Psychoneuroimmunology and the effect of mental states on immunity
Introduction to Psychoneuroimmunology
Psychoneuroimmunology represents the interdisciplinary study of the complex, bidirectional interactions among the central nervous system, the endocrine system, and the immune system, and how these interactions collectively impact physical and mental health. For much of the twentieth century, the biomedical establishment conceptualized the immune system as an autonomous, self-regulating defense network that operated entirely independently of the brain, cognitive processes, and psychological states 1. This Cartesian dualism began to permanently shift in the 1970s and 1980s, catalyzed by the foundational laboratory work of researchers such as Robert Ader and Nicholas Cohen. Their experiments demonstrated that immune responses could be classically conditioned in animal models 12. By repeatedly pairing an artificial sweetener with an immunosuppressive drug, they revealed that animals could learn to suppress their immune systems based solely on the conditioned taste, fundamentally proving that the central nervous system could directly control immune cell activity 1.
Shortly after Ader's behavioral discoveries, neuroanatomist David Felten provided the physical evidence for this connection by identifying direct neural innervations of primary and secondary lymphoid organs, including the thymus, spleen, bone marrow, and lymph nodes 13. Subsequent physiological research completely dismantled the dualistic view of mind and body by revealing that immune cells possess receptors for a wide array of neurotransmitters and neuroendocrine hormones 45. Concurrently, it was discovered that neurons and glial cells within the central nervous system express receptors for immune-signaling molecules, such as cytokines, establishing a continuous, bidirectional chemical dialogue 45.
Modern psychoneuroimmunology has evolved into a highly specialized, global field utilizing systems biology, multi-omics, and advanced functional neuroimaging to map these exact pathways 67. Current research endeavors focus on translating these biological mechanisms into clinical applications, exploring how cognitive states, chronic psychological stress, and behavioral interventions physically alter cellular immune function. Inversely, the field investigates how systemic inflammation drives psychopathology, cognitive decline, and affective disorders, challenging traditional psychiatric diagnostic boundaries 248.
Biological Pathways of Brain-Immune Communication
The translation of subjective psychological states into objective physical immunological changes is mediated by three primary pathways: the hypothalamic-pituitary-adrenal axis, the sympathetic nervous system, and the cholinergic anti-inflammatory pathway. These systems operate continuously to maintain physiological homeostasis, responding to both external psychological stressors and internal pathogenic threats.
The Hypothalamic-Pituitary-Adrenal Axis
The hypothalamic-pituitary-adrenal (HPA) axis serves as the primary systemic neuroendocrine conduit through which the brain regulates systemic inflammation and immune reactivity over prolonged periods. The cascade initiates in the brain when an individual perceives a psychological threat or experiences severe emotional distress. In response, the paraventricular nucleus of the hypothalamus synthesizes and secretes corticotropin-releasing hormone (CRH) into the hypophyseal portal system 4910. The arrival of CRH stimulates the anterior pituitary gland to release adrenocorticotropic hormone (ACTH) into the systemic blood circulation 9. ACTH subsequently travels to the adrenal glands, where it acts upon the adrenal cortex to trigger the synthesis and secretion of glucocorticoids, which in humans is primarily cortisol 910.
Glucocorticoids possess potent, broad-spectrum immunosuppressive properties that physically alter the behavior of immune cells at the genomic level. Because cortisol is lipophilic, it diffuses readily across cell membranes and binds to intracellular glucocorticoid receptors, which are abundantly expressed on virtually all leukocyte populations, including T lymphocytes, B lymphocytes, and macrophages 1112. Upon binding the hormone, the glucocorticoid-receptor complex translocates into the cell nucleus, where it alters the transcription of multiple target genes. This process fundamentally inhibits the cellular machinery responsible for producing pro-inflammatory cytokines, such as interleukin-1 beta (IL-1β), interleukin-6 (IL-6), and tumor necrosis factor-alpha (TNF-α) 101112. Concurrently, glucocorticoids suppress the rapid proliferation and cytotoxicity of natural killer (NK) cells and T lymphocytes, temporarily dampening the body's defensive capabilities 1112.
While the acute, short-term activation of the HPA axis serves as a protective regulatory mechanism to prevent excessive, self-destructive tissue damage from hyperinflammation during an injury, chronic psychological stress leads to severe HPA axis dysregulation. Prolonged, unremitting exposure to high levels of cortisol induces a phenomenon known as glucocorticoid resistance within the immune cell populations. In this state, leukocyte receptors downregulate or become desensitized to cortisol, creating a paradoxical physiological environment where the immune system is no longer effectively suppressed by the endocrine system, resulting in a state of systemic, low-grade chronic inflammation that damages local tissues 411.
The Sympathetic Nervous System Interface
While the HPA axis provides a slow, systemic hormonal blanket over the immune system, the sympathetic division of the autonomic nervous system provides a direct, highly targeted, and instantaneous neural link between the brain and immune organs. Unlike systemic endocrine distribution, the sympathetic nervous system offers regional regulation through the direct physical innervation of immune parenchyma. Sympathetic efferent nerve fibers project from the brainstem and thoracolumbar spinal cord to terminate directly within the specialized microenvironments of the spleen, thymus, lymph nodes, and bone marrow 312.
The primary neurotransmitters released by these postganglionic sympathetic fibers are catecholamines, specifically norepinephrine and epinephrine 1112. Immune cells express varying densities of both alpha- and beta-adrenergic receptors on their cell surfaces, making them exquisitely responsive to sympathetic nervous system activity. When psychological stress activates the sympathetic nervous system, commonly known as the "fight-or-flight" response, the immediate synaptic release of catecholamines dynamically alters immune cell trafficking, localized proliferation, and cytokine secretion profiles 81112.
A critical immunomodulatory consequence of sympathetic activation is the forced alteration of the T-helper 1 (Th1) and T-helper 2 (Th2) balance. Epinephrine and norepinephrine stimulation through beta-2 adrenergic receptors typically suppresses the production of Th1-type pro-inflammatory cytokines, such as interleukin-12 (IL-12) and interferon-gamma (IFN-γ), which govern cellular immunity 1112. Simultaneously, this sympathetic signaling upregulates the production of Th2-type anti-inflammatory cytokines, such as interleukin-10 (IL-10), which govern humoral immunity 1112. This stress-induced shift away from cellular immunity toward humoral immunity can be advantageous for surviving acute trauma but heavily compromises the body's defense against viral infections and the surveillance of nascent tumor cells if the psychological stress becomes a chronic condition 1112.
The Cholinergic Anti-Inflammatory Pathway
Perhaps the most significant anatomical and physiological discovery in contemporary psychoneuroimmunology is the characterization of the cholinergic anti-inflammatory pathway, frequently referred to as the vagal inflammatory reflex. Identified and mapped by Kevin Tracey and his colleagues in the early 2000s, this pathway describes a rapid, hardwired neuroimmune circuit that allows the central nervous system to instantaneously sense and suppress peripheral inflammation, protecting the organism from lethal endotoxic shock 13141516.
The sensory, or afferent, arc of this reflex begins with the vagus nerve (cranial nerve X), which continuously monitors the homeostatic state of the peripheral organs 1017. Approximately 80% of vagal nerve fibers are sensory afferents. These sensory fibers express specific receptors for pro-inflammatory cytokines, including IL-1β and TNF-α, as well as pattern recognition receptors like Toll-like receptors (TLRs) and transient receptor potential (TRP) channels such as TRPA1 and TRPV1 13181920. When peripheral immune cells detect a pathogen or tissue injury and release a surge of cytokines, these molecular signals bind to the vagal afferents, generating high-speed action potentials that are transmitted up the nerve to the nucleus tractus solitarius (NTS) located in the medulla oblongata of the brainstem 9141721.
Once the brainstem receives and integrates this inflammatory information, it initiates the motor, or efferent, arc of the reflex. Efferent signals travel back down the vagus nerve toward the abdominal organs, initiating a highly specific, multi-step biological sequence at the neuro-immune interface. This sequence heavily involves the spleen, which is a major reservoir of immune cells and a critical site for blood filtration 14202122. The efferent vagus nerve fibers do not innervate the spleen directly; rather, they synapse at the celiac superior mesenteric ganglion located in the abdomen 91523. Within the celiac ganglion, the vagal terminals release acetylcholine, which binds to nicotinic receptors on postganglionic sympathetic noradrenergic neurons that project through the splenic nerve directly into the spleen 92024.
As these splenic nerve terminals depolarize, they release norepinephrine deep within the splenic parenchyma 162024. This localized burst of norepinephrine binds to beta-2 adrenergic receptors located on a specialized subset of memory CD4+ T-lymphocytes that express the enzyme choline acetyltransferase (ChAT) 15202225. Upon noradrenergic stimulation, these unique ChAT+ T-cells synthesize and secrete acetylcholine directly into the local splenic microenvironment, acting effectively as a non-neural source of neurotransmitters 15162025. Finally, this T-cell-derived acetylcholine binds to alpha-7 nicotinic acetylcholine receptors ($\alpha 7$nAChR) located on the surface of nearby splenic tissue macrophages. This receptor activation triggers an intracellular signaling cascade that inhibits the translocation of nuclear factor kappa B (NF-$\kappa$B) and activates the JAK2-STAT3 pathway, effectively halting the synthesis and release of TNF-$\alpha$ and other highly toxic pro-inflammatory cytokines before they can cause systemic damage 101317182526.
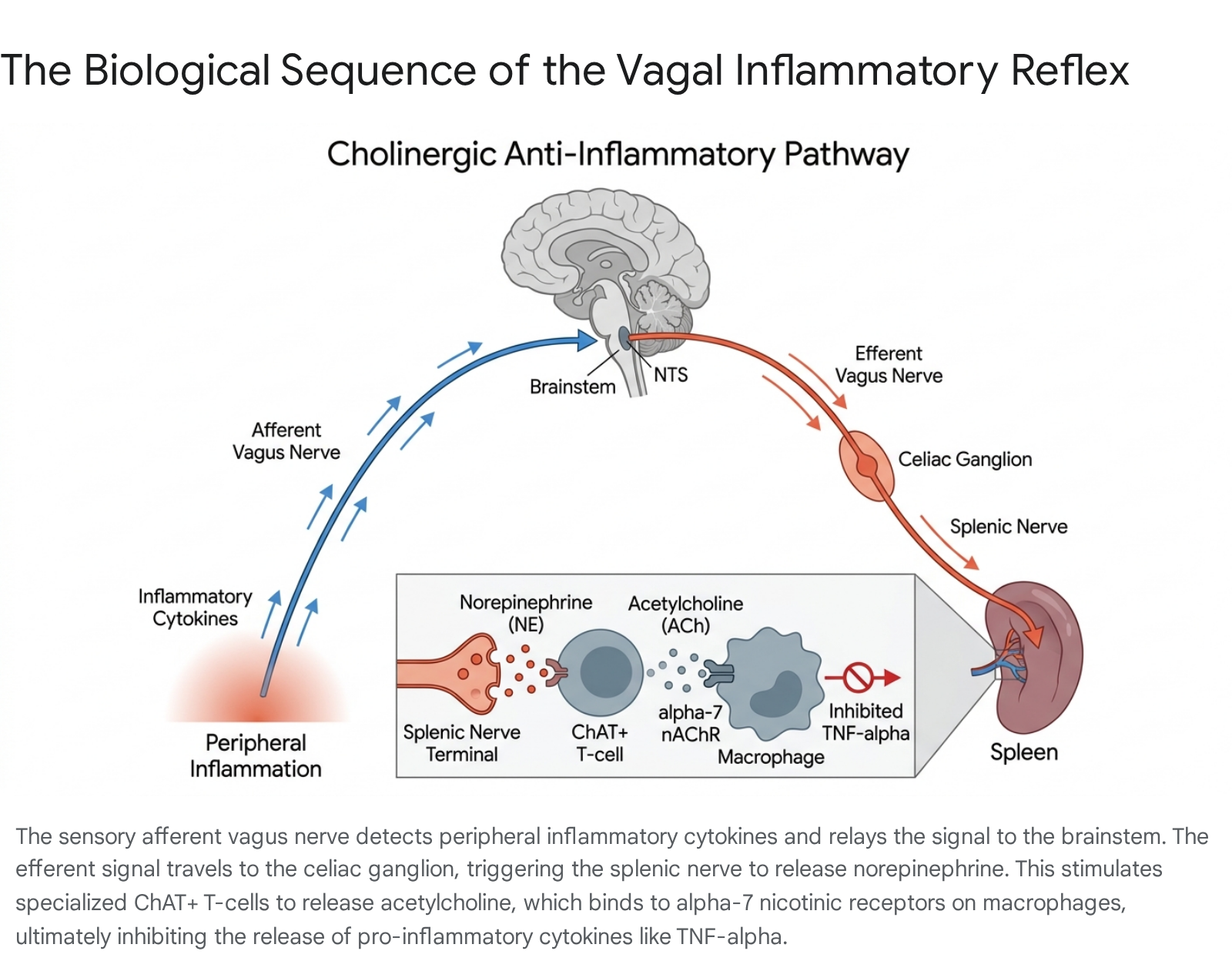
This intricate biological circuit provides a remarkably fast, localized, and highly precise mechanism for the central nervous system to apply a biological brake to the immune response, preventing systemic inflammatory shock and finely regulating the intensity of host defense mechanisms 1524.
Immune-to-Brain Signaling and Neuroinflammation
While top-down regulation explains how the brain alters immunity, psychoneuroimmunology equally encompasses the bottom-up communication pathways whereby the peripheral immune system dynamically alters brain function, cognition, and emotional states. Cytokines produced during peripheral infections, autoimmune flare-ups, or severe psychological stress are large polypeptides that do not readily cross the tight junctions of the blood-brain barrier (BBB) under normal conditions. However, the immune system successfully signals the brain through several distinct, parallel mechanisms 132326.
First, cytokines can enter the central nervous system through circumventricular organs, which are specialized brain regions (such as the area postrema) where the blood-brain barrier is intentionally porous to allow for chemical sampling of the blood 132326. Second, circulating cytokines can bind to receptors on the cerebral microvasculature, prompting these endothelial cells to synthesize and secrete secondary messengers, primarily prostaglandins, directly into the brain parenchyma 13. Third, as previously discussed, cytokines activate vagal afferent nerves, which transmit rapid neurochemical signals to the brainstem and ultimately into the higher cortical and limbic structures responsible for mood and behavior 101719.
Once the inflammatory signal successfully breaches or signals the central nervous system, it fundamentally alters local neurotransmitter metabolism - particularly decreasing the availability of serotonin and dopamine while increasing glutamate - and activates microglia, the resident resident macrophage-like immune cells of the brain 52027. Microglial activation triggers a state of active neuroinflammation, which is biologically responsible for initiating "sickness behavior" 132829. Sickness behavior is a highly conserved evolutionary response characterized by profound fatigue, anhedonia, social withdrawal, altered sleep architectures, and increased anxiety 132829. By forcing the organism to rest and withdraw, the brain conserves vital metabolic energy necessary to mount a fever and fuel the immune system to fight the presumed infection.
Recent breakthroughs in cellular tracking have further expanded our understanding of direct immune-to-brain signaling. In 2024, researchers at Mount Sinai demonstrated that during periods of severe psychosocial stress, circulating peripheral monocytes - immune cells originating in the bone marrow - are actively drawn out of the bloodstream and across the blood-brain barrier into specific brain regions that control emotional behaviors 30. Once entrenched in the brain tissue, these monocytes release an enzyme called matrix metalloproteinase 8 (MMP8) 30. MMP8 physically breaks down the extracellular matrix and restructures synaptic connections between neurons, directly impairing social behavior and reward processing 30. This structural alteration provides a tangible, physical mechanism by which peripheral immune cells can infiltrate the brain to induce depressive phenotypes following psychosocial trauma.
Psychological Stress and Immune Modulation
The immune system's physiological response to psychological stress is highly dynamic and depends entirely on the duration, intensity, and chronicity of the stressor. Within psychoneuroimmunology, a sharp biological distinction is made between acute, short-term stressors and chronic, unrelenting psychological strain.
Acute versus Chronic Stress Responses
Acute stressors, such as public speaking, taking an academic examination, or responding to a sudden emergency, generally trigger a short-term up-regulation of innate immunity. Evolutionary biology suggests this is a preparatory mechanism; the body anticipates potential physical injury during a highly stressful encounter and primes the innate immune system to facilitate immediate wound healing and prevent bacterial infection 2831. Acute stress results in the rapid mobilization of leukocytes into the bloodstream and a transient increase in the production of pro-inflammatory cytokines, such as IL-6, IL-1β, TNF-α, and IL-8 3132. Studies tracking salivary and serum biomarkers in human subjects, such as firefighters undergoing highly stressful training events, demonstrate significant and immediate spikes in these cytokines following the stressor, with the magnitude of the spike directly correlating with the individual's perceived stress levels and history of past trauma 3132.
Conversely, chronic psychological stress - such as prolonged caregiving for a spouse with dementia, occupational burnout, enduring systemic poverty, or living in a violent environment - results in profound and highly destructive immune dysregulation. Comprehensive meta-analyses demonstrate that chronic stress persistently suppresses adaptive cellular immunity 1112. It physically impairs T-cell proliferation, drastically decreases the cytolytic toxicity of natural killer cells, and severely reduces the efficacy of the immune system's response to prophylactic vaccines 1233.
Concurrently, chronic stress causes the body to lose regulatory control over baseline inflammation. Due to mechanisms like glucocorticoid receptor resistance, the body loses its hormonal ability to downregulate the inflammatory cascade. This leads to a state of chronic, low-grade systemic inflammation characterized by persistently elevated circulating levels of IL-6 and C-reactive protein (CRP), significantly elevating the risk for autoimmune disease and atherosclerosis 41134. Another critical marker of chronic allostatic load is the reactivation of latent viruses, such as the Epstein-Barr virus (EBV) or Herpes Simplex Virus type 1 (HSV-1), because the chronically stressed cellular immune system lacks the resources to keep these dormant viruses in check 3536.
| Immunological Parameter | Acute Stress Response Profile | Chronic Stress Response Profile |
|---|---|---|
| Exposure Duration | Minutes to hours | Weeks, months, or years |
| HPA Axis Activity | Transient, functional spike in cortisol secretion | Sustained elevation or blunted rhythm; severe glucocorticoid resistance |
| Innate Immunity | Rapid cellular mobilization; increased transient inflammation to prepare for injury | Sustained low-grade systemic inflammation (elevated baseline IL-6, CRP, TNF-α) |
| Adaptive Immunity | Temporary shift toward Th2; temporary redistribution of cells | Chronically suppressed T-cell proliferation; severely reduced vaccine efficacy |
| Cellular Function | Maintained or temporarily enhanced | Drastically decreased Natural Killer (NK) cell cytotoxicity |
| Viral Control | Latent viruses remain successfully suppressed | Frequent reactivation of latent pathogens (e.g., Epstein-Barr virus, HSV-1) |
| Clinical Outcomes | Adaptive preparation for physical injury | Increased susceptibility to viral infections, autoimmune flare-ups, and cardiovascular disease |
Systemic Inflammation and Psychiatric Disorders
The link between psychological stress, systemic inflammation, and severe psychiatric disorders is a cornerstone of modern psychoneuroimmunological research. Major Depressive Disorder (MDD), Post-Traumatic Stress Disorder (PTSD), and severe anxiety disorders are increasingly viewed by researchers not solely as localized neurochemical imbalances, but as systemic immuno-inflammatory conditions affecting the entire body 5313738.
Individuals with a prior diagnosis of PTSD exhibit fundamentally altered resting immune profiles. Baseline blood measurements indicate significantly higher levels of pro-inflammatory cytokines, specifically IL-6 and TNF-α, and altered overall leukocyte counts compared to healthy controls without a trauma history 31. When subjected to a new, minor acute stressor, individuals with a trauma history display an exaggerated and disproportionate inflammatory response, providing biological evidence that severe psychological trauma leaves a lasting physical imprint on immune reactivity and regulatory thresholds 31.
Experimental models of endotoxemia in human subjects provide the most direct evidence of this causal pathway. In highly controlled settings, when healthy human volunteers are administered a low, safe dose of Salmonella abortus equi endotoxin, it triggers a mild, transient spike in systemic cytokines (TNF-α, IL-6) without causing severe physical sickness or fever 39. Within hours of the peripheral immune activation, participants report significant and measurable increases in anxiety, depressed mood, and noticeable, quantifiable declines in memory performance 39. The severity of the mood and cognitive disturbances correlates directly and linearly with the volume of circulating cytokines, proving beyond doubt that peripheral immune activation physically alters emotional and cognitive states 39.
Neuroimaging and Immuno-Inflammatory Biotypes
Recent advancements combining multimodal neuroimaging and plasma proteomics have allowed researchers to map the physical footprint of systemic inflammation directly within the living human brain. In large-scale breakthrough studies published in 2024 and 2025, researchers successfully identified specific "immuno-inflammatory biotypes" that cross traditional boundaries of major psychiatric disorders 3740. By integrating functional magnetic resonance imaging (fMRI) with DNA methylation profiles of immune genes, studies have identified distinct functional connectivity markers in the brain that correlate directly with systemic immune dysfunction 37.
Patients classified into this specific immuno-inflammatory biotype display reliably elevated blood markers, including a high neutrophil-to-lymphocyte ratio and a high systemic immune-inflammatory index 3740. Clinically, these patients respond significantly worse to conventional monoaminergic antidepressant therapies compared to non-inflammatory biotypes 373840. This research establishes crucial, non-invasive neuroimaging markers of brain inflammation, laying the biological foundation for precision psychiatry. In the near future, psychiatric patients could be biologically triaged toward targeted anti-inflammatory or immunomodulatory therapies based entirely on their specific, measurable neuro-immune profile 4041.
Lifestyle Factors and Methodological Confounders
A persistent critical challenge in psychoneuroimmunology human trials is effectively isolating the direct effects of thoughts and emotions from the secondary behavioral changes that invariably accompany stress. Stressed individuals frequently and subconsciously alter their lifestyle behaviors: their sleep architectures deteriorate, their diets worsen, they exercise less, and they may increase substance and alcohol use 12. These behaviors are themselves highly potent modulators of immune function, serving as critical methodological confounders that must be controlled in rigorous human research.
The Role of Sleep Disruption
Sleep architecture is intricately and bidirectionally linked to both the central nervous system and the immune system. Normal, healthy sleep plays a vital restorative role, particularly in supporting adaptive cellular immunity and the crucial process of immunological memory consolidation 3342. Sleep disturbance - whether manifesting as clinical insomnia, chronic voluntary sleep restriction, or poor sleep quality secondary to anxiety - has a profound and highly measurable influence on systemic inflammation 334344.
Psychoneuroimmunological studies over the past decade consistently demonstrate that acute and chronic sleep restriction actively downregulates adaptive antiviral immune responses while simultaneously upregulating innate pro-inflammatory responses 3342. Chronic sleep deprivation increases cellular and genomic markers of inflammation, significantly elevating the biological risk for inflammatory-mediated morbidities such as cardiovascular disease, metabolic syndrome, and clinical depression 334244. The relationship operates bidirectionally: peripheral inflammation and elevated cytokines (specifically IL-1 and TNF-α) physically cross the blood-brain barrier to alter sleep architecture, often drastically increasing non-rapid eye movement (NREM) sleep in the acute phase of an infection to promote healing, while fragmenting sleep continuity overall 52845.
Dietary Influences and the Enteric Nervous System
Dietary composition significantly influences both objective sleep quality and systemic inflammation, acting as another vital, often overlooked variable in the mind-body matrix. Diets exceptionally high in saturated fats and refined carbohydrates are known to provoke inflammatory responses, whereas diets rich in dietary fiber, healthy unsaturated fats, and adequate protein (such as the Mediterranean diet) are associated with significantly lower systemic inflammation and improved objective sleep quality metrics as measured by polysomnography 444647.
Furthermore, the enteric nervous system (ENS) and the gut microbiome are inextricably linked to the vagal anti-inflammatory pathway. Vagal afferent sensory fibers continuously monitor the gastrointestinal tract, detecting metabolites, neuroactive compounds, and inflammatory signals produced by the microbiome flora 17484950. Dysbiosis - a severe imbalance in the microbial community often directly exacerbated by chronic psychological stress - can increase intestinal permeability 1149. This "leaky gut" phenomenon allows bacterial endotoxins to enter the systemic circulation 1149. This low-grade endotoxemia triggers a sustained, chronic inflammatory response that signals the brain via the vagus nerve, leading to secondary neuroinflammation and mood dysregulation, highlighting exactly why stringent dietary controls are absolutely vital in rigorous psychoneuroimmunology human trials 1149.
The Limits of Psychological Influence on Pathology
As the field of psychoneuroimmunology gained public awareness over the past two decades, highly nuanced scientific findings regarding stress and immunity were occasionally extrapolated into unsubstantiated, pseudoscientific claims. Most notable is the widespread public assertion that adopting a strictly "positive attitude" possesses the biological power to cure severe organic diseases, particularly cancer. This concept, often termed the "tyranny of positivity," places an immense and highly detrimental psychological burden on patients, leading them to falsely believe that experiencing natural negative emotions or distress might somehow accelerate their disease progression 515252.
Extensive peer-reviewed epidemiological and oncological research has firmly and definitively debunked this claim. A comprehensive, large-scale study published by the American Cancer Society, which rigorously analyzed over 1,000 patients suffering from head and neck cancer, demonstrated that a patient's emotional well-being, psychological state, or positive attitude had absolutely no direct or indirect effect on cancer survival rates or overall disease progression 5253. While it remains true that prolonged psychological stress suppresses immune surveillance (which is broadly detrimental to overall health), actively enforcing "positive thinking" does not possess the targeted oncological therapeutic power to reverse established tumor growth or alter the physiological prognosis of advanced, life-threatening malignancies 5354.
The scientific consensus within the discipline of psychoneuroimmunology emphasizes that while the absence of chronic toxic stress is highly beneficial for maintaining baseline immunological parameters, relentless, forced positivity is not a biological cure for organic disease 5253. Fostering realistic optimism, allowing patients to safely acknowledge distress, and utilizing effective psychological coping strategies are recommended strictly for the purpose of improving the patient's quality of life, reducing clinical anxiety, and supporting vital adherence to medical treatments, rather than serving as a primary disease-modifying intervention 545556.
Therapeutic Modulation via Behavioral Interventions
Understanding that the brain physically regulates the immune system opens critical avenues for utilizing psychological and behavioral interventions as adjunctive biological treatments. By purposefully modulating the autonomic nervous system and the HPA axis, targeted psychological therapies can attenuate destructive systemic inflammation.
Cognitive Behavioral Therapy and Cytokine Reduction
Cognitive Behavioral Therapy (CBT) is a frontline psychosocial intervention that trains individuals to identify and alter maladaptive cognitive patterns and dampens physiological stress responses. Recent rigorous meta-analyses, encompassing hundreds of randomized controlled trials and thousands of human participants, have explicitly evaluated the biological effect of CBT on peripheral immune biomarkers 3857.
These studies conclusively demonstrate that CBT, alongside other highly structured psychotherapies, is significantly associated with a measurable reduction in systemic pro-inflammatory cytokines 38575859. Specifically, engaging in standard integrated CBT yields small-to-moderate statistical effect sizes in lowering peripheral circulating levels of IL-6, C-reactive protein, and TNF-α 3857. The most robust biological reductions in inflammatory markers were reliably observed in interventions lasting eight weeks or less, suggesting that the initial acquisition of emotional regulation skills prompts a rapid and significant downregulation of the body's physiological stress cascade 57.
Furthermore, the biological benefits of CBT appear highly durable, with studies indicating that immune-related improvements and reductions in inflammation persist for at least six months post-treatment 5859. Because elevated baseline inflammation is strongly associated with treatment resistance in severe psychiatric disorders, utilizing CBT to biologically dampen inflammation presents a critical, non-pharmacological adjunct for improving overall patient outcomes 3858.
Mind-Body Interventions
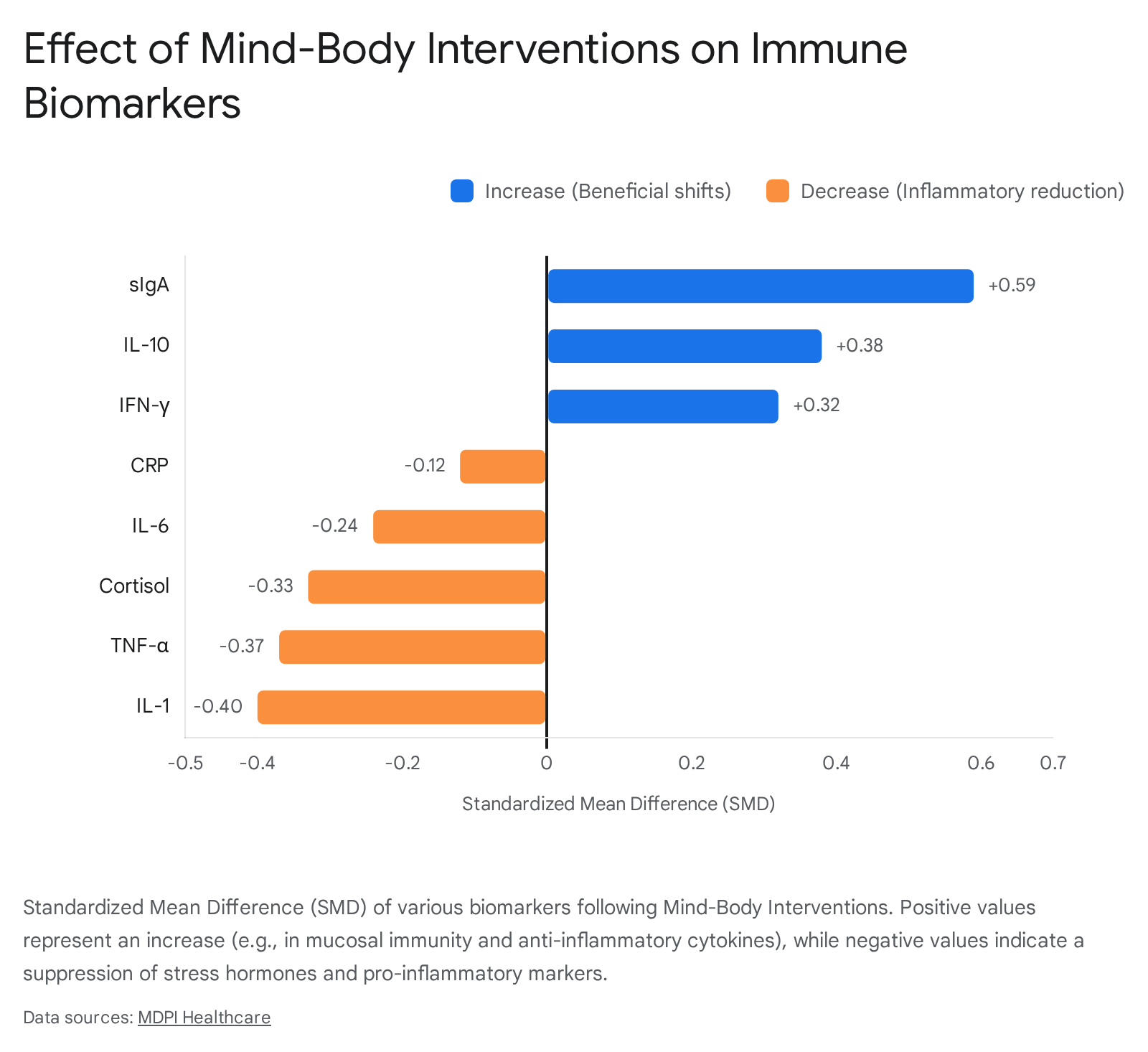
Mind-Body Interventions (MBIs) - an umbrella term encompassing highly structured practices like mindfulness meditation, yoga, Tai Chi, and Qigong - have been heavily scrutinized within psychoneuroimmunological literature for their potential to alter biology 496061. A 2025 comprehensive meta-analysis of randomized controlled trials investigated the precise physiological impact of various MBIs on a wide spectrum of immune and endocrine markers 60.
The compiled data indicate that MBIs effectively regulate psychological reactivity to stressors by significantly decreasing sympathetic tone and highly activating the parasympathetic nervous system 60. Biologically, MBIs generally decrease circulating levels of highly destructive inflammatory factors (including CRP, IL-6, TNF-α, and cortisol) while simultaneously increasing circulating levels of anti-inflammatory and antiviral factors (such as IL-10 and IFN-γ) 60. Furthermore, these interventions significantly increase levels of secretory Immunoglobulin A (sIgA), an critically important marker of mucosal immunity that defends the respiratory and gastrointestinal tracts 60.
In highly vulnerable populations dealing with severe chronic illness, such as individuals diagnosed with HIV facing high psychosocial stress, structured physical practices like Tai Chi have been shown to significantly improve appraisal-focused coping mechanisms, lower psychological distress, and improve overall quality of life 62. These practices act as a powerful biological buffer against the immuno-suppressive effects of chronic stress, providing patients with a non-pharmacological tool to regain homeostatic control.
Biofeedback and Autonomic Regulation
Biofeedback represents a technological approach to mind-body regulation. It utilizes sensitive instrumentation to provide patients with real-time, measurable data regarding their largely involuntary physiological processes - such as heart rate variability (HRV), peripheral skin temperature, respiratory rate, and skeletal muscle tension 636465. By providing continuous visual or auditory cues, individuals can learn strict operant conditioning techniques to exert voluntary, conscious control over their autonomic nervous system 6465.
Through intentional biological modulation - particularly by actively increasing parasympathetic (vagal) tone and decreasing sympathetic arousal - biofeedback forcefully interrupts the physiological stress response 63. High heart rate variability is widely considered a highly accurate index of robust vagal tone and is anatomically associated with greater prefrontal cortical inhibitory control over the amygdala, which subsequently dampens the systemic inflammatory cascade 1819. Clinical trials in populations experiencing extremely high stress, such as university students and elite athletes, have demonstrated that daily biofeedback training significantly improves clinical symptoms of anxiety, depression, and severe insomnia 6366. Crucially, these self-reported psychological improvements are biologically corroborated by measurable alterations in physiological markers and speech-derived stress parameters, confirming biofeedback's immense clinical utility in stabilizing the mind-body interface without medication 66.
Global Perspectives and Non-WEIRD Populations
Historically, psychoneuroimmunology research was predominantly conducted in Western, Educated, Industrialized, Rich, and Democratic (WEIRD) populations, severely limiting the global applicability of the findings. Recently, there has been a massive, concerted effort to globalize the field, exploring how unique cultural traditions, environmental exposures, and severe socio-economic factors shape the mind-body-immune interaction in highly diverse demographics 2767.
Psychoneuroimmunology Research in Asia
Biomedical research investment in Asia has surged over the last two decades, and with it, a rapid expansion of sophisticated psychoneuroimmunology research 27. In China, where the historical and cultural principles of Traditional Chinese Medicine (TCM) have long recognized the holistic integration of mind and body, modern researchers have heavily investigated the exact molecular basis of these ancient connections 2768.
Since the formal establishment of the regional affiliate PNIRS China in 2012, a significant portion of Chinese PNI research has focused intensely on the interactions between neuro-immune disorders mediated by microglia and central nervous system diseases 2768. Extensive local studies investigating stress-induced and inflammation-induced depression models demonstrate that emotional stress rapidly activates the sympathetic nervous system and HPA axis, suppressing leukocyte counts and specific antibody production while drastically increasing the expression of brain cytokines, particularly TNF-α, directly within the hippocampus 68. The launch of specialized academic journals, such as Brain, Behavior, and Immunity - Integrative, underscores the ongoing, successful integration of Eastern biomedical philosophies with highly rigorous Western psychoneuroimmunological scientific methodologies 69.
Stress and Inflammation in African and African-Ancestry Populations
Emerging research into the psychoneuroimmunology of African and African-ancestry populations reveals stark, highly concerning insights into how systemic, environmental stressors translate into severe physiological wear-and-tear, biologically conceptualized as allostatic load 36.
Studies examining deeply impoverished settlement communities in sub-Saharan Africa, such as those in Naivasha, Kenya, have utilized hair cortisol analysis as a highly stable, long-term biomarker for chronic stress 70. These vulnerable populations, facing unremitting chronic stressors related to severe poverty, employment instability, and highly unsafe living conditions, exhibit significantly higher hair cortisol concentrations (averaging 639 ng/g) compared to Caucasian reference groups (averaging 299 ng/g) 70.
Similarly, expansive research focusing on young, urban African Americans living in major metropolitan areas reveals that continuous exposure to community and familial violence closely correlates with dangerously elevated markers of allostatic load 36. These markers include C-reactive protein, cortisol, Immunoglobulin E (IgE), and significantly increased levels of Epstein-Barr virus (EBV) IgM antibodies 36. The reactivation of latent viruses like EBV serves as a direct, highly alarming biological proxy for severe, stress-induced cellular immunosuppression 36. Furthermore, studies in urban African American older adults demonstrate that "heightened vigilance" - the chronic, exhausting psychological anticipation of environmental threat or racial discrimination - is significantly associated with elevated systemic inflammation, specifically circulating CRP levels 34.
Interestingly, there appear to be distinct, measurable neuro-immune correlations linked directly to ancestry. Systematic reviews examining neurodegenerative diseases show that IL-6 emerges as a particularly potent pro-inflammatory marker linked to severe cognitive decline, exhibiting much stronger correlations in populations of African ancestry compared to Caucasian cohorts 71. Furthermore, data from the Heart of Detroit Study highlights that aging and moderate-to-high perceived stress synergistically interact to exacerbate "inflammaging" - the pathological, age-related increase in systemic inflammation - among older African American populations 72. These findings emphasize unequivocally that severe socioeconomic disparities, chronic discrimination, and harsh cultural contexts are deeply and permanently embedded into the biology of the immune system.
Future Directions in Systems Biology and Personalized Medicine
The future of psychoneuroimmunology relies entirely on the transition from broad observational correlations to highly targeted, personalized therapeutic interventions 64173. The groundbreaking discovery of specific, hardwired pathways, such as the vagal anti-inflammatory reflex, has catalyzed the rapid development of an entirely new medical discipline: "bioelectronic medicine" 1021.
Implantable and transcutaneous vagus nerve stimulators (VNS) are currently undergoing rigorous, late-stage clinical trials to treat severe, debilitating inflammatory conditions, including rheumatoid arthritis and treatment-refractory Crohn's disease 10212248. By utilizing highly calibrated electrical impulses to artificially mimic the brain's natural neuro-immune braking system, clinicians can effectively suppress peripheral TNF-α production and induce remission without the severe, often toxic side effects associated with systemic pharmacological immunosuppressants 2122.
Simultaneously, the deep integration of psychoneuroimmunology with precision psychiatry promises to fundamentally refine how severe mental health conditions are diagnosed and treated 404474. As the vast biological heterogeneity of disorders like clinical depression is unpacked, patients may soon be routinely and aggressively screened for inflammatory biomarkers, such as CRP, IL-6, or specific immuno-inflammatory neuroimaging signatures, upon hospital admission 3774. Those exhibiting primarily immuno-inflammatory etiologies could bypass ineffective standard treatments and be prescribed targeted anti-inflammatory biological regimens, highly structured cognitive behavioral therapy to directly downregulate autonomic arousal, or specific mind-body interventions 384041.
By definitively charting the precise molecular, neural, and behavioral mechanisms by which thoughts and emotions alter immune function, psychoneuroimmunology offers a profound, scientifically rigorous validation of mind-body integration. It underscores the undeniable reality that psychological well-being is not merely an abstract, philosophical concept, but a fundamental, measurable biological determinant of human health and longevity.