Science of Phobia Development and Exposure Therapy
Clinical Classification of Phobias
The demarcation between an evolutionary, adaptive fear response and a pathological phobia rests on the disproportionate nature of the anxiety relative to the actual threat, alongside the resultant functional impairment in social, occupational, or personal domains. Diagnostic frameworks have evolved considerably over consecutive iterations to improve inter-rater reliability, diagnostic precision, and cross-cultural applicability.
Diagnostic Evolution and Thresholds
The transition from the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) to the Fifth Edition (DSM-5) and its subsequent Text Revision (DSM-5-TR) reflects a crucial conceptual shift. The DSM-IV defined specific phobias as being characterized by a "marked and persistent fear that is excessive or unreasonable," requiring the patient to personally recognize the irrationality of their fear 12. In contrast, the DSM-5 and DSM-5-TR redefined this criterion to require "marked (intense) fear or anxiety" that is explicitly "out of proportion to the actual danger posed" 123. This modification removes the reliance on the patient's self-recognition, which is often distorted, and substitutes it with a clinical judgment of proportionality based on the sociocultural context 24.
Historically, vague terminology like "marked" and "persistent" led to reliability problems. By operationalizing these terms, the DSM-IV achieved significant improvements in diagnostic agreement, reporting a kappa of 0.86 for principal specific phobia diagnoses 14. Furthermore, the DSM-5-TR mandates a minimum symptom duration of six months across all age groups, standardizing a requirement that was previously restricted to individuals under the age of 18 in the DSM-IV 12.
The International Classification of Diseases, Eleventh Revision (ICD-11), aligns closely with the DSM-5-TR by emphasizing marked fear that is disproportionate to the actual danger, combined with consistent avoidance or endurance under intense distress 1. However, the two manuals diverge slightly on structural granularity. The DSM-5-TR formally retains five explicit subtype classifications - animal, natural environment, blood-injection-injury, situational, and other - due to distinct associations with severity and psychiatric comorbidity 14. The ICD-11 provides less explicit guidance on subtyping, prioritizing global applicability over granular descriptive categories 14.
| Diagnostic Criterion | DSM-IV Framework | DSM-5-TR Framework | ICD-11 Framework |
|---|---|---|---|
| Nature of Fear | Marked and persistent; required patient recognition of the fear as excessive. | Marked (intense) fear or anxiety; requires clinical judgment that fear is out of proportion to actual danger. | Marked and excessive fear or anxiety; out of proportion to actual danger. |
| Exposure Response | Almost invariably provokes an immediate anxiety response (often panic). | Almost always provokes immediate fear or anxiety. | Consistent provocation of intense fear or anxiety upon exposure. |
| Duration Requirement | At least 6 months (only mandated for individuals under 18). | Typically lasting for 6 months or more (applied to all age cohorts). | Lasting for several months, indicating persistence. |
| Subtype Specificity | Five explicit subtypes defined. | Five explicit subtypes retained due to differing comorbidities and onset ages. | Broader classification; less explicit subtyping guidance provided. |
Cultural Manifestations and Self-Construal
The sociocultural context of fear assessment is critical, as phobias can manifest uniquely across different populations due to varying models of self-construal 457. A prominent example of culturally bound phenotypic expression is Taijin Kyofusho (TKS), a specific expression of social anxiety widely documented in Japanese and Korean cultures since the 1920s 4567.
Unlike typical Western manifestations of social phobia, which center on the individual's fear of embarrassing themselves or facing negative evaluation, TKS is characterized by an intense fear that one's body parts, physical functions, blushing, or odors will displease, embarrass, or offend others 4568. The Japanese diagnostic system classifies TKS into four primary subtypes: sekimen-kyofu (fear of blushing), shubo-kyofu (fear of a deformed body), jikoshisen-kyofu (fear of eye-to-eye contact), and jikoshu-kyofu (fear of one's own foul body odor) 46.
This distinction highlights the profound influence of interdependent versus independent self-construals on the phenotypic expression of underlying anxiety genotypes 45. In highly collectivistic societies, the potential disruption of group harmony and the fear of bringing shame upon a social or familial unit shape the presentation of the anxiety disorder 68. To improve global diagnostic validity, the DSM-5 expanded its wording regarding the fear of negative evaluation to better encompass the unique allocentric concerns of patients exhibiting TKS 4.
Neurobiology of Fear Generation and Persistence
The acquisition, expression, and suppression of fear rely on a highly conserved neural network encompassing the amygdala, the ventromedial prefrontal cortex (vmPFC), and the hippocampus 9101112. The structural and functional dysregulation of these interconnected regions underpins the persistent fear responses observed in specific phobias and post-traumatic stress disorder (PTSD).
The Amygdaloid Complex and Threat Detection
The amygdala serves as the central hub for threat detection and the orchestration of both innate and conditioned fear 121314. During Pavlovian fear conditioning, sensory information regarding a benign conditioned stimulus (CS) and an aversive unconditioned stimulus (US) converges on the basolateral amygdala (BLA), which consists of the lateral and basal nuclei 1115. Through localized synaptic plasticity - such as the strengthening of long-term potentiation - the BLA encodes the associative memory linking the CS to the US 91114.
Upon subsequent exposure to the CS alone, the BLA drives downstream physiological responses via strong excitatory projections to the central amygdala (CeA) 11. The CeA acts as the primary output node, projecting directly to targets in the brainstem, periaqueductal gray, and hypothalamus to initiate autonomic responses, freezing, and avoidance behaviors 111315. In individuals with specific phobias and PTSD, functional neuroimaging consistently reveals amygdala hyperresponsivity when the subject is confronted with disorder-relevant stimuli 12141617. This sensitization mechanism effectively lowers the threshold for threat detection, leading to an exaggerated fear expression that resists natural habituation 14.
Prefrontal Regulation and Extinction
The medial prefrontal cortex (mPFC), specifically the ventromedial prefrontal cortex (vmPFC) in humans, provides critical top-down inhibitory control over the amygdaloid complex 91112. In rodent models, the mPFC is functionally subdivided into the prelimbic (PL) and infralimbic (IL) cortices, which execute opposing regulatory roles 1015. The PL region generally promotes fear expression by sending excitatory projections to the BLA 101518. Conversely, the IL region is vital for the consolidation, retention, and retrieval of fear extinction memory 10111518.
The vmPFC (and its rodent IL homolog) achieves fear inhibition by exciting clusters of GABAergic intercalated cells (ITCs) located between the BLA and the CeA. Once activated by the vmPFC, these ITCs send strong inhibitory projections to the CeA, effectively dampening the behavioral and autonomic expression of fear 111519.
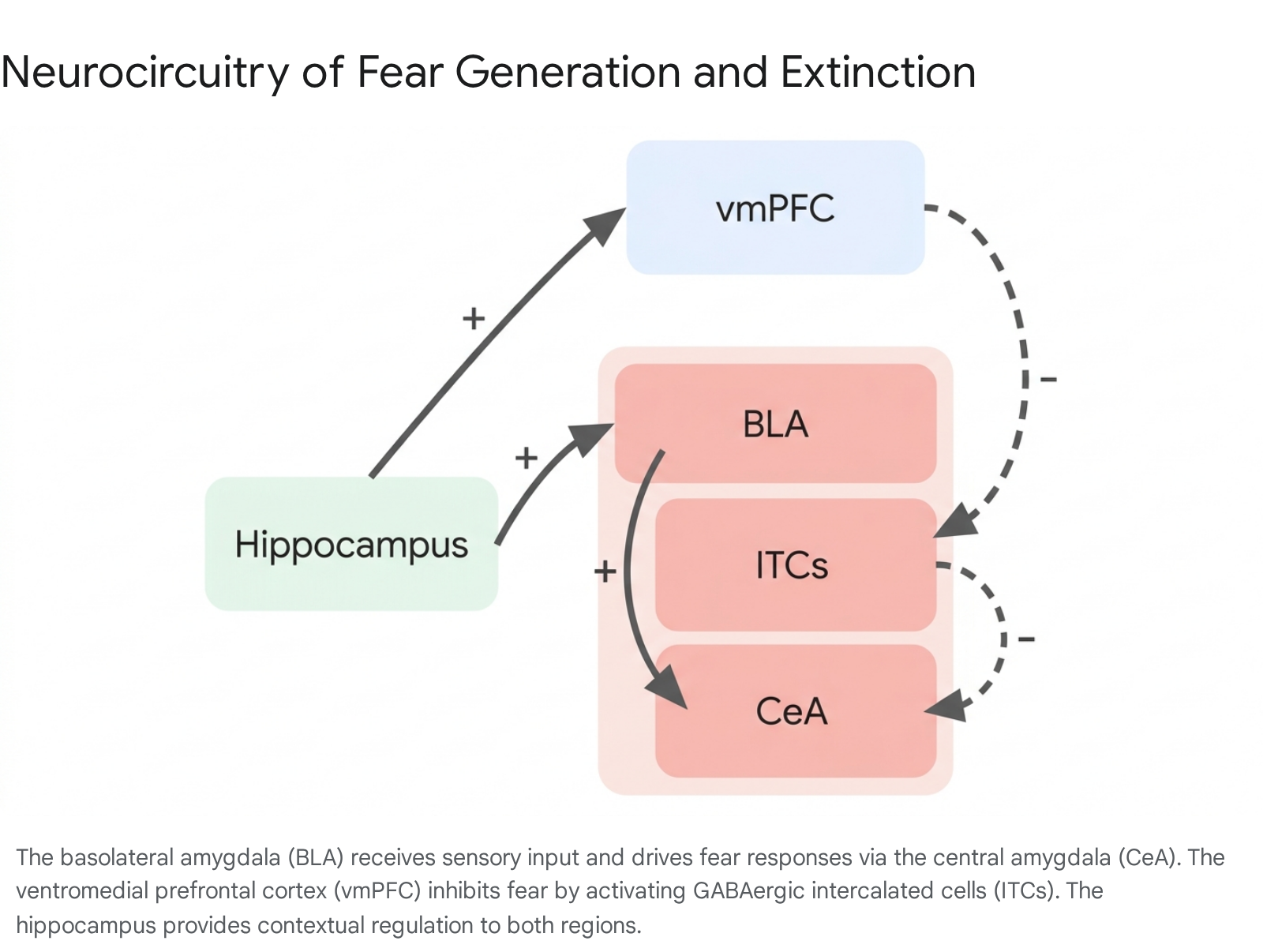
Pathological anxiety states are frequently marked by a hyporesponsive vmPFC, which fails to recruit the ITCs and thus cannot provide adequate inhibitory signaling to the hyperactive amygdala 1217. This regional imbalance severely impairs emotion regulation, threat evaluation, and the capacity for fear extinction 1218.
Hippocampal Contextual Control
The hippocampus - specifically its ventral aspect in rodents (vHPC) and the anterior region in humans - is responsible for encoding the spatial and contextual parameters within which fear conditioning and extinction occur 9101518. Fear extinction is highly context-dependent; a stimulus deemed safe in a therapeutic setting may still provoke terror in a novel environment. The hippocampus modulates this behavior by signaling whether it is appropriate to express fear or safety based on the surrounding environment, doing so through direct projections to the BLA and indirect projections via the mPFC 91011.
Recent neurobiological mapping reveals that fear renewal (the return of an extinguished fear outside the therapeutic context) relies on a distributed network. Presentation of an extinguished fear cue in a non-extinction context activates BLA-projecting neurons in both the ventral hippocampus and the prelimbic cortex 10. Furthermore, the vHPC actively suppresses the infralimbic cortex through a mechanism of feed-forward inhibition 20. Ventral hippocampal projections recruit parvalbumin-expressing interneurons within the IL cortex. This local inhibition suppresses the activity of amygdala-projecting pyramidal neurons in the IL, countering the expression of extinguished fear and directly promoting fear relapse 20.
Synaptic Mechanisms and Neural Ensembles
At the cellular level, the retention of long-term contextual fear memory requires intact perineuronal nets (PNNs) within the CA1 region of the hippocampus 21. Experimental removal of these extracellular matrix structures reduces the firing rates of parvalbumin neurons and severely impairs long-term memory retention without affecting short-term recall 21. The actual memories are stored within specific neural ensembles, or engram cells. Memory updating alters original fear engram encoding, increasing the functional overlap between fear-associated and extinction-associated cell populations within the prelimbic cortex and the basolateral amygdala 2122.
Additionally, fear generalization - where fear responses spread to novel stimuli resembling the original conditioned danger cue - demonstrates distinct neural topography. Positive generalization (increasing fear to similar cues) recruits cingulo-opercular, frontoparietal, and midbrain regions, including the locus coeruleus and periaqueductal gray 23. Negative generalization (recognizing safety as stimuli differ) is mediated by default-mode network nodes, relying heavily on the vmPFC and the hippocampus to perform pattern separation and activate fear inhibition pathways 23.
Mechanisms of Exposure Therapy
Exposure therapy - the systematic and repeated approach toward fear-provoking stimuli without the occurrence of the anticipated catastrophic outcome - remains the frontline behavioral intervention for specific phobias, PTSD, and obsessive-compulsive disorder 242526272829. While its clinical utility is undisputed, the theoretical explanations of its mechanisms of action have shifted fundamentally from early emotional processing paradigms to contemporary associative learning models.
The Emotional Processing and Habituation Model
For decades, the standard theoretical framework for exposure therapy was the Emotional Processing Theory. This model posited that therapeutic change relies on the gradual habituation of the fear response, both within a single therapy session (within-session habituation) and across sequential sessions (between-session habituation) 2527303132. Under this paradigm, clinicians encourage patients to remain in the presence of the phobic stimulus until their self-reported anxiety or physiological distress declines by a specific threshold, often targeted at a 50% reduction in Subjective Units of Distress (SUDS) 253132.
In this habituation-based model, fear reduction during the exposure trial was viewed as a necessary precursor to altering long-term cognitive beliefs about the danger of the stimulus 25. However, subsequent empirical research demonstrated critical flaws in this theory. The degree of fear reduction at the completion of an exposure session does not reliably predict the amount of fear expressed at follow-up, and patients can demonstrate profound clinical improvement even if their fear does not fully habituate during training trials 25273235.
The Inhibitory Learning Model
In response to the predictive failures of habituation models, modern clinical psychology has increasingly adopted the Inhibitory Learning Model (ILM) 25313235. Grounded in associative learning theory, the ILM argues that exposure therapy does not "erase," overwrite, or unlearn the original fear memory (the excitatory meaning) 30323536. Instead, exposure creates a secondary, competing memory trace (inhibitory learning) that indicates the stimulus is safe or tolerable 30323536.
Because the original fear memory remains neurologically intact, the clinical goal of exposure therapy under the ILM shifts from focusing on immediate anxiety reduction to prioritizing fear tolerance and the maximization of new learning 253031. The objective is to make the newly formed inhibitory association stronger and more readily retrievable than the original excitatory association across diverse contexts 2532.
To achieve this, several counterintuitive strategies are deployed to optimize inhibitory learning. These strategies are collectively termed "desirable difficulties" because they require greater cognitive effort, elevating acute distress during therapy to foster superior long-term retention of safety associations 2530.
| Optimization Strategy | Mechanism of Action within the Inhibitory Learning Model |
|---|---|
| Expectancy Violation | Designing exposures not to reduce fear, but to maximally violate the patient's specific predictions regarding the frequency, likelihood, or intensity of aversive outcomes. 2535 |
| Deepened Extinction | Combining multiple distinct feared stimuli simultaneously (e.g., presenting a previously extinguished cue alongside a novel fear cue) to deepen the cognitive learning process. 25 |
| Occasional Reinforced Extinction | Introducing occasional pairings of the feared stimulus with an actual aversive outcome (e.g., intentionally seeking minor social rejection) to increase resilience against future unpredictable negative events. 25 |
| Removal of Safety Signals | Eliminating behavioral or physical coping mechanisms (e.g., holding a cell phone, using anxiolytic medications, relying on therapist presence) that patients use to mitigate distress, as these prevent the full consolidation of inhibitory associations. 25 |
| Variability | Varying the stimulus features, duration, intensity, and inter-trial intervals. Unpredictable exposures require more active cognitive processing, which strengthens memory consolidation and generalization. 25 |
| Retrieval Cues | Employing specific physical or mental cues (e.g., a specific wristband) present during exposure training to intentionally trigger the recall of the inhibitory safety memory in novel contexts. 25 |
| Multiple Contexts | Conducting exposures in varied physical, temporal, and internal states (e.g., different rooms, varying times of day) to prevent the context-bound renewal of the original fear response. 25 |
| Affect Labeling | Requiring the patient to use linguistic processing to explicitly state their emotional experience during exposure, a process demonstrated to dampen amygdala hyperreactivity and facilitate regulation. 25 |
Interestingly, the ILM also alters traditional cognitive behavioral therapy (CBT) paradigms regarding cognitive restructuring. Traditional CBT often precedes exposure with cognitive restructuring to reduce anxiety. Under the ILM, preceding exposure with cognitive restructuring may undermine the learning process by softening the violation of the patient's catastrophic expectations; therefore, cognitive restructuring is often delayed until after the exposure work to help consolidate the new inhibitory learning 35.
Memory Reconsolidation and Fear Updating
While exposure therapy relies fundamentally on fear extinction - the formation of a parallel safety memory - advances in neuroscience have identified memory reconsolidation as an independent and highly promising therapeutic target 2122363334.
The Reconsolidation Window
Memory consolidation was traditionally viewed as a linear, permanent progression. However, modern neurobiology indicates that when a consolidated memory is retrieved, the memory trace is reactivated and temporarily enters a labile, malleable state. To be maintained in long-term storage, this trace must be restabilized via de novo protein synthesis - a dynamic process called reconsolidation 21223634.
The reconsolidation window typically remains open from 30 minutes to 6 hours post-retrieval 2236. This temporal window presents a unique neurobiological opportunity: rather than merely suppressing the fear response with a competing extinction memory, interventions can directly update, modify, or structurally weaken the original traumatic memory trace 22363335. Reconsolidation is a protein-synthesis-dependent process triggered by prediction error and relies heavily on coordinated signaling pathways, including ERK/MAPK, mTOR, and NMDA receptor activation, alongside synchronization between the hippocampus and prefrontal cortex 3635.
Extinction Versus Reconsolidation
The distinction between extinction and reconsolidation is critical for clinical application. Extinction provides reliable but often fragile benefits because the original fear memory remains intact, leaving the patient vulnerable to relapse when contexts shift 363335. Interventions targeting reconsolidation aim to alter the memory engram permanently. Disrupting the reconsolidation phase - either through behavioral means or pharmacological blockade - aims to prevent the return of fear entirely 2136333436.
One notable behavioral application is the retrieval-extinction paradigm. In this protocol, a brief exposure to the conditioned stimulus reactivates the memory. After a short delay (e.g., 10 minutes) to allow the memory to enter its labile state, full extinction training is administered within the reconsolidation window 21333436. This method has proven highly effective at erasing short-term fear memories in laboratory settings, though long-term, highly consolidated traumatic memories often require pharmacological augmentation to achieve profound structural disruption 212233.
Treatment Limitations and Attrition
Despite its status as the clinical gold standard, exposure therapy is substantially constrained by rates of non-response, patient attrition, and post-treatment relapse. Addressing these limitations is a primary focus of contemporary clinical research 1627373839.
Phenomenological Relapse
Because extinction learning is heavily context-dependent and does not erase the original fear trace, individuals who successfully undergo standard exposure therapy remain highly vulnerable to the return of fear 16253740. This phenomenological relapse typically manifests through three established associative learning mechanisms:
- Renewal: The fear response returns when the phobic stimulus is encountered in a context different from where the extinction learning occurred. For example, if a patient's fear is extinguished in the therapist's office (Context B) after being acquired in a real-world setting (Context A), the fear will rapidly return if the stimulus is encountered again in Context A (ABA renewal) or in a completely novel environment (ABC renewal) 16253740.
- Spontaneous Recovery: Conditioned fear responses re-emerge simply as a function of the passage of time following successful extinction therapy, demonstrating that the inhibitory safety memory can degrade faster than the deeply ingrained excitatory fear memory 21253337.
- Reinstatement: The fear response to a previously extinguished cue returns if the individual experiences an unexpected, unsignaled aversive event (e.g., a severe stressor), even if that event is entirely unrelated to the original phobic stimulus 212533.
These phenomena highlight a fundamental vulnerability of exposure therapy: reducing fear responses in a controlled clinical environment does not guarantee generalized safety responses in the chaotic real world 16. Furthermore, exposure therapies heavily address the associative components of fear conditioning but frequently fail to mitigate nonassociative features of severe anxiety and PTSD, such as baseline amygdala hyperactivity and generalized hyperarousal, leading to residual distress even after behavioral compliance 1640.
Attrition Rates Across Modalities
The inherently demanding nature of exposure therapy - which requires patients to repeatedly confront acute, profound psychological distress - results in severe dropout rates and widespread treatment refusal 2737384142. Meta-analytic reviews evaluating dropout rates among clinical populations, particularly within military cohorts receiving trauma-focused treatments, highlight the magnitude of this operational failure 39434445.
An analysis of 232 PTSD treatments representing over 124,000 patients found a weighted overall dropout rate of 25.6% 3944. However, this attrition rate varies dramatically across different therapeutic modalities. Weekly formats of Cognitive Processing Therapy (CPT) and Prolonged Exposure (PE) exhibit some of the highest dropout risks, at 40.1% and 34.7%, respectively 3944. Similarly, standard Virtual Reality Exposure Therapy demonstrates a high dropout rate of 37.2% in these populations 3944.
Conversely, interventions that alter the delivery format or emphasize community support demonstrate significantly superior retention. Group-based exposure therapies lower the dropout rate to a mere 6.9% 3944.
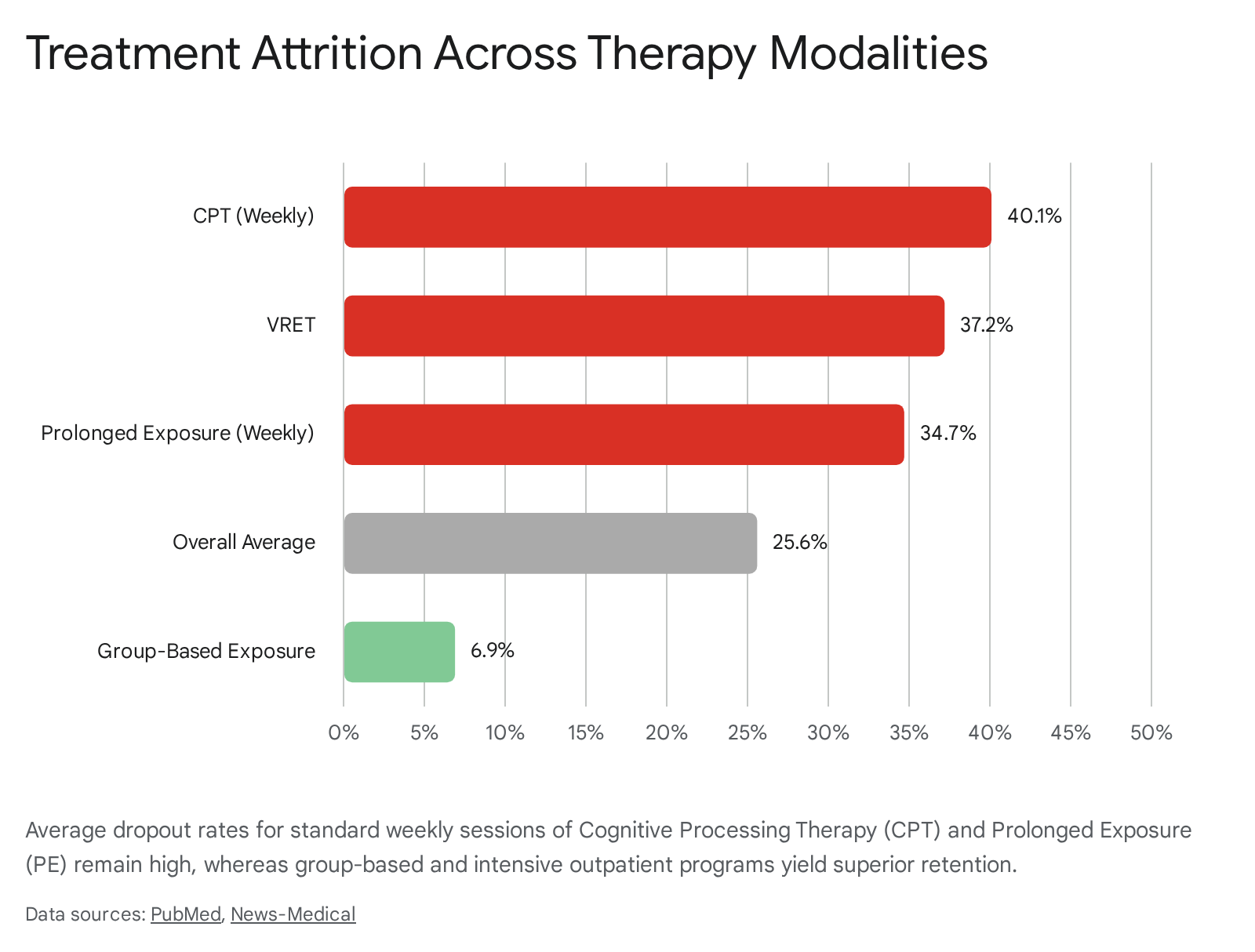
Intensive Outpatient Programs (IOPs) yielded the lowest dropout rates overall, even when IOPs included CPT (8.5%) and PE (5.5%) 39.
The timing of clinical dropout is consistently front-loaded; the highest risk of attrition occurs very early in treatment, generally between the second and sixth sessions 45. This temporal pattern underscores the initial psychological hurdle patients face when attempting to adopt the rationale of exposure therapy. To combat this, recent evidence suggests that adjusting the frequency of treatment administration may mitigate attrition. Meta-analyses demonstrate that prescribing Prolonged Exposure sessions at least twice weekly reduces the dropout rate significantly to 21%, down from 34% observed in standard, once-weekly scheduling 3843.
Technological and Pharmacological Advancements
To circumvent the logistical constraints, environmental limitations, and high refusal rates associated with traditional in vivo exposure, clinical practice has rapidly integrated immersive technologies and pharmacological adjuncts designed to accelerate neural plasticity.
Virtual and Augmented Reality
Virtual Reality Exposure Therapy (VRET) and Augmented Reality Exposure Therapy (ARET) have matured into highly efficacious tools for specific phobias, particularly acrophobia (heights), arachnophobia (spiders), and situational anxieties like aviophobia (flying) 2441424647.
VRET utilizes head-mounted displays to place the patient in a fully immersive, computer-generated environment, affording therapists absolute control over the intensity, duration, and specific parameters of the phobic stimuli 244146. ARET, conversely, overlays virtual representations of feared objects onto the patient's actual physical world, often using smartphones, tablets, or markerless projection systems 424647. This technique helps maintain the patient's grounding in their physical environment while still permitting controlled interaction with the phobic trigger 46.
Clinical outcomes for immersive therapies match or occasionally exceed those of traditional in vivo methods. VRET is associated with robust success rates ranging between 66% and 90% 24. Crucially, it provides faster desensitization, requiring an average of 8 sessions compared to 12 sessions for traditional therapy 41. The perception of a safe, highly controlled virtual environment drastically improves patient willingness to initiate and maintain therapy; dropout rates for VR-based interventions in civilian populations are frequently estimated at just 10%, compared to 25% for traditional exposure 41.
Advancements in VRET between 2023 and 2025 focus extensively on the integration of artificial intelligence and biofeedback 41. Modern VR systems continuously monitor physiological biometric markers - such as heart rate variability, galvanic skin response, and pupil dilation 41. Utilizing machine learning algorithms, these systems can dynamically and autonomously adjust the intensity of the simulated exposure in real-time based on the user's autonomic arousal. This responsive modulation optimizes the therapeutic gradient, ensuring the patient remains precisely within a productive window of distress, maximizing inhibitory learning without crossing into overwhelming panic that could derail treatment 41.
Pharmacological Augmentation of Extinction
Recognizing that exposure therapy fundamentally relies on molecular neural plasticity, researchers have sought pharmacological adjuncts to accelerate fear extinction. These adjuncts are not administered chronically as daily symptom suppressants (unlike traditional SSRIs or SNRIs), but rather as targeted, acute cognitive enhancers given directly before or after a specific exposure session 4854495051.
The most extensively researched adjunct is D-cycloserine (DCS), an antibiotic originally utilized to treat tuberculosis. In the central nervous system, DCS acts as a partial agonist at the glutamatergic N-methyl-d-aspartate (NMDA) receptor in the amygdala 264849. Because NMDA receptors are critical for synaptic plasticity and the acquisition of new memories, DCS aims to enhance the consolidation of the safety memory formed during exposure 264849.
Clinical trials across multiple anxiety disorders - including social anxiety, specific phobia, and panic disorder - indicate that low-dose DCS (typically 50 mg), administered shortly before or immediately following an exposure session, facilitates fear extinction, leading to faster symptom reduction in fewer overall sessions 264854. However, subsequent large-scale analyses revealed that the efficacy of DCS is highly contingent on the behavioral success of the exposure session itself 4952. Because DCS indiscriminately amplifies whatever learning occurs during the session, it significantly enhances therapeutic outcomes only when the exposure session ends with low fear (indicating successful inhibitory learning). Conversely, if an exposure session is unsuccessful or ends in elevated panic, administering DCS risks inadvertently consolidating and strengthening the original fear memory, potentially worsening clinical outcomes 4952.
| Pharmacological Agent | Primary Mechanism of Action | Clinical Application Target | Limitations and Efficacy |
|---|---|---|---|
| D-cycloserine (DCS) | NMDA receptor partial agonist; enhances glutamatergic neuroplasticity. 4849 | Augmentation of fear extinction memory consolidation. 485449 | Highly contingent on successful within-session fear reduction; can worsen fear if the session ends poorly. 4952 |
| Propranolol | Beta-adrenergic antagonist; blocks noradrenergic signaling in the amygdala. 36 | Disruption of memory reconsolidation post-retrieval. 36 | Mixed clinical trial results; lacks sufficient evidence for routine recommendation in PTSD. 3553 |
| Scopolamine | Anticholinergic agent; impairs hippocampal encoding. 16 | Prevention of context-specific memory encoding to limit fear renewal. 16 | Largely restricted to preclinical and highly specialized human trial models; broad side-effect profile. 16 |
| Mirdametinib / MEK Inhibitors | Blockade of the ERK/MAPK intracellular signaling pathway. 36 | Disruption of memory reconsolidation at the molecular level. 36 | Preclinical success, but requires highly specific timing and strong prediction error to induce memory labilization. |
| Intranasal Oxytocin | Neuropeptide modulation; reduces baseline amygdala hyperactivity. 5053 | Enhancement of fear extinction when paired with trauma-focused therapy. 5053 | Efficacy remains under investigation in ongoing Phase II clinical trials. 50 |
Reconsolidation Blockade and Novel Agents
Beyond enhancing extinction, psychopharmacology is actively exploring agents capable of directly disrupting the reconsolidation phase of traumatic memories 3654. The objective is to administer specific agents during the brief 6-hour window following memory retrieval, preventing the restabilization of the fear trace and permanently degrading its emotional valence 2236.
Propranolol, a beta-adrenergic antagonist, has been extensively studied for this purpose. By blocking noradrenergic signaling in the basolateral amygdala during the reconsolidation window, propranolol theoretically strips the memory of its physiological fear component 36. While it has shown promise in controlled laboratory settings and specific trauma models, broad phase 4 clinical outcomes for propranolol have been inconsistent, and current data do not support its routine clinical use for disrupting trauma memory in generalized PTSD populations 53.
Similarly, preclinical research investigates the use of scopolamine, an anticholinergic drug, to combat the pervasive problem of context renewal. By deliberately impairing hippocampal function during exposure therapy, scopolamine prevents the patient from encoding the specific clinical context (e.g., the exact room or therapist) in which the safety memory is formed. This impairment theoretically allows the newly acquired extinction memory to generalize more robustly to external, real-world environments 16. Furthermore, experimental modulation of the ERK/MAPK pathway using MEK inhibitors like mirdametinib demonstrates potential for molecular reconsolidation disruption, though such agents require strict induction of prediction error to labilize the memory prior to administration .
While the pharmacological augmentation of exposure therapy remains an area of profound translational promise, variables such as optimal dose timing, the necessity of prediction error to induce true memory labilization, and individual neurobiological variances continue to moderate clinical efficacy 3649. Validating these combinations through rigorous, standardized phase 4 interventional trials remains a critical priority for optimizing the management of severe phobic and trauma-related disorders 5455.