Scientific evidence for extreme longevity optimization
Introduction
The pursuit of extreme longevity optimization has transitioned from theoretical gerontology into a highly visible movement characterized by quantified-self protocols, most prominently exemplified by Bryan Johnson's "Blueprint." These protocols represent an aggressive departure from traditional reactive disease management, instead aiming to proactively decelerate or reverse biological aging through intensive biomarker tracking, caloric restriction mimetics, and large-scale polypharmacy. The foundational hypothesis underlying these regimens is that aging is not an immutable decline but a malleable biological process driven by interconnected cellular pathways that can be pharmacologically and nutritionally manipulated.
However, the scientific validation of these highly complex, multi-modal interventions remains a subject of intense debate among gerontologists, pharmacologists, and institutional researchers. While individual compounds - such as rapamycin, metformin, and alpha-ketoglutarate - have demonstrated profound efficacy in extending healthspan or lifespan in isolated animal models, their translation to human physiology is fraught with complexities. The practice of combining dozens of these interventions into dense "longevity stacks" introduces severe risks of inappropriate polypharmacy, adverse drug-drug interactions, and redundant or antagonistic pathway targeting.
This report provides an exhaustive, evidence-based analysis of the scientific foundations underlying extreme longevity optimization. It evaluates the validity of current aging biomarkers, the clinical efficacy of prominent pharmacological agents and endogenous metabolites, the methodological limitations of N=1 self-experimentation, and the consensus guidelines from major geriatric institutions regarding polypharmacy and systems-level healthspan extension.
The Hallmarks of Aging and Biomarker Validation
The fundamental premise of extreme longevity optimization relies heavily on the clinical distinction between chronological age, which merely records the passage of time, and biological age, which reflects the physiological, molecular, and cellular degradation of the organism. Modern geroscience categorizes this degradation into interconnected mechanisms known as the hallmarks of aging.
The Biological Framework of Aging
The hallmarks of aging function at the molecular, cellular, and systemic levels. Primary hallmarks, considered the root causes of cellular decline, include genomic instability, telomere attrition, epigenetic alterations, and a loss of proteostasis. Secondary or antagonistic hallmarks represent protective responses that eventually become detrimental, such as mitochondrial dysfunction, deregulated nutrient sensing, and cellular senescence. Finally, integrative hallmarks manifest as the downstream consequences, including stem cell exhaustion and altered intercellular communication, which ultimately drive chronic conditions such as cardiovascular disease and neurodegeneration 12.
Recent longitudinal research demonstrates that biological aging does not progress in a linear, uniform manner. A 2024 multi-cohort study tracking 108 adults over several years utilized deep multi-omic profiling - analyzing proteins, lipids, RNA, and the microbiome - to reveal that biological aging accelerates sharply during two distinct physiological stages: the mid-40s and the early 60s 3. During these specific windows, 81% of all measured molecules underwent significant shifts. The mid-40s peak was heavily linked to alterations in lipid metabolism, cardiovascular disease vulnerability, and muscle dysfunction, while the early 60s peak was associated with carbohydrate metabolism dysregulation, immune system decline, and kidney function deterioration 3. Interventions seeking to optimize longevity must theoretically mitigate these specific, non-linear surges in biological aging.
Consensus on Clinical Endpoints
A major obstacle in translating longevity research into clinical practice is the lack of a standardized definition for biological age reversal. Because aging is inherently multidimensional, isolated biomarkers capture only fragmented perspectives of the physiological landscape 4. To standardize the evaluation of longevity interventions in human trials, an international expert panel recently utilized the Delphi method to establish a consensus on reliable aging biomarkers.
Out of hundreds of proposed metrics, the panel reached an agreement on 14 specific biomarkers that are minimally invasive, highly reliable, and predict biological aging better than chronological age alone 567.
| Biomarker Domain | Validated Clinical Biomarkers | Relevance to Longevity Interventions |
|---|---|---|
| Functional and Physical | Muscle mass, muscle strength, hand grip strength, gait speed, Timed-Up-and-Go, standing balance, frailty index | Directly predicts functional independence, fall risk, and all-cause mortality; responsive to exercise and protein synthesis interventions 57. |
| Metabolic and Endocrine | Insulin-like growth factor 1 (IGF-1), Growth-differentiating factor-15 (GDF-15), HbA1c | Reflects nutrient-sensing pathway dysregulation; heavily modulated by caloric restriction mimetics and targeted pharmacology 78. |
| Inflammatory and Immune | High-sensitivity C-reactive protein (hsCRP), Interleukin-6 (IL-6) | Captures "inflammaging" and the systemic burden of senescence-associated secretory phenotypes (SASP) 78. |
| Epigenetic and Molecular | DNA methylation (Epigenetic clocks) | Tracks cumulative physiological exposure and cellular identity; utilized to assess long-term trajectory alterations 479. |
| Cardiovascular and Cognitive | Blood pressure, Cognitive health assessments | Predicts vascular vulnerability, stroke risk, and the onset of neurodegenerative pathologies 79. |
Protocols that optimize exclusively for a single molecular marker while ignoring functional metrics - such as muscle mass or cardiorespiratory fitness - fail to yield genuine, systemic healthspan extension 2. The integration of these consensus biomarkers ensures that clinical outcomes reflect true physiological resilience.
Epigenetic Clocks and the Causality Debate
The most highly publicized metric in the quantified-self and biohacking communities is the epigenetic clock. Epigenetic clocks quantify biological age by measuring DNA methylation (DNAm) patterns at specific cytosine-guanine dinucleotide (CpG) islands 49. These epigenetic marks act as molecular switches that control gene expression without altering the underlying genetic sequence, responding dynamically to environmental exposures, stress, diet, and lifestyle 11.
Generational Clock Evolution
First-generation epigenetic clocks, such as the Horvath and Hannum algorithms, were trained primarily to predict chronological age based on methylation patterns 10. However, second-generation clocks - including GrimAge, PhenoAge, and DunedinPACE - were trained on physiological outcomes, mortality risk, and functional healthspan metrics, making them substantially more sensitive to biological interventions 1011. For example, the DunedinPACE clock measures the specific rate of biological aging, with longitudinal studies demonstrating that modifiable behaviors such as smoking, high body mass index, and elevated glucose accelerate the pace of aging, while physical activity and caloric restriction decelerate it 9. Additionally, accelerated biological aging as measured by GrimAge has been directly associated with a higher risk of incident mild cognitive impairment and dementia, independent of chronological age 10.
The Mutation Paradox and Root Cause Limitation
Despite the commercial popularity of epigenetic testing to "prove" age reversal, the scientific community remains divided on whether modifying an epigenetic clock equates to reversing systemic organismal aging. A critical 2025 study from the Ideker Lab at the University of California, San Diego, challenged the prevailing assumption that epigenetic drift is a primary driver of aging. The researchers demonstrated that random DNA mutations effectively trigger widespread epigenetic changes in surrounding genes. In their analysis, mutation-based age predictions achieved a high correlation (0.67) with chronological age, while methylation-based predictions achieved a correlation of 0.83, and both methods agreed on which individuals were biologically older or younger than their chronological age .
This finding introduces a severe limitation to epigenetic-focused longevity optimization. If epigenetic alterations are merely downstream symptoms of cumulative, random DNA damage - amplified across thousands of nearby gene control switches - then interventions designed solely to "reset" epigenetic marks may be treating the symptom rather than the underlying molecular root cause . A 2025 systematic review further highlighted this ambiguity, noting substantial variation in effect sizes across 14 different epigenetic clocks and 174 disease outcomes, indicating that no single epigenetic algorithm serves as a universally superior metric for clinical trials 9. Consequently, claiming systemic "age reversal" based entirely on transient shifts in DNA methylation remains scientifically premature 912.
Pharmacological Geroprotectors and Clinical Evidence
Extreme longevity optimization heavily relies on the off-label use of pharmacological agents classified as "geroprotectors." The most prominent of these compounds target the mechanistic target of rapamycin (mTOR) and AMP-activated protein kinase (AMPK) pathways, attempting to mimic the cellular benefits of caloric restriction.
Rapamycin and mTOR Inhibition
Rapamycin (sirolimus), originally isolated from soil bacteria on Easter Island and developed as an immunosuppressant and anti-fungal agent, is widely considered the most robust pharmacological intervention for lifespan extension in mammalian models 13. By inhibiting the mTOR pathway - specifically the mTORC1 complex - rapamycin mimics a state of nutrient scarcity, which enhances cellular autophagy, reduces systemic inflammation, and improves cellular resistance to stress 81714. The National Institute on Aging's Interventions Testing Program (ITP) has repeatedly confirmed that rapamycin extends median lifespan in genetically heterogeneous mice by 10 to 25 percent, even when administered late in life 1315.
However, translating rapamycin's profound animal efficacy to humans has yielded highly nuanced results. The 2025 Participatory Evaluation of Aging with Rapamycin for Longevity (PEARL) trial represents the most significant randomized, double-blind, placebo-controlled human trial to date for healthy, normative-aging adults 132016. Over 48 weeks, 114 participants aged 50 to 85 received a placebo, 5 mg, or 10 mg of compounded rapamycin weekly.
The clinical outcomes of the PEARL trial require careful interpretation. The trial missed its primary endpoint entirely, finding no significant change in visceral adiposity (p = 0.942) across any cohort 1617. However, the trial revealed distinct, sex-specific secondary efficacy signals. Women receiving the 10 mg weekly dose demonstrated statistically significant improvements in lean tissue mass (p = 0.013) and self-reported pain reduction (p = 0.015) 131617. Meanwhile, participants in the 5 mg cohort reported modest improvements in emotional well-being and general health 1617.
A critical methodological caveat emerged during the trial: the compounded rapamycin utilized for the study exhibited roughly one-third the blood concentration bioavailability of commercial, generic sirolimus formulations, potentially muting the systemic effects of the intervention 201718. Furthermore, rapamycin carries known metabolic trade-offs. While intermittent dosing primarily targets mTORC1, chronic or excessive exposure can unintentionally suppress mTORC2, a complex required for proper insulin signaling. This suppression can disrupt glucose metabolism, elevate cholesterol, and induce a state of "pseudo-diabetes" and insulin resistance, highlighting a direct mechanistic trade-off between targeted longevity benefits and metabolic side effects 1424.
Metformin and AMPK Activation
Metformin, an FDA-approved therapeutic utilized for over 60 years to manage type 2 diabetes, is frequently incorporated into longevity protocols for its ability to activate AMPK, inhibit complex I of the mitochondrial respiratory chain, and modulate nutrient-sensing pathways 11920. Retrospective epidemiological data, notably a massive 2014 UK study analyzing 180,000 subjects, suggested that diabetics taking metformin actually outlived matched non-diabetic controls, sparking intense interest in its anti-aging potential 21.
Subsequent animal research has reinforced these geroprotective mechanisms. A rigorous 2024 longitudinal primate study published in Cell demonstrated that metformin effectively decelerated brain aging markers by approximately six years in cynomolgus monkeys, enhancing neuroplasticity, improving cerebral blood flow, and reducing neuroinflammation 19.
Despite these mechanistic validations, definitive clinical proof in healthy humans remains elusive. The epidemiological data is heavily debated, as benefits appear most pronounced in individuals already suffering from metabolic dysfunction, suggesting that metformin may work primarily by correcting existing pathology rather than fundamentally decelerating the intrinsic aging process in healthy individuals 2122. Robust clinical validation is pending the full execution of the Targeting Aging with Metformin (TAME) trial, a proposed six-year, 3,000-person study aimed at demonstrating that metformin can delay a composite onset of cardiovascular disease, cancer, and dementia in non-diabetic adults aged 65 to 79 202123. However, because metformin is a low-cost, off-patent generic drug, the trial has faced significant funding obstacles, as pharmaceutical companies lack financial incentives to sponsor the research 23.
Combinatorial Pharmacology and Polypharmacy Interventions
To counter the specific metabolic side effects of isolated pharmacological interventions, extreme longevity practitioners frequently design combinatorial regimens, stacking multiple geroprotectors to achieve synergistic effects.
The Rapamycin, Metformin, and Acarbose Triad
The most heavily researched pharmacological combination involves pairing rapamycin with acarbose or metformin. Acarbose, an alpha-glucosidase inhibitor, acts locally in the gastrointestinal tract to slow carbohydrate digestion. This mechanism blunts postprandial glucose spikes, lowers systemic IGF-1 levels, reduces systemic inflammation, and favorably remodels the gut microbiome by increasing the production of short-chain fatty acids 817.
The rationale for this combination is deeply mechanistic: acarbose's potent insulin-sensitizing effects act as a direct biological antidote to the insulin-desensitizing properties of chronic rapamycin exposure 8. The National Institute on Aging's ITP tested this specific combination rigorously in murine models. The results were unprecedented in geroscience: combining rapamycin and acarbose produced a profound synergistic effect, extending median lifespan by 28% in female mice and 34% in male mice - vastly outperforming the lifespan extension achieved by either drug administered in isolation 815.
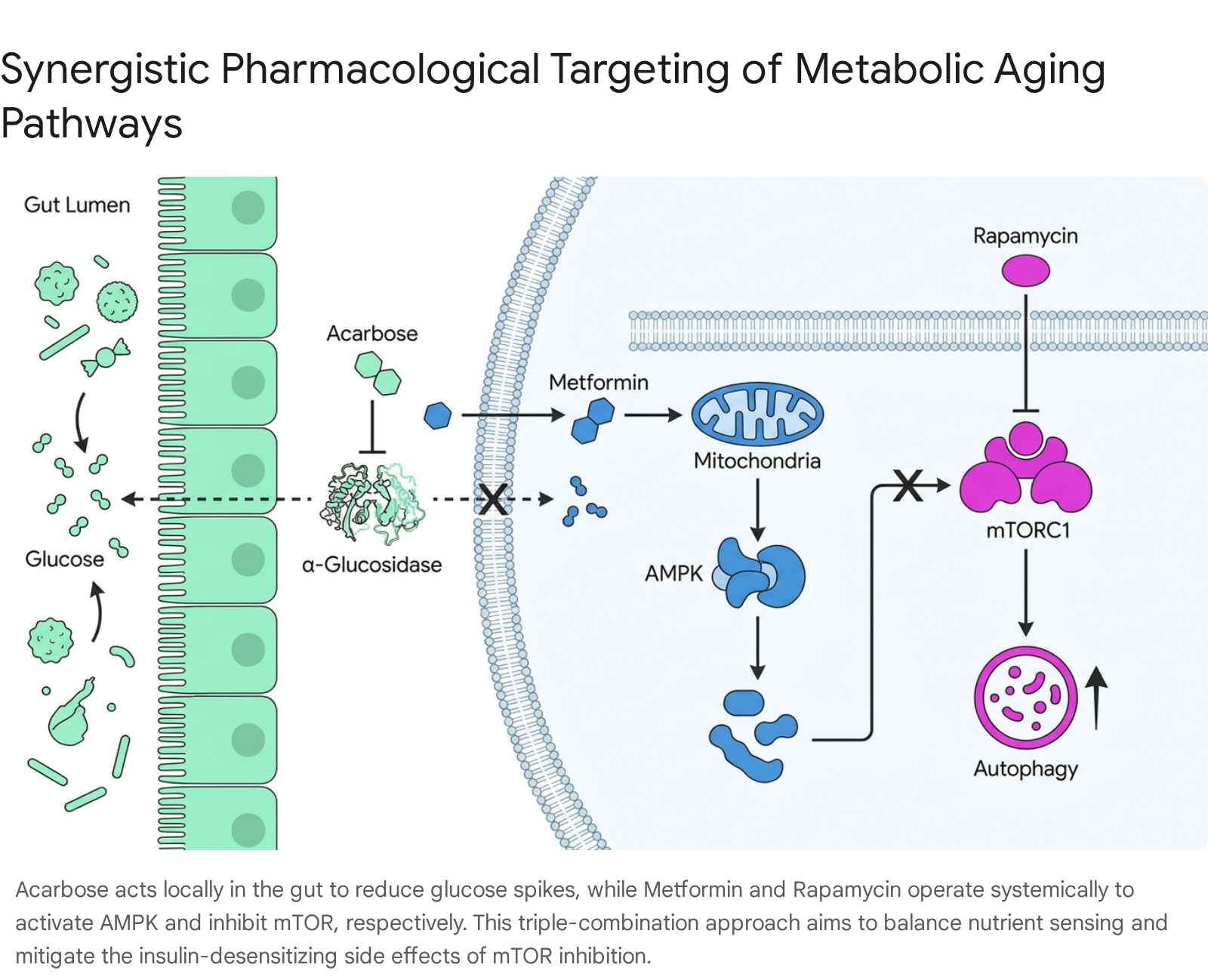
To validate multi-drug approaches in human biology, the VITAL-H trial, backed by $38 million in federal contract support, will test a triad combination of rapamycin, semaglutide, and dapagliflozin 24. Expected to enroll over 700 adults aged 60 to 65, the trial marks a shift toward measuring functional intrinsic capacity preservation using existing, FDA-approved therapeutics 24. Dapagliflozin, an SGLT2 inhibitor typically used for diabetes, is included specifically for its ability to flush excess glucose and reduce the low-grade chronic biological stress known as "inflammaging" 24.
Exogenous Metabolites and Polyamine Supplementation
Beyond prescription pharmaceuticals, extreme longevity optimization relies on the high-dose supplementation of naturally occurring cellular metabolites and polyamines intended to restore energy homeostasis and promote cellular clearance.
NAD+ Precursors: NMN and NMNH
Nicotinamide adenine dinucleotide (NAD+) is an essential coenzyme required for mitochondrial energy production, DNA repair, and the regulation of sirtuin proteins (particularly SIRT1) 252627. Systemic NAD+ levels decline precipitously with age, driving mitochondrial dysfunction and cellular senescence 26. To replete these cellular pools, practitioners utilize direct molecular precursors such as Nicotinamide Mononucleotide (NMN) or Nicotinamide Riboside (NR).
Recent human clinical trials (2024-2025) indicate that oral NMN supplementation is highly safe, well-tolerated, and successful at elevating whole-blood NAD+ concentrations 252829. Physiological benefits observed in human cohorts include improvements in aerobic capacity among amateur runners, enhanced insulin sensitivity, and reduced arterial stiffness in older adults 252628. Furthermore, a pioneering 2025 clinical trial demonstrated that nicotinamide riboside significantly improved arterial stiffness and reduced the size of severe skin ulcers in patients with Werner Syndrome, a rare disorder characterized by rapid, premature aging 30.
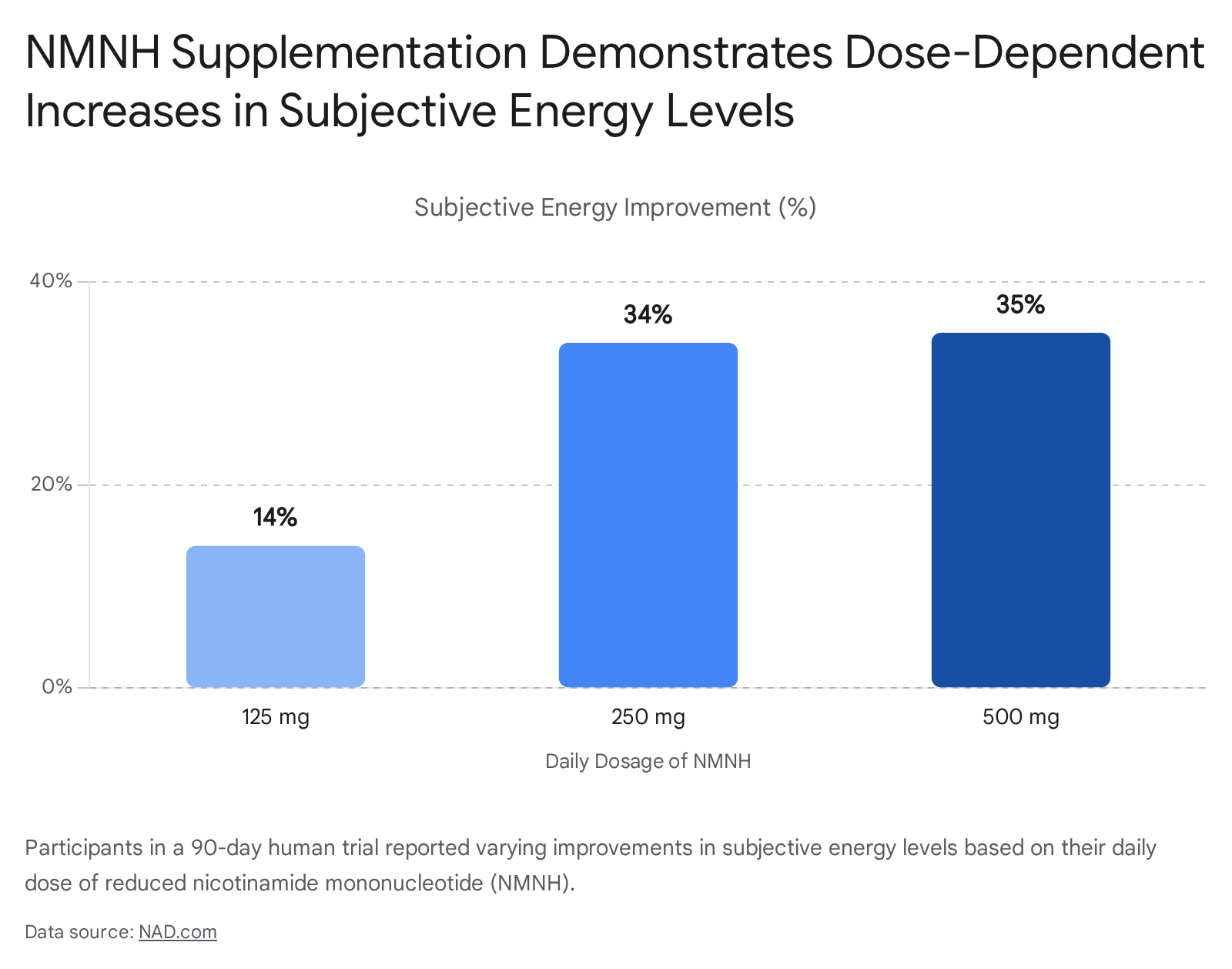
A novel, highly potent variation known as reduced nicotinamide mononucleotide (NMNH) has recently entered human testing. NMNH carries an extra hydrogen atom and two additional electrons, allowing it to bypass certain enzymatic bottlenecks. A first-of-its-kind 2026 clinical trial involving 80 healthy adults demonstrated that 500 mg of NMNH taken for 90 days effectively tripled circulating NAD+ levels 37.
Spermidine and Autophagic Clearance
Spermidine is a ubiquitous biogenic polyamine - originally discovered in human semen but heavily concentrated in dietary sources like wheat germ, soybeans, and mushrooms - that plays a critical role in cellular proliferation and intermediate metabolism 313940. In longevity science, its primary therapeutic mechanism is the induction of autophagy via the direct inhibition of the acetyltransferase activity of EP300 and the modulation of the polyamine-hypusination axis 313233. By functioning as a natural caloric restriction mimetic, spermidine facilitates the cellular clearance of misfolded proteins and damaged organelles without requiring stringent dietary starvation 314032.
Human epidemiological evidence strongly supports spermidine's geroprotective effects. The Bruneck Study, a prospective population-based cohort tracking 829 participants over 15 years (1995-2010), revealed that individuals in the highest tertile of dietary spermidine intake (>79.8 umol/d) experienced a 40% reduced risk of fatal heart failure, lower all-cause mortality, and a survival advantage equivalent to a 5.7-year reduction in chronological age compared to those in the lowest tertile 313934. Short-term interventional clinical trials have mirrored these neuroprotective benefits, demonstrating that 3.3 mg of daily oral spermidine supplementation improved memory performance and phonematic fluidity in older adults suffering from mild dementia 394032. However, systemic bioavailability is heavily dependent on individual gut microbiome synthesis, resulting in significant inter-individual heterogeneity in clinical response 3932.
Calcium Alpha-Ketoglutarate (Ca-AKG)
Alpha-ketoglutarate (AKG) is a fundamental metabolic intermediate in the mitochondrial Krebs cycle, crucial for cellular energy production. Beyond energy metabolism, AKG functions as an essential co-factor for TET (Ten-Eleven Translocation) and Jumonji enzymes, which govern DNA and histone demethylation 4435. Because endogenous AKG levels decline sharply as organisms age, targeted replenishment is theorized to restore metabolic flexibility and epigenetic stability 4436.
To overcome the poor oral bioavailability and rapid degradation of free AKG, clinical longevity protocols utilize the stabilized formulation calcium alpha-ketoglutarate (Ca-AKG). In a retrospective analysis involving 42 mid-life adults, 1 gram of daily Ca-AKG administered over seven months was associated with an average 8-year reduction in biological age as measured by DNA methylation clocks, with no serious adverse effects 4435. Controlled clinical trials have also demonstrated structural benefits; post-menopausal women taking 6 grams of Ca-AKG daily for 24 weeks experienced an 18% reduction in bone breakdown markers and an improvement in lumbar bone mineral density 4436. Furthermore, preclinical Alzheimer's disease models show that Ca-AKG restores long-term potentiation and repairs weakened synaptic plasticity, positioning it as a potent neuroprotective compound 37.
| Compound | Primary Biological Mechanism | Notable Human Clinical Evidence |
|---|---|---|
| NMN / NMNH | Replenishes NAD+ pools; supports sirtuin activity and mitochondrial energy production 2627. | Triples circulating NAD+ levels; improves aerobic capacity and reduces arterial stiffness 252837. |
| Spermidine | Inhibits EP300 acetyltransferase; acts as a caloric restriction mimetic to induce autophagy 3133. | Epidemiological link to 5.7-year chronological age survival advantage; improves memory in mild dementia 3234. |
| Ca-AKG | Krebs cycle intermediate; co-factor for TET enzymes regulating DNA demethylation 4435. | Associated with biological age reduction; improves bone mineral density and nitrogen balance 443536. |
Methodological Validity of N=1 Self-Experimentation
Extreme longevity protocols actively reject generalized, population-level health guidelines in favor of hyper-personalized, N=1 clinical methodologies. In a traditional parallel-group randomized controlled trial (RCT), researchers estimate the average effect of an intervention across a broad population. This standard approach inherently obscures the heterogeneity of treatment effects (HTE) - meaning a drug might be highly effective for a specific genetic subset of patients but entirely useless or actively harmful to others 48383951.
Advantages in Precision and Multimorbidity
An N=1 trial is defined methodologically as a multiple-period crossover experiment conducted on a single individual 383940. By repeatedly alternating between a specific treatment period and a placebo (or control) washout period, the individual acts as their own baseline and control. This design effectively eliminates inter-individual genetic, metabolic, and lifestyle confounders that plague traditional epidemiology 485141. The Oxford Centre for Evidence-Based Medicine classifies rigorously conducted N=1 trials as Level 1 evidence for the specific patient involved. This methodology is uniquely suited for managing chronic, stable conditions and navigating complex multimorbidities, as traditional RCTs often exclude patients with multiple overlapping health issues 485140.
Statistical Vulnerabilities and Regression to the Mean
Despite its theoretical superiority for individual precision medicine, N=1 biohacking is highly susceptible to profound methodological errors when executed outside of clinical supervision. Valid N=1 trials strictly require interventions with rapid onset and defined washout periods. Interventions with persistent carry-over effects - such as long-term epigenetic remodeling, microbiome shifts, or persistent cellular tissue changes - irreversibly contaminate the subsequent crossover periods, rendering longitudinal data invalid 38394041.
Furthermore, self-experimenting longevity practitioners routinely fall victim to the Regression to the Mean (RTM) fallacy. RTM is a fundamental statistical phenomenon where extreme sample observations are naturally followed by subsequent measurements that regress closer to the population average, driven purely by random biological variance and measurement error 4243. If a practitioner tests a biomarker and receives an unusually poor result (e.g., an extreme spike in inflammatory cytokines or a sudden increase in epigenetic age), immediately implements a novel supplement stack, and observes an improvement weeks later, they will almost universally attribute the success to the intervention 42. Without unmedicated baseline stabilization, rigorous blinding, and statistical adjustments, the "age reversal" celebrated in high-profile self-reported protocols often represents nothing more than natural biological fluctuation paired with powerful contextual placebo effects 4243.
Polypharmacy Risks and Geriatric Pharmacology Guidelines
A central and heavily criticized contradiction in the longevity optimization movement is the reliance on massive polypharmacy. Medically defined as the concurrent, habitual use of five or more medications, polypharmacy is a hallmark of protocols like Bryan Johnson's Blueprint, where participants frequently consume dozens of pills, powders, and injections daily 4445.
The Clinical Dangers of Unregulated Stacking
From the perspective of clinical pharmacology, incrementally higher numbers of daily prescribed medications strictly correlate with exponential increases in the risk of hospitalization, adverse drug events, and all-cause mortality 44454647. The interaction between standard longevity drugs highlights the severity of this danger. For instance, rapamycin is heavily metabolized by the cytochrome P450 enzyme CYP3A. Concurrent ingestion of potent CYP3A inhibitors - such as cannabidiol (CBD), grapefruit compounds, or high doses of certain herbal extracts - drastically increases circulating levels of mTOR inhibitors in the bloodstream, severely raising the risk of acute toxicity, immunosuppression, and liver damage 1447.
Furthermore, many longevity supplements exhibit mechanistic redundancy that can inadvertently damage the organism. Stacking multiple compounds that independently inhibit insulin signaling or suppress oxidative stress can induce harmful physiological consequences 4849. Oxidative stress is not universally detrimental; mild oxidative signaling is absolutely necessary for hormesis, the process by which cells build adaptive resilience. Completely neutralizing reactive oxygen species (ROS) through the excessive consumption of free-radical scavenging antioxidants will actively block the beneficial, adaptive cellular effects of physical exercise and stress responses, rendering the organism weaker rather than younger 4849.
Institutional Guidelines on Deprescribing
Major European medical institutions, which manage the world's most rapidly aging populations, actively caution against the over-accumulation of treatments in aging cohorts. The European Geriatric Medicine Society (EUGMS) and the British Geriatrics Society (BGS) view the uncontrolled proliferation of medications as a systemic failure of modern medicine's fragmented, symptom-specific approach 44475051.
In a 2023 consensus position paper, the EUGMS Special Interest Group on Pharmacology advocated strongly for widespread "deprescribing" - defined as the supervised, intentional withdrawal of inappropriate medications to minimize iatrogenic harm, reduce the daily pill burden, and improve holistic functional outcomes 525354.
| Clinical Paradigm | Approach to Therapeutics | View on Aging and Biological Resilience |
|---|---|---|
| Extreme Biohacking / Quantified Self | Aggressive polypharmacy; combinatorial "stacking" of highly experimental agents to force molecular shifts 4849. | Aging is an engineering problem solved by the continuous addition of molecular interventions and pathway suppressions. |
| Geriatric Medicine (EUGMS / BGS) | Rigorous deprescribing; targeting minimal effective dosing; establishing pragmatic therapeutic goals 47525355. | Aging alters pharmacokinetics and renal function; reducing drug burden minimizes the risk of cascading systemic failure. |
Geriatricians emphasize that smart prescribing in older age requires periodically reviewing all medications to prioritize treatments that have a real, demonstrable impact on functional quality of life, rather than merely chasing optimized clinical parameters on a blood panel 44515657.
Institutional Perspectives on Resilience Engineering
Leading global gerontology research centers draw a sharp, uncompromising distinction between evidence-based "longevity medicine" and consumer-driven "biohacking."
Critiques of Redundant Protocols
Institutions such as the Max Planck Institute for Biology of Ageing and the UCL Institute of Healthy Ageing maintain that while foundational lifestyle interventions - such as sleep optimization, protein maintenance, and rigorous physical activity - are scientifically unassailable, the commercial longevity market is plagued by specious claims and uncalibrated experimentation 8585960.
True longevity medicine is defined by clinical risk stratification, advanced genetic testing, precise metabolic optimization (e.g., maintaining insulin sensitivity and lipid profiles), and the use of targeted pharmaceuticals under rigorous medical supervision 58. In stark contrast, extreme biohacking frequently lacks regulatory oversight and chases short-term optimization metrics using unregulated peptides or redundant supplement stacks that completely lack longitudinal human safety data 85860. Cardiologist and researcher Eric Topol's analysis of "Super Agers" - individuals over 80 who exhibit no major chronic diseases - revealed that these individuals possess no secret genetic advantages. Their resilience is driven by a web of fundamental factors: exercise, sleep quality, social connection, and immune system health, proving that extreme pharmacological intervention is not a prerequisite for maximizing healthspan 60.
The Systems Biology View of Aging
Global gerontology institutions, including the National Center for Geriatrics and Gerontology (NCGG) in Japan and the European Society of Preventive Medicine, are fundamentally shifting away from viewing aging as a simple collection of isolated biological errors 616263. As presented at the 2026 International Conference on Targeting Longevity in Berlin, leading researchers now define aging as a systems-level failure - a gradual loss of coordination and communication between mitochondria, the gut microbiota, metabolic regulation, and immune signaling networks 6364.
If aging represents a breakdown in vast, interconnected network coordination, bombarding the biological system with dozens of isolated molecular targets via extreme polypharmacy may yield rapidly diminishing or actively antagonistic returns 4864. Researchers hypothesize that future longevity therapies must shift away from trying to reverse individual cellular defects and instead focus on "resilience engineering." This paradigm prioritizes stabilizing biological complexity and maintaining functional capacity to withstand stress, rather than solely attempting to maximize chronological lifespan via molecular brute force 264.
Conclusion
The scientific architecture underpinning extreme longevity optimization rests on valid, cutting-edge biological principles but frequently overextends current clinical evidence. The conceptual shift from treating the symptoms of chronological age to targeting the root causes of biological age is well-supported by advances in epigenetic tracking and molecular biomarkers. Furthermore, specific interventions - such as rapamycin's targeted inhibition of mTOR, metformin's activation of AMPK, and the autophagic induction driven by spermidine - possess incredibly robust mechanistic backing and compelling lifespan extension data in mammalian models.
However, the leap to massive, self-directed human polypharmacy is not currently validated by controlled clinical trials. Human studies such as the PEARL trial and the epidemiological data surrounding metformin demonstrate modest, highly nuanced healthspan benefits - such as localized pain reduction and lean mass preservation - rather than dramatic, systemic lifespan extension. The strategy of "stacking" dozens of experimental interventions is heavily criticized by clinical geriatric pharmacologists due to the severe, documented risks of adverse drug-drug interactions, redundant pathway targeting, and the dangerous blunting of necessary hormetic stressors. Additionally, the N=1 methodology championed by biohackers is statistically vulnerable to the regression-to-the-mean fallacy, frequently confusing natural biological variance and placebo effects with therapeutic success.
Ultimately, while the underlying geroscience is rapidly advancing toward actionable, disease-modifying interventions, true longevity optimization requires a paradigm shift away from extreme, unregulated polypharmacy. The future of the field, as championed by major gerontological institutions, lies in precision resilience engineering, rigorous clinical validation, targeted deprescribing, and the holistic mitigation of functional decline to maximize human healthspan.