered in the UK under ISRCTN85957759) involving 19 participants evaluated the effects of D+Q administration over six months 284546. Surprisingly, the study found significant increases in epigenetic age acceleration measured by first-generation and mitotic epigenetic clocks, alongside a notable decrease in telomere length at three and six months 2846.
Building upon these potentially deleterious findings, a subsequent investigation evaluated a multi-compound protocol combining D+Q with fisetin (DQF). The addition of fisetin to the senolytic regimen appeared to mitigate the adverse impacts of D+Q on epigenetic aging, resulting in non-significant increases in age acceleration and favorable shifts in immune cell proportions 2846. This suggests that complex, multi-compound senolytic/senomorphic cocktails may be necessary to balance clearance efficacy with the maintenance of stem cell and epigenetic health. Furthermore, fisetin is increasingly being evaluated in combinatorial oncology trials, such as NCT07025226, which combines fisetin, quercetin, dasatinib, and temozolomide to target residual disease and senescent cell accumulation in previously treated glioma patients 47.
Safety Profiles and Long-Term Clinical Considerations
Fisetin and quercetin are widely available as dietary supplements and are designated as "Generally Recognized as Safe" (GRAS) for standard nutritional consumption. However, their safety profiles at the highly concentrated doses utilized in clinical senolytic protocols require rigorous, long-term evaluation.
Off-Target Toxicity and Anticoagulant Risks
In preclinical trials and early human pilot studies, intermittent high-dose fisetin (frequently tested at 20 mg/kg/day) and quercetin (up to 2000 mg/day) have been remarkably well-tolerated 1836. They demonstrate a highly favorable therapeutic margin compared to synthetic, oncology-grade senolytics. For instance, navitoclax (a pharmaceutical BCL-2/BCL-xL inhibitor) is associated with severe, dose-limiting thrombocytopenia because platelets rely heavily on BCL-xL for survival 111536.
However, natural flavonoids are not devoid of side effects. Both fisetin and quercetin exhibit mild anticoagulant properties. Concurrent administration with pharmaceutical blood thinners - such as Warfarin, Aspirin, or novel oral anticoagulants - could theoretically elevate the risk of hemorrhage 1439. Furthermore, as both compounds are metabolized via hepatic cytochrome P450 pathways, they carry a distinct risk of pharmacokinetic drug-drug interactions, particularly when co-administered with CYP3A4 inhibitors 2748. Chronic administration of high-dose flavonoids has prompted researchers to recommend periodic monitoring of liver enzyme levels to guard against hepatotoxicity 18.
Long-Term Stem Cell Exhaustion and Knowledge Gaps
Despite rapid translational progress, significant clinical gaps remain as of 2026. The medical consensus actively discourages the off-label, over-the-counter use of concentrated senolytics for general "anti-aging" purposes outside of heavily monitored, IRB-approved trials 62734.
A primary obstacle is the lack of reliable, non-invasive clinical biomarkers capable of accurately quantifying a patient's senescent cell burden before treatment 273448. Without robust assays, it is impossible to identify the specific patient cohorts that will benefit from therapy, risking unnecessary exposure to off-target effects. Additionally, the long-term physiological consequences of repeatedly depleting senescent cells over decades remain entirely unknown. While senescent cells drive chronic inflammation, they also play vital roles in initiating physiological wound healing and preventing unchecked tumorigenesis. It is currently unclear whether continuous cycles of senolysis over a 10-to-20-year period could eventually exhaust the progenitor stem cell pool, impair acute injury repair, or result in fibrotic tissue replacement rather than healthy regeneration 61114.
Conclusion
The evidence supporting fisetin and quercetin as potent senolytic and senomorphic agents is robust at the preclinical level and is rapidly generating compelling, albeit early, human clinical data. By transiently targeting redundant SCAP survival networks - specifically the BCL-2 family proteins and PI3K/AKT/mTOR pathways - these flavonoids exploit intrinsic apoptotic priming to induce selective apoptosis in damaged, pro-inflammatory senescent cells while largely sparing healthy tissue. Fisetin has emerged as the most potent naturally occurring senolytic monotherapy, capable of broad-tissue clearance and robust SASP suppression via p38 MAPK inhibition. Quercetin demonstrates its highest efficacy when combined synergistically with dasatinib to ensure comprehensive SCAP network coverage.
Clinical pilot studies have validated the core therapeutic hypothesis: intermittent "hit-and-run" dosing of these compounds effectively reduces the senescent cell burden and lowers systemic inflammation in humans without causing severe acute toxicity. However, advanced Phase 2 trials indicate that senolytics are not a panacea for universal aging; their efficacy is highly specific, benefiting individuals with a demonstrably high burden of localized senescence, such as those with idiopathic pulmonary fibrosis, diabetic kidney disease, or chemotherapy-induced functional decline. Future advancements depend on overcoming the bioavailability paradox through advanced hydrogel and nanoparticle formulations, establishing reliable clinical biomarkers to quantify senescence, and completing longitudinal trials to elucidate the long-term safety and epigenetic impact of clearing the cellular hallmarks of aging.