Scientific evidence on dietary patterns and mental health
Nutritional psychiatry represents an evidence-based, transdisciplinary investigation into how diet and nutrients influence mental and brain health across the life course. Historically, psychiatric treatment paradigms have relied almost exclusively on pharmacotherapy, psychotherapy, and neuromodulation. However, an expansive body of epidemiological, preclinical, and clinical evidence now positions diet quality as a highly modifiable risk factor for psychiatric morbidity 123.
Despite a surge in clinical application and public interest, the field is characterized by a tension between robust observational data, complex physiological mechanisms, and the rigorous constraints of randomized controlled trials (RCTs) 456. This report synthesizes current scientific knowledge regarding the biological pathways linking diet and mental health, evaluates the efficacy of dominant dietary patterns, details the methodological limitations inherent in nutritional research, and outlines the integration of this evidence into clinical practice guidelines.
Foundational Biological Mechanisms
The pathways through which nutrition influences mental health extend far beyond simple caloric intake or macronutrient ratios. Research indicates that dietary components modulate neurobiology through several interconnected systemic pathways, primarily involving the microbiota-gut-brain axis, systemic inflammation, neurotransmitter synthesis, and mitochondrial energy metabolism 178.
The Microbiota-Gut-Brain Axis
The human gastrointestinal tract is colonized by trillions of microorganisms that play a critical role in food digestion, vitamin synthesis, and immune system regulation. The microbiota-gut-brain axis is a bidirectional communication network connecting the enteric nervous system to the central nervous system (CNS) via the vagus nerve, immune signaling, and circulating microbial metabolites 19.
Dietary patterns dictate the composition, resilience, and diversity of the gut microbiome. Diets high in dietary fiber, prebiotics, and polyphenols - found abundantly in plant-based whole foods - promote the proliferation of beneficial bacterial taxa 310. These bacteria ferment indigestible carbohydrates into short-chain fatty acids (SCFAs), such as butyrate, acetate, and propionate. SCFAs maintain the structural integrity of the intestinal epithelial barrier, exert systemic anti-inflammatory effects, and can cross the blood-brain barrier (BBB) to influence neuroplasticity 34911.
Conversely, dietary patterns characterized by high intakes of refined sugars, saturated fats, and artificial additives can rapidly induce gut dysbiosis 112. Dysbiosis compromises the intestinal barrier, a condition frequently termed increased intestinal permeability or "leaky gut." This degradation allows endotoxins, such as lipopolysaccharides (LPS), and microbial metabolites to translocate into the systemic circulation 713.
Systemic Inflammation and Oxidative Stress
The translocation of endotoxins triggers a systemic immune response, elevating circulating pro-inflammatory cytokines such as interleukin-6 (IL-6), interleukin-17 (IL-17), and C-reactive protein (CRP) 81415. Chronic, low-grade systemic inflammation is now a well-established risk factor for the development of major depressive disorder (MDD) and anxiety disorders 4.
In the CNS, systemic inflammation activates microglia, the brain's resident immune cells. Activated microglia release local inflammatory mediators that impair neurogenesis and neuroplasticity, particularly in the hippocampus 4. Furthermore, inflammation reduces the availability of Brain-Derived Neurotrophic Factor (BDNF), a critical protein responsible for the survival and growth of neurons 416. Diets rich in antioxidants and phytochemicals - such as those containing olive oil, berries, and nuts - are hypothesized to neutralize reactive oxygen species, thereby mitigating oxidative stress and preserving neural network integrity 317.
Tryptophan Metabolism and Serotonin Synthesis
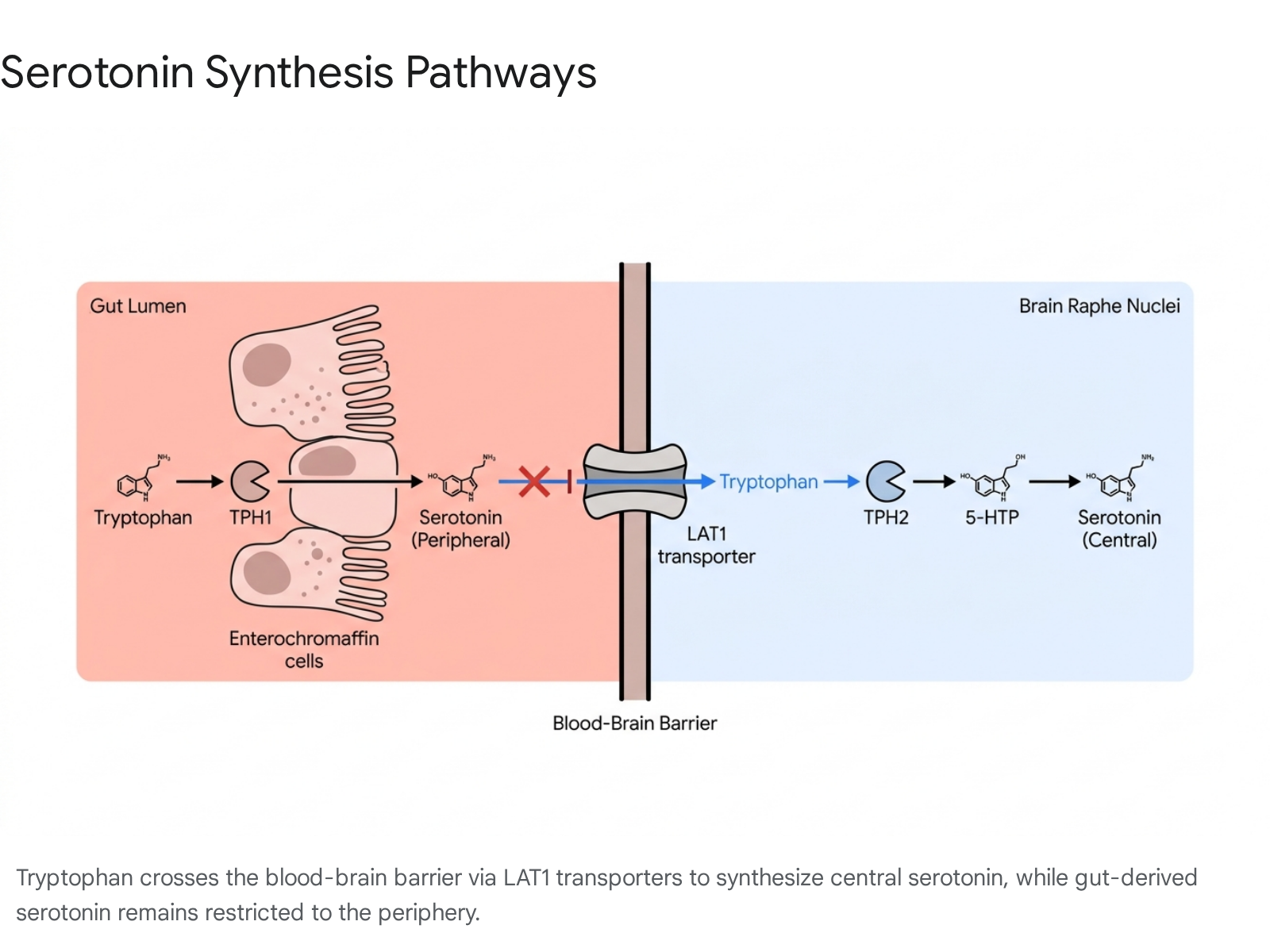
A prevalent misconception in popular nutrition is the direct, unmediated relationship between gut serotonin and mood. It is frequently cited in public health literature that 90% to 95% of the body's serotonin is produced in the gastrointestinal tract 1819. While factually accurate, this gut-derived serotonin functions primarily as a peripheral hormone, regulating gastrointestinal motility, vascular tone, and platelet function 1318.
Crucially, peripheral serotonin cannot cross the blood-brain barrier 91819. The central nervous system must synthesize its own serotonin de novo from the essential amino acid tryptophan, which must be obtained exclusively through dietary intake 913.
The synthesis of serotonin relies on two distinct isoenzymes separated by the BBB: 1. Tryptophan Hydroxylase 1 (TPH1): Expressed in the gut enterochromaffin cells, responsible for synthesizing peripheral serotonin 911. 2. Tryptophan Hydroxylase 2 (TPH2): Expressed in CNS neurons, serving as the rate-limiting step for central serotonin synthesis 911.
Dietary tryptophan crosses the BBB via the Large Amino Acid Transporter 1 (LAT1) 918. However, tryptophan must compete with branched-chain amino acids (BCAAs) for this specific transporter. Furthermore, during states of chronic inflammation, pro-inflammatory cytokines activate the indoleamine 2,3-dioxygenase (IDO) enzyme. IDO shunts tryptophan away from the serotonin synthesis pathway and redirects it down the kynurenine pathway 13. Overactivation of the kynurenine pathway depletes central tryptophan and produces neurotoxic metabolites, which correlate strongly with depressive symptoms and cognitive decline 413. Therefore, diets that reduce systemic inflammation indirectly preserve the brain's capacity to synthesize serotonin by preventing IDO overactivation.
Brain Energy Metabolism and Mitochondrial Function
Emerging research in metabolic psychiatry identifies mitochondrial dysfunction and impaired glucose metabolism as core pathophysiological features in severe psychiatric disorders, including MDD, bipolar disorder, and schizophrenia 1920. The brain is a highly energy-demanding organ, accounting for a disproportionate amount of the body's metabolic requirements.
When neurons experience insulin resistance or mitochondrial deficits, cellular energy production drops, leading to oxidative stress and impaired neural network connectivity 20. Dietary interventions that alter brain energy substrates - specifically shifting cellular metabolism from glucose to ketone bodies - are currently under intensive investigation for their ability to bypass defective glycolytic pathways and restore neural energy homeostasis 1920.
Evaluation of Specific Dietary Patterns
Scientific evaluation of nutrition has largely shifted away from analyzing isolated micronutrients (e.g., assessing Vitamin D or Zinc supplementation in a vacuum) toward assessing holistic dietary patterns. The synergistic effect of food matrices, fiber content, and macronutrient interactions exerts a far more profound impact on human psychobiology than isolated compounds 7821.
Ultra-Processed Foods and Western Dietary Patterns
The epidemiological evidence regarding the psychiatric harms of ultra-processed foods (UPFs) is among the most robust in nutritional psychiatry. UPFs are industrially formulated edible substances characterized by refined carbohydrates, modified fats, minimal dietary fiber, and the inclusion of artificial additives, emulsifiers, and preservatives 2223.
A comprehensive 2024 umbrella review of epidemiological meta-analyses demonstrated a clear, dose-response association between UPF exposure and the risk of common mental disorders 2224. The aggregated data indicates that for every 10% increase in UPF consumption, there is an approximate 11% higher risk of developing depression 10.
The mechanisms underlying this deleterious association are multifold. UPFs displace nutrient-dense foods, leading to inadequate intake of polyphenols, omega-3 fatty acids, and essential micronutrients required for optimal brain function 1023. The lack of dietary fiber in UPFs starves beneficial gut microbes, driving rapid dysbiosis 10. Additionally, the high glycemic load of UPFs induces rapid fluctuations in blood glucose and insulin levels, causing mood lability and promoting oxidative stress 22. Across global cohorts, the nutritional transition away from traditional food systems toward Westernized, UPF-heavy diets is strongly correlated with rising rates of psychiatric morbidity and non-communicable diseases 16232526.
The Mediterranean Diet
Conversely, dietary patterns rich in minimally processed whole foods consistently demonstrate protective effects against psychiatric distress. The Mediterranean Diet (MD) is currently the most extensively researched pattern, emphasizing high consumption of extra-virgin olive oil, vegetables, legumes, whole grains, nuts, and fish, with moderate poultry and dairy, and highly restricted red meat and sweets 121623.
The landmark PREDIMED (Prevención con Dieta Mediterránea) trial is central to this evidence base. While primarily designed as a cardiovascular primary prevention trial, secondary analyses evaluated the incidence of depression among participants. In the overall cohort of older adults at high cardiovascular risk, the reduction in depression risk for those assigned to the MD supplemented with nuts was not statistically significant (Hazard Ratio [HR] 0.78; 95% Confidence Interval [CI] 0.55 to 1.10) 272829. However, in a prespecified sub-analysis restricted to participants with Type 2 Diabetes, the MD supplemented with nuts resulted in a statistically significant 41% reduction in the risk of developing depression compared to the low-fat control diet (HR 0.59; 95% CI 0.36 to 0.98) 2728.
Recent 2025 meta-analyses of RCTs confirm that MD interventions significantly reduce depressive symptoms among adults with major or mild depression, though the overall certainty of evidence remains low to moderate due to methodological heterogeneity between studies 3031. Furthermore, while the evidence for depression is supportive, the evidence for the MD alleviating clinical anxiety remains highly uncertain and largely inconclusive 173032.
Traditional Agrarian Diets of the Global South
Beyond the Mediterranean framework, traditional agrarian diets of the Global South - including precolonial African, South American, and Iranian dietary patterns - exhibit comparable mental health benefits 212533. These diets share a foundational reliance on high-quality plant-based whole foods, local pulses, indigenous grains (such as sorghum, millet, teff, and amaranth), and fermented foods 2533.
Meta-analyses focusing on non-Western populations indicate that adherence to these traditional, high-diversity diets is inversely associated with depression and psychological distress 212334. A key component of these traditional diets is high dietary diversity, which ensures a broad spectrum of bioavailable micronutrients and supports microbiome resilience 2334. For example, adherence to a "Modern Iranian" or "Flexitarian" pattern has been associated with significantly lower anxiety and stress scores compared to Westernized patterns 21. However, as industrialization and globalization encroach, these traditional food systems are being rapidly replaced by imported ultra-processed foods, threatening public health and exacerbating the dual burden of malnutrition and mental illness 2533.
Ketogenic and Metabolic Therapies
The ketogenic diet (KD) is a high-fat, adequate-protein, and extremely low-carbohydrate regimen explicitly designed to induce hepatic ketogenesis. Originally developed over a century ago as a highly effective, evidence-based treatment for pediatric medication-resistant epilepsy, the KD is now being rigorously evaluated as a metabolic therapy for severe psychiatric disorders 1935.
By replacing glucose with ketone bodies (such as beta-hydroxybutyrate) as the primary neural fuel source, the KD may bypass defective glycolytic pathways common in psychiatric disorders, enhance mitochondrial respiration, upregulate gamma-aminobutyric acid (GABA, an inhibitory neurotransmitter), and reduce neuroinflammation 1920.
Clinical evidence for the KD in psychiatry is growing rapidly but remains preliminary compared to epilepsy data 35. A comprehensive 2026 meta-analysis of 50 studies evaluating KDs for mental health outcomes found modest but statistically significant improvements in depressive symptoms (Standardized Mean Difference [SMD] -0.48), particularly in trials that verified adherence via biochemical ketone monitoring 36. However, mirroring the Mediterranean diet data, evidence regarding the KD's efficacy for anxiety remains inconclusive (SMD -0.03) 36. Pilot clinical trials are currently underway evaluating KDs for bipolar disorder and schizophrenia, with early findings showing notable symptom reduction and improved systemic metabolic markers 203837.
The Carnivore Diet
At the extreme end of the carbohydrate-restriction spectrum is the carnivore diet, an entirely animal-based dietary pattern (meat, fish, eggs, dairy) that strictly eliminates all plant matter 383940. The diet has gained immense popularity through social media channels, with proponents claiming extraordinary benefits, including the remission of severe autoimmune conditions, depression, and even isolated case reports of schizophrenia remission 384142.
A highly cited 2021 survey of over 2,000 self-identified carnivore diet adherents revealed that 95% reported improvements in mood, mental clarity, and anxiety 4142. However, the scientific evidence supporting these psychiatric claims is overwhelmingly anecdotal, and the long-term physiological safety of the diet is severely contested by the medical community 404344.
A 2026 scoping review of the carnivore diet concluded that the overall quality of evidence is "very limited" due to an absolute absence of control groups, reliance on subjective self-reporting, and short study durations 38. While short-term adherence may yield benefits such as rapid weight loss, stable blood glucose, and the elimination of dietary triggers for certain gastrointestinal issues, the systemic risks are substantial 394445.
Eliminating plant foods completely eradicates dietary fiber and phytonutrients, posing severe long-term risks to gut microbiome diversity and structural integrity 404446. Furthermore, rigorous nutritional analyses of typical carnivore meal plans reveal critical deficiencies in essential micronutrients, particularly Vitamin C, magnesium, folate, and calcium 38434547. This is frequently accompanied by alarming increases in saturated fat intake and low-density lipoprotein (LDL) cholesterol, raising significant long-term cardiovascular risks 38434547. Consequently, no major medical or psychiatric body currently endorses the carnivore diet for mental health management 384048.
Table 1: Comparison of Dominant Dietary Patterns and Mental Health Evidence
| Dietary Pattern | Core Nutritional Components | Psychiatric Association | Proposed Biological Mechanisms | Certainty of Evidence |
|---|---|---|---|---|
| Western / UPF | High refined sugar, modified fats, chemical additives, low fiber | Dose-response increased risk of depression, anxiety, and distress | Induces low-grade inflammation, gut dysbiosis, high glycemic variability | High (Observational Cohorts / Umbrella Reviews) |
| Mediterranean | Extra-virgin olive oil, nuts, legumes, fish, vegetables, whole grains | Reduced depressive symptoms, potential prevention of depression onset | High polyphenols reduce oxidative stress; omega-3s support neuroplasticity | Moderate (RCTs and Meta-analyses) |
| Traditional (Global South) | Local pulses, millets, whole grains, fermented foods, high diversity | Inversely associated with anxiety and depression prevalence | Dietary diversity supports microbiome resilience; rich in bioavailable micronutrients | Low to Moderate (Cross-sectional and Cohorts) |
| Ketogenic | Very high fat, adequate protein, <50g carbohydrates/day | Modest improvement in depressive symptoms; early data in bipolar/schizophrenia | Shifts neural fuel to ketones, bypasses impaired glycolysis, upregulates GABA | Low to Moderate (Emerging RCTs and Pilot Studies) |
| Carnivore | Exclusively animal-derived (meat, eggs, dairy); zero plant matter | Anecdotal reports of improved mood and clarity; no controlled psychiatric data | Elimination of inflammatory plant antigens; stable blood glucose; extreme ketosis | Very Low (Self-reported surveys, Case studies) |
Clinical Trial Evidence and Methodological Evaluation
The transition of nutritional psychiatry from observational epidemiology to clinical intervention is heavily reliant on randomized controlled trials. While observational data demonstrates strong correlations, rigorous RCTs are required to establish causality and quantify therapeutic efficacy.
The SMILES Trial and Landmark Interventions
The Supporting the Modification of lifestyle In Lowered Emotional States (SMILES) trial, published in 2017, represents a watershed moment for the discipline. It was the first RCT specifically designed to test whether a structured dietary intervention could function as a primary treatment strategy for clinical depression 4952.
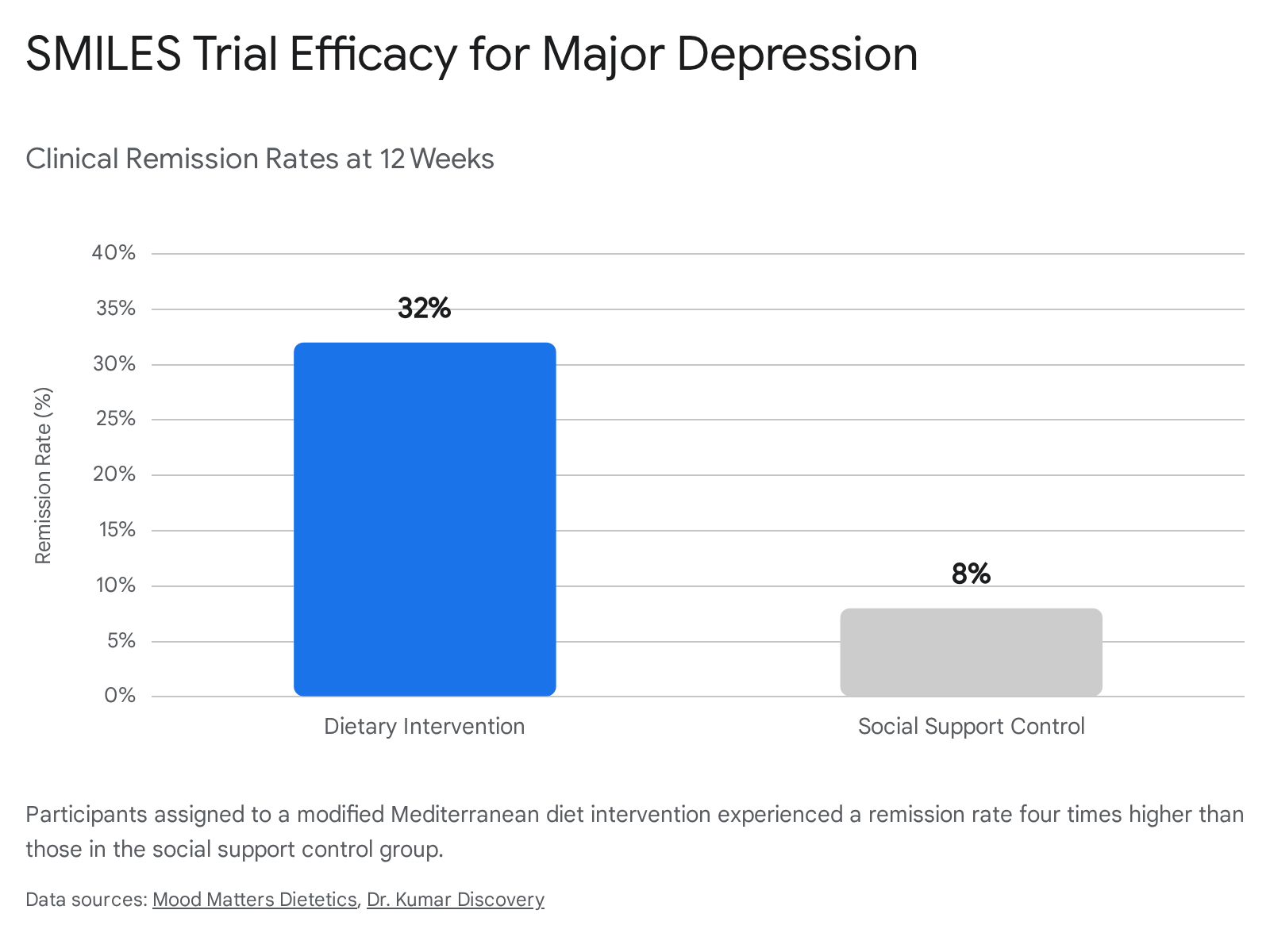
In this trial, 67 adults diagnosed with moderate to severe major depression were randomized to either a 12-week modified Mediterranean diet intervention (delivered by trained clinical dietitians) or a social support control group (a "befriending" protocol designed to match clinical contact time without providing dietary advice) 525051.
The results were clinically profound. The dietary intervention group demonstrated an average 7.1-point reduction on the clinician-rated Montgomery - Åsberg Depression Rating Scale (MADRS), compared to a 3.9-point reduction in the social support control group 52. Remarkably, 32% of participants in the diet group achieved full clinical remission of depression (defined as a MADRS score below 10), compared to only 8% in the control group 5251. This yielded a Number Needed to Treat (NNT) of 4.1, indicating that for every four patients treated with the dietary protocol, one achieved full remission 51.
Despite its groundbreaking outcomes, the SMILES trial has faced intense methodological scrutiny. Critics point out that the sample size was relatively small and that the profound effect size (Cohen's d of 1.2, vastly exceeding the typical effect sizes of SSRIs or CBT) may be artificially inflated by expectation bias and a failure of blinding 5052. Because participants were aware they were receiving a highly publicized "healthy diet," they may have expected to feel better. Furthermore, interviewer-rated improvements (MADRS) were significantly higher than self-reported improvements, suggesting that the clinical raters may have subconsciously deciphered treatment allocation 50. The authors of the SMILES trial acknowledge these limitations but maintain that a strong internal correlation between the degree of dietary adherence and the degree of symptom improvement supports a causal biological mechanism rather than mere placebo 4952.
Efficacy of Nutraceuticals and Isolated Supplements
Research evaluating specific nutritional supplements - or nutraceuticals - has yielded mixed and generally underwhelming results. Interventions utilizing isolated prebiotics and probiotics designed to target the gut-brain axis have demonstrated modest efficacy in enhancing specific cognitive domains, such as working memory and attention 4.
However, a 2025 scoping review noted that these single-agent interventions exhibit a "modest effect size" and generally fail to produce significant, durable improvements in core psychiatric symptoms 4. This reinforces a growing consensus in nutritional neuroscience: isolated nutrients act as marginal modulators, whereas broad-spectrum, whole-dietary patterns provide the synergistic substrates necessary for systemic psychobiological repair 78.
Methodological Limitations in Nutritional Psychiatry
The challenges in proving diet-mental health relationships stem from the inherent complexities of conducting nutritional RCTs, which differ vastly from traditional pharmacological trials 56.
Expectation Bias and Blinding Challenges
In a pharmaceutical trial, patients and clinical raters can be easily and effectively blinded using identical placebo pills. In nutritional psychiatry, true double-blinding is virtually impossible; participants invariably know what food they are eating, and preparing indistinguishable "placebo diets" over months is logistically unfeasible 64950. This lack of blinding introduces significant expectation bias, especially given widespread societal beliefs regarding "healthy eating."
Trial Design Standards and ISNPR Recommendations
To address these chronic methodological flaws, the International Society for Nutritional Psychiatry Research (ISNPR) published comprehensive clinical guidelines in 2024, detailing 49 specific recommendations for future dietary trials 6.
Key directives include utilizing active comparator groups (rather than passive waitlists) and matching expectancy and engagement between groups 6. The ISNPR strongly recommends prioritizing weight-neutral trial designs to successfully isolate the neurobiological effects of diet quality from the secondary psychological and physiological effects of weight loss 6. Furthermore, the guidelines mandate that dietary interventions be delivered exclusively by Accredited Practising Dietitians or Registered Dietitians, rather than general research staff, to ensure intervention fidelity and manage psychiatric comorbidities such as disordered eating 6.
Clarification on Non-Psychiatric Dietary Trials
It is vital to distinguish between trials designed to measure psychiatric outcomes and those measuring physical morbidity. For instance, the highly cited AMALFI trial evaluated a mail-based electrocardiogram (ECG) patch (Zio LTCM) for the remote screening of atrial fibrillation (AFib) in older adults 53545556. While cardiovascular health and mental health are deeply interconnected through inflammatory and vascular pathways, large-scale remote screening trials like AMALFI focus exclusively on cardiac diagnosis and anticoagulation exposure, providing no direct data on dietary intake or psychiatric morbidity 575859. The lack of similarly scaled, remotely deployed, and universally accessible diagnostic trials specifically for nutritional psychiatry highlights a significant infrastructure gap in the field.
The Future of Precision Nutrition
The future of nutritional psychiatry relies on moving past generic, population-level dietary guidelines toward "precision nutrition." Individuals possess highly varied metabolic phenotypes, genetic predispositions, and gut microbiome compositions, meaning that a dietary pattern beneficial for one patient may be biologically ineffective for another 16061.
Between 2025 and 2026, research in precision nutrition has accelerated rapidly through the integration of artificial intelligence (AI) and machine learning 6061. These technologies analyze vast, multidimensional datasets - including continuous glucose monitoring, individual microbiome profiles, and digital meal tracking - to generate highly personalized dietary interventions 606263.
Innovations such as "MealMeter" utilize multimodal sensing (heart rate, blood glucose, inertial motion) combined with machine learning to estimate real-time macronutrient intake, bypassing the notorious inaccuracy of self-reported food diaries 62. While preliminary trials indicate that AI-supported personalized nutrition can effectively manage physiological markers like postprandial glucose and lipid profiles, the data regarding long-term behavioral adherence and psychiatric outcomes remains firmly in a translational stage 6063.
Integration into Clinical Practice Guidelines
As the evidence base matures, global psychiatric bodies and medical associations are increasingly incorporating structured dietary recommendations into formal treatment algorithms. The integration of these lifestyle interventions marks a distinct paradigm shift from a purely biomedical approach to a comprehensive biopsychosocial-lifestyle model of psychiatric care 6465.
Table 2: Summary of Institutional Guidelines on Diet and Mental Health
| Institution / Body | Guideline Document | Clinical Stance on Dietary Intervention | Level of Evidence Cited |
|---|---|---|---|
| Royal Australian and New Zealand College of Psychiatrists (RANZCP) | Mood Disorders Clinical Practice Guidelines (2015, 2020 / 2024 Updates) 656667 | Diet and lifestyle modifications are categorized as foundational "actions" (essential step 1) and "choices" across the depressive spectrum 656768. | Evidence is integrated alongside pharmacotherapy, drawing heavily on major trials (e.g., PREDIMED, SMILES) 6568. |
| American Psychiatric Association (APA) | 2024 Position Updates / CME Directives 6970 | Highlights dietary interventions (specifically reducing UPFs and increasing whole foods) as having the strongest experimental evidence for the role of nutrition in mental health 69. | Acknowledges robust epidemiological and emerging trial data; emphasizes Mediterranean patterns 1269. |
| American College of Lifestyle Medicine (ACLM) | Expert Consensus Statement (2026) 4871 | Strongly endorses whole-food, plant-predominant dietary patterns for MDD. Explicitly advises against very low-carbohydrate/ketogenic diets due to perceived health risks 4871. | Consensus based on review of 71 statements; cites favorable 'Number Needed to Treat' for nutrition vs. SSRIs 48. |
The RANZCP was the first major international body to formally mandate lifestyle and dietary interventions as a non-negotiable first step (classified as "Actions") in the clinical management of mood disorders 6567. Similarly, the APA now emphasizes that psychiatrists and allied clinicians do not need to be specialized dietitians to assess a patient's diet; rather, they should confidently encourage shifts away from ultra-processed foods toward Mediterranean-style eating as a standard component of care 6970.
However, institutional guidelines remain sharply divided regarding metabolic and extreme diets. While metabolic psychiatry researchers and clinical pioneers advocate for the use of therapeutic ketogenic diets based on strong physiological rationales (e.g., mitochondrial bypass, inflammation reduction) 20, broader institutions like the ACLM formally advise against low-carbohydrate diets 4871. This institutional resistance is primarily due to historically perceived cardiovascular risks and a lack of longitudinal psychiatric safety data 4871. This significant discrepancy highlights the critical need for well-powered, long-term RCTs specifically comparing divergent dietary patterns head-to-head.
Conclusions
Science has definitively established that dietary patterns profoundly influence mental health. The elucidated neurobiological mechanisms - spanning the microbiota-gut-brain axis, systemic inflammation, tryptophan metabolism, and mitochondrial energy dynamics - provide a robust physiological basis for the observed clinical phenomena.
Epidemiological consensus is virtually unanimous that ultra-processed foods significantly elevate the risk of psychiatric disorders. Conversely, whole-food dietary patterns, particularly the Mediterranean diet and diverse traditional agrarian diets, confer substantial protective benefits. Clinical trials, notably the SMILES trial, demonstrate that targeted, dietitian-led dietary improvements can yield remission rates in clinical depression that rival or exceed traditional pharmacotherapy.
However, the evidence falls short in several critical domains. First, the inherent methodological limitations of dietary RCTs - such as the impossibility of blinding, pervasive expectation bias, and small sample sizes - temper the certainty of causal claims, particularly regarding anxiety disorders, where the data remains persistently inconclusive. Second, extreme dietary interventions like the ketogenic and carnivore diets are characterized by a severe mismatch between enthusiastic anecdotal reports and rigorous, long-term clinical safety data. Finally, while the field is rapidly advancing toward AI-driven precision nutrition, clinical application remains constrained by technological and translational bottlenecks.
As major clinical guidelines officially begin to mandate nutritional assessment as a standard of psychiatric care, the future of the field requires large-scale, methodologically rigorous trials that move beyond generic dietary advice toward personalized, metabolically informed interventions.