Reproductive aging and ovarian reserve diagnostics and extension
Female reproductive aging represents one of the most profound physiological transformations in human biology, serving as the earliest manifestation of systemic organismal senescence. Historically, clinical perspectives on ovarian decline have been narrowly focused through the prism of procreation, fertility preservation, and the management of climacteric symptoms. However, an accumulating body of evidence demands a paradigm shift: the trajectory of reproductive aging is a critical determinant of midlife and late-life systemic health. The cessation of ovarian function, culminating in natural menopause, precipitates a cascade of physiological alterations that dramatically influence longevity and morbidity profiles. Advancements in genomic analysis, precision diagnostics, and novel therapeutic interventions are currently redefining the boundaries of the reproductive lifespan. This report provides an exhaustive analysis of the contemporary landscape of reproductive aging, explicitly differentiating the utility of ovarian reserve biomarkers, elucidating the genetic architecture of menopause across diverse populations, and critically evaluating the frontiers of fertility extension, including pharmacological mTOR inhibition, cryopreservation, and in vitro gametogenesis.
The Systemic Ramifications of Reproductive Senescence
The menopausal transition is not an isolated reproductive event but a systemic physiological inflection point. Emerging epidemiological data indicate that the temporal onset of natural menopause, alongside prior reproductive milestones such as the age at menarche and parity, fundamentally shapes long-term health trajectories 12. Female reproductive decline occurs decades before the onset of other systemic age-related dysfunctions, driven by the continuous depletion of the ovarian follicle pool and a precipitous, concurrent decline in oocyte quality 1.
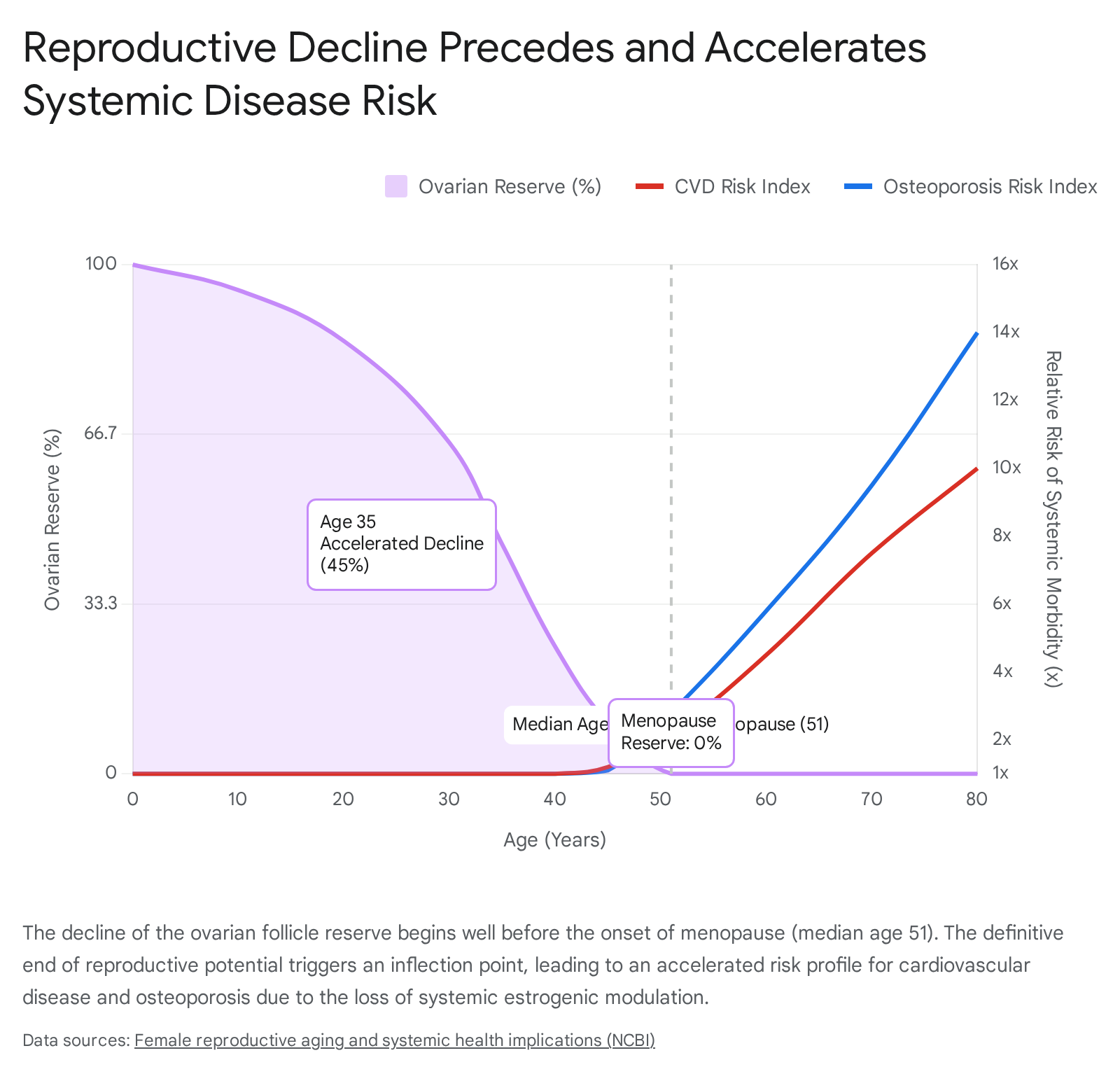
The loss of ovarian endocrine function, primarily the withdrawal of circulating estradiol and inhibin, initiates widespread pathobiological changes that increase a woman's vulnerability to chronic, age-related diseases.
The cardiovascular implications of reproductive aging are particularly profound. Cardiovascular disease (CVD) remains the leading cause of mortality in aging women, and the risk escalates significantly following the menopausal transition 1. This elevated risk is mediated by unfavorable alterations in cardiometabolic health, including the development of dyslipidemia, increasing insulin resistance, and a marked redistribution of body fat characterized by a sharp accumulation of visceral adipose tissue 13. Comprehensive genetic analyses utilizing Mendelian randomization have demonstrated causal relationships between reproductive factors and cardiovascular outcomes. An earlier first birth, a higher number of live births, and early menarche are genetically linked to a higher risk of atrial fibrillation, coronary artery disease, heart failure, and stroke 2. Much of the increased risk associated with early menarche appears mediated by higher body mass index, suggesting that metabolic factors are tightly intertwined with early reproductive maturation 2. Furthermore, women who suffer from conditions that disrupt normal reproductive transitions, such as Premature Ovarian Insufficiency (POI) or Polycystic Ovary Syndrome (PCOS), face compounded cardiometabolic risks. PCOS, for instance, confers a higher risk of developing hypertension post-menopause and is intimately linked to non-alcoholic fatty liver disease (NAFLD) with elevated rates of fibrosis 1. Pregnancy complications, such as preeclampsia and other hypertensive disorders of pregnancy, also unmask underlying cardiovascular vulnerabilities, with affected women showing significantly higher long-term risks of myocardial infarction and heart failure 3.
Beyond the cardiovascular system, reproductive aging dictates skeletal and neurological health outcomes. Menopause is the single greatest risk factor for osteoporosis, with bone mineral density loss beginning approximately one year prior to the final menstrual period and accelerating rapidly during the transition 1. This imbalance between bone formation and resorption results in irreversible deficits in bone microarchitecture, affecting millions of women globally and resulting in high morbidity due to fragility fractures 14.
Neurologically, post-menopausal women exhibit a heightened vulnerability to cognitive dysfunction and neurodegenerative pathologies, most notably Alzheimer's disease. The prevalence of Alzheimer's disease is significantly higher in women aged 65 and older compared to men, a disparity heavily influenced by the loss of the neuromodulatory and neuroprotective functions of estrogen 1. Large-scale cohort studies, such as retrospective analyses of 273,240 women from the UK Biobank, indicate that events related to shorter cumulative exposure to endogenous estrogen, such as older age at menarche, early onset of menopause, and hysterectomy, strictly correlate with higher all-cause dementia risks 5. Conversely, longer reproductive spans, pregnancy, and older age at natural menopause are associated with a degree of neuroprotection, underscoring the critical necessity of viewing the ovaries as central regulators of systemic healthspan rather than strictly reproductive organs 15.
Diagnostic Modalities and the Divergent Utility of Ovarian Reserve Markers
The clinical assessment of the ovarian reserve has become a cornerstone of modern reproductive endocrinology. The most frequently utilized biomarkers include Anti-Müllerian Hormone (AMH), Antral Follicle Count (AFC) assessed via transvaginal ultrasonography, and early follicular phase (day-3) Follicle-Stimulating Hormone (FSH) alongside Estradiol. However, the interpretation of these markers is frequently fraught with clinical misunderstanding. It is imperative to explicitly differentiate between the capacity of these markers to predict quantitative responses to exogenous pharmacological stimulation versus their profound inability to predict the qualitative likelihood of natural, unassisted conception.
Predictive Efficacy in Assisted Reproductive Technology (ART)
In the context of in vitro fertilization (IVF) and controlled ovarian hyperstimulation (COH), the ovarian reserve markers operate as highly robust quantitative predictors. The ultimate goal of COH is to rescue a cohort of follicles from normal physiological atresia to achieve multifollicular development, thereby maximizing oocyte yield for retrieval. According to comprehensive 2023 and 2024 meta-analyses, AFC and AMH are the most accurate and reliable indicators of ovarian response in COH candidates 67.
AMH is a dimeric glycoprotein belonging to the transforming growth factor-$\beta$ (TGF-$\beta$) family, exclusively secreted by the granulosa cells of growing preantral and small antral follicles 89. Because AMH production diminishes as follicles mature and become FSH-dependent, its serum concentration directly reflects the size of the recruitable follicular pool 8. Furthermore, AMH levels are largely independent of the menstrual cycle phase, making it a highly convenient clinical tool. AFC, defined as the total number of follicles measuring 2 - 10 mm in both ovaries, similarly provides a direct morphological assessment of the recruitable cohort.
While previous literature debated the superiority of AMH versus AFC, contemporary meta-analyses - such as a 2024 review utilizing the Der Simonian-Laird random-effects model across 26 studies - indicate that both markers possess high diagnostic accuracy for anticipating poor (typically $\le$ 3 oocytes) or excessive (typically >15 oocytes, leading to OHSS risk) retrieval outcomes 6710. Comparison of summary estimates shows that AFC is marginally superior in detecting high-response categories and slightly outperforms AMH overall, while AMH demonstrates an exceptionally high diagnostic odds ratio (DOR) and slightly higher sensitivity for predicting poor response, particularly in limited-resource environments 67. Utilizing predictive multivariate models that combine AMH and AFC yields the highest predictive Area Under the Curve (AUC) for tailoring gonadotropin dosages, outperforming either metric alone 610.
Conversely, basal day-3 FSH is significantly inferior as a predictive biomarker for COH. FSH levels fluctuate continuously based on negative feedback from ovarian estradiol and inhibin B, and frequently remain within normal parameters until the late phases of diminishing ovarian reserve (DOR) 6. Consequently, FSH is deemed an unreliable single definitive test for identifying poor responders in modern ART protocols compared to the direct assessments provided by AMH and AFC 610.
The Fallacy of Predicting Natural Fecundability
While AMH and AFC are unparalleled in predicting oocyte quantity upon massive exogenous stimulation, they hold virtually zero predictive value for oocyte quality or the probability of natural conception. In an unmedicated, natural menstrual cycle, conception relies on the physiological selection, dominance, and ovulation of a single competent oocyte. This qualitative biological phenomenon is fundamentally disconnected from the total number of resting or recruitable follicles in the ovarian reserve.
Guidelines established by the American Society for Reproductive Medicine (ASRM) and the American College of Obstetricians and Gynecologists (ACOG) strictly caution against the use of AMH to counsel non-infertile women regarding their current fecundability or future reproductive potential 910. Landmark prospective cohort studies definitively support this stance. The Time to Conceive study, which followed women aged 30 to 44 without known infertility risk factors, demonstrated that women with severely low AMH levels (<0.7 ng/mL) or high FSH values (>10 IU/L) achieved identical cumulative natural pregnancy rates after 6 and 12 cycles of attempting pregnancy compared to women with normal reserve markers 910. Extended follow-up of this cohort over three years confirmed that low AMH did not correlate with worse future fecundability, lower live birth rates, or higher rates of future infertility 9.
Similarly, the EAGER (Effects of Aspirin in Gestation and Reproduction) trial, which enrolled 1,202 women with a history of one or more miscarriages, revealed that a low AMH (<1 ng/mL) did not negatively impact the subsequent time required to conceive naturally compared to women with normal values (1.0 - 3.5 ng/mL) 910. A 2024 retrospective analysis of patients with unexplained infertility matched with severe male-factor infertility controls corroborated that the frequency of low AMH (<0.7 ng/mL) did not statistically differ between groups, confirming that a low ovarian reserve does not inherently reduce the chance of natural pregnancy provided the woman remains ovulatory 11. Ultimately, patient age remains the singular, most accurate prognosticator of oocyte chromosomal competence and natural conception probability.
Diagnostic Criteria for Premature Ovarian Insufficiency (POI)
The diagnostic application of these biomarkers differs significantly when evaluating pathological reproductive aging, such as Premature Ovarian Insufficiency (POI). POI is characterized by the loss of ovarian activity before the age of 40. According to the 2024 and 2025 ASRM evidence-based guidelines, the primary diagnostic criteria for POI are strictly clinical and endocrine: the presence of disordered menstrual cycles (spontaneous amenorrhea or oligomenorrhea) lasting for at least four months, paired with an elevated FSH concentration greater than 25 IU/L 12. This FSH elevation must be confirmed with a secondary measurement taken four to six weeks later if there is diagnostic uncertainty.
Crucially, the ASRM emphasizes that AMH should not be utilized as a primary diagnostic test for POI. AMH testing may hold secondary utility to confirm a POI diagnosis only in cases where FSH results are equivocal or inconclusive, and the results must be interpreted within the broader clinical context 12. Healthcare professionals are specifically advised against routinely performing AMH testing to predict the future onset of POI in healthy populations due to insufficient evidence of long-term diagnostic accuracy 12.
Structured Biomarker Comparison
The distinct roles of these primary ovarian reserve markers are synthesized in the table below, distinguishing their utility in assisted reproductive technologies from their limitations in natural family planning and diagnostic medicine.
| Biomarker | Physiological Source | Utility in IVF / COH Prediction | Utility in Natural Conception / Menopause Prediction | Clinical Limitations |
|---|---|---|---|---|
| Anti-Müllerian Hormone (AMH) | Granulosa cells of preantral and small antral follicles. | High. Accurate predictor of poor and hyper-response. Guides initial gonadotropin dosing and OHSS risk mitigation. | None. Does not predict natural fecundability, time to pregnancy, or clinical pregnancy rates. | Cannot predict oocyte quality. Suppressed temporarily by oral contraceptives or intense mTOR inhibition. |
| Antral Follicle Count (AFC) | Transvaginal ultrasound visual count of follicles measuring 2 - 10 mm. | High. Marginally superior to AMH for identifying high responders. Highly accurate alongside AMH. | None. Irrelevant for predicting unassisted pregnancy rates in ovulatory females. | Highly operator-dependent. Requires specialized ultrasound equipment and trained sonographers. |
| Basal FSH / Estradiol (Day 3) | Anterior pituitary (FSH) responding to negative feedback from ovarian estradiol/inhibin. | Low/Moderate. Late indicator of diminished reserve. Less sensitive than AMH/AFC. | Diagnostic for POI. FSH > 25 IU/L (repeated) defines premature ovarian insufficiency alongside amenorrhea. | Highly variable across and within cycles. Masked by early elevation of estradiol. |
Genetic Architecture of Natural Menopause Timing: Diverse Populations and Recent GWAS Discoveries
The timing of the Age at Natural Menopause (ANM) is a complex, polygenic trait characterized by substantial heritability, estimated between 44% and 85% via twin studies 13. While environmental and epidemiological factors - most notably cigarette smoking - exhibit a clear dose-response relationship in accelerating menopause onset, the fundamental temporal bounds of the reproductive lifespan are genetically encoded 1316. Historically, understanding the genetic determinants of ANM was severely constrained by a reliance on candidate gene association studies and genome-wide association studies (GWAS) that overwhelmingly restricted their cohorts to women of European ancestry 141516.
However, prioritizing 2023 and post-2023 literature reveals a critical expansion into multi-ethnic and trans-ethnic genetic mapping, illuminating shared mechanisms and ethnic heterogeneities. Recent large-scale GWAS meta-analyses encompassing over 200,000 women have expanded the number of identified independent genomic loci associated with ANM to 290, strengthening the understanding of the complex trait 1317. Crucially, investigations analyzing these loci across diverse populations - including African, Hispanic/Latina, Asian American, Native Hawaiian, and American Indian/Alaskan Native cohorts - have confirmed the transferability of European-discovered loci 13141517. For example, the multi-ethnic Population Architecture using Genomics and Epidemiology (PAGE) Study, analyzing over 45,000 women of diverse ancestries, identified significant trans-ethnic associations at both novel and established loci 1518. Assessment of nearly 78,000 women of East Asian descent successfully identified these genetic signals, though notable heterogeneities in allele frequencies and effect sizes were observed between Asian and European populations 1317. A focused replication study among 3,533 Chinese women successfully extended multiple GWAS-identified genetic susceptibility loci to Asian-ancestry populations, validating associations in specific genes 19. Furthermore, analyses in Korean populations identified synergistic interactions between intronic SNPs on ADAMTS9 and SMAD3 genes for age at natural menopause 14.
The Primacy of DNA Damage Repair (DDR) Pathways
The most profound biological insight generated by recent GWAS data is the overwhelming implication of the DNA Damage Repair (DDR) pathway in the regulation of the ovarian lifespan. Pathway analyses reveal that nearly two-thirds of the known ANM-associated single nucleotide polymorphisms (SNPs) reside within or regulate genes integral to maintaining genomic integrity, such as EXO1, HELQ, UIMC1, FAM175A, FANCI, BRSK1, MCM8, and POLG 1719202421.
The prominence of DDR pathways fundamentally rewrites the biological narrative of ovarian aging. The primordial follicle pool is established entirely during fetal development and remains arrested in prophase I of meiosis for decades. Over this prolonged, dormant period, the oocytes are highly susceptible to the accumulation of DNA double-strand breaks and oxidative genomic damage. The capacity of a woman's biological machinery to repair this damage dictates the rate of follicular atresia. Individuals carrying genetic variants that confer highly efficient DNA repair mechanisms exhibit a delayed age of natural menopause, whereas minor defects in these pathways accelerate follicular depletion, resulting in earlier menopause or POI 161720.
A sophisticated 2023 two-stage mixed linear-association analytical model applied to over 173,000 UK Biobank participants uncovered that almost three-quarters of genetic variants tied to menopause timing exhibit age-specific effects 22. This research, validated against an Estonian Biobank cohort, highlighted that early menopause features and ovarian depletion were heavily driven by these DNA damage processes 22. Conversely, late menopause variants heavily overlapped with altered genetic risk pathways for systemic conditions such as heart failure, hyperlipidemia, and breast cancer, underscoring the tight causal interplay between reproductive longevity and overall somatic health 22.
Furthermore, contemporary research has moved beyond static genomic sequences to incorporate functional metabolomics. A 2024 metabolome-wide Mendelian randomization study cross-referenced 684 circulating metabolites with GWAS data from the ReproGen consortium to identify causal metabolic pathways governing ANM 23. The analysis isolated 76 prioritized metabolites, heavily highlighting the glycerophosphocholine class. Specifically, elevated levels of omega-3 and polyunsaturated fatty acids were identified as playing a direct causal role in delaying the age of menopause 23. This intersection of genomics and metabolomics underscores that while the baseline reproductive lifespan is genetically anchored via DDR efficacy, downstream metabolic environments actively modulate the rate of physiological decline.
Established and Maturing Fertility Preservation Strategies
In response to the physiological inevitability of reproductive aging, medical science has established sophisticated methodologies to uncouple the genetic quality of gametes from chronological age. These methodologies rely heavily on the utilization of extreme low-temperature physics - specifically, ultra-rapid cooling through vitrification - to halt biological time within reproductive tissues, preventing the formation of damaging intracellular ice crystals that plagued earlier slow-freezing techniques 28.
Oocyte Cryopreservation: Medically Indicated vs. Elective
Mature oocyte cryopreservation (OC) is recognized by the ASRM as a fully established, standard-of-care fertility preservation technique for postpubertal females 24. The process requires controlled ovarian stimulation followed by the surgical retrieval of oocytes, which are immediately vitrified 24. OC provides significant reproductive autonomy compared to embryo cryopreservation, as it circumvents the immediate necessity for a sperm source and grants the patient singular control over future gamete disposition 24.
The indications for OC have bifurcated into two distinct clinical pathways: medically indicated OC and planned (elective) OC. Medically indicated OC is deployed for patients facing highly gonadotoxic therapies, such as systemic chemotherapy or pelvic irradiation for malignancies, or for individuals with genetic predispositions to premature ovarian failure 24. Because cancer therapies severely deplete the primordial follicle pool and increase the risk of subsequent aneuploidy, expedited OC prior to treatment initiation is critical. Post-treatment attempts at OC yield significantly compromised outcomes; clinical data suggest higher cycle cancellation rates, fewer retrieved oocytes, and elevated risks of birth defects if gametes are exposed to gonadotoxins prior to preservation 24.
Conversely, planned OC - often termed "social egg freezing" - is utilized by healthy women intending to circumvent age-related fertility decline 2526. The ASRM Ethics Committee has firmly established that planned OC is ethically permissible, affirming its role in supporting reproductive autonomy 25. Clinical outcome data for planned OC emphasize that chronological age at the time of retrieval and the total number of mature oocytes vitrified are the overriding determinants of future live birth efficiency 2427. Retrospective cohort analyses aggregating cycles from multiple centers illustrate a stark age-related decline in efficiency. For women under 30 years of age, the estimated live birth efficiency per warmed oocyte is roughly 7.4%. This efficiency drops to 7.0% for women aged 30 - 34, 6.5% for women 35 - 37, and plummets to 5.2% for women over 38 2627. Consequently, mathematical modeling dictates that a woman under 35 may require approximately 15 to 20 vitrified oocytes to secure a high probability of one live birth, whereas a woman over 35 would require a significantly larger cohort - often necessitating multiple expensive stimulation cycles 2627. Despite these age-related constraints, thaw survival rates, fertilization, and clinical pregnancy rates from autologous vitrified oocytes are now statistically comparable to cycles utilizing fresh oocytes within equivalent age brackets, with fertilization rates hovering around 70% 2628.
Ovarian Tissue Cryopreservation (OTC)
The most notable recent shift in clinical guidelines concerns Ovarian Tissue Cryopreservation (OTC). As of the newly released ASRM 2026 committee opinions, OTC is officially no longer considered an experimental procedure and is established as an acceptable clinical technique 2429.
OTC involves the surgical excision and vitrification of the ovarian cortical tissue, which harbors thousands of dormant primordial follicles. The tissue can subsequently be autotransplanted - typically orthotopically back to the ovarian fossa - to restore both endocrine function and natural fertility 2430. This technique represents a monumental advancement for specific patient populations. It is the primary, recommended strategy for prepubertal females facing gonadotoxic therapy, as their immature hypothalamic-pituitary-gonadal axis precludes standard hormonal stimulation for oocyte retrieval 24. Furthermore, OTC is indicated for adult women requiring immediate, urgent oncological intervention where the two-to-three-week delay required for standard controlled ovarian stimulation is clinically unacceptable 24. The removal of the experimental label from OTC in 2026 solidifies its integration into comprehensive, medically indicated fertility preservation programs, broadening the accessibility of reproductive safeguarding, while acknowledging that institutional capacity dictates the choice between slow freezing and vitrification methods 30.
Next-Generation Fertility Extension: Pharmacological and Cellular Interventions
While cryopreservation relies on ex vivo tissue storage, an aggressive new wave of research focuses on in vivo therapeutic interventions to delay the depletion of the ovarian reserve and rejuvenate oocyte competence. These pharmacological approaches leverage modern longevity science, targeting the cellular hallmarks of aging.
mTOR Inhibition: The Rapamycin Revolution
The mechanistic target of rapamycin (mTOR) pathway serves as a master regulator of cellular metabolism, growth, and proliferation. In the ovary, the mTOR complex 1 (mTORC1) acts as the primary metabolic sensor that triggers the activation and recruitment of dormant, primordial follicles into the growing pool 9. Hyperactivation of this pathway with age leads to the accelerated "burnout" of the ovarian reserve. Consequently, pharmacological inhibition of mTOR using rapamycin is emerging as a highly promising intervention for ovarian life extension.
Translational research has rapidly progressed to human clinical trials. The landmark VIBRANT trial (Validating Benefits of Rapamycin for Reproductive Aging Treatment), conducted at Columbia University by researchers Dr. Zev Williams and Dr. Yousin Suh, represents the vanguard of this effort 363132. Initiated in perimenopausal women (aged 35 - 45), the trial utilizes a low, pulsatile dose of rapamycin (5 mg weekly) over 12 weeks 936. Preliminary 2024 and 2025 institutional data indicate that this specific mTOR inhibition protocol can effectively slow the rate of follicular loss by approximately 20% 3631. While an untreated ovary typically exhausts roughly 50 follicles per month during this age bracket, rapamycin administration theoretically restricts this attrition to 15 to 40 follicles, physically elongating the reproductive window and potentially delaying the onset of menopause 931.
Furthermore, rapamycin demonstrates efficacy in improving the qualitative outcomes of existing fertility treatments. A pivotal 2025 randomized clinical trial published in Cell Reports Medicine evaluated a daily 1 mg dose of rapamycin administered for 3-4 weeks during the ovarian stimulation phase in 100 women with prior failed IVF cycles 36. This low-dose intervention resulted in significantly higher yields of quality zygotes and tripled the rate of blastocyst development (27.5% vs 7.7%) 36. Clinical pregnancy rates doubled compared to the placebo control (50% vs 28%) 36. However, clinicians note that systemic mTOR inhibition suppresses the recruitment of follicles that produce AMH, rendering intra-treatment AMH draws clinically useless for assessing absolute ovarian reserve; accurate baseline measurements must be taken via transvaginal ultrasound prior to initiating the drug, and washout protocols are required prior to natural conception or unmedicated IVF 9.
NAD+ Precursors and Sirtuin Activation
The age-related decline in Nicotinamide Adenine Dinucleotide (NAD+) represents a core mechanism driving mitochondrial dysfunction and oxidative stress within the aging oocyte. Preclinical data heavily suggested that augmenting NAD+ via dietary precursors, such as Nicotinamide Mononucleotide (NMN) or Nicotinamide Riboside (NR), could reverse these defects by enhancing mitochondrial metabolism and activating sirtuins (specifically SIRT1), which are critical for DNA repair and longevity 1633.
In 2025, a crucial retrospective analysis published in Human Reproduction validated these pre-clinical hypotheses in a human cohort. The study evaluated 50 young women (under 35) suffering from Diminished Ovarian Reserve (DOR) who were pretreated with oral NMN 33. The NMN cohort exhibited vastly improved follicular health; follicle size improved significantly (from 12.02 $\pm$ 1.38 mm to 20.38 $\pm$ 1.46 mm), and follicular vascularity was notably higher (77.6% Grade 4 vs. 59% Grade 3 in controls) 33. Furthermore, endometrial thickness was significantly enhanced (10.02 $\pm$ 1.03 mm vs. 7.74 $\pm$ 0.67 mm) 33.
Most significantly, patients required a drastically lower total dose of exogenous gonadotropins (267 $\pm$ 37.99 IU vs. 471 $\pm$ 98.07 IU) and fewer injections to achieve stimulation, and realized a 48% positive clinical pregnancy rate (12 pregnancies) compared to just 24% (6 pregnancies) in the control group 33. The temporal efficacy was also enhanced, with the NMN cohort conceiving within an average of three months, halving the time to conception compared to the 6-9 months seen in controls, alongside a reduced abortion rate (8.33% vs 16.66%) 33. Although large-scale randomized controlled trials are required to define exact dosing parameters and long-term consequences, NMN supplementation has cemented its role as a viable adjuvant therapy to restore metabolic homeostasis in aging gametes.
Senolytics, Spermidine, and Metformin
Cellular senescence - the accumulation of damaged cells that cease division but actively secrete pro-inflammatory cytokines (the Senescence-Associated Secretory Phenotype, or SASP) - drives ovarian tissue fibrosis and follicular atresia.
Senolytics: Drugs designed to selectively induce apoptosis in senescent cells represent a major longevity frontier. Early human Phase 1 trials utilizing the combination of Dasatinib and Quercetin (D+Q) successfully demonstrated a reduction in senescent cell burden in human adipose tissue, specifically lowering p16INK4A and p21CIP1-expressing cells within 11 days of a 3-day oral course 4041. While systemic "anti-aging" claims remain premature, the biological proof-of-concept exists in humans, and targeted senolytic clearance in fibrotic ovarian environments is an active area of investigation 4034.
Spermidine: A naturally occurring polyamine that induces autophagy - the cellular "recycling" mechanism crucial for clearing damaged mitochondria and denatured proteins. A 2026 comprehensive review in Nature highlighted spermidine's ability to extend mammalian lifespan by 10-15% and noted its high safety profile in human clinical trials (e.g., 40 mg/day) 4344. Human data from the Bruneck Study showed that higher spermidine intakes correlated with a 40% reduced risk of fatal heart failure and lower all-cause mortality 44. By enhancing autophagy, spermidine preserves the integrity of the oocyte's complex mitochondrial network, which is vital for maintaining competence during embryo division 44.
Metformin: Extensively used as a glucose metabolism modulator, metformin has shown profound anti-aging effects independent of glycemic control. In mammalian models, prolonged metformin administration decreases ovarian fibrosis, maintains regular estrous cycles, and elevates systemic E2 levels 1635. Crucially, metformin exerts these effects by upregulating SIRT1 expression and significantly lowering markers of oxidative damage (8-OHdG, 4-HNE) and senescence (P16) within the ovarian microenvironment, expanding the primordial follicle pool 35.
In Vitro Gametogenesis (IVG): The Frontier of Reproductive Science
The most radical and technologically ambitious approach to circumventing reproductive aging is In Vitro Gametogenesis (IVG). This technology aims to generate fully functional human gametes (sperm or eggs) from somatic, non-reproductive tissues, completely bypassing the physiological limitations of the biological ovarian reserve 3637. By utilizing induced pluripotent stem cells (iPSCs) or direct cellular reprogramming, IVG holds the theoretical potential to allow post-menopausal women, cancer survivors devoid of gonadal tissue, and even same-sex couples to produce genetically related offspring 363839.
While IVG in murine models has successfully yielded viable offspring - including mice with two biological fathers through the manipulation of imprinted genes - translating this technology to human biology has proven exceptionally difficult due to the rigorous complexity of chromosomal mechanics and meiosis 38404142.
The 2025 "Mitomeiosis" Breakthrough and its Limitations
A landmark proof-of-concept study published in Nature Communications in late 2025 by researchers at Oregon Health & Science University (OHSU) marked a significant, albeit highly imperfect, step toward human IVG 3953. The researchers bypassed the lengthy and complex iPSC differentiation pathway entirely. Instead, they utilized Somatic Cell Nuclear Transfer (SCNT) - the technique famously used to clone Dolly the Sheep - to insert the nucleus of a diploid human skin cell (46 chromosomes) into a donor human egg that had been stripped of its own genetic material 5343.
The primary biological hurdle is ploidy reduction: human gametes must be haploid (23 chromosomes) to ensure a euploid embryo upon fertilization. To force the somatic cell to discard half its genome, the researchers developed an experimental division technique termed "mitomeiosis" 43. The experimental mitomeiosis process involves somatic cell nuclear transfer directly into an enucleated Metaphase II oocyte. The residual metaphase activity within the cytoplasm forces premature chromosomal condensation, deliberately bypassing the replication S-phase 4344. Following artificial activation using a selective cyclin-dependent kinase inhibitor and fertilization with sperm, the eggs successfully shed a portion of their chromosomes into a polar body 4445.
Despite creating 82 functional oocytes and demonstrating that experimental halving of a diploid genome is physically feasible, the technique is fraught with profound genomic instability 3945. Comprehensive chromosomal tracing via sequencing revealed that mitomeiosis completely failed to recreate the precise homologous pairing and crossover recombination steps that define natural meiosis 4445. Without this homologous pairing, chromosomes segregate randomly, statistically resembling Monte Carlo simulations 43. This random segregation results in rampant, near-universal aneuploidy. While roughly 9% of the resulting embryos progressed to the blastocyst stage (the critical stage for clinical transfer), they exhibited massive chromosomal abnormalities, carrying the wrong number or combination of chromosomes, rendering them entirely unsuitable for human reproduction 394345.
The OHSU team explicitly noted that while a major scientific milestone, mitomeiosis IVG remains strictly a laboratory proof-of-concept 3744. Perfecting the bioengineering required to force somatic chromosomes to pair and segregate evenly will require at least a decade of rigorous optimization, compounded by significant ethical and regulatory oversight regarding the creation and destruction of human embryos and the implications for multiplex parenting 36373945.
Synthesis and Strategic Outlook
The landscape of reproductive aging is rapidly evolving from a narrative of inevitable, irreversible decline to one of precise quantification, genomic understanding, and active clinical intervention. Biomarkers such as AMH and AFC offer unmatched precision in navigating the artificial environment of assisted reproduction, guiding gonadotropin dosing and risk mitigation, yet must be strictly decoupled from prognosticating natural fecundability. The integration of trans-ethnic GWAS and metabolomic data has fundamentally shifted the understanding of ovarian aging toward a model dictated by DNA damage repair efficacy and lipid metabolism, deeply intertwining the reproductive window with somatic longevity.
Simultaneously, the therapeutic frontier is expanding exponentially. While ex vivo cryopreservation strategies have transitioned into safe, highly reliable standards of care for both elective and medical indications, the future lies in in vivo cellular manipulation. The clinical pipeline spans immediate-horizon pharmacological therapies to distant-horizon bioengineering feats. The following evidence table synthesizes the current status and developmental trajectory of these diverse fertility extension interventions.
Evidence Table: Modalities for Reproductive Extension and Preservation
| Intervention / Modality | Biological Mechanism | Current Application Status | Primary Limitations / Clinical Hurdles |
|---|---|---|---|
| Oocyte Cryopreservation (Elective & Medically Indicated) | Ex vivo vitrification of mature, unfertilized oocytes to halt cellular aging. | Standard of Care (ASRM approved). | Highly age-dependent efficiency (<6.5% per oocyte over age 35). Requires invasive, costly hormonal stimulation. |
| Ovarian Tissue Cryopreservation (OTC) | Surgical excision and vitrification of ovarian cortex to preserve primordial follicles. | Standard of Care (ASRM 2026 update; no longer experimental). | Requires surgical excision and autotransplantation. Theoretical risk of reseeding malignant cells in cancer patients. |
| NAD+ Precursors (NMN, NR) | Restoration of mitochondrial homeostasis and activation of SIRT1 for DNA repair. | Clinical Adjuvant. Supported by 2025 human retrospective trials for IVF outcomes. | Lacks large-scale, multi-center randomized controlled trials to optimize dosing parameters. |
| mTOR Inhibition (Rapamycin) | Suppression of mTORC1 to prevent hyperactivation and premature recruitment of dormant follicles. | Active Clinical Trials. VIBRANT trial (Phase 2 equivalent) showing 20% reduced follicle loss. | Long-term systemic effects of continuous use unknown. Rebound kinetics upon drug cessation require mapping. |
| Senolytics (e.g., D+Q) & Autophagy Inducers (Spermidine) | Clearance of pro-inflammatory senescent cells (SASP) and recycling of damaged cellular organelles. | Early Clinical Proof-of-Concept. Validated in humans for adipose/kidney tissue; theoretical in ovaries. | Difficult to achieve localized efficacy without off-target effects. Optimal combinations and tissue-specific delivery remain unsolved. |
| In Vitro Gametogenesis (Mitomeiosis / SCNT) | Halving a diploid somatic genome to generate functional haploid oocytes, bypassing normal meiosis. | Pre-clinical Laboratory Proof-of-Concept. (2025 Nature Comms). | Severe genomic instability. Random chromosome segregation leads to near-universal aneuploidy. Estimated 10+ years from clinical trials. |
The ultimate goal of extending the reproductive lifespan is no longer solely focused on circumventing age-related infertility. Instead, by preserving the endocrine output and genomic integrity of the ovaries, the scientific community is targeting the most fundamental upstream regulator of women's cardiovascular, skeletal, and neurological health, aiming to synchronize the female reproductive lifespan with the modern human healthspan.