Safety and efficacy of rapamycin for human longevity
Mechanistic Target of Rapamycin Biology
Origin and Pharmacological Discovery
Rapamycin, known clinically as sirolimus, is a macrolide compound with a rich pharmacological history. It was first isolated in 1975 from a soil sample collected on Easter Island, known indigenously as Rapa Nui, originating from the bacterium Streptomyces hygroscopicus 12. Initial pharmacological evaluations identified rapamycin as a potent antifungal agent, but subsequent investigations revealed marked antiproliferative, anti-tumorigenic, and immunosuppressive properties 13. These findings pivoted its clinical trajectory, leading to its eventual approval by the U.S. Food and Drug Administration (FDA) in 1999 as a prophylactic agent to prevent allograft rejection in renal transplant patients 34.
Decades after its discovery, researchers investigating yeast resistance to the compound identified its central biological target: the mechanistic target of rapamycin (mTOR) 15. The mTOR protein is an evolutionarily conserved serine/threonine protein kinase that functions as a master regulatory hub for cellular metabolism, growth, proliferation, and survival across eukaryotic organisms 56. The mTOR kinase forms the catalytic core of two structurally and functionally distinct multiprotein complexes: mTOR Complex 1 (mTORC1) and mTOR Complex 2 (mTORC2) 6. The differentiation between these two complexes forms the biochemical basis for both the longevity-enhancing benefits and the adverse metabolic side effects of rapamycin administration 56.
mTOR Complex 1 and Cellular Aging
The mTORC1 complex is acutely responsive to environmental and intracellular cues, functioning as a primary nutrient sensor. It is activated by the presence of abundant amino acids, glucose, cholesterol, and oxygen, alongside growth factors like insulin 6. When nutrients are plentiful, mTORC1 activation drives anabolic processes, orchestrating protein synthesis, lipid synthesis, and cellular proliferation through the phosphorylation of downstream substrates such as the eukaryotic translation initiation factor 4E-binding proteins (4E-BPs) and ribosomal protein S6 kinase beta-1 (S6K1) 67.
Crucially, the activation of mTORC1 simultaneously inhibits catabolic processes, most notably macroautophagy, by phosphorylating and inhibiting the Unc-51 like autophagy activating kinase 1 (ULK1) complex 6. In the context of biological aging, chronic overactivation of the mTORC1 pathway prevents cells from clearing damaged organelles, misfolded proteins, and toxic aggregates 58. This accumulation of cellular detritus is a primary hallmark of aging, leading to a decline in tissue homeostasis, the induction of cellular senescence, and heightened vulnerability to age-related pathologies 589.
When rapamycin is introduced, it forms a complex with the intracellular protein FKBP12, which directly binds to and allosterically inhibits mTORC1 5. This specific pharmacological inhibition deceives the cell into a state of perceived nutrient scarcity, effectively mimicking the physiological effects of dietary restriction without a reduction in caloric intake 101112. By attenuating S6K1 activity and lifting the inhibition on ULK1, rapamycin restores robust autophagic clearance, improves proteostasis, and shifts cellular priorities from aggressive growth toward maintenance and stress resistance 68.
mTOR Complex 2 and Metabolic Homeostasis
While mTORC1 inhibition mediates the geroprotective effects of rapamycin, the secondary complex, mTORC2, presents a significant pharmacological challenge. Unlike mTORC1, mTORC2 is relatively insensitive to acute nutrient fluctuations and primarily responds to growth factors, playing a critical role in regulating the actin cytoskeleton, cell survival, and metabolic homeostasis via the activation of the protein kinase Akt 67.
Short-term or low-dose rapamycin administration is highly selective for mTORC1 6. However, chronic, high-dose administration - such as the regimens utilized in solid organ transplantation - leads to the unintended suppression of mTORC2 assembly 5. The inhibition of mTORC2 disrupts downstream PI3K/Akt insulin signaling pathways 67. Preclinical rodent models demonstrate that this chronic suppression removes a critical negative feedback loop, triggering severe glucose intolerance, insulin resistance, and the upregulation of hepatic gluconeogenic master genes, including PEPCK and G6Pase, along with the transcriptional coactivator PGC-1α 7. This unintended metabolic disruption underscores the necessity for precise dosing strategies to uncouple the longevity benefits of mTORC1 inhibition from the metabolic toxicity of mTORC2 suppression 56.
Preclinical Evidence and Lifespan Extension
Vertebrate Meta-Analysis Data
The foundational evidence supporting rapamycin as a gerotherapeutic originates from extensive preclinical modeling. Rapamycin was the first small pharmacological molecule definitively shown to extend both the median and maximal lifespan in murine models, a finding robustly replicated across independent laboratories, including the rigorous Interventions Testing Program (ITP) sponsored by the National Institute on Aging 1256. Furthermore, lifespan extension has been documented in evolutionary divergent model organisms, including yeast, nematode worms, and Drosophila, confirming the highly conserved nature of the mTOR pathway 46.
To quantify the aggregate magnitude of these geroprotective effects, Ivimey-Cook et al. published an exhaustive meta-analysis in Aging Cell in 2025 1314. This analysis aggregated 911 effect sizes from 167 distinct scientific papers covering eight different vertebrate species, including fish, mice, rats, and rhesus macaques 101415. The researchers compared the lifespan-extending efficacy of three primary interventions: dietary restriction (DR, utilizing both caloric reduction and intermittent fasting), rapamycin, and metformin 151617.
The meta-analysis established that both forms of dietary restriction reliably extended lifespan across the analyzed species, confirming DR as the biological gold standard for longevity intervention 1617. Crucially, the analysis revealed that rapamycin extended lifespan to a statistically comparable degree 1017. When adjusted for bias, the estimated log-response ratios translated to an approximate 19% longer lifespan with dietary restriction and a 24% longer lifespan with rapamycin compared to control cohorts 13.
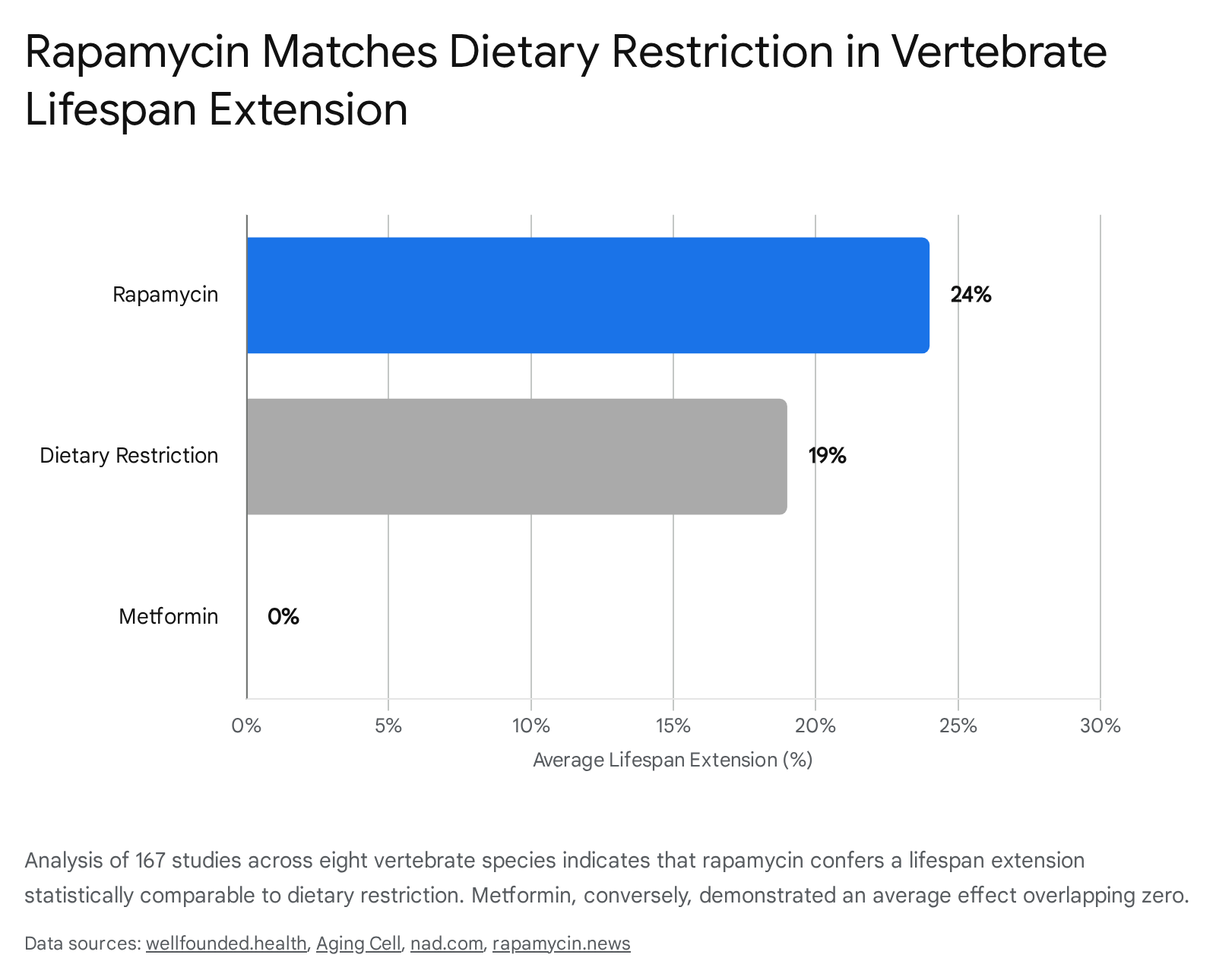
The statistical difference between the efficacy of dietary restriction and rapamycin was not significant, indicating that rapamycin successfully mirrors the lifespan extension of dietary restriction in vertebrates 101317.
Interventional Comparisons
The findings regarding metformin, an antidiabetic compound frequently prescribed off-label for anti-aging, provided a stark contrast. While metformin activates AMPK - a kinase that acts upstream to inhibit mTOR - its average effect on lifespan extension across vertebrate models overlapped with zero, showing a significantly smaller and more inconsistent effect than both rapamycin and dietary restriction 101317. While metformin maintains utility in metabolic indications, the meta-analysis suggests it does not possess the same intrinsic, cross-species longevity properties as rapamycin 1013.
Additionally, the analysis evaluated potential sex-specific responses. Historically, some individual rodent studies indicated that transient, short-term rapamycin treatment extended lifespan in both sexes at low doses, but only in males at higher doses 515. However, the aggregate data spanning 167 studies demonstrated no consistent, statistically significant effect of sex across all log-response measures, suggesting that both males and females derive comparable proportional longevity benefits from mTOR inhibition 101415.
The robust preclinical data also highlights the temporal flexibility of mTOR inhibition. Unlike many theoretical anti-aging interventions that require lifelong adherence, transient rapamycin treatment initiated in late middle age - roughly equivalent to a 60-year-old human - yielded significant extensions in lifespan and indices of healthspan, including cardiac markers and motor function 28.
Clinical Pharmacokinetics and Dosing Paradigms
Translating rapamycin from preclinical animal models to human gerotherapeutics represents a complex pharmacological challenge, primarily due to the drug's narrow therapeutic index and potent immunomodulatory profile. The safety and efficacy of rapamycin are fundamentally dictated by the dosing protocol, necessitating a strict clinical demarcation between high-dose transplant regimens and low-dose longevity regimens 218.
Renal Transplant Immunosuppression Protocols
The FDA-approved indication for sirolimus is the prophylaxis of organ rejection in patients receiving renal transplants 192019. In this high-acuity clinical setting, the pharmacological objective is the profound and continuous suppression of T-cell and B-cell proliferation to prevent the recipient's immune system from attacking the allograft 220.
To achieve continuous immunosuppression, patients are typically administered an initial loading dose of 10 to 15 mg, followed by a daily maintenance dose ranging from 2 to 5 mg 192122. The primary metric of treatment success is the maintenance of whole-blood trough concentrations between 5 and 15 ng/mL 1921. For patients at a high immunologic risk - such as those with a history of allograft loss or high-panel reactive antibodies - target trough concentrations are often pushed to 16 to 24 ng/mL for the first year post-transplant 19. This daily dosing ensures that circulating drug levels never fall below the immunosuppressive threshold, resulting in the chronic inhibition of both mTORC1 and mTORC2 56. It is this persistent inhibition that generates the severe adverse event profile associated with the drug, including metabolic dysfunction, impaired tissue repair, and cytopenia 35.
Off-Label Longevity Protocols
In stark contrast, the clinical application of rapamycin for human longevity aims to selectively inhibit mTORC1 to stimulate autophagy and proteostasis, while sparing mTORC2 to maintain metabolic and immunological function 21819. This objective is achieved through an intermittent, pulsatile dosing strategy 223.
Because rapamycin has a relatively long half-life of approximately 60 hours (2.5 days), daily administration leads to drug accumulation and chronic pathway suppression 226. Longevity physicians bypass this accumulation by prescribing once-weekly or bi-weekly dosing schedules 21819. Typical off-label regimens range from a conservative starting dose of 2 to 3 mg weekly to a more standard dose of 5 to 10 mg weekly for an average 70 kg adult 21819. This translates roughly to the 0.075 to 0.15 mg/kg once-weekly equivalent utilized in companion dog longevity trials 19.
This intermittent approach generates a high peak blood concentration that transiently suppresses mTORC1 24. Over the subsequent week, the drug is naturally cleared from the system, allowing blood levels to drop well below the 5 ng/mL immunosuppressive threshold 219. This biological "washout" period permits the recovery of normal immune and metabolic function before the next dose is administered, drastically altering the systemic toxicity profile compared to transplant patients 218.
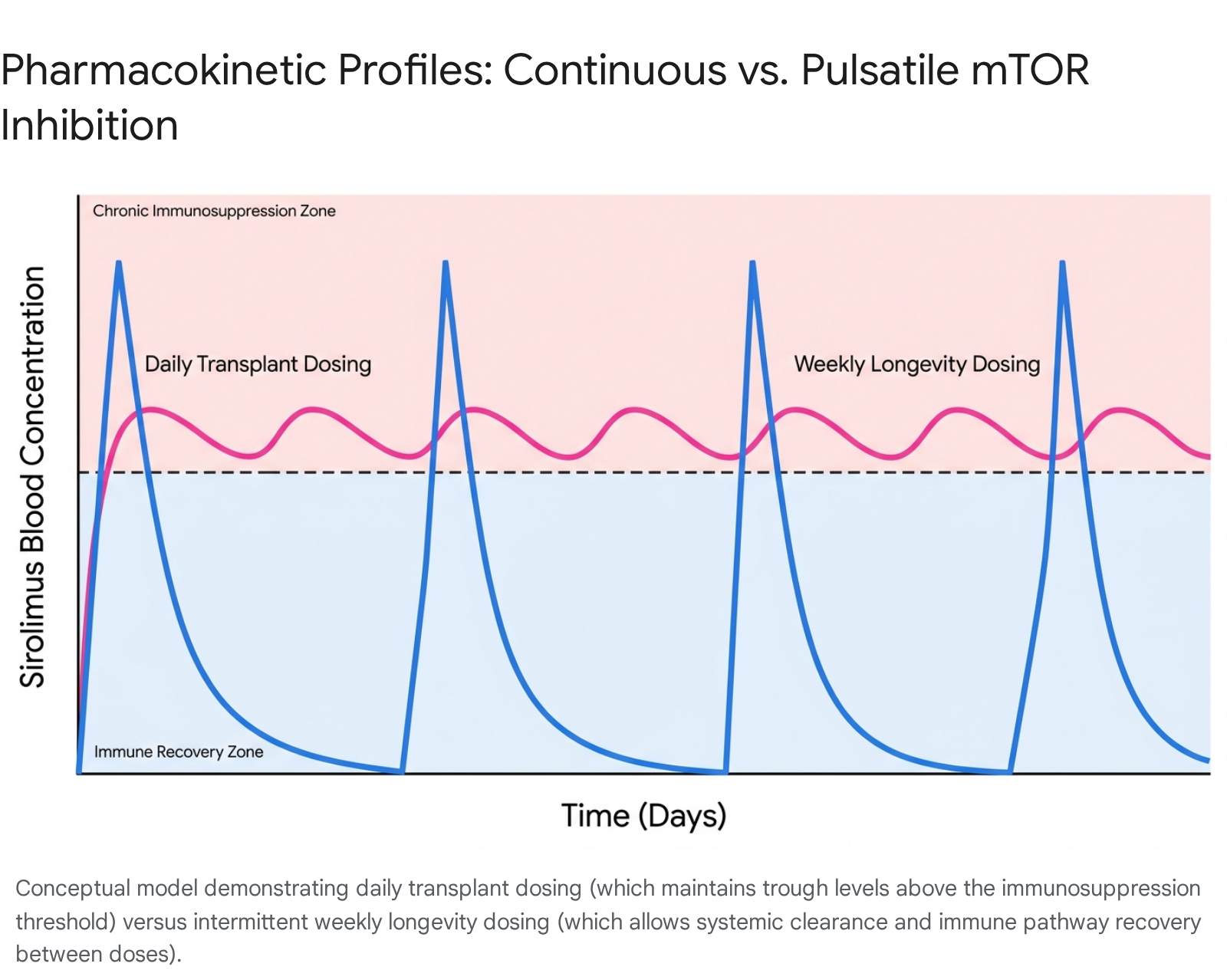
| Clinical Parameter | Renal Transplant Protocol | Off-Label Longevity Protocol |
|---|---|---|
| Dosing Frequency | Daily (chronic continuous exposure) 1919 | Intermittent (once weekly or bi-weekly) 21819 |
| Typical Dosage | 2 to 5 mg daily (after a 10 - 15 mg loading dose) 1921 | 3 to 10 mg weekly (no loading dose utilized) 21819 |
| Pharmacokinetic Goal | Maintain steady trough levels of 5 - 15 ng/mL 192122 | High peak concentration followed by rapid systemic clearance 218 |
| Pathway Target | Sustained systemic inhibition of both mTORC1 and mTORC2 56 | Transient inhibition of mTORC1; functional preservation of mTORC2 2623 |
| Clinical Objective | Profound immunosuppression to prevent renal allograft rejection 419 | Targeted autophagy induction, proteostasis, and immune modulation 2811 |
Bioavailability and Drug Interactions
A critical variable in assessing off-label longevity protocols is the absolute bioavailability of the rapamycin formulation. Rapamycin exhibits inherently low systemic availability; when administered orally, the mean bioavailability of commercial standard tablets is approximately 27% in healthy subjects 22.
However, real-world longevity practice frequently involves compounded formulations of rapamycin to accommodate specific micro-dosing intervals. Recent pharmacokinetic estimates indicate that compounded rapamycin can exhibit drastically reduced absorption, showing approximately 31% relative bioavailability compared to commercial formulations 18. As revealed in recent clinical trials, a prescribed compounded dose of 10 mg weekly may effectively deliver the biological equivalent of only 2.9 mg of standard commercial rapamycin 2825. This variability complicates cross-study comparisons and underscores the need for standardized dosing formulations in future anti-aging research.
Furthermore, rapamycin is extensively metabolized by the cytochrome P450 enzyme system in the liver, specifically CYP3A4, rendering it highly sensitive to pharmacokinetic interactions 526. Concomitant use of CYP3A4 inhibitors - ranging from pharmaceutical agents like ketoconazole and erythromycin to common supplements like cannabidiol (CBD) - can drastically reduce rapamycin clearance, elevating circulating blood levels and increasing the risk of unexpected toxicity 526. Conversely, CYP3A4 inducers, such as rifampin or St. John's wort, accelerate metabolism, potentially subverting the drug's gerotherapeutic efficacy 26. Patients utilizing off-label rapamycin with any degree of polypharmacy require stringent clinical oversight to navigate these complex metabolic pathways 19.
Human Clinical Trial Data
While vast epidemiological datasets exist for chronically ill transplant recipients, robust data concerning rapamycin's efficacy in healthy human populations has historically been sparse. However, several landmark clinical trials published between 2024 and 2025 have begun to map the phenotypic outcomes of low-dose mTOR inhibition in normative-aging cohorts.
Musculoskeletal Outcomes in the PEARL Trial
The Participatory Evaluation of Aging with Rapamycin for Longevity (PEARL) trial represents a pivotal milestone in human gerotherapeutics. Initiated as a 48-week, randomized, double-blind, placebo-controlled, decentralized study, PEARL aimed to evaluate the safety and efficacy of intermittent rapamycin in mitigating clinical signs of aging 263127. The trial included 114 participants aged 50 to 85, who were randomized into three cohorts: 5 mg of compounded rapamycin weekly, 10 mg weekly, and a placebo arm 282728.
The primary predefined clinical endpoint of the trial was the reduction of visceral adiposity as measured by Dual-energy X-ray Absorptiometry (DXA) scans 2634. The results indicated that visceral adiposity did not change significantly across the aggregate cohort (p = 0.942) 2627.
Despite the failure to meet the primary endpoint, the secondary outcomes revealed highly significant, sex-dependent anatomical shifts. Female participants assigned to the 10 mg weekly dose demonstrated an average 4.5% to 5% increase in lean tissue mass over the 48-week period (p = 0.013) compared to baseline 25262734. This cohort of women concurrently reported statistically significant reductions in generalized aches and pain (p = 0.015) 2627. Conversely, male participants receiving the 10 mg dose exhibited an average 1.4% increase in bone mineral content over the trial duration 282531.
Further subjective improvements were noted in the 5 mg weekly cohort, where participants reported significant enhancements in overall emotional well-being (p = 0.023) and general health self-ratings (p = 0.004) 2627. It is critical to contextualize these findings within the drug's previously discussed bioavailability constraints: due to the formulation used, the 5 mg and 10 mg cohorts experienced actual systemic exposures equivalent to roughly 1.4 mg and 2.9 mg of commercial rapamycin 2825. The observation of significant morphological improvements in lean mass at such low biological exposures highlights the profound sensitivity of the human musculoskeletal system to mTOR modulation.
Immunomodulation and the Novartis Studies
The conceptual framework underlying rapamycin use frequently wrestles with its paradoxical immune effects. In transplant medicine, high doses purposefully induce severe immunosuppression 24. However, in aging biology, immunosenescence - the progressive dysfunction of the immune system - leads to chronic low-grade inflammation ("inflammaging"), decreased vaccine efficacy, and heightened vulnerability to pathogens 82029.
Evidence that low-dose mTOR inhibition reverses, rather than exacerbates, immunosenescence originated from a seminal 2014 clinical trial led by Dr. Joan Mannick under Novartis 24. The six-week, placebo-controlled trial included 218 older adult volunteers aged 65 and above, randomized to receive placebo, 0.5 mg daily, 5 mg weekly, or 20 mg weekly of everolimus (RAD001), a structural analog of rapamycin 24. Following the six-week treatment period, participants were administered an influenza vaccine 2.
The results were paradigm-shifting. Participants receiving the 0.5 mg daily and 5 mg weekly regimens exhibited a 20% enhancement in antibody titers in response to the influenza vaccine relative to the placebo group 1219. Furthermore, phenotypic analysis of circulating immune cells demonstrated a reduction in PD-1 positive CD4 and CD8 T-cells - markers associated with immune cell exhaustion 12. This indicated a functional recalibration of the immune system toward a more resilient, youthful state 124. Notably, the cohort receiving the highest dose (20 mg weekly) experienced a higher incidence of adverse effects and reduced antibody titers, reaffirming that the immunological benefits of mTOR inhibition follow a hormetic, dose-dependent curve where lower exposures yield optimal immunomodulation 4.
Reproductive Aging and the VIBRANT Trial
Recent clinical focus has expanded to the female reproductive system, particularly the ovaries, which represent the fastest-aging organs in the human body 3031. Ovarian senescence not only terminates fertility but also precipitates the systemic physiological decline associated with menopause, increasing susceptibility to cardiovascular disease, bone density loss, and cognitive decline 313239.
The Validating Benefits of Rapamycin for Reproductive Aging Treatment (VIBRANT) trial at Columbia University aims to ascertain if pharmacological mTOR inhibition can safely delay ovarian aging 303133. Early pilot data published in 2024 evaluated a cohort of women aged 35 to 45 who were in late stage 3a of reproductive aging (immediately preceding the onset of perimenopause) 3241. Participants were randomized to receive 5 mg of rapamycin weekly or a placebo for 12 weeks 323941.
Preliminary findings indicate that rapamycin slowed the rate of natural follicular atresia by approximately 20% 313241. Under standard physiological conditions, a woman loses an average of 50 eggs per month; ultrasound and endocrine biomarker analysis revealed that the rapamycin-treated cohort lost approximately 15 eggs per month 313941. Crucially, the 5 mg weekly dosage preserved normal menstrual cycling and did not suppress ovulation, contrasting with the anovulatory effects seen at higher, continuous doses 3039. The extension of the fertility window by a theoretical five years, if replicated in larger Phase 2 cohorts, positions rapamycin as a highly novel intervention for modulating reproductive aging without relying on exogenous hormone replacement therapies 303132.
Dermatological and Cardiovascular Markers
Beyond musculoskeletal and immunological parameters, rapamycin has demonstrated localized geroprotective effects in the integumentary system. Exploratory, prospective human trials applying topical rapamycin to the skin successfully reduced specific cellular markers of senescence, notably decreasing the expression of the p16INK4A protein 91234. This topical intervention simultaneously improved structural parameters by increasing collagen VII levels, offering evidence that mTOR inhibition can rejuvenate tissue morphology at the local level without necessitating systemic drug exposure 12.
Systemically, while broad cardiovascular disease outcomes require longitudinal studies, ongoing trials and early data suggest moderate benefits in vascular regulation and metabolic resilience 1935. However, these parameters must be carefully weighed against the known metabolic toxicities of the drug.
Systematic Safety Profile
The ethical framework surrounding off-label gerotherapeutics mandates that interventions prescribed to healthy individuals carry minimal risk of severe morbidity 5. While the low-dose, intermittent regimens utilized in the PEARL and VIBRANT trials demonstrated high tolerability with no severe adverse events reported, a comprehensive safety profile must account for the specific vulnerabilities associated with mTOR inhibition across varying populations 3132.
Mucosal Ulceration and Stomatitis Incidence
The most uniformly reported side effect of rapamycin, spanning both high-dose transplant cohorts and low-dose longevity users, is mTOR inhibitor-associated stomatitis (mIAS) 3637. These lesions manifest as aphthous-like mucosal ulcers within the oral cavity and digestive tract, presenting identically to recurrent aphthous stomatitis with ovoid ulcerations surrounded by erythematous halos 2337.
In oncology and renal transplant populations, mIAS is remarkably prevalent, affecting 44% to 60.8% of patients receiving continuous mTOR inhibitor therapy, with severe (Grade 3/4) toxicity requiring dose interruptions occurring in up to 9% of cases 3738. The onset is rapid, typically emerging within 4 to 10 days of treatment initiation 3738.
In contrast, the incidence and severity of mIAS in longevity cohorts are significantly attenuated. A 2024 secondary data analysis of 333 healthy adults utilizing off-label rapamycin (average dose ~6 mg weekly) found that while approximately 26% of users reported some degree of oral health change, the presentation was overwhelmingly benign 36. Only 4 users reported persistent ulcers, whereas 50 users experienced transient, intermittent sores that resolved despite continued drug usage 36. Intriguingly, statistical analysis revealed that the incidence of these ulcers in the off-label cohort was not strictly dose-dependent; users on lower doses occasionally reported more persistent symptoms, indicating an individualized mucosal susceptibility to mTOR inhibition rather than a purely concentration-driven toxicity 36.
An analysis of the FDA's Adverse Event Reporting System (FAERS) utilizing Bayesian confidence propagation algorithms further corroborates the high signal associated with mTOR inhibitors 47. Everolimus and palbociclib (a CDK4/6 inhibitor) ranked among the top drugs likely to induce aphthous ulcers in real-world clinical data, specifically in patients over the age of 60 and females 47. Consequently, longevity protocols highly recommend routine oral health monitoring for users initiating therapy 36.
Metabolic Disruptions and Starvation Pseudo-Diabetes
The regulation of systemic metabolism is arguably the most complex safety domain for human rapamycin use. As previously established, rapamycin functionally replicates the state of nutrient scarcity 623. When administered continuously or at doses that breach individual tolerability thresholds, this artificial starvation signal triggers a defensive physiological response termed "starvation pseudo-diabetes" (SPD) 2324.
In SPD, the body mobilizes lipids and heavily suppresses peripheral glucose uptake to preserve energy for vital organs, resulting in systemic hyperlipidemia (elevated LDL cholesterol and triglycerides) and varying degrees of glucose intolerance 2324. Among renal transplant recipients taking combination regimens, significantly increased serum triglyceride levels - ranging from 11.7 to 42.0 mmol/L - are frequently observed within months of initiation 48.
While severe hypertriglyceridemia is largely confined to transplant regimens, off-label users remain susceptible to mild lipid and glycemic alterations. Systematic reviews indicate that patients with preexisting age-related metabolic dysfunctions - such as pre-diabetes or unmanaged hyperlipidemia - are at a significantly elevated risk of exacerbating these conditions when initiating rapamycin 3435. However, the PEARL trial noted no significant differences in markers of metabolic health, including HbA1c, liver function (ALT/AST), or renal function between the 5 mg, 10 mg, and placebo cohorts over 48 weeks, suggesting that healthy adults utilizing strict intermittent dosing can successfully avoid SPD 282528.
Oncological Risk and Protection Vectors
Because rapamycin induces immunosuppression at high continuous doses, concerns regarding an elevated risk of opportunistic infections and secondary malignancies persist. However, the mechanistic reality of mTOR inhibition complicates this assumption.
A robust meta-analysis of 39,039 kidney transplant recipients (evaluating 20 randomized controlled trials and 2 observational studies) assessed the oncological impact of sirolimus 2039. The analysis found that sirolimus use was associated with an overall decrease in cancer incidence (Incidence Rate Ratio [IRR] = 0.71) compared to alternative immunosuppressants like calcineurin inhibitors 2039. This protective effect was intensely driven by a substantial reduction in non-melanoma skin cancer (IRR = 0.49) and a 60% reduction in the incidence of new kidney cancers (IRR = 0.40) 203940. These findings align with rapamycin's origin as a highly effective antiproliferative and anti-angiogenic agent 20.
Conversely, the data indicated a concerning association with a higher incidence of prostate cancer (IRR = 1.85) among male transplant recipients utilizing sirolimus 203940. Furthermore, while systemic cancer rates dropped, the generalized state of profound immunosuppression in transplant cohorts did result in higher occurrences of specific viral and respiratory tract infections, alongside rare drug-induced pneumonitis 340. While the magnitude of these risks is exponentially lower in healthy individuals adhering to once-weekly longevity dosing, the clinical directive dictates that any patient utilizing rapamycin should temporarily discontinue the medication during an active acute infection or prior to undergoing major surgical procedures due to impaired wound healing mechanisms 252841.
System-Level Synthesis of Human Evidence
To provide a definitive, evidence-based assessment of rapamycin's utility in healthy aging, Lee et al. published a comprehensive systematic review in The Lancet Healthy Longevity in early 2024 1335. The researchers synthesized data from 19 unique human trials that evaluated rapamycin and its derivatives across thousands of adult participants, assessing both healthy volunteers and those suffering from established age-related diseases 35.
The findings definitively parse the physiological domains where mTOR inhibition exhibits proven clinical efficacy versus systems where evidence remains neutral or entirely unassessed.
| Physiological System | Clinical Impact of Rapamycin / Rapalogs in Human Trials | Evidence Confidence |
|---|---|---|
| Immune System | Significant functional enhancement. Demonstrated improvements in response to influenza vaccination and partial reversal of immunosenescence markers (e.g., PD-1+ cell reduction) in older adults. | High 133542 |
| Cardiovascular System | Measured functional improvements associated with biological aging parameters, though definitive morbidity reduction requires longer trials. | Moderate 93542 |
| Integumentary (Skin) | Reduced cellular markers of senescence (p16INK4A) and improved structural proteins (collagen VII) via topical administration protocols. | Moderate 9123542 |
| Musculoskeletal System | Mixed outcomes. Localized improvements in lean tissue mass and bone mineral content documented in subsets (e.g., PEARL), but no universal benefit established across all systemic trials. | Low-to-Moderate 29354243 |
| Endocrine & Neurological | No statistically significant systemic effects demonstrated in current human trial designs. Cognitive improvement established in preclinical AD models, but unverified in humans. | Low 354142 |
| Respiratory & Renal | Insufficient data. The impacts on respiratory function and renal decline in the context of healthy aging cohorts remain largely unassessed. | Unassessed 3542 |
The systematic review concluded that while rapamycin undeniably modifies age-related physiological changes - particularly within the immune and cardiovascular networks - it cannot currently be classified as an intervention that drives total, whole-body rejuvenation in humans 1335. Importantly, the review noted no serious adverse events attributed to rapamycin in healthy individuals, reinforcing the short-term safety of the drug, while noting that participants with existing age-related metabolic or immunological pathologies experienced increased incidence of infections and dyslipidemia 35.
Ongoing Clinical Trials and Future Directions
The current frontier of geropharmacology is aggressively pursuing longer-term, specialized trials to close the empirical gaps surrounding human mTOR inhibition. The scientific consensus is shifting from observational studies of off-label users toward robust, placebo-controlled clinical investigations 1133.
Notable ongoing studies include the Rapamycin - Effects on Alzheimer's and Cognitive Health (REACH) trial 44. Initiated at the University of Texas Health Science Center, this 12-month study (NCT04629495) evaluates the safety and feasibility of oral rapamycin in older adults presenting with amnestic mild cognitive impairment (aMCI) and early-stage Alzheimer's disease 44. This trial directly tests preclinical findings suggesting that mTOR inhibition can correct structural neurological abnormalities and reduce toxic protein aggregates in the aging brain 1241.
Additionally, the Everolimus Aging Study (NCT05835999) aims to determine whether low-dose mTORC1 inhibition can improve the molecular hallmarks of aging in older adults possessing metabolic risk factors 45. Concurrent studies, such as the RAP PAC pharmacology trial, are mapping precise dose-finding strategies to establish a definitive, optimized safety profile for intermittent dosing protocols 45. In 2026, the University of Arizona Coit College of Pharmacy is slated to launch a large-scale, $12 million Phase 3 clinical trial examining rapamycin's ability to maintain physical and immunologic function in adults aged 65 and older over a three-year period 20.
Regulatory Developments and Trial Registries
The global regulatory environment is also adapting to facilitate longevity research. In late 2025, China's National Medical Products Administration (NMPA) formalized a new 30-day review and approval pathway for Investigational New Drug (IND) applications pertaining to innovative drugs with significant clinical value, specifically encouraging global multi-center trials 46. Such regulatory streamlining accelerates the deployment of novel gerotherapeutics, ensuring that rigorous, Phase 3 longevity data is generated at an unprecedented pace 4657. Concurrently, Chinese interventional registries are cataloging structured lifestyle and pharmacological trials aimed at reversing neuroinflammation and metabolic dysfunction in Alzheimer's cohorts, broadening the multi-modal approach to extending human healthspan 47.
Conclusions
Based on exhaustive preclinical data, human clinical trials, and systematic safety reviews, rapamycin stands as the most highly validated, pharmacologically viable intervention for extending mammalian healthspan currently available. Preclinical meta-analyses conclusively demonstrate that rapamycin mimics the potent lifespan-extending effects of dietary restriction across vertebrate species.
However, translating these biological mechanisms to healthy human cohorts necessitates a paradigm shift in pharmacological application. The robust safety of rapamycin is strictly contingent upon intermittent, low-dose administration - typically 5 to 10 mg delivered once weekly. This protocol allows for the targeted, transient inhibition of mTORC1, thereby initiating cellular autophagy and immune recalibration, while permitting systemic clearance to prevent the chronic suppression of mTORC2. Continuous daily dosing, as utilized in organ transplantation, invariably leads to severe immunodeficiency, mucosal ulceration, and debilitating metabolic disruptions, including hyperlipidemia and insulin resistance.
Recent human clinical trials, specifically PEARL and VIBRANT, validate the short-term safety of weekly off-label dosing protocols. Furthermore, they provide preliminary evidence of profound physiological benefits, including the preservation of ovarian reserve, the mitigation of immunosenescence, and sex-specific improvements in lean tissue mass and subjective well-being. Nevertheless, comprehensive systematic reviews emphasize that rapamycin is not a uniform systemic rejuvenator. Evidence regarding its effects on human neurological decline and long-term all-cause mortality remains incomplete.
Therefore, while the clinical evidence supports the biological plausibility and acceptable safety profile of off-label rapamycin for longevity, it remains an experimental intervention. Its application requires stringent, personalized physician oversight, rigorous baseline metabolic testing, and continuous monitoring to circumvent the narrow threshold between geroprotective autophagy and systemic toxicity. As major Phase 3 trials continue to mature throughout the late 2020s, the precise parameters for optimizing mTOR inhibition to extend human lifespan will move definitively from theoretical biology to evidence-based medical practice.