Quantum Tunneling in Olfaction
The Foundational Mechanisms of Olfactory Reception
Olfaction represents one of the most evolutionarily ancient sensory modalities, enabling organisms to detect, discriminate, and interpret volatile chemical compounds in their environment. In mammals, this complex process begins in the olfactory epithelium, where odorant molecules dissolve into the nasal mucosa and bind to olfactory receptors (ORs) located on the cilia of olfactory sensory neurons 123. The molecular basis of this system was definitively established in 1991 when Richard Axel and Linda B. Buck discovered the multigene family encoding mammalian olfactory receptors, a breakthrough that earned the 2004 Nobel Prize in Physiology or Medicine 1345. The human genome contains approximately 339 complete, functional genes coding for these receptor proteins 45.
Olfactory receptors belong to the class A G protein-coupled receptor (GPCR) superfamily 126. Structurally, these are seven-transmembrane (7TM) integral membrane proteins 78. The canonical model of olfactory signal transduction relies on the "shape theory," also known as the docking theory. Under this paradigm, an odorant acts as a specific ligand that docks into the orthosteric binding site of an OR based on molecular volume, steric conformation, electrostatic interactions, hydrogen bonding, and van der Waals forces - analogous to a key fitting into a lock 3910.
The binding of a structurally compatible odorant stabilizes a specific active conformation of the transmembrane helical bundle. This conformational change propagates to the intracellular domains of the receptor, enabling the recruitment and activation of a heterotrimeric G-protein, specifically the olfactory-specific G-protein, $\text{G}_{\text{olf}}$ 21112. The dissociation of the G-protein subunits subsequently activates adenylyl cyclase, leading to the conversion of adenosine triphosphate (ATP) into cyclic adenosine monophosphate (cAMP). The ensuing elevation in intracellular cAMP opens cyclic nucleotide-gated ion channels, resulting in an influx of calcium and sodium ions. This cascade depolarizes the olfactory sensory neuron, generating an action potential that transmits the olfactory signal to the glomeruli within the olfactory bulb, and ultimately to the olfactory cortex for central processing 451213.
Despite the molecular elegance and widespread acceptance of the shape theory, the model has historically encountered difficulties in explaining specific anomalies in structure-odor relationships. Empirical observations have repeatedly demonstrated that molecules with highly similar shapes can exhibit drastically different odors, while structurally disparate molecules can evoke nearly identical olfactory percepts 914. A classic example is the metallocene experiment: ferrocene and nickelocene possess identical molecular geometries - characterized by a metal atom sandwiched between two cyclopentadienyl rings - yet they emit completely distinct odors 9. Similarly, the shape theory struggles to account for the observation that certain boranes smell intensely sulfurous despite entirely lacking sulfur atoms 9. Furthermore, enantiomers - chiral molecules that are non-superimposable mirror images with identical physical properties - frequently exhibit distinct odors, such as the (S)- and (R)-enantiomers of carvone smelling of caraway and spearmint, respectively. While docking theory addresses chirality through asymmetric receptor binding pockets, the cumulative anomalies have continually spurred the development of alternative biophysical models.
The Vibration Theory of Olfaction
To address the predictive limitations of the shape theory, researchers have periodically proposed that the olfactory system detects the molecular vibrations of odorants. The vibration theory of olfaction was first formally articulated by Malcolm Dyson in 1938, who suggested that the olfactory apparatus detects the thermalized vibrational frequencies of molecules, specifically within the infrared range of 1,400 to 3,500 $\text{cm}^{-1}$ 1314. R.H. Wright subsequently expanded upon this concept in 1977, attempting to mathematically correlate odor character with specific vibrational spectra 1713. However, early iterations of the vibration theory lacked a plausible biological transduction mechanism; the human body lacks the mechanisms to conduct conventional infrared spectroscopy, rendering the detection of subtle molecular vibrations in a warm, aqueous cellular environment highly improbable under classical biophysics.
Inelastic Electron Tunneling Mechanism
The vibration theory was revitalized in 1996 when biophysicist Luca Turin proposed a specific, quantum mechanical mechanism for vibrational detection: inelastic electron tunneling (IET) 9131517. Turin's framework, frequently termed the "swipe card" model, does not discard the necessity of molecular shape; it posits that shape is a necessary but insufficient condition for olfactory activation 9.
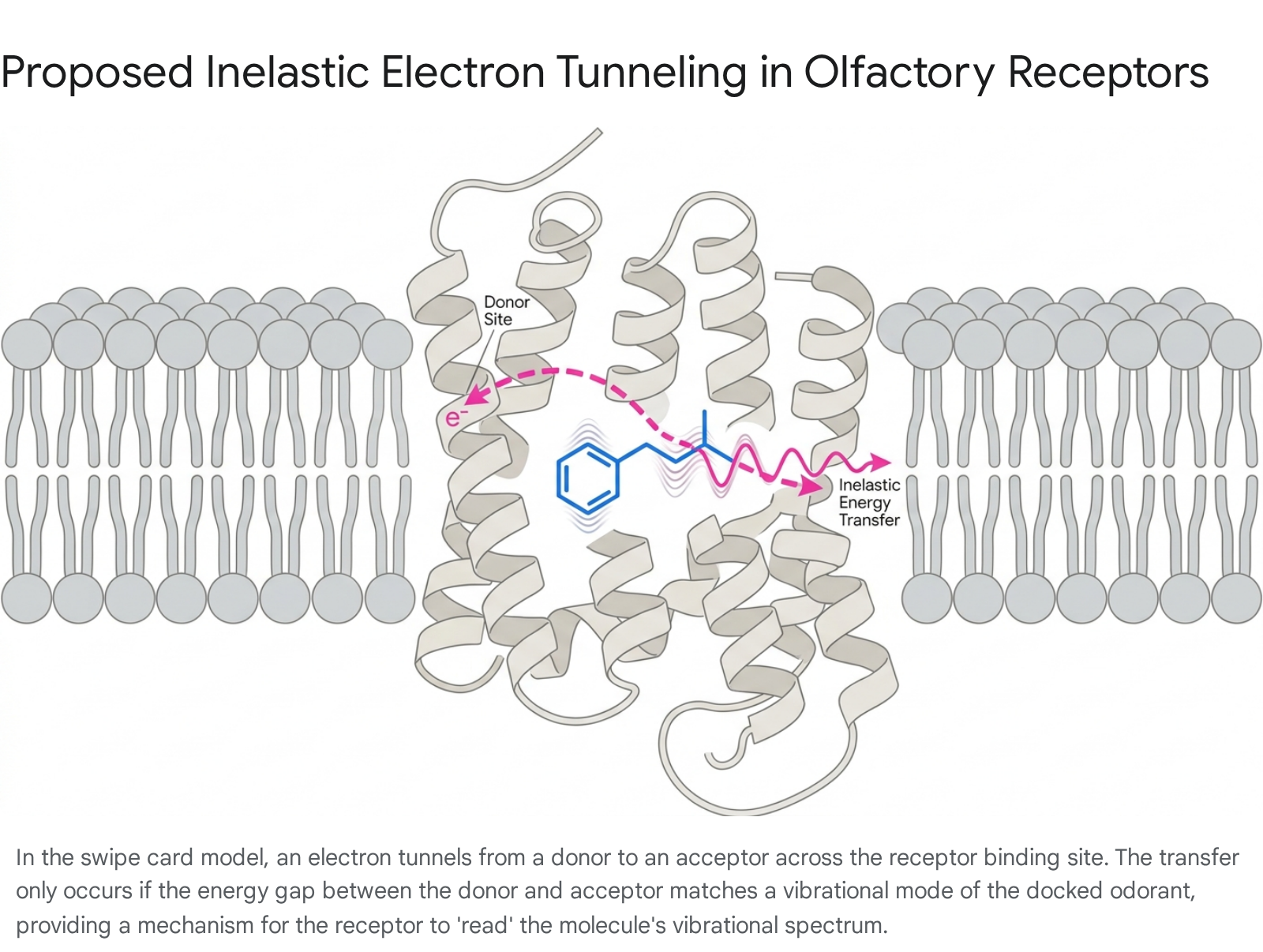
The odorant molecule must first physically fit into the receptor's binding site. However, the subsequent activation of the receptor relies on a quantum tunneling event 913.
In quantum mechanics, electron tunneling describes the phenomenon wherein an electron traverses a potential energy barrier that it lacks the classical kinetic energy to cross. If the electron maintains its exact energy state during the transfer, the tunneling is elastic. Inelastic electron tunneling occurs when the electron donates a discrete quantum of energy during the transfer process 71315.
Turin proposed that olfactory GPCRs function as biological electron tunneling junctions. In an empty receptor, an electron donor site maintains a higher energy level than an electron acceptor site, separated by a narrow physical gap across the orthosteric binding pocket 71115. Because biological electron transfer requires strict energy conservation, an electron cannot tunnel across the gap unless a mechanism exists to absorb the exact energy differential ($\Delta E$) between the donor and acceptor sites. When an odorant docks, it acts as a molecular bridge. If the odorant possesses an intramolecular vibrational mode with an energy quantum that perfectly matches the energy gap ($E_{\text{acceptor}} - E_{\text{donor}} = \Delta E$), the electron undergoes inelastic tunneling 71516. The electron's lost energy is transferred into the odorant, exciting its specific vibrational mode 1115.
This flow of electrons through the protein backbone is theorized to be the catalyst for receptor activation. Specifically, the transfer of electrons might reduce a disulfide bridge linking the receptor to the associated G-protein, thereby releasing the G-protein for further intracellular signal transduction 11. Consequently, the combinatorial coding of odors in the brain would be derived from the aggregate responses of hundreds of receptor types, each tuned to different vibrational frequencies, operating much like a biological spectroscope 91519.
Theoretical Plausibility and the Role of Transition Metals
The biophysical feasibility of sustained, coherent electron transfer within biological macromolecules - which generally function as wide-bandgap insulators - has been a primary point of contention 20. However, in 2007, researchers at University College London, including Marshall Stoneham, published a detailed numerical analysis in Physical Review Letters confirming that Turin's proposed mechanism is consistent with the established laws of physics 915. By modeling the dissipative, odorant-mediated inelastic electron tunneling mechanism, they concluded that the predicted tunneling rates and physical parameters could viably support signal transduction within the physiological timescales of olfaction 1315.
A critical requirement of the tunneling mechanism is the presence of suitable electron donor and acceptor moieties within the receptor structure. Transition metals are highly efficient mediators of biological electron transfer. Turin hypothesized that a zinc ion ($\text{Zn}^{2+}$) could serve as the requisite electron acceptor 1121. Zinc exhibits a +2 oxidation state and possesses high charge density, allowing it to interface with redox-active amino acids like cysteine 1120. Computational screening of the primary sequence of $\text{G}_{\text{olf}}$ revealed a conserved zinc-binding motif (His-Tyr-Cys-Tyr-Pro-His) exposed on the G-protein surface, which could facilitate the formation of a transient disulfide bridge with the receptor 11.
Further support for the role of transition metals in GPCR activation was derived from high-resolution structural analyses of rhodopsin, the evolutionary ancestor of olfactory receptors. Density-functional theory calculations identified an electron donor-bridge-acceptor (DBA) complex within the rhodopsin core 1620. The complex features a conserved tryptophan serving as the electron donor and a tightly bound zinc ion - coordinated by histidine, glutamate, and a secondary tryptophan in a highly unusual, non-bisecting geometric plane - functioning as the acceptor 1620. The retinal molecule acts as the bridge. Upon protonation, the retinal aldimine enables a complete electron charge to transfer from the tryptophan to the zinc complex, functionally operating as a molecular "tunnel triode" 20.
Experimental physiological evidence has also implicated zinc in olfactory sensitivity. An in vivo study demonstrated that the introduction of zinc metal nanoparticles - clusters of 40 to 300 metallic zinc atoms measuring 1 to 2 nanometers - in picomolar concentrations profoundly enhanced the electrical responses of rat olfactory sensory neurons to odorants, as measured by electroolfactograms and whole-cell patch-clamp recordings 12. Conversely, equivalent concentrations of ionic zinc ($\text{Zn}^{2+}$), as well as nanoparticles of copper, silver, or gold, failed to produce the enhancement effect, or actively diminished the response. These results support the hypothesis that highly localized metallic or electron-dense interfaces are involved in the immediate transduction events between receptor proteins and G-proteins 12.
Behavioral Psychophysics: The Isotope Effect
A fundamental prediction of the vibration theory of olfaction is the "isotope effect." If ORs operate as molecular spectroscopes detecting vibrational frequencies, substituting an atom in an odorant with a heavier isotope will decrease the frequency of the associated chemical bonds 3917. The most pronounced biological isotope effect involves replacing hydrogen with deuterium. The carbon-deuterium (C-D) bond possesses roughly twice the reduced mass of a carbon-hydrogen (C-H) bond, which drastically shifts the bond's fundamental stretching frequency downward by approximately 800 $\text{cm}^{-1}$ in the infrared spectrum 3918. Crucially, isotopic substitution exerts a negligible impact on the molecule's electrostatic profile, van der Waals volume, and overall geometric shape 1919. Therefore, if an organism can behaviorally discriminate between a hydrogenated odorant and its fully deuterated isotopologue, it provides strong circumstantial evidence that the olfactory system detects parameters beyond molecular docking alone 919.
Human Behavioral Trials
The capacity of humans to distinguish isotopologues has been the subject of conflicting psychophysical research. In 2001, a study by Haffenden et al. reported that human subjects were capable of distinguishing between standard benzaldehyde (which smells of bitter almonds) and fully deuterated benzaldehyde 917. However, this study was criticized for methodological flaws, specifically concerning the purity of the chemical samples and the implementation of non-standard, unblinded sensory testing protocols 919.
In 2004, researchers Keller and Vosshall published a rigorous rebuttal in Nature Neuroscience. Utilizing strictly controlled, double-blind testing, they demonstrated that naive human subjects could not differentiate between standard acetophenone (a sweet-smelling compound) and its fully deuterated analogue (d8-acetophenone) 9171920. This finding was broadly interpreted as a refutation of the vibration theory in mammalian olfaction 9.
The debate was re-ignited in 2013 when Turin and a team of researchers conducted further human trials. They acknowledged the negative results regarding acetophenone but hypothesized that the molecule's relatively small size - containing only eight hydrogen atoms - produced a weak vibrational shift that fell below the absolute threshold for human detection 920. To circumvent this, the team tested macroscopic musk compounds, specifically cyclopentadecanone, which features 28 hydrogen atoms, thereby amplifying the theoretical isotopic divergence 1920. In a double-blind forced-choice protocol involving both trained perfumers and untrained subjects, the panel successfully discriminated between standard cyclopentadecanone and deuterated d28-cyclopentadecanone 19. Participants reported that the heavily deuterated musk lost its defining musky olfactory character, presenting instead with "overtones of burnt candle wax" 20. Statistical analysis of 132 aggregated trials yielded 119 correct identifications, producing a p-value of $5.9 \times 10^{-23}$, indicating a near-zero probability of accidental occurrence 13.
Invertebrate Behavioral Models
Insects have provided more robust behavioral evidence for isotopic discrimination. Because insects generally possess significantly fewer olfactory receptor variants compared to mammals, it is theorized that they may leverage secondary detection layers, such as vibrational spectroscopy, to maintain high discriminatory acuity across diverse chemical classes 21.
In 2011, Turin, Skoulakis, and colleagues demonstrated that Drosophila melanogaster (fruit flies) could reliably differentiate between standard acetophenone and deuterated acetophenone in a behavioral T-maze assay 91720. Furthermore, the fruit flies generalized the odor of the deuterated acetophenone to other distinct chemical compounds, such as nitriles, which share analogous spectral vibrational bands in the low-energy infrared region. This cross-generalization strongly implied that the insects were categorizing the scents based on spectral signatures rather than molecular shape 917.
Subsequent studies extended these findings to Apis mellifera (honeybees). Utilizing electroantennogram amplitudes and proboscis extension reflex conditioning, researchers demonstrated that honeybees could be trained to discriminate common odorants from their deuterated counterparts 11721. The bees successfully differentiated isotopologues of 1-octanol, benzaldehyde, and isoamyl acetate across varying levels of deuteration, correlating behavioral avoidance with measurable activity differences at specific glomeruli in the honeybee brain 12122.
| Organism Model | Target Odorant | Successful Discrimination | Primary Citation Source |
|---|---|---|---|
| Drosophila melanogaster (Fruit Fly) | Acetophenone (d8) | Yes | Turin, Skoulakis et al. (2011) 920 |
| Apis mellifera (Honeybee) | 1-octanol, Benzaldehyde | Yes | Paoli et al. (2016) 11721 |
| Human | Benzaldehyde (d6) | Yes (Contested) | Haffenden et al. (2001) 917 |
| Human | Acetophenone (d8) | No | Keller & Vosshall (2004) 91719 |
| Human | Cyclopentadecanone (d28 Musk) | Yes | Gane, Turin et al. (2013) 192023 |
| Melon Fly | Known Attractants | No | Doolittle et al. 1 |
| Florida Harvester Ant | 4-methyl-3-heptanone | No | Blum et al. 1 |
Despite these positive results, isotopic discrimination is not uniform across all invertebrate species. Studies exposing melon flies to deuterated variants of species-specific attractants, and Florida harvester ants to deuterated ketone alarm pheromones, failed to elicit any differential behavioral responses 1.
Moreover, critics of the vibration theory caution against interpreting systemic behavioral differentiation as definitive proof of inelastic electron tunneling at the primary receptor level. "Perireceptor events" - biological processes that occur before the odorant successfully docks with the OR - can be heavily influenced by isotopic substitution 131923. The kinetic isotope effect establishes that enzymes process heavier isotopes at significantly different reaction rates. Consequently, deuterated odorants might be degraded differently by cytochrome P450 enzymes in the nasal mucosa, or transported at different velocities by odorant-binding proteins through the hydrophilic mucus layer, leading to distinct concentration gradients and novel metabolic byproducts reaching the receptors 11719. Such mechanisms could easily account for variations in perceived odor character independent of quantum tunneling.
Receptor-Level Biophysics and Molecular Evidence
To isolate the mechanism of primary activation and bypass the confounding physiological variables of whole-organism testing, research has shifted toward directly assaying isolated mammalian olfactory receptors in controlled in vitro environments.
Heterologous Expression and Isotope Screening
In 2015, a landmark biophysical study led by Eric Block aimed to rigorously test the vibration theory at the receptor level. Rather than observing organismal behavior, the team utilized a heterologous expression system to screen a library of 330 human olfactory receptors against various odorants 1319. They successfully identified OR5AN1 as the specific human receptor tuned to recognize macrocyclic musk compounds, responding robustly to both cyclopentadecanone and muscone. Simultaneously, they isolated the mouse receptor MOR244-3, which is highly tuned to (methylthio)methanethiol, a sulfurous thiol compound 91923.
With the primary receptors identified, Block et al. conducted extensive dose-response assays comparing the activation dynamics of the standard hydrogenated ligands against their fully deuterated (and carbon-13 substituted) isotopologues 1923. The in vitro results were unequivocal: the receptors failed to distinguish between the isotopomers. The activation curves, efficacy, and binding affinities for normal and deuterated cyclopentadecanone at the OR5AN1 receptor were virtually identical 91923. The same null result was observed for MOR244-3 exposed to isotopologues of its primary thiol ligand 1923.
The experimental data was supported by quantum mechanics/molecular mechanics (QM/MM) simulations. The computational models indicated that electron transfer through the bound ligand has a negligible energetic impact on the potential energy landscape of the receptor 1923. Furthermore, theoretical analyses demonstrated that the proposed inelastic electron tunneling mechanism would likely be overwhelmed and suppressed by the quantum noise and structural vibrations inherent to the non-odorant background amino acids comprising the receptor pocket 923. The researchers concluded that the exhaustive lack of correlation between OR-level signaling and isotopic manipulation rendered the vibration theory biophysically implausible in mammalian systems, strongly reinforcing the traditional shape theory 91923.
High-Resolution Structural Biology: Cryo-Electron Microscopy
The most definitive evidence regarding the activation mechanism of olfactory receptors emerged in 2023, marking a transformative milestone in sensory biology. For over three decades, structural analysis of mammalian ORs had been hindered by their extreme instability and low surface expression when isolated from native tissues, necessitating a heavy reliance on predictive computational homology models 161824. This barrier was finally overcome when a multi-institutional team utilized advanced cryogenic electron microscopy (cryo-EM) to resolve the first high-resolution atomic structure of a human olfactory receptor, OR51E2, bound to its primary ligand, the short-chain fatty acid propionate 24252627.
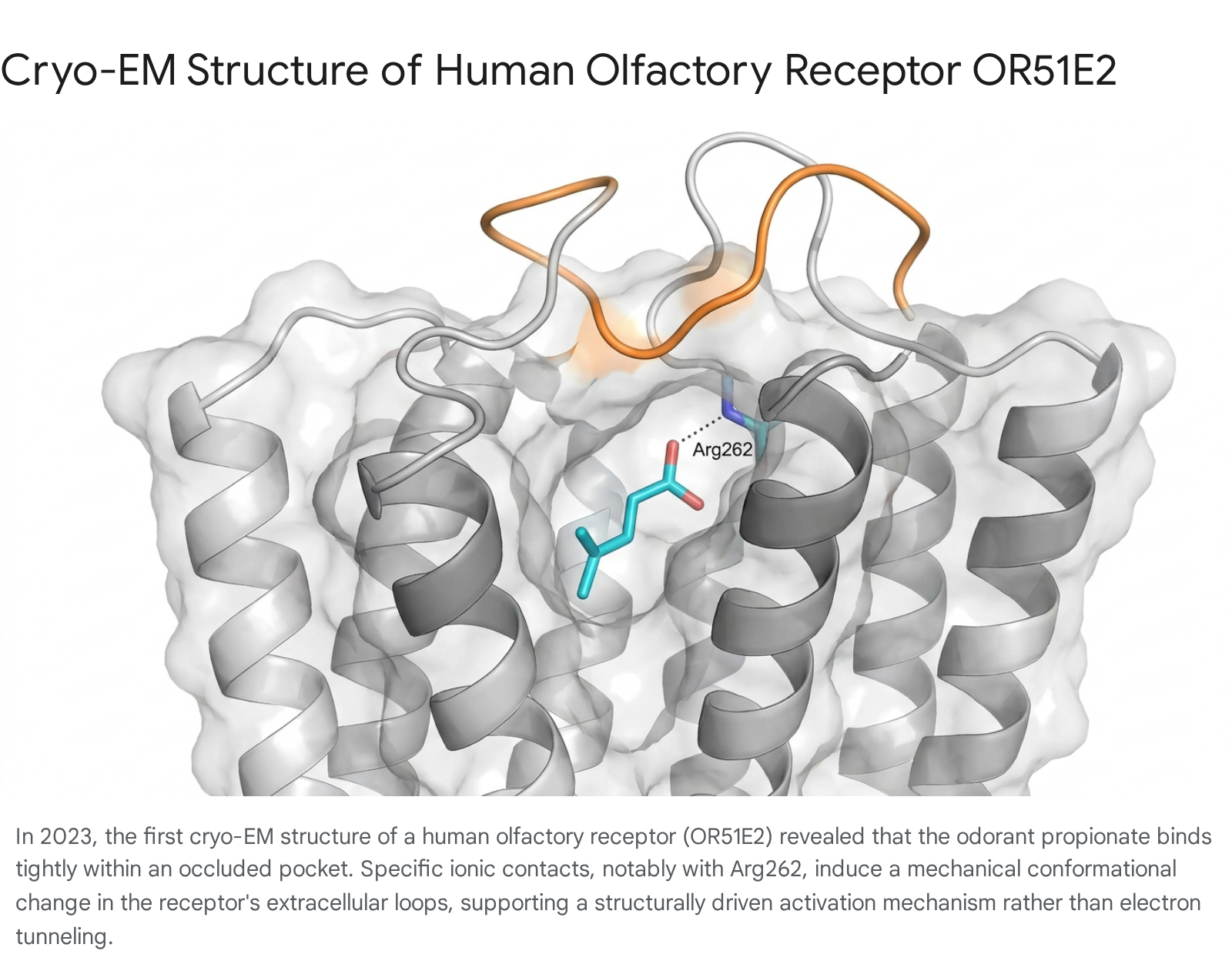
The cryo-EM maps provided unprecedented clarity regarding the mechanics of odor recognition. Propionate docks deeply within an occluded binding pocket primarily defined by transmembrane helices 3, 4, 5, and 6 2434. In direct contrast to the theoretical models proposed by the vibration theory, the activation of the receptor is entirely mediated by classical steric packing and electrostatic interactions. The critical interaction occurs when the carboxylic acid moiety of the propionate molecule forms a strong, direct ionic bond with a specific arginine residue ($\text{Arg262}^{6.59}$) located on Extracellular Loop 3 (ECL3) 82434.
Molecular dynamics simulations of the resolved structure elucidated the sequence of activation. In the absence of an odorant, ECL3 and its associated intracellular structural motifs (such as the FYGx motif) exhibit significant rotational flexibility and conformational heterogeneity 834. When propionate binds, the ionic anchoring physically pulls and stabilizes ECL3, breaking existing hydrogen bonds (e.g., between Tyr251 and Ser111) and triggering a mechanical shift in transmembrane helix 6 242734. This cascading steric reorientation opens the intracellular binding pocket, facilitating the coupling of the heterotrimeric G-protein 252627. A similar activation mechanism was verified via the engineering and cryo-EM structural resolution of a synthesized consensus receptor, consOR51, which validated the highly conserved nature of these steric binding motifs across the receptor family 2528. No evidence of a functional zinc coordination site specifically positioned to mediate an inelastic electron tunneling junction through the ligand was identified in these structures 2434.
In 2024, researchers applied advanced computational infrared spectroscopy directly to the 3D atomic coordinates of the OR51E2 structure to assess whether any subtle vibrational detection was occurring 18. The simulations monitored the precise infrared spectrum of propionate and its deuterated variants as they sat within the physiological confines of the active receptor over a 500-nanosecond timescale. The results indicated that while the high-frequency C-H modes of the molecule predictably shifted by roughly 800 $\text{cm}^{-1}$ upon deuteration, the vibrational modes associated specifically with the carboxyl group remained almost completely unaffected 18. Because propionate binds and activates the OR51E2 receptor almost exclusively via this carboxyl end, the portion of the molecule responsible for the functional ionic coupling undergoes no meaningful vibrational shift upon isotopic substitution. Consequently, the researchers concluded that a vibrational component to propionate detection by OR51E2 is highly unlikely, fully corroborating the shape theory at the atomic level 18.
Expanding Theoretical Horizons in Quantum Biology
While the structural and in vitro evidence strongly disfavors the vibration theory within the primary human olfactory system, the core concepts of quantum mechanics continue to shape theoretical biophysics and adjacent fields of neuropharmacology.
Extrapolating Vibration Detection to Other Receptors
Current biophysical research has hypothesized that if inelastic electron tunneling does not mediate olfaction, it may instead play a role in other pharmacological and neurological signaling systems. A 2024 review published in Bioessays outlines theoretical frameworks extending the principles of the vibration theory to ligand-gated ion channels and metabotropic receptors 61436. Researchers emphasize that while the classical lock-and-key model effectively explains how a ligand binds, the precise mechanism by which the physical binding event is translated into channel activation or allosteric modulation remains, in some instances, inadequately characterized 14. By utilizing deuterated ligand analogs and analyzing the vibrational spectra of pharmacological agents - such as the effects of nicotine and cotinine in planarian behavioral models - investigators are probing whether quantum tunneling events might facilitate the rapid state-changes observed in complex neural receptors 1436.
Alternative Quantum Theories of Smell
Within the specific domain of olfaction, highly theoretical frameworks continue to be published in an attempt to completely rewrite the energetic processing of sensory data. In 2023 and 2024, Jan Myjkowski published the "Quantum Theory of Smell," a model diverging significantly from Turin's framework. Myjkowski proposes that olfactory receptors do not merely detect the kinetic or vibrational oscillations of a molecule; rather, they serve as conduits for the transfer of the total quantized energy of the odorant - encompassing its kinetic, potential, and electronic states - directly into the central nervous system 45. The theory challenges fundamental tenets of standard electrophysiology by arguing that cellular depolarization acts on an "all-or-nothing" principle and thus cannot accurately encode complex, binned olfactory information 4537. Consequently, the theory posits that continuous, proportional energy streams must be transferred from the odorant to the receptor, through the G-protein, and into intracellular energy metabolism pathways to establish an accurate olfactory image in the brain 4537.
Similarly, a 2023 hypothesis titled the "Luminescence Hypothesis of Olfaction" attempts to merge electron tunneling with photobiology 1329. This model agrees with the initial premise of the vibration theory - that an odorant undergoes an inelastic electron tunneling event upon binding. However, it proposes that this tunneling elevates the odorant molecule to an excited, high-energy state. As the molecule naturally decays back to its ground state, it releases energy via fluorescence luminescence, emitting multiple radiative photons 13. These photons purportedly carry roughly 2.5 eV of energy and pass through the surrounding sustentacular cells to activate adjacent olfactory neurons in a highly synchronized, near-simultaneous pattern 13. Proponents suggest this 1:3 stoichiometric emission of photons provides the temporal basis necessary for the brain to decode complex combinatorial odors, and theorize it may account for clinical phenomena such as COVID-19-induced anosmia 1329. Both Myjkowski's total energy framework and the Luminescence Hypothesis remain highly speculative components of the literature, lacking the rigorous in vitro structural validation that currently defines the field.
Differentiating Quantum Biology from Quantum Consciousness
It is necessary to contextualize the ongoing research into quantum olfaction within the broader, rapidly maturing discipline of quantum biology 3031. The field rigorously investigates processes where non-trivial quantum phenomena - such as long-lived coherence, quantum entanglement, and highly optimized tunneling - are fundamental to biological function in warm, wet environments 3032. Well-documented examples include the utilization of exciton coherence in the Fenna-Matthews-Olson (FMO) complex during photosynthesis to achieve near-perfect energy transfer efficiency, and the radical pair mechanism underpinning magnetoreception in migratory birds like the European robin, where entangled electrons act as highly sensitive magnetic compasses 303233.
The existence of these verified quantum biological mechanisms ensures that theories like the vibration theory of olfaction are treated as biophysically plausible hypotheses worthy of investigation, rather than pseudoscience 32. However, researchers in the field strictly delineate these evidence-based mechanisms from the speculative domains of "quantum consciousness" 443435. While quantum olfaction proposes a highly specific, localized tunneling event at a receptor site to detect a chemical, theories of quantum consciousness - such as the Orchestrated Objective Reduction (Orch-OR) theory involving cellular microtubules - extrapolate macroscopic cognitive awareness and subjective experience from subatomic wave-function collapses 324434. Mainstream quantum biology generally views the latter as lacking empirical foundation, while focusing on the precise molecular interactions that drive enzyme catalysis, vision, and sensory transduction 3234.
Conclusion
The mechanism by which the olfactory system discriminates among an almost infinite array of chemical odorants remains a central challenge in molecular biology. The vibration theory of olfaction, principally championed by Luca Turin, provided a theoretically elegant framework to address the anomalies inherent in classical docking models. By proposing that olfactory receptors function as nanoscale electron tunneling spectrometers, the theory offered a mathematically viable, quantum mechanical solution to structure-odor discrepancies 9131517.
However, the current scientific consensus overwhelmingly favors the classical shape theory of olfaction 1181923. While behavioral studies demonstrating isotopic discrimination in insects initially bolstered the vibration theory, subsequent analyses suggest these results are likely products of systemic perireceptor events, such as differing enzymatic metabolic rates and mucosal diffusion profiles related to the kinetic isotope effect 11719. Crucially, exhaustive in vitro testing of isolated human olfactory receptors has consistently failed to demonstrate any sensitivity to isotopic substitution or vibrational shifts 1923.
The debate has been largely settled at the molecular level by the 2023 advent of cryogenic electron microscopy in olfactory research 82427. The structural resolution of the human OR51E2 receptor unequivocally demonstrates that odorant recognition and subsequent receptor activation are mediated entirely by classical steric packing, electrostatic interactions, and induced-fit mechanical shifts 242634. Advanced computational models utilizing these physical structures confirm that the vibrational modes relevant to tunneling are functionally absent at the primary binding interface 18. While quantum mechanics is integral to all fundamental biochemical interactions, the specialized inelastic electron tunneling mechanism proposed by the vibration theory is not the driver of primary olfactory transduction in mammals.