Role of taurine in the biology of aging
The pursuit of interventions capable of delaying the physiological decline associated with aging has driven geroscience into a new era of metabolic exploration. For decades, the biological hallmarks of aging - ranging from genomic instability and telomere attrition to cellular senescence and mitochondrial dysfunction - have been meticulously cataloged. However, the identification of endogenous circulating factors that actively dictate the pace of these systemic declines has remained an elusive frontier. Historically, age-associated metabolic shifts were largely viewed as passengers of the aging process: downstream consequences of cellular degradation rather than the primary drivers of it. This paradigm was abruptly challenged by a resurgence of interest in taurine (2-aminoethanesulfonic acid), a ubiquitous, semi-essential micronutrient.
First isolated from ox bile in 1827 by Tiedemann and Gmelin, taurine's physiological functions remained poorly understood for nearly 150 years 1. It was not until the 1970s, when commercial feline diets deficient in taurine led to widespread retinal degeneration and blindness in cats, that its critical role in mammalian biology was recognized 1. Today, taurine is known to facilitate a wide array of functions, including the regulation of glucose levels, conjugation of bile acids, detoxification, membrane stabilization, osmoregulation, and neuromodulation 2.
This comprehensive research report evaluates the current scientific consensus surrounding taurine's role in aging, anchored by the catalytic 2023 Science study by Singh et al. and the subsequent critical reevaluations published in 2025. By interrogating the translational gap between murine models and human clinical realities, the intricacies of allometric mega-dosing, the mapping of taurine to the biological hallmarks of aging, and the epidemiological realities of regional dietary patterns, this report provides an exhaustive, expert-level synthesis of taurine's therapeutic potential. Furthermore, it examines the dietary supplement industry's volatile response to shifting scientific narratives, evaluating the oversight of institutional watchdogs and federal regulatory frameworks.
The 2023 Paradigm Shift: Singh et al. and the Taurine Deficiency Hypothesis
In June 2023, a landmark investigation published in Science by a massive international consortium led by Parminder Singh and Vijay Yadav fundamentally altered the discourse surrounding amino acid metabolism and longevity. The study was predicated on a comprehensive multi-species metabolomic screening designed to identify circulating molecules that reliably decline with chronological age 234.
The researchers observed a precipitous, age-related decline in serum taurine concentrations across distinct mammalian species. Specifically, the researchers reported that in 15-year-old rhesus macaques, circulating taurine levels were 85% lower than those observed in their 5-year-old counterparts 25. A highly similar trajectory was observed in human cohorts, where the researchers claimed that individuals over the age of 60 exhibited taurine levels that were more than 80% lower than those found in children and young adults 46. This observation prompted a critical hypothesis: If taurine deficiency correlates so strongly with advancing age, could the restoration of youthful taurine levels delay the aging process? To answer this, the researchers initiated comprehensive supplementation protocols in multiple model organisms.
The empirical results of the Singh et al. intervention were highly compelling. The administration of high-dose oral taurine reversed age-associated deficiency and yielded significant extensions in both lifespan and healthspan across species. In middle-aged mice (14 months old, roughly equivalent to 45 human years), taurine supplementation at 500 and 1000 milligrams per kilogram of body weight per day exhibited a median lifespan extension of 10% to 12% 27. Female mice demonstrated a slightly more pronounced longevity benefit of 12%, compared to males at 10%, translating to an additional three to four months of life - a profound extension in the context of murine biology 78. Moreover, the life expectancy for these mice at 28 months of age increased by 18% to 25% 1011.
Beyond mere survival, the supplemented mice displayed marked improvements in functional physiological metrics. They exhibited reduced age-associated weight gain, increased energy expenditure, augmented bone mineral density, superior muscle endurance on rotarod tests, and mitigated depression-like and anxiety-driven behaviors 689. The murine subjects also demonstrated a more youthful immune system profile and improved cognitive functions, specifically in spatial memory assessed via Y-maze performance 610.
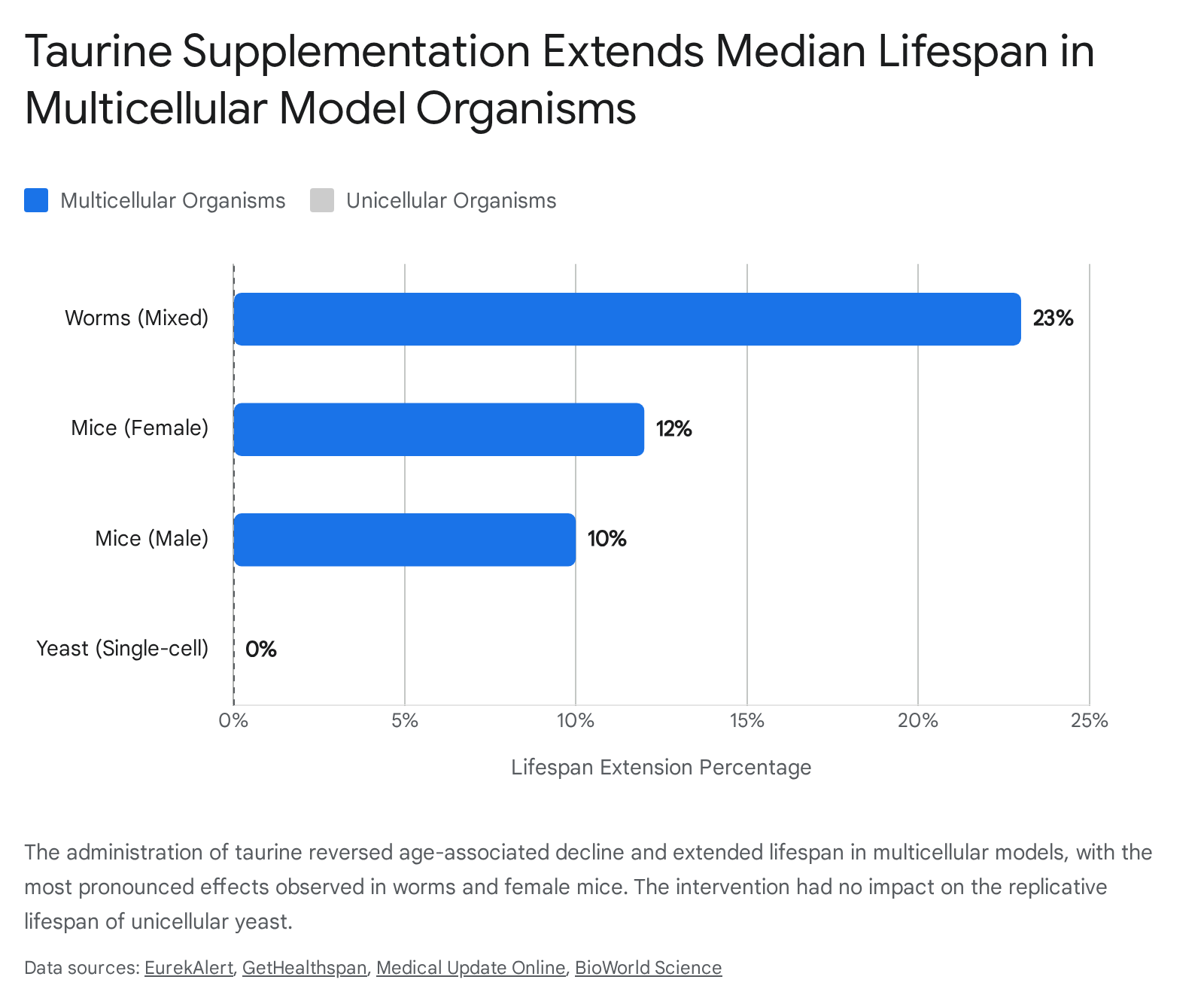
The translational efficacy was further corroborated in non-human primates. Middle-aged rhesus macaques subjected to a six-month taurine supplementation regimen positively modulated bone mineral density in the spine and legs, reduced fasting blood glucose levels, decreased biomarkers of liver damage, and improved overall immunological health profiles 27. In invertebrate models, taurine supplementation increased the median lifespan of multicellular worms (Caenorhabditis elegans) by 10% to 23% 211. Interestingly, the intervention failed to alter the replicative lifespan of unicellular yeast (Saccharomyces cerevisiae). This null result in yeast suggests that taurine's anti-aging mechanisms rely on complex intercellular networks or specific metabolic pathways that diverged later in the evolutionary tree, effectively requiring a multicellular architecture to manifest longevity benefits 41213.
| Model Organism | Cohort Specification | Dosage / Intervention | Lifespan Extension | Healthspan Improvements |
|---|---|---|---|---|
| Mus musculus (Mice) | 14-month-old Females | 500-1000 mg/kg/day | +12% Median | Reduced adiposity, increased bone mass, enhanced rotarod performance, reduced SASP |
| Mus musculus (Mice) | 14-month-old Males | 500-1000 mg/kg/day | +10% Median | Improved Y-maze memory, reduced insulin resistance, enhanced stem cell pools |
| Rhesus macaques | 15-year-old Mixed Sex | 250 mg/kg/day | N/A (Ongoing) | Increased spinal/leg bone density, reduced fasting glucose, lower inflammatory cytokines |
| C. elegans (Worms) | Multicellular Nematode | Media Supplementation | +10% to +23% Median | Maintained physical motility |
| S. cerevisiae (Yeast) | Unicellular Fungi | Media Supplementation | 0% (No effect) | None observed |
The summation of these findings led Singh et al. to formally categorize taurine deficiency as a fundamental driver of aging, postulating that the restoration of youthful taurine levels constitutes a highly promising anti-aging intervention 91117.
Mechanistic Mapping: Taurine and the Biological Hallmarks of Aging
To understand how taurine exerts such profound systemic effects, it is necessary to map its biochemical activity to the established biological hallmarks of aging. Normal aging is characterized by a progressive loss of physiological integrity, driven by a series of interconnected cellular deteriorations, including genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, deregulated nutrient sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, and altered intercellular communication 181415. The 2023 study, supplemented by broader biochemical research, demonstrates that taurine operates as a pleiotropic agent, modulating several of these hallmarks simultaneously. Data indicates that taurine acts dynamically across the cellular architecture, translocating into the mitochondria to facilitate transfer RNA translation, rescuing Complex I function, and reducing reactive oxygen species. This upstream stabilization prevents downstream DNA damage and suppresses the secretion of pro-inflammatory cytokines, effectively halting the progression of cellular senescence 91315.
Mitochondrial Dysfunction and Oxidative Stress
Mitochondria produce the majority of cellular adenosine triphosphate (ATP) but are also the primary source of reactive oxygen species (ROS). During aging, mitochondrial efficiency declines, and the leakage of ROS damages cellular macromolecules, accelerating systemic decay 1516. Taurine functions as a potent, mitochondria-specific antioxidant, though its action is far more sophisticated than simple free radical scavenging; it actively buffers mitochondrial matrix dynamics.
Biochemically, a specific pool of cytosolic taurine translocates into the mitochondria, where it binds directly to the uridine region of transfer RNA (tRNA) at the wobble position 13. This taurinylation of tRNA is strictly required for the accurate translation of the ND6 protein, a core functional subunit of the electron transport chain (ETC) Complex I 13. In aged tissues, tRNA taurinylation drops by up to 60%, severely impairing ETC function, crippling ATP synthesis, and exacerbating ROS leakage 13. Exogenous taurine supplementation rescues this taurinylation process, restoring ND6 expression, rehabilitating Complex I activity, and significantly suppressing mitochondrial oxidative stress 213. Furthermore, in the cytosol, taurine neutralizes the highly reactive hypochlorite ion, forming the stable, non-toxic antioxidant N-chlorotaurine, thereby protecting the broader cellular architecture from oxidative degradation 6.
Cellular Senescence and Genomic Instability
Cellular senescence occurs when cells permanently arrest their cell cycle in response to stress, DNA damage, or telomere attrition. Instead of undergoing apoptosis, these cells linger and secrete a toxic, pro-inflammatory cocktail known as the Senescence-Associated Secretory Phenotype (SASP) 915. These "zombie cells" degrade neighboring tissue architecture and are a primary driver of age-related systemic inflammation, commonly referred to as "inflammaging."
Taurine supplementation drastically reduces the accumulation of senescent cells in aged tissues 910. RNA sequencing of taurine-supplemented cells revealed a marked suppression of the genetic networks that dictate SASP release, effectively silencing the pro-inflammatory signaling loop 1113. Concurrently, by aggressively mitigating ROS generation at the mitochondrial level, taurine indirectly prevents the accumulation of DNA lesions. This upstream protection preserves genomic stability and increases cellular survival rates even under conditions of severe telomerase deficiency 910.
Epigenetic Alterations and Stem Cell Exhaustion
Aging induces profound epigenetic drifts, including DNA hypermethylation and altered histone modifications, which alter chromatin architecture, silence critical regenerative genes, and exhaust stem cell reserves 1822. For instance, in aging skeletal muscle, the enhancer mark H3K27ac becomes heavily deregulated, driving a fibrogenic conversion of satellite cells (muscle stem cells) and depleting the regenerative niche necessary to combat sarcopenia 22.
Taurine exhibits a distinct capacity to influence chromatin conformation. While not a direct methyltransferase, its role as a metabolic buffer influences the broader Taurine/Serine/Methionine (TSM) ratio, impacting the availability of substrates for transmethylation reactions 17. The epigenetic impact of aging on muscle is profound; a recent study examining the skeletal muscle epigenetic clock in an Asian population identified 20 age-associated CpG markers, with accelerated methylation widespread across genomic regions regulating muscle structure and stress response 1819. Taurine's metabolic buffering appears to counteract some of this epigenetic degradation. Supplementation in aged murine models demonstrably increased local stem cell populations in both muscle and intestinal tissues (e.g., LGR5+ stem cells), effectively rescuing the stem cell exhaustion hallmark and promoting youthful tissue regeneration 913.
Loss of Proteostasis and Sarcopenia
Proteostasis - the maintenance of protein homeostasis through folding, trafficking, and degradation - collapses during aging, contributing heavily to skeletal muscle atrophy (sarcopenia) 1820. Sarcopenia is characterized by chronic low-grade inflammation and mitochondrial dysfunction. Taurine is highly expressed in skeletal muscle, where it regulates ion channels and intracellular calcium handling. In vitro studies utilizing myogenic L6 cells exposed to Tumor Necrosis Factor alpha (TNF-α) to simulate senescent muscle inflammation demonstrated that high-dose taurine administration stimulates the differentiation process by downregulating inflammatory pathways and modulating autophagy and apoptosis, protecting against the catabolic pathways that dissolve muscle mass 2021.
Altered Intercellular Communication: The GABAergic Axis
Aging inherently disrupts neuroendocrine signaling and intercellular communication networks. Taurine is highly concentrated in excitable tissues, notably the brain and central nervous system, where it acts both as an osmoregulator and a critical neuromodulator. Crucially, taurine functions as a natural agonist on GABA(A) and glycine receptors 622. As systemic inflammation rises with age, neuronal hyper-excitability often leads to excitotoxicity and cognitive decline. Taurine release provides an opposing, inhibitory, and neuroprotective influence by activating these classical neuronal receptors 22. Interestingly, pharmacological derivatives such as homotaurine (HT) possess up to 100 times greater potency at GABA(A) receptors than endogenous taurine, suggesting that the anti-aging benefits of taurine in mitigating neuro-inflammation and preserving cognitive networks are intimately tied to this GABAergic modulation 22.
The Translational Gap: Pharmacokinetics, Allometric Scaling, and Renal Physiology
While the murine and primate data published in 2023 was transformative, a severe translational gap exists between animal models and human pharmacology. Translating the therapeutic success of taurine in a mouse to a human clinical application requires navigating complex pharmacokinetic barriers, the mathematical realities of allometric mega-dosing, and distinct interspecies differences in renal and hepatic physiology.
The Mathematics of Allometric Mega-Dosing
In the Singh et al. study, the profound anti-aging effects in mice were achieved using extraordinarily high oral doses of 500 to 1,000 milligrams per kilogram of body weight per day 26. A common, critical misconception in consumer longevity communities is that animal dosages map at a 1:1 ratio to humans. However, physiological time, metabolic rate, and body surface area (BSA) dictate that smaller mammals require vastly larger doses per unit of body weight to achieve equivalent systemic exposure 2923.
The U.S. Food and Drug Administration (FDA) provides strict pharmacokinetic guidance for converting animal doses to a Human Equivalent Dose (HED) utilizing BSA normalization. The standard allometric scaling formula utilizes a species-specific correction factor ($K_m$) 2931:
$$HED = \text{Animal Dose (mg/kg)} \times \left( \frac{K_m \text{ Animal}}{K_m \text{ Human}} \right)$$
| Species | Reference Body Weight (kg) | $K_m$ Factor | Factor to Convert Animal Dose (mg/kg) to HED |
|---|---|---|---|
| Mouse | 0.02 | 3 | Divide by 12.3 (or multiply by 0.081) |
| Rat | 0.15 | 6 | Divide by 6.2 (or multiply by 0.162) |
| Macaque | 3.0 | 12 | Divide by 3.1 (or multiply by 0.324) |
| Human | 60.0 | 37 | N/A (Baseline) |
To calculate the HED from the successful murine dose of 1,000 mg/kg utilized by Singh et al.:
$$HED = 1000 \times \left(\frac{3}{37}\right) = 81.08 \text{ mg/kg}$$
For a standard 60 kg human adult, this translates to an oral dose of approximately 4.86 grams to 6.0 grams of pure taurine daily to replicate the peak physiological saturation seen in the murine models 3124. While regulatory bodies like the European Food Safety Authority (EFSA) generally regard taurine as safe at doses up to 6 grams per day, maintaining such extreme physiological saturation over decades poses entirely unknown long-term risks regarding renal clearance, transporter down-regulation, and broader amino acid imbalances 33. As noted by longevity researchers Joseph McGaunn and Joseph Baur, "large, long-term human safety trials are lacking, and the equivalent doses used in the Singh et al. study would be very high in humans... supplementation should be approached with caution" 28.
Interspecies Discordance in Taurine Metabolism and Renal Excretion
The translational gap is further widened by inherent biological differences in how humans and mice synthesize, utilize, and excrete taurine. In mammalian cells, taurine is synthesized endogenously from the amino acid cysteine via the rate-limiting enzyme cysteine sulfinic acid decarboxylase (CSAD) 234. Mice possess a vastly more robust endogenous synthetic capacity than humans. Consequently, the baseline taurine concentrations in the bloodstream of mice and rhesus monkeys can surpass natural human levels by up to ten-fold 8. Therefore, a mouse experiencing an "age-related decline" is starting from a biochemical baseline fundamentally alien to human physiology. Because humans rely much more heavily on dietary intake (exogenous sourcing) to maintain taurine pools, human cellular architecture may be evolutionarily adapted to function optimally at much lower baseline concentrations 835.
Furthermore, the human kidney plays a central, aggressive role in maintaining taurine body pool size and homeostasis. Renal handling of taurine is heavily regulated by the TauT transporter protein, located primarily in the proximal tubule 25. Taurine acts as a vital intracellular osmolyte in the kidney, protecting medullary cells from the extreme hyperosmotic stress of urine concentration. When dietary taurine intake spikes (such as through 6-gram mega-dosing), renal reabsorption pathways rapidly downregulate, and the excess is aggressively excreted in the urine 2526. This robust homeostatic excretion mechanism raises significant questions about whether humans can actually maintain the elevated, "youthful" serum concentrations achieved in the Singh murine models without triggering compensatory renal dumping.
The 2025 Scientific Correction: Challenging the Biomarker Hypothesis
The intense media coverage surrounding the 2023 publication led many in the geroscience community to prematurely coronate taurine as a definitive, universal blood-based biomarker of biological aging. However, the scientific method demands rigorous, independent replication. In 2025, two major studies systematically dismantled the premise that taurine uniformly declines with human aging, exposing critical methodological limitations in the original epidemiological assumptions.
Fernandez et al. (Science, 2025): The Longitudinal Reality
In June 2025, a consortium of researchers led by Maria Emilia Fernandez, Rafael de Cabo, and Luigi Ferrucci at the National Institutes of Health's National Institute on Aging (NIA) published a devastating rebuttal to the taurine deficiency hypothesis in Science 272829. The fundamental flaw identified in the human data presented by Singh et al. was its reliance on cross-sectional analysis - comparing different people of different ages at a single, isolated point in time 3031.
Fernandez et al. instead utilized comprehensive longitudinal data, tracking the exact same individuals repeatedly over their lifetimes. They aggregated data from three massive, geographically distinct cohorts: the Baltimore Longitudinal Study of Aging (BLSA, ages 26 - 100), the Predictive Medicine Research (PREMED) cohort (ages 20 - 68), and the Balearic Islands Study of Aging (ages 20 - 85) 272832. To ensure cross-species validity, they also tracked longitudinal aging data in rhesus monkeys (ages 3 - 32) and mice (ages 9 - 27 months) 28.
The results were entirely antithetical to the 2023 narrative. Circulating taurine concentrations did not universally decline with age. In fact, in most cohorts studied, taurine levels either remained entirely stable or exhibited a steady increase as the subjects aged 283031. The study revealed that intra-individual variance (how much a single person's taurine levels fluctuate day-to-day based on acute diet, stress, and exercise) vastly exceeded any overarching, inter-individual, age-associated trends 2831.
Crucially, the Fernandez study highlighted profound sex-based divergences. Across virtually all demographics, women maintained higher baseline taurine levels than men, and their levels tended to increase more erratically over time 27. In the Emory PREMED cohort and the Longitudinal Aging in Mice study, male taurine levels remained completely flat over their entire lifespans, contradicting the massive declines reported two years prior 2731. Based on these robust, multi-cohort longitudinal findings, Fernandez et al. definitively concluded that low circulating taurine is not a universal feature of aging, formally disqualifying it as a reliable biological clock or biomarker 2829.
Marcangeli et al. (Aging Cell, 2025): The Phenotypic Disconnect
Simultaneously, a separate 2025 investigation by Marcangeli et al., published in Aging Cell, sought to correlate circulating taurine with tangible, functional phenotypic markers of aging. The study evaluated 137 healthy, active, and inactive men ranging from 20 to 93 years of age 443334. The researchers measured an exhaustive battery of physical metrics, including maximal isometric handgrip strength, maximal isometric knee extension, lower limb power, the Timed Up and Go test, step tests, muscle cross-sectional area, and direct cellular assessments of skeletal muscle mitochondrial respiration and ROS production 4434.
Mirroring the NIA findings, Marcangeli and colleagues found absolutely zero correlation between circulating taurine levels and chronological age 44. More damningly, they found no association between baseline taurine levels and muscle mass, structural strength, functional decline, or mitochondrial capacity 3334. The researchers concluded that while taurine may possess profound cytoprotective qualities in a controlled in vitro environment or in genetically isolated, mega-dosed mice, systemic taurine deficiency is definitively not a primary, overarching driver of sarcopenia or biological aging in healthy human populations 3334.
| Study / Year | Study Design & Cohort | Primary Finding regarding Taurine and Aging | Scientific Conclusion |
|---|---|---|---|
| Singh et al. (2023) | Cross-sectional (Mice, Monkeys, EPIC-Norfolk Humans) | Precipitous 80%+ decline in circulating taurine with age across all species. | Taurine deficiency is a primary driver of aging; supplementation extends lifespan. |
| Fernandez et al. (2025) | Longitudinal (BLSA, PREMED, Balearic Islands, Mice, Monkeys) | Taurine remains stable or increases with age. High intra-individual variability. | Taurine is not a universal biomarker of aging. Effects are highly context-dependent. |
| Marcangeli et al. (2025) | Cross-sectional Functional (137 Men, ages 20-93) | No correlation between taurine levels and age, muscle mass, strength, or mitochondrial function. | Challenges the hypothesis that taurine deficiency drives human functional decline. |
These 2025 corrections suggest that taurine therapy is likely highly context-dependent. While reversing an acute deficiency in a highly specific environment - such as in an undernourished, frail population, or a specific metabolic disorder - might yield therapeutic healthspan benefits, blind supplementation in healthy, well-nourished adults is unlikely to universally delay the fundamental aging process 3035.
Epidemiological Insights: Regional Dietary Patterns and Longevity
If circulating taurine is shaped more by environmental exposure and diet than by a strict chronological biological clock, epidemiological data becomes paramount to understanding its long-term health impact. Taurine is found almost exclusively in animal proteins - predominantly seafood, dark poultry meat, and dairy products - and is virtually absent in a strict vegan or plant-based diet 1036. Therefore, evaluating the dietary patterns of the world's longest-lived populations (the "Blue Zones") provides critical real-world insight into the effects of lifelong taurine exposure.
The Okinawan Paradigm vs. The Mediterranean Diet
The traditional Okinawan diet and the Mediterranean dietary pattern (MDP) are the two most heavily researched and validated longevity diets globally. Both diets share foundational similarities: they emphasize plant-based, minimally processed, sustainable foods that foster a robust microbiome and mitigate inflammation 4937. However, their macronutrient distributions and specific micronutrient profiles differ significantly. The Mediterranean diet derives substantial cardiovascular benefits from plant-derived polyphenols and monounsaturated fats (primarily olive oil and nuts), but it is relatively moderate in taurine content due to lower overall seafood consumption, particularly in inland Mediterranean regions 4937.
Conversely, the traditional Okinawan diet presents a fascinating paradox. While it is heavily skewed toward carbohydrates (approximately 85% of caloric intake derived from sweet potatoes, root vegetables, and leafy greens) and notoriously low in fat (6%), it is uniquely rich in specific, high-taurine marine foods, such as local seaweeds, specific fish, and squid 38.
A massive, 20-year epidemiological undertaking known as the CARDIAC study (Cardiovascular Disease and Alimentary Comparison) investigated the relationship between diet and cardiovascular mortality across 61 populations in 25 countries 35. The study utilized 24-hour urinary taurine excretion as a definitive biomarker for seafood intake. The results revealed that native Japanese Okinawans maintained the highest global intake of dietary taurine. This peak taurine consumption correlated directly with the world's lowest rate of ischemic heart disease (a mere 31 deaths per 100,000 people, compared to 110 in the United States) and the highest overall life expectancy 35.
The Impact of Dietary Migration: The Brazilian Cohort
The most striking evidence from the CARDIAC study arose from migration tracking. Researchers examined native Okinawans alongside genetically identical Okinawan emigrants living in Brazil. Two distinct Brazilian cohorts were tracked: one in coastal Sao Paulo, and another living more than 600 miles inland in Campo Grande 35.
The inland Campo Grande cohort, largely isolated from sources of fresh seafood, adopted a localized diet high in roasted terrestrial meats, consuming fish on average only once every two weeks 35. Consequently, their 24-hour urinary taurine levels plummeted. Tragically, the lifespan of this specific Brazilian-Okinawan population was an astounding 17 years shorter than their counterparts living in Japan. This precipitous drop in longevity was driven almost entirely by a massive surge in cardiovascular mortality, directly linked to low fish consumption and the resulting severe reduction in taurine intake 35.
These epidemiological findings align with recent Japanese cohort studies, such as the National Institute for Longevity Sciences-Longitudinal Study of Aging (NILS-LSA). Tracking adults over a ten-year follow-up, researchers found that individuals with an estimated average daily taurine intake of roughly 200 mg to 360 mg per day exhibited significantly better maintenance of knee extension muscle strength into their later years compared to those with lower intakes 3953. Thus, while mega-dosing synthetic taurine remains clinically unproven for extending maximum lifespan, continuous dietary consumption of taurine-rich foods is unequivocally linked to profound cardiovascular resilience and the preservation of functional healthspan.
Human Clinical Horizons: Evidence, Trials, and Efficacy
With the "universal aging biomarker" hypothesis heavily moderated by the 2025 longitudinal data, the clinical focus of taurine research has rightly shifted away from lifespan extension and toward targeted, disease-specific applications focused on healthspan.
Metabolic and Cardiovascular Validation
Where the clinical data remains overwhelmingly positive is in targeted metabolic and cardiovascular support. A rigorous 2024 meta-analysis encompassing 25 randomized controlled trials (RCTs) and over 1,024 adult participants evaluated human taurine supplementation at doses ranging from 500 mg to 6,000 mg per day over durations spanning 5 to 365 days 3354.
The results confirmed that taurine operates as a highly effective, mild metabolic modulator in humans. Supplementation consistently resulted in statistically significant reductions in both systolic and diastolic blood pressure, lowered fasting blood glucose, and improved lipid profiles (reduced circulating triglycerides and LDL cholesterol, alongside elevated HDL) 3355. Furthermore, taurine demonstrated potent anti-inflammatory effects in human subjects, specifically driving down levels of high-sensitivity C-reactive protein (hs-CRP), a critical biomarker of chronic, low-grade systemic inflammation intricately linked to the pathology of cardiovascular disease 3355. In the realm of sports nutrition, independent meta-analyses confirm that pre-workout taurine supplementation provides modest endurance gains and reliably reduces post-exercise markers of acute muscle damage, such as creatine kinase (CK) and lactate dehydrogenase (LDH) 54.
The TauAge Trial
To definitively bridge the gap between targeted metabolic relief and broader indices of systemic aging, the scientific community eagerly anticipates the completion of the TauAge Study (German Clinical Trials Register: DRKS00035066) 40. This prospective, double-blind, randomized, placebo-controlled human intervention trial is specifically designed to evaluate the anti-aging efficacy of taurine mega-dosing 40.
The trial is evaluating the effects of a substantial 4-gram daily dose of taurine (administered as 2g in the morning and 2g in the evening) over a continuous six-month period in healthy older adults aged 55 to 75 4057. Crucially, the trial's primary endpoints are explicitly focused on measuring shifts in biological age biomarkers, global metabolic function, cognitive retention, and physical fitness metrics 4057. Having completed recruitment in late 2025, the results, expected in mid-to-late 2026, will provide the first definitive, high-dose RCT data regarding taurine's ability to manipulate biological aging clocks in a healthy human population, serving as the ultimate clinical test of the 2023 Singh et al. hypotheses.
The Supplement Industry, Regulatory Dynamics, and Misconceptions
The 2023 Science publication triggered an immediate and explosive commercial response. Dietary supplement brands rapidly co-opted the Singh et al. data, leveraging the prestigious journal publication to market taurine as a breakthrough "longevity pill." This aggressive marketing drove a staggering 300% surge in taurine product sales within a single month 41. However, the rapid translation of nuanced murine metabolic science into consumer marketing has generated significant public misconceptions and escalating regulatory friction.
Debunking the Energy Drink Myth
The most pervasive public association with taurine is its ubiquitous presence as a primary ingredient in popular energy drinks like Red Bull. Because these beverages are consumed and marketed for acute neurological stimulation, a widespread public myth exists that taurine itself is a stimulant. Biochemically, this is entirely false.
As established, taurine operates as a GABAergic agonist - an inhibitory neurotransmitter modulator that fundamentally calms the central nervous system, reducing neuronal hyper-excitability 622. Its inclusion in energy drink formulations is not to stimulate, but rather to serve as a physiological buffer, mitigating the jittery side-effects, anxiety, and cardiovascular stress induced by the massive doses of caffeine and simple sugars present in the beverage. Consuming commercial energy drinks in an attempt to achieve longevity is a profound biological paradox; the severe metabolic damage and oxidative stress inflicted by high glycemic loads and chronic sympathetic nervous system stimulation completely negates any theoretical cellular benefits of the trace amounts of taurine included in the can 4243.
Regulatory Scrutiny and the Leukemia Controversy
The supplement industry's aggressive marketing of taurine encountered severe headwinds in mid-2025. Just weeks prior to the Fernandez et al. longitudinal refutation, a highly publicized preclinical study published in Nature revealed a potential oncological risk. The researchers discovered that elevated taurine levels could drive glycolysis and actively induce leukemogenesis in acute myeloid leukemia cells via the sodium- and chloride-dependent taurine transporter (TAUT) 32. The researchers included a brief, cautious warning suggesting that supplementary taurine should be carefully reconsidered in leukemia patients undergoing chemotherapy 43.
Mainstream media outlets immediately sensationalized these in vitro findings, publishing alarming headlines asserting that common energy drink ingredients "feed blood cancer" 43. This forced aggressive defensive posturing from industry watchdogs. The Natural Products Association (NPA), a leading trade organization led by CEO Dr. Daniel Fabricant, vehemently criticized the media for irresponsibly extrapolating a highly specific, in vitro oncology finding into a blanket safety warning for healthy consumers 43. The NPA formally rebutted the panic, emphasizing that taurine possesses an exceptionally well-established safety profile, is critical for bone marrow stem cell function, and is globally recognized as safe by health authorities under normal dietary conditions 43.
The Regulatory Landscape: FDA's Human Foods Program
Amidst this turbulence, the federal regulatory environment governing dietary supplements is undergoing a massive structural shift. In late 2024 and early 2025, the U.S. Food and Drug Administration (FDA) implemented its unified Human Foods Program (HFP), effectively restructuring and absorbing the former Center for Food Safety and Applied Nutrition 4462.
The HFP has explicitly intensified surveillance over new dietary ingredients, focusing heavily on safety substantiation, manufacturing purity, and labeling accuracy in a supplement market now exceeding $60 billion 62. While taurine remains legally protected and broadly permissible under the foundational Dietary Supplement Health and Education Act (DSHEA) of 1994, the FDA strictly prohibits marketers from making "disease claims" 6245. Supplement companies are legally barred from claiming that taurine cures, treats, or prevents diseases - which technically includes the physiological pathology of aging itself.
The independent, scientific watchdog consensus on taurine's marketing, as synthesized by evaluation platforms like Longevity or Bullshit, is stark: "Real ingredient, fake longevity story" 54. These watchdogs correctly assess that while the specific, sensational claim that taurine deficiency actively drives human aging collapsed within two years under the weight of the NIH's longitudinal data, the amino acid itself remains highly legitimate for mild metabolic regulation and exercise support 54. The longevity branding, however, is deemed an overreach designed to command premium pricing.
Conclusion
The trajectory of taurine research over the past three years serves as a profound, illuminating case study in the complexities of geroscience and the perils of translational medicine. The 2023 Singh et al. study undeniably demonstrated that high-dose taurine administration can drastically improve physiological resilience, stabilize mitochondrial function, and suppress cellular senescence in highly controlled, genetically homogenous animal models. However, the subsequent 2025 longitudinal data unequivocally proved that these specific murine metabolic realities do not seamlessly extrapolate to human biology. Circulating taurine does not universally decline with human age, and mega-dosing synthetic amino acids lacks the long-term safety validation required to recommend it as a universal, population-wide anti-aging panacea.
Yet, dismissing taurine's biological utility is equally flawed. Epidemiological evidence derived from the world's longest-lived populations, coupled with rigorous human clinical meta-analyses, confirms that a diet naturally rich in taurine - or targeted supplementation in the explicit presence of metabolic syndrome - yields highly significant cardiovascular, muscular, and anti-inflammatory benefits. Taurine may not be the elusive, master clock of systemic aging that the media initially portrayed, but it remains a vital metabolic buffer and a cornerstone of cellular homeostasis. Until the upcoming TauAge clinical trial provides definitive answers regarding mega-dosing, the scientific consensus suggests that maintaining optimal taurine levels, preferably through a balanced, pescatarian-leaning diet, remains a scientifically sound, low-risk pillar of long-term cardiovascular health and functional preservation.