Scientific assessment of longevity escape velocity
Longevity escape velocity, occasionally referred to as actuarial escape velocity or biological escape velocity, represents a theoretical demographic and biotechnological threshold. The concept posits a future scenario wherein biomedical interventions can extend an individual's remaining life expectancy at a rate faster than chronological time passes 12. Formally conceptualized by David Gobel and biogerontologist Aubrey de Grey in a 2004 paper, though present in futurist literature such as Robert Anton Wilson's writings in the 1970s, the premise relies on a mathematical analogy to physical escape velocity 2. Just as an object must achieve a specific minimum speed to break free from a planetary body's gravitational pull, longevity escape velocity suggests that medical science must advance at a specific, compounding pace to outstrip the biological accumulation of age-related cellular damage 23.
The coherence of this concept is intrinsically tied to the biological feasibility of the Strategies for Engineered Negligible Senescence (SENS), a framework proposed by de Grey that categorizes biological aging into seven distinct types of molecular and cellular damage. SENS posits that the targeted, periodic repair of these damage classes can indefinitely sustain human health and lifespan, theoretically enabling longevity escape velocity 456. Evaluating the scientific coherence of Aubrey de Grey's concept requires a rigorous examination of the mathematical and actuarial models underlying mortality, the clinical progress of SENS-aligned therapeutics as of 2026, the contrast between SENS and the widely accepted Hallmarks of Aging, and the biological and evolutionary constraints that challenge the plausibility of radical life extension.
Actuarial and Demographic Foundations
The theoretical framework for longevity escape velocity is grounded in the mathematics of population mortality and the dynamics of adult life expectancy. Proponents of the concept emphasize remaining life expectancy rather than life expectancy at birth, as infant and childhood mortality improvements have largely exhausted their capacity to mathematically boost population-wide lifespans in high-income nations 127.
The age-specific death rate in adult human populations is most commonly modeled using the Gompertz-Makeham law of mortality. Within this model, the age-specific death rate $\mu(a)$ can be approximated by the equation $\mu(a) \approx B + G e^{\gamma a}$, where $B$ represents the background, age-independent mortality rate, $G$ serves as a baseline scaling factor, and $\gamma$ represents the exponential rate of aging 1. In contemporary human populations, $\gamma$ is approximately 0.085 per year, reflecting a biological reality where mortality risk roughly doubles every eight years following the age of 30 13.
To achieve longevity escape velocity, biomedical interventions must compress the rate of aging and reduce the age-specific mortality risk $\mu(a, t)$ rapidly enough to offset chronological decay. Mathematically, escape velocity is achieved when the partial derivative of remaining life expectancy $e(a, t)$ with respect to chronological time $t$ exceeds unity: $\frac{\partial e}{\partial t} > 1$ 1. Under this condition, for every calendar year an individual survives, their remaining life expectancy increases by more than one year 12.
De Grey has calculated that reaching this actuarial threshold requires a sustained, annual reduction in mortality rates across mid-to-late adult ages of approximately ten percent 148. If such a reduction is achieved and maintained indefinitely, the result is a geometric expansion of life expectancy. De Grey theorized that under a continuous ten percent annual mortality reduction, the first individual to reach 1,000 years of age would likely be only a decade or two younger than the first individual to reach 150 years of age. This is because the rapid compounding of rejuvenation therapies would increasingly distance survivors from the Gompertzian force of biological frailty, granting them sufficient time to benefit from second- and third-generation medical advancements 248.
Demographers further analyze this dynamic through the lenses of lifetable entropy and lifespan disparity. Entropy equations, such as $H = e_1 / e_0$, capture the relative magnitude of baseline mortality levels against the exponential rate of aging 9. The realization of longevity escape velocity would manifest mathematically as a continuous, unprecedented compression of mortality followed by a perpetual rightward shift of the survival curve 910. Despite the rigorous mathematical framework, the timeline for achieving such velocity remains highly speculative. Futurist Ray Kurzweil has projected that humanity will reach longevity escape velocity between 2029 and 2035, aided by artificial intelligence simulating biological processes, while de Grey has estimated a fifty percent probability that humanity will cross this threshold by the mid-to-late 2030s 1211.
The SENS Framework
Aubrey de Grey's proposition for reaching longevity escape velocity relies entirely on shifting the gerontological focus away from metabolic manipulation toward comprehensive damage repair 46. This engineering paradigm argues that cellular metabolism is vastly too complex to fundamentally alter without inducing catastrophic side effects. However, the resulting structural damage of that metabolism is finite, categorizable, and potentially reversible 410.
The SENS framework categorizes age-related degeneration into seven primary classes of damage, pairing each with a theoretical, biotechnology-driven intervention strategy 4613. Evaluating the coherence of de Grey's concept requires assessing the clinical and preclinical progress of these seven pillars.
Cell Loss and Tissue Atrophy
The RepleniSENS category addresses the gradual loss of cells and tissue atrophy that accompanies aging, proposing stem cell therapies and tissue engineering to restore depleted cell populations 611. This domain aligns closely with mainstream regenerative medicine and has seen substantial clinical advancement. Phase II clinical trials of mesenchymal stem cells have demonstrated functional improvements in elderly patients suffering from frailty. For instance, Longeveron's Lomecel-B mesenchymal stem cell infusions have shown measurable improvements in walking distance and reductions in inflammatory markers in older adults, providing proof-of-concept that exogenous cellular reinforcements can partially ameliorate age-related physical decline 611. Research into the regeneration of the thymus - a critical organ for immune function that normally involutes with age - represents a highly pursued goal within this category, with early clinical data suggesting that pharmacological and cellular interventions can partially restore youthful immune profiles 11.
Senescence and Death-Resistant Cells
ApoptoSENS targets senescent cells and other death-resistant cellular populations. Cellular senescence occurs when cells permanently exit the cell cycle in response to sublethal genotoxic stress, yet remain metabolically active 1516. According to the threshold model of autophagy, below a critical damage threshold, robust autophagic flux suppresses senescence by maintaining mitochondrial integrity. However, once this threshold is exceeded, autophagy is functionally reprogrammed to sustain the massive biosynthetic demands of senescent cells 17. These cells subsequently secrete a highly pro-inflammatory cocktail known as the senescence-associated secretory phenotype (SASP), driven by the activation of cGAS-STING and NF-κB innate immune pathways 1712.
The targeting of cellular senescence has become a cornerstone of biogerontology. Senolytics - pharmacological agents that selectively induce apoptosis in dysfunctional senescent cells - have advanced rapidly. The combination of dasatinib and quercetin (D+Q), as well as flavonoids like fisetin, has shown efficacy in reducing SASP inflammation in clinical models of lung and kidney disease 615. The precision of senolytic targeting advanced significantly in 2025 with the development of SenSCOUT, an integrated single-cell imaging and machine learning framework introduced by the National Institutes of Health. By analyzing over 50,000 single cells, researchers identified 11 distinct fibroblast subtypes, pinpointing specific morphologies (C7, C10, and C11) that score highest for pathological senescence 13. This degree of high-resolution mapping is critical, as aggressive, non-specific senolytic clearance can impair localized tissue repair or accelerate stem cell exhaustion if the stem cell niche is continually forced to regenerate cleared tissue 11220.
Intracellular Aggregates
LysoSENS is designed to address the accumulation of indigestible intracellular waste, such as lipofuscin, which congregates within the lysosome. Over decades, this stubborn waste causes lysosomes to swell, diluting their hydrolytic enzymes, crowding out other organelles, and triggering inflammatory cascades when the lysosomal membrane becomes damaged 21. The original SENS strategy proposed mining the metagenomes of soil bacteria to identify exogenous hydrolases capable of breaking down these aggregates, which would then be introduced into human cells 61122.
However, recent clinical translation has shifted toward harnessing and amplifying endogenous lysosomal biology. In May 2026, Lysoway Therapeutics initiated a Phase I clinical trial in Melbourne, Australia, for LW-1017, a highly brain-penetrant, small-molecule TRPML1 agonist 24. TRPML1 is a vital lysosomal ion channel regulating the autophagy-lysosomal pathway; its activation represents a differentiated approach to restoring cellular clearance systems impaired in neurodegenerative diseases like Alzheimer's and Parkinson's . Concurrently, targeted protein degraders (PROTACs) have expanded into lysosomal pathways. Arvinas reported first-quarter 2026 Phase 1 clinical data for ARV-102, an oral PROTAC LRRK2 degrader, demonstrating greater than fifty percent degradation of LRRK2 in the cerebrospinal fluid of Parkinson's patients, coupled with a notable reduction in endolysosomal and neuroinflammatory biomarkers such as CD68 and GPNMB .
Extracellular Aggregates
The AmyloSENS category addresses misfolded proteins and aggregates in the extracellular space, most notably amyloid-beta and pathological tau proteins associated with neurodegeneration 61415. The SENS paradigm historically advocated for immunotherapy and catalytic antibodies (catabodies) to clear these aggregates 2215. This approach has merged heavily with mainstream neuroscience, though clinical outcomes for amyloid-clearing monoclonal antibodies have historically yielded mixed results regarding cognitive restoration 1416.
A highly promising evolution in extracellular clearance strategies utilizes small extracellular vesicles (sEVs) and exosomes. Exosomes, ranging from 30 to 150 nanometers in diameter, are natural intercellular messengers that can be engineered to cross the blood-brain barrier and deliver therapeutic cargo, including siRNAs, miRNAs, or autophagic promoters 1718. By 2025 and 2026, exosome-based clinical pipelines matured significantly. Regulatory advancements, such as the FDA clearance of Investigational New Drug (IND) applications for products like ExoFlo for inflammatory indications and AGLE-102 for tissue repair, demonstrate the clinical viability of utilizing these nanometer-scale particles to mediate extracellular environment repair 1819. Furthermore, research led by the University of Ottawa in 2026 demonstrated that specific sEVs naturally target specific organ tissues, allowing for highly precise delivery of aggregate-clearing gene therapies without relying on synthetic nanoparticles 20.
Extracellular Crosslinks
GlycoSENS targets advanced glycation end-products (AGEs) that form pathological crosslinks between long-lived extracellular matrix proteins, primarily collagen and elastin 62122. As tissues age, these crosslinks increase the mechanical stiffness of blood vessels, impairing renal and cardiac function, and hardening the extracellular matrix against natural turnover processes 21. Glucosepane is the most prevalent and problematic crosslink in aged human tissues 2135.
Early pharmacological agents, such as alagebrium (a thiazolium halide), proved largely ineffective because they were only capable of cleaving a minor subset of sugar-derived alpha-diketone bridges, leaving the robust glucosepane structures intact 21. The GlycoSENS strategy, heavily reliant on philanthropic funding, successfully synthesized glucosepane in the laboratory to allow for the identification of bacterial enzymes capable of cleaving it 1122. As of late 2025, Revel Pharmaceuticals reported success in identifying lead candidate bacterial enzymes and small-molecule breakers that cleaved up to forty percent of glucosepane crosslinks in ex vivo assays using aged human dermal fibroblasts 611. Despite this in vitro progress, in vivo translation remains a severe bottleneck. The dense extracellular matrix presents a formidable physical barrier, and it remains uncertain whether engineered enzymes can be delivered effectively throughout the human vasculature without inducing toxicity or compromising the structural integrity of healthy collagen 61621.
Mitochondrial DNA Mutations
MitoSENS addresses the age-related accumulation of mutations and deletions within mitochondrial DNA (mtDNA) 61336. Because mitochondria reside outside the cell nucleus, they lack robust DNA repair mechanisms and are highly susceptible to oxidative damage 15. Rather than attempting to improve mitochondrial repair systems, the SENS engineering approach proposes allotopic expression: engineering backup copies of the 13 protein-encoding mitochondrial genes, integrating them into the safety of the nuclear genome, and ensuring the synthesized proteins are successfully targeted and imported back into the mitochondria 3523.
While utilizing allotopic expression as a widespread anti-aging intervention remains confined to preclinical laboratory settings, the fundamental concept has achieved clinical validation in the treatment of rare monogenic disorders. Gene therapies such as rAAV2-ND4 utilize allotopic expression to treat Leber Hereditary Optic Neuropathy (LHON), demonstrating that transferring mitochondrial gene functions to the nucleus is biologically executable in humans 3524. Parallel engineering approaches are also advancing; researchers at Texas A&M University have explored the use of inorganic molybdenum disulfide nanoflowers to stimulate massive mitochondrial biogenesis within stem cells, generating energized cells that can transfer healthy mitochondria to aging tissues .
Nuclear Mutations and Epimutations
OncoSENS addresses the accumulation of nuclear mutations, which de Grey views primarily through the lens of oncogenic risk 635. Because accumulated DNA damage allows cancer cells to escape normal proliferative limits, SENS proposes a highly radical preventative protocol: Whole-body Interdiction of Lengthening Telomeres (WILT) 615. The original WILT concept requires the genetic knockout of telomerase and Alternative Lengthening of Telomeres (ALT) mechanisms in all somatic cells. This would impose a strict replicative limit on all cells, theoretically rendering lethal tumor overgrowth impossible, but it would require the continuous, whole-body reseeding of engineered stem cells to maintain tissue renewal 611.
Due to the insurmountable complexity and risk of whole-body telomerase ablation, WILT has never advanced to human trials 611. De Grey has subsequently pivoted toward a modified approach colloquially termed "WILT 2.0," which utilizes suicide-gene therapies like 6-thio-2-deoxyguanosine (THIO). Rather than preventing telomere lengthening universally, THIO turns telomerase expression into a suicide trigger, rapidly selectively killing highly proliferative cancer cells before they can divide into lethal tumors 16.
Comparative Analysis: SENS Versus The Hallmarks of Aging
While the SENS framework was highly influential in the early 2000s, mainstream biogerontology largely organized itself around the "Hallmarks of Aging" framework, originally published in a landmark 2013 paper by López-Otín et al., and subsequently expanded in a 2023 update in the journal Cell 10152526. The updated framework defines twelve interconnected hallmarks: genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, disabled macroautophagy, deregulated nutrient-sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, altered intercellular communication, chronic inflammation, and dysbiosis 152526.
The division between the Hallmarks of Aging (HoA) and SENS is deeply philosophical, rooted in divergent epistemological and teleological approaches to human biology 510. The HoA framework is primarily descriptive; it seeks to map the fundamental, interconnected biological mechanisms driving senescence to optimize biological function and extend healthspan 51027. SENS, conversely, is strictly prescriptive and interventionist. It views the underlying mechanisms of aging as largely irrelevant, provided the resulting structural damage can be repaired or bypassed to radically prolong lifespan 510.
A direct conceptual mapping reveals where the SENS engineering approach intersects with established biological mechanisms, and highlights critical areas omitted by de Grey's original theories.
| Hallmarks of Aging (2023 Update) | Corresponding SENS Category | Conceptual Overlap and Distinction |
|---|---|---|
| Cellular Senescence / Chronic Inflammation | ApoptoSENS | Both frameworks identify senescent cell accumulation as heavily pathogenic. SENS focuses strictly on targeted ablation (senolytics), whereas HoA also explores SASP modulation and immune communication. |
| Loss of Proteostasis / Disabled Macroautophagy | LysoSENS / AmyloSENS | HoA investigates the failure of protein folding and recycling mechanisms. SENS ignores the failure mechanisms to focus on exogenous enzymatic degradation of the resulting aggregates. |
| Mitochondrial Dysfunction | MitoSENS | HoA investigates reactive oxygen species production and metabolic failure. SENS focuses exclusively on bypassing the dysfunction via nuclear allotopic expression. |
| Stem Cell Exhaustion | RepleniSENS | Highly aligned. Both frameworks advocate for cellular replacement, stem cell therapies, and regenerative medicine to treat tissue atrophy. |
| Genomic Instability / Telomere Attrition | OncoSENS | HoA views these as primary causes of widespread cellular damage. SENS views nuclear mutations predominantly as a cancer risk, addressable via telomerase interdiction. |
| Epigenetic Alterations / Dysbiosis / Deregulated Nutrient-Sensing | No direct SENS equivalent | These hallmarks lack explicit SENS engineering interventions. SENS historically assumed epigenetic drift was a downstream consequence of structural damage, rather than a primary driver of aging. |
The Epigenetic Paradigm Shift
The most profound theoretical challenge to the SENS framework stems from its historical omission of epigenetic alterations. De Grey originally hypothesized that epimutations - changes in gene expression regulation that do not alter the underlying DNA sequence - were secondary, downstream effects of explicit structural damage, and therefore did not require a dedicated pillar of intervention 3527.
However, the advent of partial epigenetic reprogramming has forced a paradigm shift in biogerontology. The discovery that the transient expression of Yamanaka transcription factors (OCT4, SOX2, KLF4, and c-MYC) can restore youthful epigenetic methylation patterns without erasing cellular identity suggests that cellular aging can be reversed purely through regulatory reprogramming, independent of localized structural repair 284429.
By 2026, this concept moved from theory to regulated clinical testing. The U.S. Food and Drug Administration cleared the Investigational New Drug (IND) application for Life Biosciences' ER-100 443031. Originating from Harvard researcher David Sinclair's Information Theory of Aging, ER-100 is an OSK-based partial epigenetic reprogramming therapy designed to restore vision in patients suffering from non-arteritic anterior ischemic optic neuropathy (NAION) and open-angle glaucoma 443031. Initiating Phase 1 human trials in early 2026, ER-100 represents the first clinical attempt at targeted cellular rejuvenation using epigenetic reprogramming 4430. While partial reprogramming operates via genomic regulation rather than SENS-style structural repair, proponents argue it achieves the ultimate objective of reversing age-related decline, demonstrating that repairing discrete categories of physical "garbage" may not be the sole pathway to rejuvenation 4429.
Demographic Critiques of Radical Life Extension
Despite the rapid acceleration of specific biotechnological interventions, longevity escape velocity relies heavily on the assumption that medical progress will translate directly into exponential gains in aggregate human survival. Sweeping actuarial and demographic analyses published in 2024 and 2025 heavily critique this assumption, providing empirical evidence that challenges the plausibility of achieving escape velocity in the near future.
A landmark 2024 study published in Nature Aging by S. Jay Olshansky et al. analyzed mortality data from 1990 to 2019 across the world's longest-lived populations (including Japan, South Korea, France, and Spain) 323334. The data revealed a sharp deceleration in life expectancy gains 323334. Olshansky concluded that the radical life extension witnessed over the 20th century was a unique demographic anomaly, driven almost entirely by the elimination of early-life mortality and infectious diseases 323536. Because mortality reduction has now shifted to older ages, where biological frailty and concurrent multi-organ failure dominate, medicine is encountering a point of diminishing returns. The study asserts that modern medicine is yielding incrementally smaller improvements in longevity despite occurring at "breakneck speed" 333537.
Olshansky's actuarial models project that without a fundamental intervention that slows the underlying biological clock, survival to age 100 will cap at approximately fifteen percent for women and five percent for men 333436. This effectively cements a biological "glass ceiling" on human lifespan somewhere between 85 and 90 years 3235.
This decelerating trend was further corroborated by Héctor Pifarré i Arolas, José Andrade, and Carlo Giovanni Camarda in a comprehensive 2025/2026 study published in the Proceedings of the National Academy of Sciences (PNAS). Utilizing data from the Human Mortality Database across 23 high-income countries, the researchers demonstrated that the rate of life expectancy gains per generation has plummeted drastically 738.
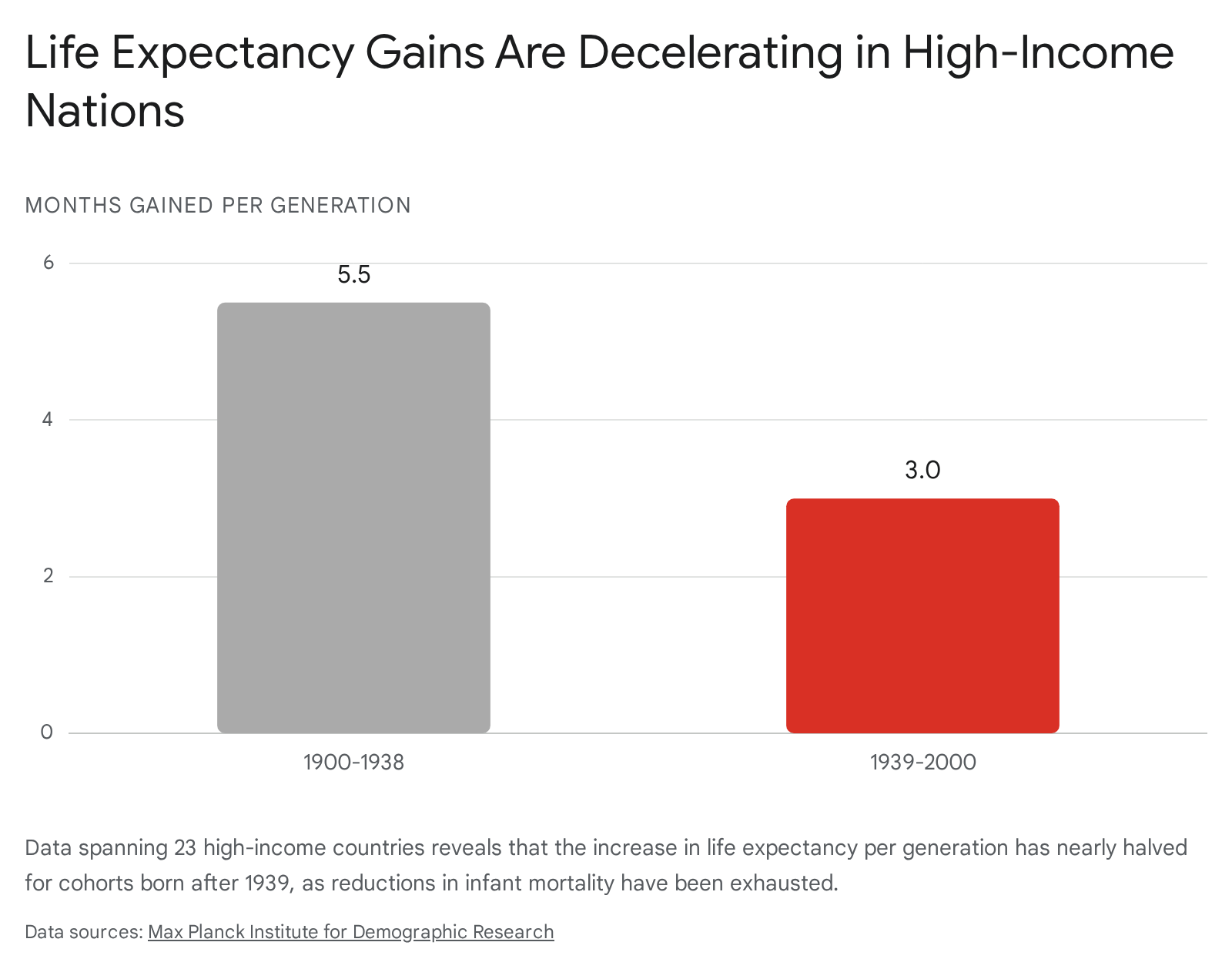
The PNAS analysis asserts that even if adult survival rates were to miraculously improve at twice the rate currently predicted, it would still be insufficient to match the rapid increases achieved in the early 20th century 73839. The authors unequivocally project that no generation born after 1939 will reach an average lifespan of 100 years, rendering longevity escape velocity statistically highly improbable under current therapeutic paradigms 73839. While researchers such as Dmitri Jdanov and Domantas Jasilionis at the Max Planck Institute for Demographic Research argue that optimistic alternative scenarios involving future breakthroughs should not be entirely ignored, the prevailing demographic consensus points toward a firm compression of late-life mortality 40.
Evolutionary Constraints and Systemic Complexity
Beyond actuarial deceleration, the scientific coherence of achieving longevity escape velocity faces profound scrutiny from evolutionary biology. Critics argue that achieving indefinite lifespan extension requires overcoming deeply entrenched biological constraints governed by evolutionary trade-offs, primarily antagonistic pleiotropy.
The duality hypothesis and antagonistic pleiotropy posit that alleles conferring high developmental and reproductive fitness during an organism's early life become systemic liabilities in post-reproductive life, as natural selection lacks the mechanisms to optimize for macroevolutionary timeframes beyond reproductive age 1541. The SENS framework treats the seven forms of biological damage as independent engineering targets. However, empirical biological data reveals that these systems are profoundly interconnected 127.
For example, extracellular matrix stiffening (GlycoSENS) drives intracellular mechanical stress that directly exacerbates mitochondrial dysfunction (MitoSENS). This dysfunction in turn produces reactive oxygen species that accelerate telomere attrition (OncoSENS) 11027. Consequently, interventions targeting isolated damage classes often trigger cascading failures elsewhere. A highly aggressive senolytic clearance protocol, while successfully reducing SASP-related inflammation, may inadvertently impair localized tissue repair or accelerate stem cell exhaustion if the stem cell niche is repeatedly forced to undergo regenerative division to replace the cleared tissues 11720.
Furthermore, the physical limitations of somatic cells present a formidable barrier. The Hayflick limit governs the finite replicative capacity of human cells, generally permitting 40 to 60 divisions before inducing senescence due to telomere shortening 111. Attempting to circumvent this limit via SENS protocols like whole-body telomerase ablation requires an impossible degree of flawlessly calibrated cellular replacement, demanding trillions of engineered stem cells to be integrated without provoking immune rejection, fibrosis, or multi-organ failure 16. Biologists contend that the "engineer's paradigm" driving the SENS framework severely oversimplifies the systemic complexity, feedback loops, and homeostasis required to sustain a living biological organism 156.
Conclusion
Longevity escape velocity remains a mathematically elegant concept that clearly defines the exact rate of mortality reduction required to achieve indefinite human lifespan extension. Aubrey de Grey's SENS framework provides a conceptually coherent, albeit highly reductionist, engineering roadmap to reach this velocity by prioritizing the targeted clearance and repair of accumulated damage over the infinitely complex alteration of fundamental metabolism.
As of 2026, specific pillars of SENS - most notably senolytics, lysosomal clearance agonists, and emerging epigenetic reprogramming therapies - have successfully transitioned from radical theory to regulated human clinical trials, proving that the reversal of discrete elements of biological aging is technologically feasible. However, sweeping demographic evidence confirms that overall human life expectancy gains are currently decelerating, encountering a firm biological ceiling governed by the interconnected frailty of old age. Until the isolated victories in discrete damage repair can be safely synthesized into a systemic, whole-body intervention that overrides evolutionary pleiotropy without triggering compensatory biological failure, longevity escape velocity remains a mathematically speculative horizon rather than an imminent clinical reality.