Relationship between muscle mass, sarcopenia, and longevity
Skeletal muscle mass constitutes approximately 40% of total body weight in healthy adults. It serves not only as the primary mechanical engine for human locomotion but also as a critical metabolic sink for glucose disposal and a reservoir for amino acids. Sarcopenia, originally defined in 1989 as a geriatric syndrome characterized by the progressive decline of skeletal muscle mass and strength, has increasingly been recognized as a central driver of human aging and a primary risk factor for all-cause mortality 1. The condition is no longer viewed merely as an inevitable consequence of aging, but rather as a highly modifiable pathology heavily influenced by dietary patterns, mechanical loading, and intracellular nutrient-sensing pathways 2.
The preservation of muscle tissue presents a unique biological dilemma in clinical gerontology. The biochemical pathways responsible for synthesizing muscle tissue - most notably the mechanistic target of rapamycin (mTOR) and the insulin-like growth factor 1 (IGF-1) axis - are the identical pathways that, when chronically activated, are implicated in accelerated cellular aging and oncogenesis 34. This fundamental conflict has given rise to divergent clinical paradigms regarding optimal daily protein intake and nutritional strategies for older adults. Resolving the tension between promoting anabolism to prevent terminal frailty and limiting nutrient signaling to extend molecular lifespan remains one of the most heavily researched frontiers in longevity science 5.
Epidemiological Prevalence and Diagnostic Frameworks
Estimating the global burden of sarcopenia is inherently complicated by the lack of a universally applied diagnostic consensus. Clinical prevalence varies dramatically depending on the specific diagnostic framework utilized, the demographic characteristics of the studied cohort, and the primary measurement modalities employed to quantify muscle mass and functional capacity 3.
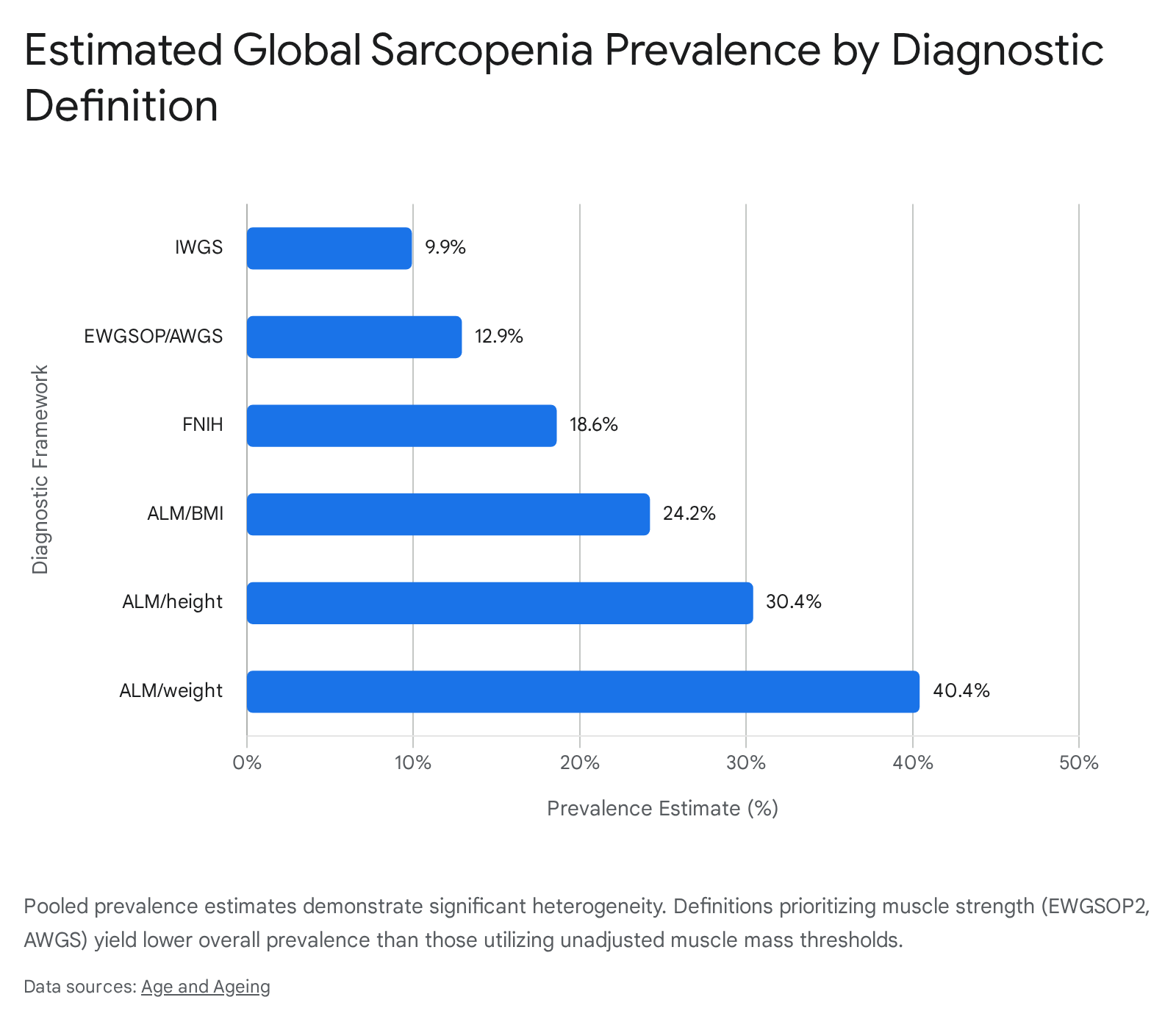
Variability Across Diagnostic Criteria
Early definitions of sarcopenia focused exclusively on low muscle mass, often defined relative to a younger reference population. Modern diagnostic frameworks have evolved to require evidence of functional impairment - specifically low muscle strength or poor physical performance - in addition to low appendicular lean mass (ALM) 4.
The transition from the initial European Working Group on Sarcopenia in Older People (EWGSOP1) guidelines to the updated EWGSOP2 criteria in 2019 shifted the primary diagnostic focus from absolute muscle mass to muscle strength. Under EWGSOP2, low grip strength is utilized as the primary indicator for "probable sarcopenia," with dual-energy X-ray absorptiometry (DXA) used secondarily to confirm the diagnosis 4. Consequently, epidemiological studies applying the EWGSOP2 criteria consistently yield significantly lower prevalence estimates compared to older definitions. In the multinational DO-HEALTH European trial evaluating community-dwelling older adults, the prevalence of sarcopenia was recorded at a mere 0.7% using EWGSOP2 criteria, but skyrocketed to 16.8% when utilizing older definitions like the Delmonico criteria .
The American Foundation for the National Institutes of Health (FNIH) introduced alternative criteria that adjust ALM by body mass index (BMI) rather than height squared. This methodology specifically aims to account for the growing prevalence of sarcopenic obesity, ensuring that excess adiposity does not mask dangerous deficits in underlying skeletal muscle 4. Meanwhile, the Asian Working Group for Sarcopenia (AWGS 2019) introduced population-specific cut-offs, acknowledging that Asian cohorts exhibit distinct anthropometric baselines, dietary histories, and muscle mass decline trajectories 56. The Sarcopenia Definition and Outcomes Consortium (SDOC) further complicates the landscape by defining sarcopenia strictly through the dual presence of muscle weakness and slowness, independent of DXA-derived lean mass, arguing that lean mass unadjusted for functional capacity has no consistent connection with adverse health outcomes like falls and mortality 4.
| Diagnostic Framework | Target Population | Muscle Mass Thresholds (DXA) | Strength Thresholds | Functional Severity Indicator |
|---|---|---|---|---|
| EWGSOP2 (2019) | European / Global | ALM/height2 <7.0 kg/m2 (M) / <5.5 kg/m2 (W) | Grip Strength <27 kg (M) / <16 kg (W) | Gait speed ≤0.8 m/s |
| AWGS (2019) | Asian | ALM/height2 <7.0 kg/m2 (M) / <5.4 kg/m2 (W) | Grip Strength <28 kg (M) / <18 kg (W) | Gait speed <1.0 m/s or SPPB ≤9 |
| FNIH (2014) | North American | ALM/BMI <0.789 (M) / <0.512 (W) | Grip Strength <26 kg (M) / <16 kg (W) | Gait speed <0.8 m/s |
| SDOC (2020) | Global | Not explicitly required | Grip Strength <35.5 kg (M) / <20 kg (W) | Gait speed <0.8 m/s |
Summary of international working group consensus statements on sarcopenia diagnostic thresholds across key demographic regions 47.
Regional Variations and Environmental Disparities
A comprehensive systematic review of 52 studies involving over 70,000 community-dwelling older adults estimated the overall global prevalence of sarcopenia at 18.8%, though with significant clinical heterogeneity 8. Regional variations are profound and heavily influenced by the diagnostic tool utilized. Across Asian countries utilizing the AWGS 2019 criteria, the pooled prevalence among community-dwelling older adults sits at 16.5%, with "possible sarcopenia" present in up to 28.7% of the demographic and severe sarcopenia affecting 4.4% 6.
In contrast, studies in Europe relying on the more stringent EWGSOP2 criteria frequently report prevalence rates as low as 10% 5, though older meta-analyses utilizing diverse definitions estimate European prevalence closer to 23.4% 8. Urban-rural disparities further shape the epidemiological landscape. Meta-analyses involving over 433,000 participants globally demonstrate that sarcopenia is significantly more prevalent in rural settings (20%) compared to urban populations (16%). This gap is primarily attributed to disparities in nutritional access, long-term physical labor exhaustion, and limited healthcare infrastructure for early intervention 9.
Comorbidities and Associated Mortality Risks
Sarcopenia rarely exists in isolation. It is an intersectional pathology that frequently co-occurs with, and exacerbates, other chronic age-related conditions. The prevalence of sarcopenia rises sharply in specific patient populations: 18% in those with diabetes mellitus, 21.6% in individuals with chronic obstructive pulmonary disease (COPD), 24.5% in patients with chronic kidney disease (CKD), and up to 35% in individuals with diverse cardiovascular diseases 5. Within specific acute conditions, such as acute decompensated heart failure, prevalence can soar to 61%, while unresectable esophageal cancer populations demonstrate prevalence rates of 66% 510.
The clinical consequences of sarcopenic decline extend far beyond localized weakness. Loss of muscle mass is robustly correlated with cognitive impairment, metabolic syndrome, and systemic inflammation 5. In surgical and oncological settings, patients presenting with sarcopenia face a dramatically higher risk of postoperative complications, prolonged hospitalization, and poorer disease-progression-free survival rates. For instance, in patients undergoing emergency laparotomy, preexisting sarcopenia increases the odds ratio (OR) of mortality by 3.50 5. General frailty resulting from sarcopenia increases the risk of falls by a factor of 1.5 to 3 and elevates general mortality risk by 29% to 51% 5. The economic and physical toll is immense; currently affecting over 50 million individuals globally, epidemiological models project sarcopenia to impact over 200 million individuals within the next forty years as global demographics skew older 2.
Cellular Pathophysiology of Age-Related Muscle Decline
The biological mechanisms driving sarcopenia are distinct from those driving cachexia. While cachexia involves acute muscle degradation largely mediated by extreme cytokine cascades associated with diseases like terminal cancer or immunodeficiency, sarcopenia is a gradual, multifactorial deterioration driven by intrinsic cellular aging 12.
Mitochondrial Dysfunction and Oxidative Stress
A primary mechanism of sarcopenia is the age-related decline in mitochondrial oxidative capacity. As skeletal muscle ages, the efficiency of the electron transport chain diminishes, leading to heightened production of reactive oxygen species (ROS). This elevated ROS production creates a state of chronic oxidative stress that destabilizes mitochondria within skeletal muscle fibers, rendering them highly susceptible to apoptotic stimuli and downregulating crucial pathways related to mitochondrial biogenesis 1. Furthermore, oxidative stress directly drives proteolysis, upregulates tumor necrosis factor-alpha (TNF-α), and inhibits muscle cell differentiation 1.
Stem Cell Depletion and Cellular Senescence
The role of muscle stem cells - known as satellite cells or MuSCs - in the pathogenesis of sarcopenia remains an area of active debate. Satellite cells reside in the muscle niche and are responsible for repairing muscle fiber damage 11. While early hypotheses suggested that a linear depletion of MuSCs was the primary cause of sarcopenia, recent studies introduce significant nuance. Evidence indicates that while older individuals do experience a decrease in MuSC content (particularly in Type II fast-twitch fibers), geriatric models with profound sarcopenic symptoms do not always show a direct correlation between total MuSC depletion and the onset of muscle atrophy 1. Some researchers suggest that MuSC dysfunction and sarcopenia may be separate entities, noting that sarcopenic atrophy often precedes measurable impairments in MuSC function 1.
Conversely, cellular senescence is unequivocally linked to muscle decline. The accumulation of senescent cells - which cease dividing but resist apoptosis - results in the Senescence-Associated Secretory Phenotype (SASP). SASP involves the chronic release of pro-inflammatory cytokines, chemokines, and proteases into the local tissue microenvironment, contributing to persistent low-grade inflammation that suppresses muscle regeneration 112.
Neuromuscular Degeneration and Adipose Infiltration
Sarcopenia is also driven by changes external to the muscle fiber itself. The neuromuscular junction (NMJ), the critical synapse connecting motor neurons to skeletal muscle, undergoes significant morphological deterioration with age. The denervation of muscle fibers, particularly fast-twitch Type II fibers, leads directly to the loss of strength and explosive power (dynapenia) that precedes gross muscle mass loss 113.
Simultaneously, aging muscle undergoes profound changes in tissue composition. Fibroadipogenic progenitors (FAPs) located within the muscle interstitium lose their ability to support muscle repair and instead differentiate into adipocytes. This process results in the accumulation of intramuscular adipose tissue (IMAT). Elevated IMAT physically disrupts muscle architecture, secretes local inflammatory adipokines, induces insulin resistance, and directly impairs physical mobility 1.
The Dietary Protein and Longevity Paradigm Conflict
Given the profound mortality risks associated with muscle atrophy and the biological reality of cellular aging, nutritional interventions have become central to preventative gerontology. However, clinical consensus regarding optimal daily protein intake remains highly elusive. The current scientific discourse is anchored by two fundamentally competing models: a low-protein paradigm optimized for maximizing molecular lifespan, and a high-protein paradigm optimized for extending functional healthspan and preventing frailty.
Caloric Restriction and the Low-Protein Lifespan Model
The low-protein longevity model, championed prominently by gerontology researchers such as Dr. Valter Longo of the University of Southern California, posits that restricting dietary protein - specifically branched-chain amino acids (BCAAs) like leucine, and methionine - is necessary to suppress pro-aging genetic signaling pathways 1415. This model draws heavily from evolutionary biology, extensive data on yeast and rodent survival models, and observational data derived from centenarian populations in global "Blue Zones" such as Okinawa and Sardinia, where traditional diets are overwhelmingly plant-based and relatively low in protein 1415.
When dietary protein is abundant, it serves as a potent signaling molecule that activates the mechanistic target of rapamycin complex 1 (mTORC1) and elevates circulating levels of insulin-like growth factor 1 (IGF-1) 414. While these signals successfully drive cellular growth and muscle synthesis, their chronic activation is fundamentally antagonistic to organismal longevity. High mTOR and IGF-1 activity suppress autophagy - the vital cellular "housekeeping" process that degrades damaged organelles and misfolded proteins - and stimulate cellular proliferation, thereby increasing the risk of oncogenesis and accelerating cellular senescence 416.
Epidemiological support for this model includes a landmark 18-year longitudinal study tracking over 6,000 adults. The findings demonstrated that high protein intake in individuals aged 50 to 65 was associated with a 74% higher risk of all-cause mortality and a more than fourfold increase in cancer-related death, effects largely driven by animal protein consumption rather than plant protein 17. Based on these molecular and epidemiological findings, the longevity diet model recommends a moderate-to-low protein intake of approximately 0.31 to 0.36 grams per pound of body weight (roughly 0.68 to 0.79 g/kg/day) - aligning very closely with the current US Recommended Dietary Allowance (RDA) of 0.8 g/kg/day - until the age of 65 1718.
Crucially, this model acknowledges a demographic inflection point. Beyond age 65, the epidemiological pattern reverses. In older adults, low protein intake is associated with heightened mortality, primarily due to catastrophic muscle loss and the inability to recover from acute illness. Therefore, the Longo model advises a deliberate increase in protein intake after age 65, incorporating greater quantities of fish, eggs, and goat-derived dairy, to mitigate the risk of sarcopenia 1718. The protocol also relies heavily on periodic Fasting Mimicking Diets (FMD) - a low-protein, low-calorie, plant-based regimen utilized several times a year to force the body into a state of deep autophagy without the risks of complete water fasting 419.
Muscle Preservation and the High-Protein Healthspan Model
In stark contrast, the high-protein paradigm, popularized by clinicians focusing on proactive "Medicine 3.0" such as Dr. Peter Attia, asserts that the existential risks of sarcopenia, metabolic dysfunction, and physical frailty far outweigh the theoretical oncogenic risks of elevated dietary protein 2021. Medicine 3.0 represents a medical philosophy focused on aggressive, early-stage prevention and the optimization of physical and cognitive healthspan, explicitly rejecting the reactive "Medicine 2.0" framework that waits until chronic diseases are deeply entrenched before intervening 2728.
Proponents of this model argue that the established RDA of 0.8 g/kg/day represents a mere baseline required to maintain nitrogen balance and prevent severe malnutrition (like kwashiorkor), rather than the optimal dose required for a human to thrive biologically 2029. Instead, this paradigm advocates for a daily protein intake between 1.6 and 2.2 grams per kilogram of body weight (roughly 0.75 to 1.0 g/lb) across adult life stages 21.
The clinical rationale is rooted in the inevitability of age-related functional decline. Because individuals lose muscle mass at an alarming rate as they age, maximizing a "reserve" of muscle mass, explosive strength, and bone density in midlife acts as a critical buffer against late-life frailty 2030. A loss of balance resulting in a hip fracture is a leading cause of terminal decline in the elderly, making robust muscular infrastructure a matter of survival. Furthermore, skeletal muscle is the primary organ for glucose disposal; abundant, highly active muscle tissue radically improves insulin sensitivity and defends against type 2 diabetes and metabolic syndrome 22.
The high-protein model challenges the assertion that chronic dietary protein restriction is the optimal method for modulating mTOR in humans. Attia and aligned researchers argue that transient mTOR activation - such as the acute metabolic spike triggered by a high-protein meal consumed immediately after heavy resistance exercise - is brief, highly localized to skeletal muscle, and absolutely essential for tissue repair and maintenance 3. In this view, longevity data derived from nutrient-deprived rodents confined to sterile cages translates poorly to humans living in a dynamic physical world where functional capacity dictates quality of life 32.
| Clinical Focus | Low-Protein Lifespan Model (e.g., Longo) | High-Protein Healthspan Model (e.g., Attia) |
|---|---|---|
| Primary Biological Goal | Minimize pro-aging signaling (mTOR, IGF-1); maximize systemic autophagy. | Maximize muscle reserve, metabolic capacity, and bone density; prevent terminal frailty. |
| Target Intake (Age < 65) | ~0.7 to 0.8 g/kg/day (0.31 - 0.36 g/lb) | 1.6 to 2.2 g/kg/day (~0.75 to 1.0 g/lb) |
| Target Intake (Age > 65) | Moderate increase (~1.0 - 1.2 g/kg/day) | 1.6 to 2.2 g/kg/day (Adjusted for impaired absorption) |
| Preferred Protein Sources | Predominantly plant-based (legumes, nuts); strictly limited animal protein. | High-bioavailability animal proteins (whey, eggs, meat, venison). |
| View on mTOR Activation | Chronic or frequent activation universally accelerates cellular aging and increases cancer risk. | Transient, exercise-induced activation is safe, highly localized, and metabolically necessary for repair. |
Comparative overview of the two dominant nutritional paradigms dictating protein intake in preventative gerontology 3182021.
Anabolic Resistance and Muscle Protein Synthesis Dynamics
The human physiological capacity to convert dietary amino acids into functional skeletal muscle tissue changes profoundly across the lifespan. Resolving the debate on optimal protein volume requires a deep understanding of how skeletal muscle interprets metabolic signals at the cellular level.
Mechanisms of Anabolic Resistance
Sarcopenia is primarily driven by an increasing imbalance between muscle protein synthesis (MPS) and muscle protein breakdown. While basal (fasted) rates of MPS remain surprisingly stable throughout life, aging skeletal muscle develops a pronounced unresponsiveness to anabolic stimuli - a phenomenon clinically termed "anabolic resistance" 22.
Anabolic resistance means that skeletal muscle becomes desensitized to post-absorptive hyperaminoacidemia (the presence of amino acids in the blood after eating) and mechanical loading from exercise 22. In healthy young adults, the ingestion of roughly 20 grams of high-quality protein (approximately 0.24 g/kg of body weight) is entirely sufficient to maximally stimulate MPS 23. In stark contrast, the senescent muscle of older adults requires a significantly higher threshold of circulating amino acids, particularly leucine, to trigger the same synthetic response. Studies indicate that older adults require nearly double the dose - approximately 0.40 g/kg per meal, or roughly 30 to 40 grams of high-quality protein - to overcome anabolic resistance and fully saturate the MPS machinery 23.
This resistance is exacerbated by physical inactivity, systemic low-grade inflammation, and splanchnic extraction (where the gut and liver sequester a higher proportion of ingested amino acids in older adults before they can reach peripheral muscle tissue) 22. Consequently, a standard diet that provides adequate total daily protein but delivers it in sub-threshold doses may entirely fail to prevent sarcopenic decline.
Protein Dosing: Bolus, Distribution, and Pulsed Intake
The recognition of anabolic resistance has forced researchers to investigate not just the total quantity, but the temporal distribution of protein intake. The traditional Western dietary pattern is notoriously skewed: a small proportion of daily protein is consumed at breakfast (e.g., 10-15g) and lunch (e.g., 15-20g), with the vast majority consumed in a single large bolus at dinner (e.g., 65g) 23.
Because the human body does not possess a dedicated storage reservoir for excess amino acids analogous to glycogen (for carbohydrates) or adipose tissue (for lipids), protein consumed in massive excess of the per-meal MPS ceiling is largely oxidized for energy or converted into urea and excreted. Consequently, a highly skewed dietary pattern results in only one maximal stimulation of MPS per day 2023.
Emerging nutritional strategies advocate for an "even" or "pulsed" distribution of protein. This involves consuming distinct, optimally dosed boluses of high-quality protein (e.g., 30 - 40 grams) spaced evenly across three or four daily meals 23. This approach ensures that the leucine threshold necessary to activate the mTORC1 pathway in skeletal muscle is repeatedly breached, generating multiple discrete spikes in MPS throughout the day without necessitating an extreme total caloric intake 34.
Clinical trials assessing targeted amino acid formulations strongly support this pulsed approach. In a 2024 human study, the ingestion of a precise 3.6-gram composition of essential amino acids (EAAs) heavily enriched with leucine successfully stimulated a 48.9% increase in muscle protein fractional synthesis rate (FSR) in older adults compared to basal states 35. These concentrated, pulsed interventions demonstrate that the specific profile and temporal delivery of amino acids, rather than merely gross total protein volume, primarily govern muscle preservation.
Conversely, extreme "grazing" strategies - such as consuming 10 grams of protein every 1.5 hours - fail to optimize anabolism. Trials show that this trickle-feeding approach fails to breach the required leucine threshold in older adults, resulting in higher systemic amino acid oxidation rather than directed muscle protein synthesis 23. Recent complex studies by Trommelen et al. have pushed the boundaries further, showing that a massive post-exercise bolus of 100g of milk protein prolonged the postprandial anabolic window up to 12 hours in young males compared to a 25g dose 24. However, the translation of such extreme single-bolus strategies to older, anabolically resistant populations remains uncertain, reinforcing the current consensus that achieving 30-40g multiple times a day is the most reliable clinical target for combating sarcopenia 2324.
The mTOR Paradox: A Unifying Biochemical Framework
The apparent contradiction between the longevity benefits of suppressing mTOR globally and the functional necessity of activating mTOR to prevent sarcopenia has perplexed researchers for years. However, groundbreaking multi-omic profiling studies published in Nature Aging in 2025 have illuminated a highly specific mechanism known as the "mTOR paradox" in skeletal muscle aging. This framework provides a unifying theory that bridges the low-protein and high-protein paradigms 252627.
Branched-Chain Amino Acid Catabolism in Healthy Muscle
In healthy human physiology, branched-chain amino acids (BCAAs: leucine, isoleucine, and valine) derived from dietary protein are utilized for muscle protein synthesis. Any excess BCAAs are efficiently broken down by highly specific mitochondrial catabolic enzymes within the skeletal muscle 28.
Recent extensive human multi-omic profiling (involving cohorts from Singapore, the UK, and Jamaica) combined with murine studies reveals that in age-related sarcopenia, this fundamental mitochondrial BCAA catabolism fails 2526. Specifically, aging muscle exhibits a profound deficiency in Ppm1k, a critical positive regulator of the BCAA catabolic cascade, alongside an overexpression of BCKDK, which inhibits catabolism 2527. Because the aged, sarcopenic muscle physically cannot metabolize excess BCAAs, these amino acids accumulate to toxic levels within the intracellular environment and the systemic bloodstream 27.
Pathological mTORC1 Hyperactivation
This intracellular accumulation directly triggers the mTOR paradox. The persistent presence of uncatabolized BCAAs locks the mTORC1 pathway into a state of chronic, pathological hyperactivation 27.
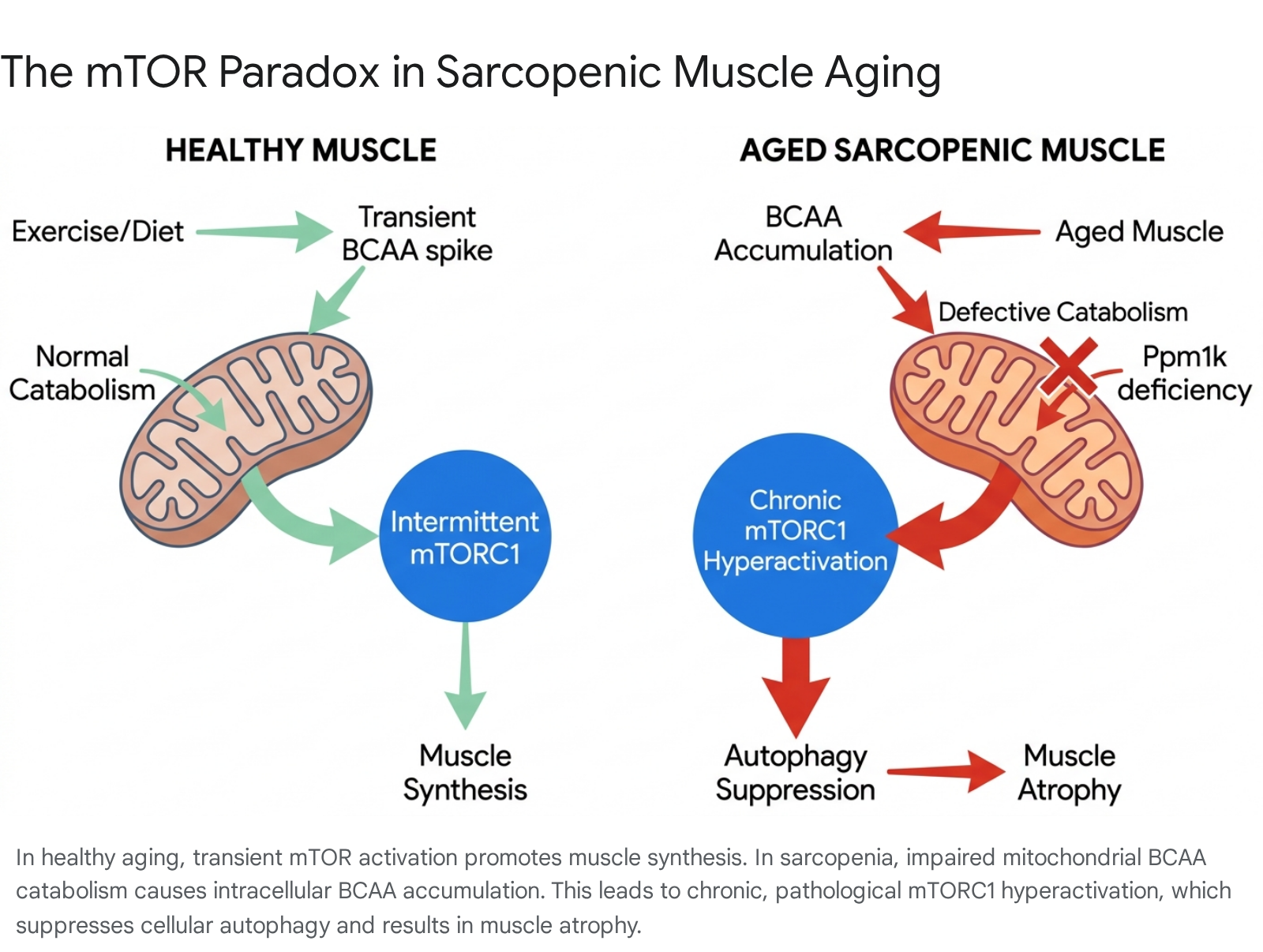
Rather than driving healthy muscle synthesis, this unrelenting mTORC1 signal paralyzes the cell's regenerative capacity. It forcibly suppresses autophagy (via the inhibition of ULK1) and drives further mitochondrial deterioration by creating a highly oxidative environment 2729. Furthermore, the accumulation of branched-chain keto acids (BCKAs) directly inhibits insulin receptor substrate 1 (IRS-1), severely impairing local insulin sensitivity 27.
Therefore, in pathological aging, mTORC1 hyperactivation is not a sign of anabolic strength; it is a signal of metabolic gridlock. The muscle physically atrophies because it cannot recycle damaged proteins, clear dysfunctional mitochondria, or properly utilize glucose 27.
Reconciling the Paradigms: Intermittent Nutrient Stimulation
Understanding the mTOR paradox cleanly reconciles the seemingly contradictory advice of longevity experts. Dr. Longo's observation that high protein intake correlates with mortality in sedentary, aging populations is biochemically sound. In a sedentary individual with declining mitochondrial function, constant nutrient influx without catabolic expenditure results in BCAA pooling. This promotes the exact chronic mTOR activation that suppresses autophagy and drives cellular aging 427.
Conversely, Dr. Attia's protocol of high protein intake is biologically viable and highly effective primarily because it is inextricably linked to rigorous mechanical loading. Vigorous resistance exercise restores mitochondrial flexibility, actively drains the intracellular BCAA pool for energy and repair, and ensures that mTORC1 activation remains strictly intermittent. The pathway spikes to drive acute repair, and then rapidly silences, permitting the crucial autophagic clearance to resume 2730.
The scientific literature increasingly supports periodic nutrient stimulation as the optimal strategy for human longevity 5. Interventions that combine pulsed essential amino acid intake to overcome age-related anabolic resistance, coupled with periods of fasting or caloric restriction to ensure deep autophagic clearance, mimic the natural, evolutionary rhythms of feast and famine 45.
Emerging Therapeutics and Multimodal Interventions
As the molecular understanding of sarcopenia expands, novel pharmaceutical and physical therapies are emerging to complement dietary interventions, targeting the specific pathological bottlenecks of muscle aging.
Pharmacological Agents
Significant momentum has gathered behind drugs capable of modulating the myostatin/activin signaling pathway. Myostatin is a natural negative regulator of muscle growth. Clinical trials using bimagrumab (an ActRIIB antibody) demonstrated a 6 - 8% increase in lean muscle mass within a single month, accompanied by concurrent fat mass reduction, suggesting high efficacy for sarcopenic obesity without the need for strict caloric restriction 11.
Furthermore, targeted therapies addressing the mTOR paradox directly are entering clinical phases. PF-07328948, a selective BCKDK inhibitor, is currently undergoing clinical trials. By degrading BCKDK, the drug enhances the muscle's ability to catabolize BCAAs, lowering toxic systemic levels of branched-chain ketoacids and alleviating the chronic mTORC1 pressure that drives sarcopenic atrophy 31.
The Gut Microbiome and Chrono-Nutrition
Emerging research highlights the profound influence of the gastrointestinal microbiome on skeletal muscle health, proposing a "gut-muscle axis." Age-related dysbiosis reduces the microbial production of short-chain fatty acids (SCFAs), which are critical for gut health and systemic anti-inflammatory signaling 32. Diets rich in fiber, such as the Mediterranean and Baltic Sea diets, promote SCFA-producing bacteria and are strongly correlated with better grip strength and locomotion in older adults, suggesting that plant-based diversity is essential alongside adequate protein intake 3233.
Chrono-nutrition - the alignment of food intake with circadian rhythms - is also proving vital. Research indicates that muscle protein synthesis is subject to circadian regulation, and consuming high-quality protein predominantly during the active morning phase (e.g., breakfast) aligns with optimal genetic clock expression, preventing weakness more effectively than late-evening consumption 34. Time-restricted eating (TRE) models have shown benefits for systemic glucose metabolism, though care must be taken to ensure the compressed eating window does not inadvertently restrict total daily protein intake below the anabolic threshold 35.
Physical and Technological Interventions
While resistance training remains the gold standard, non-pharmacological adjuncts are showing efficacy for populations unable to perform heavy mechanical loading. Low-intensity pulsed ultrasound and pulsed electromagnetic field (PEMF) therapies have demonstrated an ability to attenuate muscle atrophy and mitigate pain. PEMF treatments specifically target senescent bone marrow mesenchymal stromal cells, altering the inflammatory niche and promoting functional recovery in cohorts suffering from sarcopenic obesity and chronic back pain 136.
Conclusion
Sarcopenia is not merely a localized deterioration of physical strength, but a systemic metabolic failure inextricably linked to the biology of aging. The global prevalence of the condition, ranging from 10% to nearly 29% of the older adult population depending on regional diagnostic criteria, underscores an escalating public health crisis 568.
The tension between preventing sarcopenia and extending cellular lifespan is resolved by understanding the nuances of nutrient signaling. While constant caloric and protein abundance in a sedentary state accelerates aging pathways via the mTOR paradox, severe protein restriction in older adulthood virtually guarantees terminal frailty. The optimal intervention lies in targeted, intermittent metabolic modulation.
To combat age-related anabolic resistance, older adults require higher individual per-meal doses of protein - specifically distributed evenly as distinct biological pulses of 30 to 40 grams - to adequately stimulate muscle protein synthesis 23. Crucially, this high-protein intake must be coupled with rigorous mechanical loading. By driving healthy mitochondrial BCAA catabolism, exercise prevents the chronic hyperactivation of mTORC1, allowing the muscle to harness amino acids for transient repair while preserving the critical periods of cellular autophagy necessary for true longevity 2527. Future clinical directives must pivot from debating total daily protein volume toward optimizing the temporal delivery of nutrients, physical exertion, and restorative fasting to preserve functional autonomy in global aging populations.