Role of the gut microbiome in human aging and longevity
The biological process of aging is driven by a complex interplay of genetic, epigenetic, and environmental factors that cumulatively result in the progressive degradation of physiological functions. While the traditional hallmarks of aging - such as genomic instability, telomere attrition, cellular senescence, and mitochondrial dysfunction - have long been the focus of gerontological research, the mammalian gut microbiome has recently been proposed as a critical, distinct hallmark of the aging process 123. The human gastrointestinal tract harbors an immensely diverse and metabolically active ecosystem of trillions of microorganisms. This microbial community functions essentially as a highly integrated endocrine and immune organ, dictating nutrient extraction, pathogenic defense, and systemic metabolic signaling 456.
As the human host ages, the homeostatic relationship between the intestinal mucosa and the microbiome undergoes profound shifts, characterized by altered microbial composition, reduced ecological resilience, and a modified output of bioactive metabolites 78. A central question in modern biogerontology is whether these age-associated shifts represent a passive, correlational downstream consequence of host physiological decline, or if gut dysbiosis actively and causally drives the aging phenotype. Utilizing multi-omics profiling, metagenomic sequencing across diverse centenarian cohorts, and heterochronic fecal microbiota transplantation (FMT) in murine models, contemporary research provides compelling evidence that the microbiome is an upstream, modifiable, and causal modulator of healthspan and lifespan 91011.
Lifespan Trajectories of the Human Gut Microbiome
The human gut microbiome is a dynamic ecosystem that continuously evolves from birth through senescence. Following rapid colonization and diversification during the first three years of life, the adult microbiome stabilizes into a relatively resilient state 412. However, as individuals enter their sixth and seventh decades, this ecological stability typically deteriorates, giving way to significant compositional and functional restructurings 1314.
Ecological Drift and Biological Age Clocks
In a healthy adult, a high degree of alpha diversity (the richness and evenness of microbial species within an individual) is a universally recognized marker of metabolic and immunological robustness 5. As the host ages, chronological progression is frequently accompanied by a decline in this alpha diversity and an increase in beta diversity (inter-individual variation) 11215. This phenomenon is driven by a combination of intrinsic physiological changes - such as reduced gastrointestinal motility, altered gastric acid secretion, and immunosenescence - as well as extrinsic factors like polypharmacy, dietary modifications, and changing dentition 1617.
Advanced machine-learning models and multi-omics integrations have established "microbiome aging clocks" that accurately predict biological age based on microbial composition 418. A large-scale prospective cohort study integrating transcriptome, metabolome, and microbiome data across thousands of individuals revealed that a microbiome age diverging negatively from a healthy reference strongly correlates with increased mortality, heightened systemic inflammation, and diminished physical function 18. Specifically, the gut microbiome clock demonstrated profound predictive capacity for multimorbidity, showing strong associations with 14 out of 16 common non-cancer diseases, particularly metabolic disorders such as fatty liver disease and hypertension 18.
Interestingly, a longitudinal analysis of over 9,000 individuals ranging from 18 to 101 years of age demonstrated that in instances of healthy aging, the gut microbiome becomes increasingly unique and divergent from the general population starting in mid-to-late adulthood 1920. This individualized drift correlates with a steady decline in the dominance of core bacterial genera, such as Bacteroides, which are typically shared across younger humans 20. Older adults exhibiting this high degree of compositional uniqueness tend to display enhanced mobility, faster walking speeds, and better overall survival rates compared to peers whose microbiomes remain stagnant or shift toward pathobiont dominance 1920.
Microbiome Transitions in Frailty
When aging is accompanied by frailty - a multidimensional geriatric syndrome reflecting cumulative physiological decline - the degradation of the gut microbiome is highly pronounced. A multi-omics analysis of 1,821 older adults mapping the Frailty Index (FI) against metagenomic and plasma metabolomic data identified distinct microbial signatures of suboptimal aging 11. Frail older adults exhibit a marked depletion of commensal taxa critical for maintaining epithelial integrity and immunoregulatory capacity 7.
Populations of short-chain fatty acid (SCFA)-producing bacteria, including Faecalibacterium prausnitzii, Eubacterium rectale, Roseburia hominis, and members of the Lachnospiraceae family, are consistently diminished in frail subjects 81421. The loss of these protective taxa is frequently offset by the proliferation of opportunistic pathobionts, particularly within the phylum Proteobacteria 12223. Increased abundances of Streptococcus, Escherichia, Shigella, and sulfate-reducing bacteria like Desulfovibrio are regularly observed 223. This compositional dysbiosis compromises the intestinal epithelial barrier, leading to increased intestinal permeability ("leaky gut") and the subsequent translocation of microbial products, such as lipopolysaccharides (LPS), into the systemic circulation 1424. This endotoxemia activates innate immune receptors, precipitating a state of chronic, low-grade systemic inflammation clinically defined as "inflammaging" 71417.
To summarize the structural evolution of the gut microbiome across human development and aging, the following table synthesizes the dominant microbial trajectories:
| Life Stage / Cohort | Alpha Diversity | Core Commensal Abundance (e.g., Faecalibacterium, Bacteroides) | Pathobiont Abundance (e.g., Proteobacteria) | Gut Barrier Integrity | Systemic Inflammatory Status |
|---|---|---|---|---|---|
| Healthy Adult (20-50 yrs) | High | Highly Abundant | Low / Suppressed | Intact | Low / Regulated |
| Normal Aging (65-80 yrs) | Moderate to Low | Declining | Moderately Elevated | Vulnerable | Mild Inflammaging |
| Frail Elderly (70-90+ yrs) | Very Low | Severely Depleted | Highly Enriched | Compromised (Leaky Gut) | Chronic High (Overt Inflammaging) |
| Successful Aging (100+ yrs) | High (Re-diversified) | Distinctive Unique Patterns | Suppressed / Controlled | Intact / Resilient | Balanced (Anti-inflammatory offset) |
Microbial Signatures of Extreme Longevity
The hypothesis that microbiome degradation strictly follows chronological age is subverted by populations exhibiting extreme longevity. Centenarians (aged 100 years or older) and semi-supercentenarians (aged 105 or older) represent natural models of exceptionally successful aging, characterized by delayed onset or complete avoidance of the chronic diseases that typically account for late-life morbidity 825. Metagenomic sequencing of centenarian cohorts globally has revealed that their gut microbiomes do not exhibit the dysbiotic collapse seen in frail octogenarians; rather, they harbor distinct, youth-associated, or uniquely protective microbial architectures 2627.
Taxonomic Markers of Centenarians
Contrary to the general elderly demographic, centenarians frequently maintain an overall microbial diversity that equals or surpasses that of healthy individuals decades their junior 16212327. This elevated alpha diversity provides a high degree of ecological plasticity and functional redundancy, enabling the centenarian gut to resist environmental stressors, dietary fluctuations, and pathogenic colonization 2128.
Cross-sectional studies spanning varied geographic "blue zones" have isolated several overlapping taxonomic signatures of longevity. The mucin-degrading bacterium Akkermansia muciniphila is overwhelmingly enriched in long-lived populations in Italy, China, and Korea 81629. This bacterium plays an essential role in preserving mucosal barrier function, dampening systemic inflammation, and enhancing host metabolic pathways, including insulin sensitivity 2529. Furthermore, centenarian microbiomes frequently display an enrichment of Bifidobacterium adolescentis and bacteria from the Christensenellaceae family, alongside an unusual stability in Bacteroidetes populations 21262830.
Geographic and Cohort-Specific Variations
While universal patterns exist, the longevity microbiome is heavily influenced by regional diets, environmental exposures, and host genetics, resulting in distinct geographic phenotypes: * The Sardinian Cohort: In the blue zone of Sardinia, Italy, centenarians displayed a reorganized taxonomic structure characterized by a relative depletion of Faecalibacterium prausnitzii and Eubacterium rectale, contrasted with a massive enrichment of Bifidobacterium adolescentis and the methanogenic archaeon Methanobrevibacter smithii 21. The enrichment of methanogens like M. smithii may facilitate optimal microbial fermentation by acting as hydrogen sinks, thereby improving energy extraction from dietary fibers in the aging gut 21. * The Chinese Cohorts: Extensive analyses across China have validated these unique trajectories. A cohort study in Guangxi Province involving 1,575 individuals found that those aged 100 and above maintained a Bacteroides-dominated enterotype with increased species evenness, protecting the ecosystem from chaotic disruptions 2628. In Hainan, researchers observed sex-specific microbiome patterns, where male centenarians exhibited higher overall diversity compared to females, with different species enriched by sex 1426. Functional annotations of Chinese centenarians consistently indicate a preserved capacity for glycolysis, SCFA fermentation, and enhanced xenobiotic (foreign chemical) degradation 2130. * The Japanese Cohort: Research investigating Japanese centenarians (average age 107) discovered a microbiome uniquely enriched with bacterial species capable of generating novel, highly potent secondary bile acids, most notably isoallo-lithocholic acid (isoalloLCA) 1331.
The Gut Virome and Mycobiome in Longevity
While bacteriological profiling dominates microbiome aging research, the broader microbial ecosystem - including the virome (specifically bacteriophages) and the mycobiome (fungi) - plays a critical regulatory role. Recent virome analyses demonstrate that while newborns possess extremely high viral diversity that progressively declines into old age, centenarians reverse this trend, harboring surprisingly rich and diverse gut viral communities 2627. These bacteriophages are not merely parasitic; they carry auxiliary metabolic genes that support specific bacterial functions, including sulfate, methionine, and taurine metabolic pathways 2627. This suggests that the virome actively shapes the functional output of the centenarian gut ecosystem and regulates bacterial host density to maintain long-term homeostasis.
Metabolite-Mediated Mechanisms of Aging
The causal mechanisms by which the gut microbiome influences systemic host aging are fundamentally biochemical. The microbiome operates as an auxiliary metabolic organ, biotransforming dietary substrates and host-derived end-products into thousands of bioactive compounds, collectively known as gut microbiota-derived metabolites (GMDMs) 73233. These GMDMs permeate the intestinal epithelium, enter the enterohepatic circulation, and exert profound immunomodulatory, neuroendocrine, and epigenetic effects across multiple organ systems 73234.
The balance between protective and pathogenic GMDMs dictates whether the microbiome buffers the host against age-related decline or actively accelerates it.
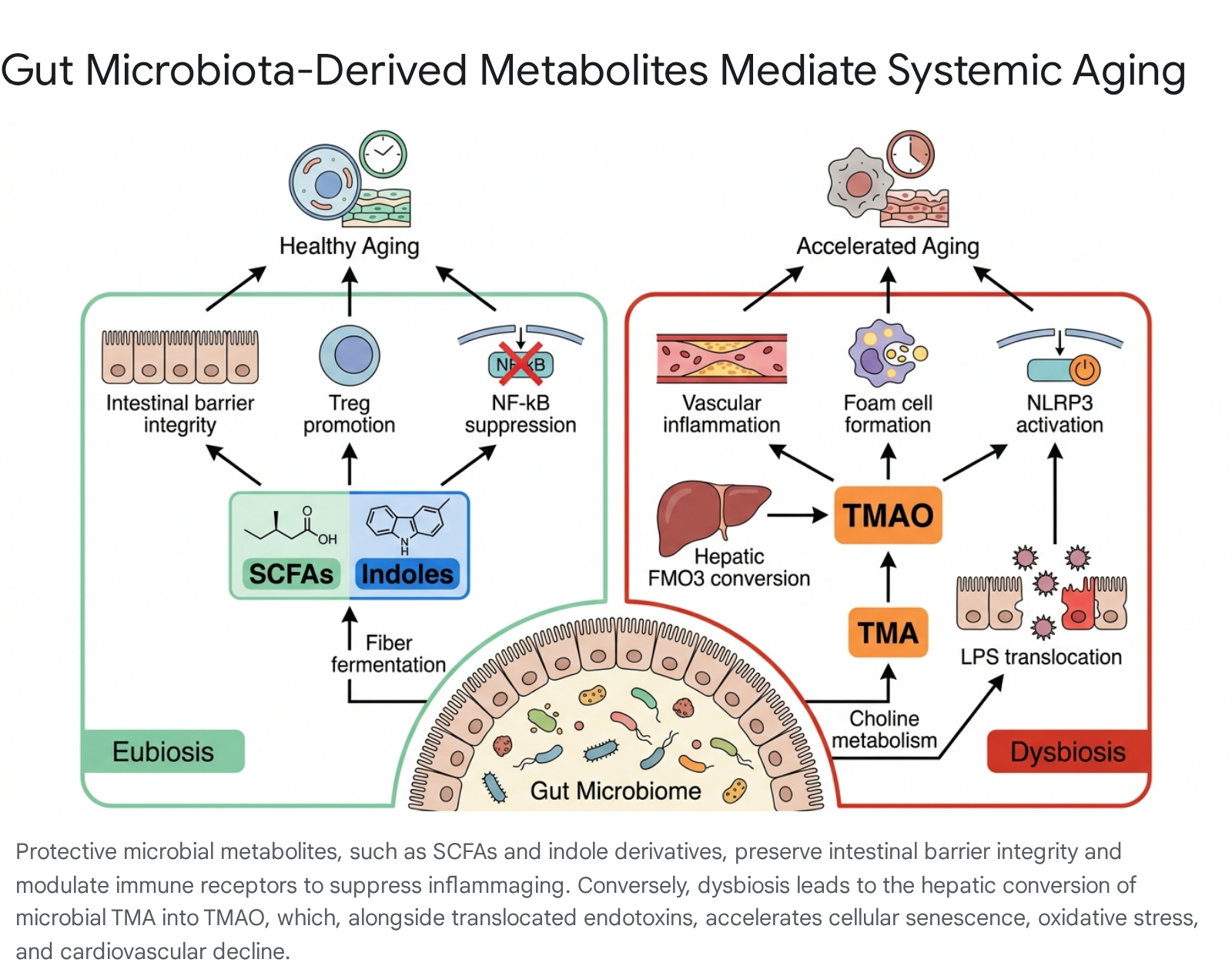
Short-Chain Fatty Acids and the Inhibition of Inflammaging
Short-chain fatty acids (SCFAs), predominantly acetate, propionate, and butyrate, are generated through the anaerobic fermentation of indigestible dietary fibers by specific taxa within the Bacteroidota and Bacillota (Firmicutes) phyla 735. In healthy states, SCFAs - particularly butyrate - serve as the primary energetic substrate for colonocytes, promoting the secretion of protective mucins and maintaining tight junction integrity in the intestinal epithelium 3536.
Systemically, SCFAs act as potent anti-aging immunomodulators. They function as specific ligands for host G protein-coupled receptors (e.g., GPR43, GPR41, FFAR2/3) expressed on diverse immune cells, including macrophages and T lymphocytes 3637. Mechanistically, SCFAs (especially butyrate and propionate) operate as endogenous inhibitors of histone deacetylases (HDACs). This epigenetic regulation actively suppresses the activation of pro-inflammatory transcription factors, primarily Nuclear Factor-kappa B (NF-κB) and Mitogen-Activated Protein Kinases (MAPKs) 73637. The downstream effect is a profound suppression in the synthesis of senescence-associated secretory phenotype (SASP) components, including Interleukin-6 (IL-6), Interleukin-1β (IL-1β), and Tumor Necrosis Factor-alpha (TNF-α), paired with an upregulation of the anti-inflammatory cytokine IL-10 73637. Furthermore, SCFAs promote the differentiation and proliferation of regulatory T cells (Tregs) and regulatory B cells (Bregs), enhancing peripheral immune tolerance 738. The severe age-associated depletion of SCFA-producing taxa removes this critical immunometabolic brake, directly precipitating the systemic inflammaging phenotype 734.
Trimethylamine N-oxide and Accelerated Cardiovascular Senescence
Conversely, age-associated dysbiosis frequently upregulates the production of pro-aging, pathogenic catabolites. Trimethylamine N-oxide (TMAO) is the most rigorously characterized microbiota-derived driver of cardiovascular aging. TMAO biogenesis requires strict host-microbe co-metabolism: gut microbes metabolize specific dietary precursors - such as choline, L-carnitine, betaine, and phosphatidylcholine (abundant in red meat and dairy) - into the intermediate gas trimethylamine (TMA) 3239. TMA rapidly enters the portal circulation and is oxidized in the host liver by flavin-containing monooxygenase 3 (FMO3) to form TMAO 73240.
Circulating TMAO levels increase progressively with chronological age and constitute an independent predictive biomarker for atherosclerosis, hypertension, incident heart failure, and stroke 394142. TMAO accelerates cardiovascular aging through several intersecting molecular pathways: 1. Endothelial Senescence and Dysfunction: Elevated TMAO induces profound oxidative stress and cellular senescence in vascular endothelial and smooth muscle cells. It activates the p53/p21WAF1/Cip1/Rb signaling axis and promotes reactive oxygen species (ROS)-dependent p38-MAPK/NF-κB activation, triggering the release of pro-inflammatory SASP components that damage vascular walls 3341. 2. Accelerated Foam Cell Formation: TMAO actively promotes the oxidation of low-density lipoproteins (LDL) while simultaneously upregulating scavenger receptors (CD36 and SRA) on macrophages. This results in the rapid engulfment of oxidized lipids, accelerating foam cell formation and the development of atherosclerotic plaques 334344. Furthermore, TMAO inhibits reverse cholesterol transport and downregulates hepatic bile acid synthesis (via inhibition of CYP7A1), leading to dangerous systemic cholesterol accumulation 4344. 3. NLRP3 Inflammasome Activation: TMAO directly triggers the assembly and activation of the nucleotide-binding oligomerization domain-like receptor protein 3 (NLRP3) inflammasome, further destabilizing cardiovascular homeostasis, promoting fibrosis, and inducing prothrombotic platelet hyper-reactivity 73743.
Indole Derivatives and Tryptophan Catabolism
The microbial catabolism of the essential amino acid tryptophan yields a variety of indole derivatives, including indole-3-propionic acid (IPA) and indole-3-acetic acid (IAA) 73336. These specific metabolites function as highly potent endogenous ligands for the host Aryl hydrocarbon receptor (AhR) 733. Activation of AhR by IPA in youthful or healthy systems mitigates atherosclerosis, tightens the intestinal barrier by upregulating junctional proteins, and exerts significant neuroprotective effects against oxidative stress 3233.
However, tryptophan metabolism exhibits context-dependent pleiotropy during aging. In states of severe dysbiosis or specific pain sensitization pathways, AhR activation by certain indole derivatives can stimulate the release of IL-6 and TNF-α, which subsequently activate the JAK-STAT3 signaling pathway in dorsal root ganglion (DRG) neurons, exacerbating chronic pain 36. Furthermore, other amino acid derivatives associated with the aging microbiome, such as phenylacetylglutamine (PAGln), have been identified as pathogenic. Elevated PAGln induces DNA damage and mitochondrial dysfunction, driving host cellular senescence via adrenergic receptor-AMPK signaling pathways 3236.
Secondary Bile Acids in Longevity
Primary bile acids, synthesized from cholesterol in the liver and secreted to aid lipid digestion, are biochemically transformed by the colonic microbiota into secondary bile acids 3245. Specific bacteria expressing bile salt hydrolase (BSH) enzymes - such as Methanobrevibacter - deconjugate and modify these structures 45. Secondary bile acids act as signaling molecules targeting host nuclear and membrane receptors, most notably the farnesoid X receptor (FXR) and the G-protein-coupled bile acid receptor (TGR5) 745. Activation of hepatic FXR by microbial bile acids suppresses de novo lipogenesis and promotes fatty acid β-oxidation, preserving metabolic homeostasis 45.
Crucially, the unique microbiome of Japanese centenarians is characterized by an abundance of microbial strains capable of synthesizing a specific secondary bile acid known as isoallo-lithocholic acid (isoalloLCA) 1331. In vitro and murine in vivo assays demonstrate that isoalloLCA exhibits potent antimicrobial properties, strongly inhibiting the growth of dangerous, multidrug-resistant pathogens such as Clostridioides difficile 31. This specialized metabolic output provides a targeted chemical defense mechanism that helps the centenarian gut resist life-threatening infections common in geriatric populations.
The following table summarizes the primary microbiota-derived metabolites and their mechanisms of action in the context of host aging:
| Metabolite Class | Primary Microbial Source | Dietary Precursor | Key Host Receptors | Physiological Mechanism in Host Aging |
|---|---|---|---|---|
| Short-Chain Fatty Acids (Butyrate, Propionate) | Firmicutes, Bacteroidota | Indigestible Fibers | GPR43, FFAR2/3 | Protective: Inhibits HDACs; suppresses NF-κB and SASP cytokines; enhances mucosal barrier; promotes Tregs. |
| TMA / TMAO | Proteobacteria, specific Firmicutes | Choline, L-carnitine | Intracellular cascades (p53, MAPK) | Pathogenic: Induces vascular cellular senescence; upregulates CD36 causing foam cell formation; activates NLRP3 inflammasome. |
| Indole Derivatives (IPA, IAA) | Tryptophan-metabolizing taxa | Tryptophan | Aryl hydrocarbon receptor (AhR) | Context-Dependent: Mitigates atherosclerosis and neuroinflammation; however, specific AhR pathways may exacerbate localized pain sensitization. |
| Secondary Bile Acids (e.g., isoalloLCA) | BSH-expressing bacteria (Methanobrevibacter) | Primary Bile Acids (Host-derived) | FXR, TGR5 | Protective: Suppresses hepatic lipogenesis; unique variants strongly inhibit colonization by multidrug-resistant pathogens (C. difficile). |
| Phenylacetylglutamine (PAGln) | Phenylalanine-metabolizing taxa | Phenylalanine | Adrenergic receptors | Pathogenic: Induces DNA damage and mitochondrial dysfunction; triggers cellular senescence via AMPK signaling. |
Establishing Causality Through Murine FMT Models
While human epidemiological and multi-omics data successfully identify correlations between dysbiosis and biological aging, determining true causality necessitates in vivo manipulation. Murine models utilizing heterochronic Fecal Microbiota Transplantation (FMT) - the transfer of gut microbiota between hosts of vastly different biological ages - have served as the gold standard for proving that the microbiome is an active driver of the aging clock, rather than just a passive reflection of it 7910.
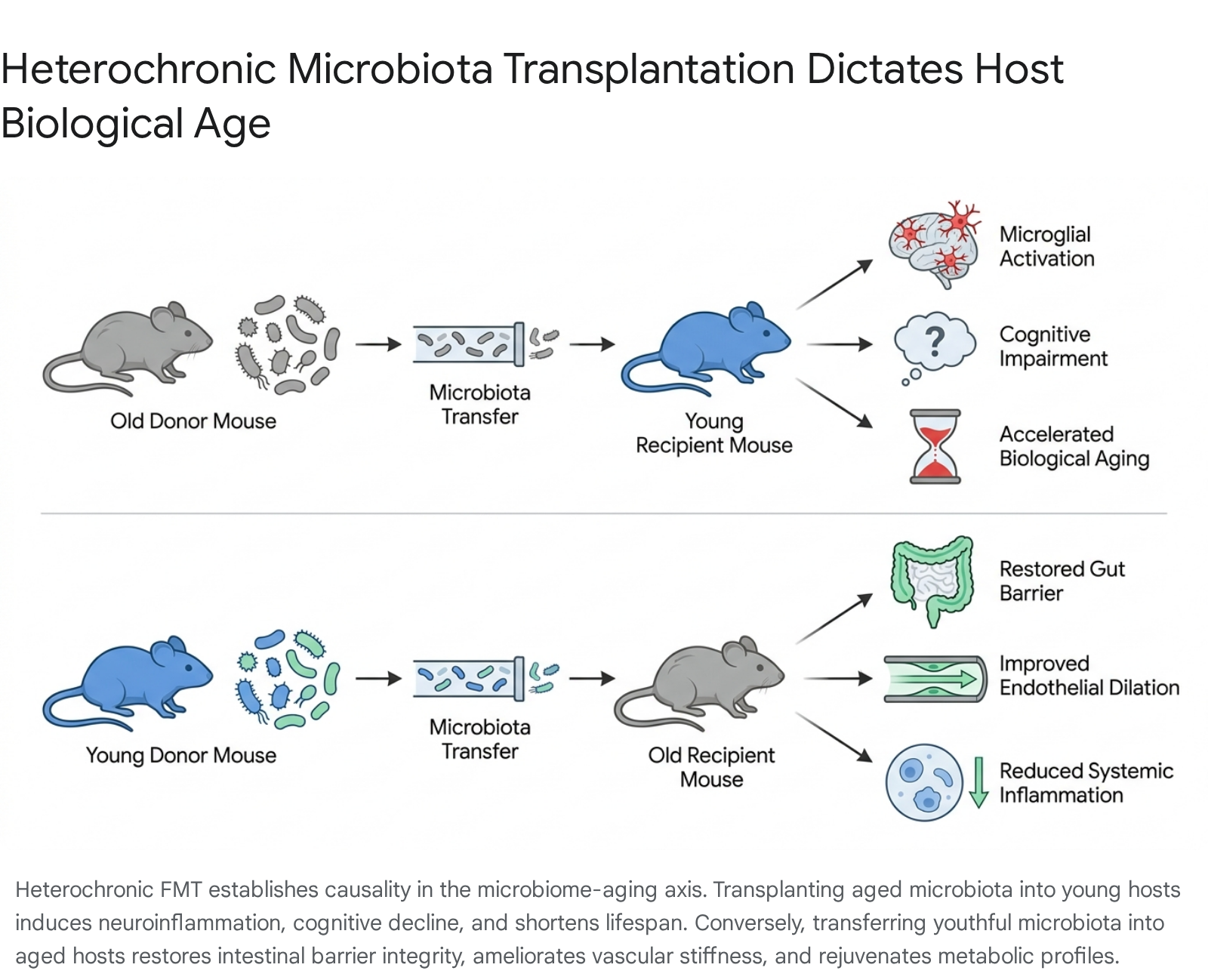
Accelerated Aging via Old-to-Young FMT
When microbiota harvested from aged murine donors is transplanted into young, healthy recipient mice (rendered susceptible via germ-free derivation or broad-spectrum antibiotic clearing), the young hosts rapidly develop localized and systemic phenotypes of advanced biological age 1046.
The central nervous system is highly susceptible to this dysbiotic insult via the gut-brain axis. Old-to-Young FMT accelerates neuroinflammation, characterized by a significant proliferation and activation of Iba-1+ microglia in both the cortex and the corpus callosum, mirroring the neuroinflammatory pathology of natural aging 10. This microglial overactivation translates directly to behavioral and cognitive deficits. In standardized murine cognitive assays, young mice receiving aged microbiota perform poorly in the Y-maze (evaluating spatial working memory) and the Novel Object Location/Recognition tasks (ISO-NOL) 31048. Furthermore, aged microbial grafts induce profound behavioral anxiety, demonstrated by significantly reduced exploratory head-dipping during holeboard test evaluations 46. Beyond the nervous system, the transfer of old microbes induces systemic immune dysfunction, compromises intestinal barrier integrity, increases chronological aging clocks by the equivalent of several human years, and demonstrably shortens the absolute lifespan of the young recipients 46.
Rejuvenation via Young-to-Old FMT
Conversely, introducing youthful microbiota into aged hosts (Young-to-Old FMT) exerts potent, multi-systemic rejuvenating effects 2224. Engraftment of young microbes successfully overrides the entrenched dysbiosis of the aged gut, resulting in an immediate restoration of alpha diversity and a transition toward a youthful metabolomic output 347.
The cardiovascular and metabolic benefits of Young-to-Old FMT are particularly striking. Aged mice receiving young microbiota demonstrate reversed age-associated vascular dysfunction; specifically, they show improved carotid artery endothelium-dependent dilation (mediated by increased nitric oxide bioavailability) and significantly reduced aortic stiffness, linked to the restoration of structural elastin proteins in the arterial wall 2224. The procedure also dramatically reduces vascular oxidative stress by normalizing aortic superoxide production and upregulating the antioxidant enzyme superoxide dismutase 22.
Systemically, young microbiota downregulates the accumulation of age-associated long-chain fatty acids in the blood - a biomarker inversely correlated with longevity - while upregulating beneficial amino acid metabolism 3. The structural integrity of the aging gut is repaired, evidenced by a sharp reduction in serum intestinal fatty-acid binding protein (I-FABP), which limits the translocation of bacterial endotoxins and resolves systemic endotoxemia 2448. At the transcriptomic level, bulk RNAseq analysis of the brain's amygdala in aged recipients reveals that young FMT successfully downregulates inflammatory pathways and upregulates oxidative phosphorylation, effectively ameliorating depression and anxiety-like behaviors 347.
The following table summarizes the divergent systemic outcomes observed in heterochronic murine FMT studies, solidifying the causal role of the microbiome in age-related physiological modulation:
| Biological System | Old-to-Young FMT (Accelerated Aging) | Young-to-Old FMT (Rejuvenation) |
|---|---|---|
| Cognition & Behavior | Impaired spatial memory (Y-maze); increased anxiety (decreased head-dipping) | Improved cognitive retention; alleviation of depressive/anxiety behaviors |
| Neuroinflammation | Massive increase in Iba-1+ microglia in cortex and corpus callosum | Downregulation of neuroinflammatory pathways in the amygdala |
| Cardiovascular Health | Induction of endothelial dysfunction and vascular oxidative stress | Restored endothelium-dependent dilation; reduced aortic stiffness |
| Gut Integrity | Increased intestinal permeability (leaky gut) | Decreased serum I-FABP; tightened intestinal barrier; reduced LPS leakage |
| Metabolism | Shifts toward obesogenic profile; elevated TMAO | Reduced circulating long-chain fatty acids; improved lipid profiles |
| Overall Lifespan | Measurable shortening of absolute lifespan | Improved frailty indices and physical grip strength |
Translational Challenges and Clinical Misconceptions
While preclinical murine models present an optimistic framework for microbiome-mediated rejuvenation, extrapolating these findings directly to clinical human applications presents monumental challenges. The murine gut and human gut differ anatomically, immunologically, and metabolically 23. Furthermore, laboratory mice exist in highly controlled, specific-pathogen-free environments, fed uniform diets, which fails to capture the immense complexity of human lifestyle, genetics, and lifetime pathogen exposure 215.
Engraftment Resistance in the Aged Human Gut
A critical limitation in translating FMT to elderly humans is the reduced ecological plasticity of the aged gut. While germ-free mice readily accept microbial grafts, the human gut is a densely populated, highly entrenched environment. Human-to-mouse translation studies reveal significant engraftment bottlenecks; when microbiota from healthy or frail elderly humans was transplanted into germ-free mice, only 51% to 66% of donor genus-level taxa successfully colonized the recipients after one week 15.
In human clinical contexts, single-dose FMT interventions rarely result in permanent compositional shifts. The established aged microbiome exhibits "colonization resistance," actively outcompeting newly introduced taxa 215. To achieve the rejuvenative effects observed in lifelong murine models - where subjects received repeated transplants every eight weeks alongside antibiotic conditioning - human interventions would likely require sustained, long-term administration, raising concerns regarding feasibility, antibiotic resistance, and regulatory approval 248.
Prevalent Clinical Misconceptions
The rapid popularization of the microbiome in consumer health has generated pervasive clinical misconceptions that impede evidence-based geromedicine. A foundational fallacy is the pursuit of a singular "universal" healthy microbiome profile 549. Scientific consensus acknowledges vast inter-individual heterogeneity; a microbial architecture that confers longevity in an Okinawan centenarian may be biologically incompatible with the dietary and genetic background of an older adult in North America 212649.
Several quantitative paradigms have also been discarded by the scientific community. The long-cited 10:1 ratio of bacterial to human cells is obsolete, and the Firmicutes-to-Bacteroidetes (F/B) ratio - once heavily promoted as a definitive biomarker for obesity and metabolic disease - is no longer considered a reliable or universal diagnostic metric due to extreme strain-level variations 5.
The reductive categorization of microbes as universally "good" or "bad" is similarly inaccurate, as microbial metabolic effects are highly context-dependent 5. For example, while SCFAs are overwhelmingly protective against cardiometabolic disease, emerging evidence indicates that in specific neurodegenerative contexts - such as specific Alzheimer's disease models - high levels of SCFAs may actually exacerbate amyloid-β plaque deposition 25. Similarly, the activation of the AhR receptor by indole derivatives is highly protective in the gut, but can promote neuroinflammatory pain sensitization in dorsal root ganglion neurons 36.
Finally, the assumption that over-the-counter probiotic supplements are universally safe and beneficial for older adults remains unproven and potentially hazardous. In geriatric populations, particularly those experiencing reduced gastrointestinal motility or immune compromise, the indiscriminate introduction of probiotic strains can precipitate or exacerbate Small Intestinal Bacterial Overgrowth (SIBO), leading to severe malabsorption, bloating, and systemic distress 552.
Nutritional Modulations and Gerobiotics
Despite the complexities of FMT, non-invasive interventions utilizing dietary modulations, precision probiotics (gerobiotics), and postbiotics represent the most viable clinical pathways for mitigating age-related dysbiosis.
Dietary Interventions and the NU-AGE Trial
Diet fundamentally sculpts the structural and functional composition of the gut microbiome 3852. The most comprehensive evidence for human microbiome modulation stems from the NU-AGE trial, a large, multi-center, one-year clinical intervention that provided community-dwelling older adults with a specialized Mediterranean-style diet 434. Adherence to this high-fiber, polyphenol-rich diet successfully induced systemic microbiome remodeling. Participants experienced a significant enrichment of health-associated taxa, a reduction in markers of systemic inflammaging, and a measurable decrease in physical frailty 43850. By providing the necessary indigestible carbohydrate substrates, such diets directly fuel the restoration of SCFA-producing bacteria, bypassing the need for exogenous microbial transplants.
Precision Probiotics and Postbiotics
While generic consumer probiotics lack robust clinical validation for lifespan extension, specifically isolated strains - termed "gerobiotics" - show immense promise in targeted applications. Clinical and preclinical trials have identified specific strains of Lactobacillus and Bifidobacterium (e.g., L. rhamnosus, L. paracasei) that effectively reduce chronic inflammatory markers and improve host lipid profiles 27.
In advanced murine frailty models, researchers have successfully engineered synthetic microbial consortia to treat dysbiosis. The "S7" consortium - a defined group of seven health-promoting bacterial taxa (including Barnesiella intestinihominis and Eubacterium rectale) isolated from healthy aging humans - was administered to germ-free mice colonized with a frail human microbiome 15. The S7 species successfully engrafted, resulting in increased systemic acetate levels, reduced colon permeability, and significant reductions in plasma TNF-α concentrations, demonstrating the feasibility of rationally designed live biotherapeutics 15.
Simultaneously, the development of "postbiotics" - the direct administration of beneficial microbial metabolites, inactive bacterial cellular components, or synthesized human milk oligosaccharides (HMOs) - offers a mechanism to bypass the colonization resistance of the aged gut entirely 22752. By delivering the bioactive end-products directly, clinicians can exact precise metabolic control without relying on the unpredictable ecological dynamics of live bacterial engraftment 2738.
Conclusion
The vast body of contemporary research definitively answers the question of causality: the gut microbiome is an active, fundamental driver of the human aging process, not a mere correlational biomarker of physiological decline. Age-associated dysbiosis initiates a pathogenic cascade characterized by the breakdown of the intestinal barrier, the systemic depletion of immunoregulatory short-chain fatty acids, and the dangerous accumulation of pro-atherosclerotic and pro-senescent metabolites such as TMAO and phenylacetylglutamine. This microbial deterioration directly fuels the chronic, low-grade inflammaging that precipitates cardiovascular disease, neurodegeneration, and physical frailty. Conversely, the uniquely diverse and metabolically specialized microbiomes of centenarians - capable of neutralizing multidrug-resistant pathogens and maintaining robust barrier integrity - demonstrate that preserving ecological resilience within the gut is paramount for achieving exceptional longevity. While translating the striking rejuvenative effects of murine fecal microbiota transplantation to humans faces significant biological and regulatory hurdles, the rapid advancement of precision nutrition, defined microbial consortia, and targeted postbiotics presents a transformative frontier. Modulating the gut microbiome offers one of the most accessible and profound therapeutic avenues for extending human healthspan and mitigating the global burden of age-related disease.