Psychedelic interventions for mental health
Introduction
The integration of psychedelic compounds into psychiatric practice represents one of the most substantial and heavily debated shifts in psychopharmacology in the twenty-first century. For decades, the treatment of major depressive disorder (MDD), treatment-resistant depression (TRD), and post-traumatic stress disorder (PTSD) has relied predominantly on monoaminergic antidepressants, such as selective serotonin reuptake inhibitors (SSRIs), alongside various modalities of trauma-focused cognitive behavioral therapy. While these standard-of-care treatments demonstrate efficacy for a subset of the population, a significant proportion of patients do not achieve clinical remission. Up to 40% to 60% of patients with PTSD do not respond adequately to first-line SSRIs, and approximately one-third of individuals with major depressive disorder eventually develop treatment-resistant depression, resulting in prolonged suffering and profound socioeconomic burden 12.
In response to this unmet clinical need, neuroscientists and clinicians have revived investigations into classical psychedelics (such as psilocybin and lysergic acid diethylamide [LSD]), atypical psychedelics and empathogens (such as 3,4-methylenedioxymethamphetamine [MDMA]), and dissociative anesthetics (such as ketamine and esketamine). Early-phase clinical trials yielded unprecedented effect sizes, leading the United States Food and Drug Administration (FDA) to grant Breakthrough Therapy designations to MDMA for PTSD in 2017 and to psilocybin for TRD in 2018 and MDD in 2019 345.
However, the transition from controlled clinical trials to regulatory approval and real-world clinical implementation has proven exceedingly complex. The psychiatric field is currently grappling with profound methodological challenges regarding clinical trial blinding, controversies over the necessity and standardization of psychological support, severe health economic constraints, and varying regulatory decisions that have halted some compounds while accelerating others. This report provides an exhaustive analysis of the current research on psychedelics and mental health, examining neurobiological mechanisms, clinical efficacy, safety profiles, health economics, and recent regulatory milestones to determine the true trajectory of psychedelic-assisted therapy.
Neurobiological Mechanisms of Action
To comprehend the therapeutic potential of psychedelics, it is necessary to examine their distinct neurobiological mechanisms, which depart significantly from conventional psychiatric medications. While traditional antidepressants primarily modulate synaptic neurotransmitter concentrations over prolonged, multi-week periods, psychedelics induce rapid and sustained structural and functional changes in the brain within hours.
Psychoplastogens and Synaptic Plasticity
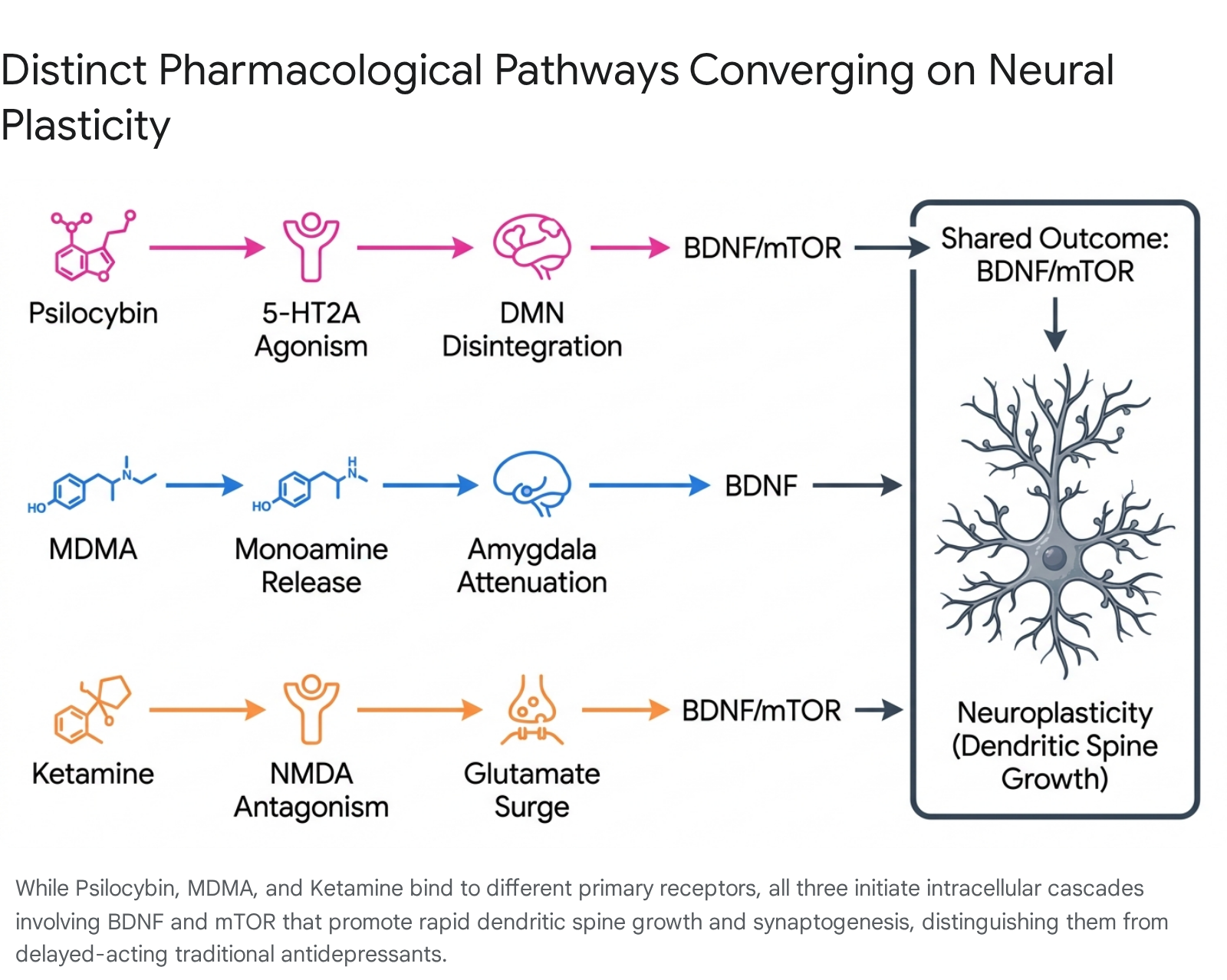
The therapeutic efficacy of psychedelics is increasingly attributed to their role as "psychoplastogens" - compounds capable of rapidly promoting structural and functional neural plasticity 56. Classical psychedelics, such as psilocybin and LSD, exert their primary psychoactive effects through agonism of the serotonin 5-HT2A receptor, which is densely expressed in the mammalian neocortex, particularly within the prefrontal cortex 78.
Activation of the 5-HT2A receptor initiates a complex intracellular signaling cascade that upregulates the expression of plasticity-related genes. Key pathways include the brain-derived neurotrophic factor (BDNF) and the mammalian target of rapamycin (mTOR) pathways 689. Preclinical in vitro and in vivo studies demonstrate that a single administration of a psychedelic produces rapid changes in dendritic complexity, spinogenesis, and synaptogenesis 9. In adult murine models, psilocybin and ketamine rapidly increase dendritic spine density in the prefrontal cortex within days - a process that typically takes weeks with traditional SSRIs 10. Furthermore, precise genetic knockout studies in adult mice indicate that the serotonin 2A receptor is critical for psilocybin's neuroplastic effects; removing this receptor completely eliminates the dendritic spine growth 10.
These neuroplastic changes significantly outlast the acute pharmacological clearance of the drugs. A single dose induces a "critical period" of heightened neural plasticity, spanning from hours to several weeks, during which the brain is highly receptive to environmental, psychological, and behavioral inputs 58. This biological window provides the theoretical foundation for combining psychedelic administration with structured psychotherapy, allowing for the potential unlearning of maladaptive cognitive patterns and rigid fear responses 4.
Default Mode Network and Macroscopic Dynamics
Beyond cellular and synaptic mechanisms, advanced neuroimaging studies indicate that psychedelics profoundly alter macroscopic brain network dynamics. Psilocybin has been consistently shown to decrease functional connectivity and resting-state activity within the Default Mode Network (DMN). The DMN is a network of interacting brain regions (including the medial prefrontal cortex and posterior cingulate cortex) associated with self-referential processing, ego identity, mental time travel, and rumination 78. Hyperactivity and hyperconnectivity within the DMN are common neurobiological correlates of depression, anxiety disorders, and obsessive-compulsive traits.
By inducing temporary disintegration of the DMN and simultaneously increasing global connectivity between typically segregated brain networks, psilocybin induces an acute state of "ego dissolution" or a mystical-type experience 7. This temporary neurological flexibility allows patients to experience a profound disruption of rigid, pathological thought loops. Following the acute experience, the DMN typically reconstitutes, but often with a "reset" baseline connectivity that correlates with sustained reductions in depressive symptomatology 7.
Monoaminergic and Glutamatergic Pathways
MDMA, classified as an empathogen-entactogen, operates through a distinctly different primary mechanism than classical psychedelics. It acts as a potent presynaptic reuptake inhibitor and releasing agent for serotonin, norepinephrine, and dopamine, while concurrently stimulating the release of neurohormones such as oxytocin 711. Neurally, MDMA decreases amygdala reactivity, thereby attenuating acute fear responses while enhancing emotional openness and interpersonal trust 1213. This highly specific profile makes MDMA uniquely suited for trauma-focused therapy; it enables patients with severe PTSD to re-engage with deeply traumatic memories without experiencing overwhelming autonomic arousal or psychological numbing 4714.
Conversely, ketamine and its enantiomer esketamine act primarily as non-competitive N-methyl-D-aspartate (NMDA) receptor antagonists within the glutamatergic system 715. By blocking NMDA receptors on inhibitory GABAergic interneurons, ketamine induces a rapid surge in extracellular glutamate. This glutamate burst subsequently activates AMPA receptors, triggering the release of BDNF and stimulating the mTOR pathway, leading to rapid synaptogenesis 56.
Clinical Efficacy and Trial Outcomes
The clinical efficacy of these compounds has been evaluated across multiple psychiatric indications. However, interpreting these results requires careful consideration of the specific compound utilized, the chronicity of the targeted disease, and the comparative efficacy against established baselines.
Psilocybin for Depressive Disorders
Psilocybin has been extensively studied for the treatment of both MDD and TRD. Clinical trials have demonstrated that a single high dose of psilocybin (typically 25 mg), combined with psychological support, can yield rapid antidepressant effects. In several trials, 71% of individuals with major depressive disorder responded to psilocybin treatment, with over 50% entering and sustaining remission at six months, and up to 58% maintaining remission at 12 months 12.
A pivotal question in clinical psychopharmacology is how psilocybin compares directly to standard-of-care SSRIs. A landmark double-blind randomized controlled trial published in the New England Journal of Medicine directly compared psilocybin to escitalopram over a six-week period for patients with long-standing major depressive disorder 161718. In this trial, the primary outcome measure - the 16-item Quick Inventory of Depressive Symptomatology Self-Report (QIDS-SR-16) - showed a mean change from baseline of - 8.0 in the psilocybin group versus - 6.0 in the escitalopram group. This 2.0-point difference did not achieve statistical significance (P = 0.17) for the primary endpoint 18.
Despite the primary outcome miss, secondary outcomes consistently favored psilocybin. A QIDS-SR-16 clinical response at 6 weeks occurred in 70% of the psilocybin group compared to 48% in the escitalopram group, and remission rates were substantially higher at 57% versus 28%, respectively 18. Other secondary measures, including the Warwick-Edinburgh Mental Wellbeing Scale (WEMWBS) and Spielberger's Trait Anxiety Inventory (STAI), also indicated larger magnitude improvements for the psilocybin cohort, though the authors noted these were not corrected for multiple comparisons 1618.
A comprehensive 2024 systematic review and network meta-analysis published in the BMJ further elucidated these comparisons by evaluating 19 placebo-controlled studies. By strictly distinguishing between the placebo response in psychedelic trials versus standard antidepressant trials to avoid estimation bias, researchers found that high-dose psilocybin was superior to placebo in antidepressant trials and showed a larger relative effect size compared to 10 mg and 20 mg of escitalopram 171920. However, the authors noted that the overall absolute effect size was small to moderate, cautioning that the mandatory psychotherapy accompanying psilocybin administration complicates the isolation of its pure pharmacological efficacy 19.
Ketamine and Esketamine
While classical psychedelics remain investigational in most jurisdictions, dissociative agents acting on the glutamate system are already heavily integrated into clinical practice. Esketamine received FDA approval (marketed as Spravato) in 2019 for TRD in conjunction with an oral antidepressant, and for MDD with acute suicidal ideation 152122.
In a significant regulatory expansion in January 2025, esketamine achieved FDA approval as a monotherapy for adults with TRD. This approval was supported by data demonstrating robust, rapid improvements in the Montgomery-Asberg Depression Rating Scale (MADRS) within 24 hours of administration 2123. At week four, 22.5% of patients taking esketamine achieved full remission (MADRS total score ≤ 12) compared to just 7.6% of patients taking placebo 23.
Ketamine and esketamine currently provide the most rapid intervention available in modern psychiatry, capable of producing relief within hours, making them ideal for acute suicidality 24. However, a fundamental limitation of ketamine therapy is the transience of its clinical effects. The antidepressant response often diminishes within one to two weeks, necessitating repeated, ongoing maintenance sessions 2426. This contrasts sharply with psilocybin, where a single session can sustain antidepressant effects for months or longer 724. Meta-analyses directly comparing intravenous ketamine to psilocybin for depression show comparable initial effect sizes, though psilocybin generally demonstrates a stronger mediating effect correlated with the subjective psychological experience of the patient (R2 values ranging from 5 - 10% for ketamine versus 24% for psilocybin) 25.
MDMA for Post-Traumatic Stress Disorder
The development pipeline for MDMA-assisted therapy (MDMA-AT) has been focused almost exclusively on severe, chronic PTSD. The primary sponsor, Lykos Therapeutics (formerly MAPS Public Benefit Corporation), conducted two pivotal Phase 3 randomized, double-blind, placebo-controlled trials (MAPP1 and MAPP2) 2826.
The clinical results of these trials were statistically robust. In the MAPP2 trial, which included 104 participants with moderate to severe PTSD (with average symptom duration of 16.2 years), 71.2% of participants receiving MDMA-AT no longer met the diagnostic criteria for PTSD at the conclusion of the trial, compared to 47.6% in the placebo-plus-therapy group 12. Furthermore, 46.2% of the MDMA cohort achieved complete clinical remission, compared to 21.4% in the control arm 12.
Despite these highly compelling metrics, the regulatory interpretation of these outcomes has been severely contested, driven by intense methodological criticisms regarding clinical blinding, expectation bias, and therapeutic integrity, which are analyzed in subsequent sections of this report.
| Compound | Drug Class / Primary Mechanism | Target Psychiatric Indication | Time to Peak Effect | Duration of Acute Effects | Standard Treatment Model |
|---|---|---|---|---|---|
| Psilocybin | Classical Psychedelic (5-HT2A receptor agonist) | Major Depressive Disorder (MDD), Treatment-Resistant Depression (TRD) | 1 - 2 hours | 4 - 6 hours | 1 - 2 high-dose sessions surrounded by preparatory and integration psychotherapy 327. |
| MDMA | Empathogen / Entactogen (Monoamine reuptake inhibitor & releasing agent) | Post-Traumatic Stress Disorder (PTSD) | 1 - 2 hours | 6 hours | 2 - 3 medication sessions (8 hours each) integrated into a 4-month psychotherapy protocol 2728. |
| Esketamine | Dissociative Anesthetic (NMDA receptor antagonist) | TRD, MDD with acute suicidal ideation | 30 - 40 minutes | 1 - 2 hours | Twice weekly induction phase, transitioning to weekly or bi-weekly maintenance dosing 1529. |
The Methodological Crisis: Blinding and Expectation Bias
The advancement of psychedelic medicine is currently hindered by a severe methodological crisis. Traditional psychopharmacological research relies on the double-blind, randomized, placebo-controlled trial as the gold standard to isolate a drug's pharmacological effect from the placebo effect. In psychedelic trials, maintaining this blind is exceptionally difficult.
Functional Unblinding in Clinical Trials
The subjective, psychoactive effects of high-dose psychedelics - including profound alterations in perception, mood, and suggestibility - are so pronounced that both participants and clinical raters can almost always accurately guess treatment allocation 3031. This phenomenon is known as "functional unblinding."
An analysis of modern psychedelic RCTs found that among studies reporting on blinding integrity, 83% reported significant (>50%) functional unblinding 31. In the Lykos MDMA Phase 3 trials, approximately 80% of participants in the active arm were completely certain they had received MDMA, while nearly two-thirds of the placebo group correctly guessed they had received the placebo 35. Consequently, participants enrolled in these trials, who often have long-standing, treatment-resistant conditions and hold highly optimistic beliefs regarding psychedelic therapies, experience a massive expectation bias 283031.
When a participant realizes they have received the active drug, the resulting placebo effect acts synergistically with the pharmacological effect to artificially inflate the reported efficacy 303132. Conversely, a "nocebo" effect occurs in the control group; upon realizing they received an inert placebo, patients often experience acute disappointment, artificially suppressing the baseline response and widening the apparent efficacy gap between the two arms 37. Some trials have attempted to mitigate this by using active placebos (such as low-dose psychedelics or niacin), but these have proven insufficient to perfectly mask the profound experiential differences of a high dose 3138.
Expectation Bias and Equal Unblinding
The magnitude of this expectation bias was clearly demonstrated in a pivotal 2026 meta-analysis published in JAMA Psychiatry. Researchers specifically sought to compare psychedelic therapy to open-label traditional antidepressants under "equal unblinding" conditions - scenarios where patients in both groups were fully aware of the treatment they were receiving 33.
The study found that when both groups are unblinded, the statistical difference in efficacy between psychedelic-assisted therapy and traditional antidepressants diminishes dramatically. Under these conditions, the estimated difference on the 17-item Hamilton Depression Rating Scale (HAM-D 17) was only 0.3 points, favoring open-label antidepressants (P = 0.73) 33. The investigators concluded that the efficacy of psychedelic therapy has likely been consistently overestimated in previous literature by failing to account for blinding-related issues 3233. While this does not imply that psychedelic therapy is ineffective, it suggests that the true pharmacological effect size is smaller than early, highly optimistic narratives indicated 3733.
The Role of Psychological Support
A central tenet of modern psychedelic research is that the drug itself is merely a catalyst; the therapeutic context - often referred to as "set and setting" - and the accompanying psychological support are strictly necessary for optimal clinical outcomes 3435.
Synergistic Effects of Therapy and Pharmacotherapy
Psychedelic-assisted therapy (PAT) is inherently a combined treatment model. Clinical protocols for psilocybin and MDMA mandate multiple non-drug psychotherapy sessions prior to administration (preparation) and following administration (integration) 3436. The preparation phase is critical for building the therapeutic alliance, which naturalistic and clinical studies have heavily correlated with positive clinical outcomes, including the occurrence of emotional breakthroughs and mystical-type experiences 41335.
During the active dosing session, patients enter a vulnerable, hyper-plastic state. Clinicians typically employ non-directive counseling, encouraging the patient to follow an inner-directed process rather than engaging in rigid cognitive-behavioral interventions during the acute drug effects 1334. The integration phase is subsequently required to help patients make sense of the profound cognitive and emotional shifts experienced, anchoring these acute insights into durable, long-term behavioral changes 3437. Observational studies warn that a lack of structured psychological support following high-dose psychedelic use can lead to ontological shock and extended psychological difficulties, underscoring the absolute necessity of clinical facilitation 35.
Paradigmatic Friction in Regulatory Oversight
The mandatory inclusion of psychotherapy in these protocols complicates the traditional regulatory process. Governing bodies such as the FDA are chartered to evaluate the safety and efficacy of pharmacological agents, not the practice of psychotherapy 3435.
Consequently, evaluating a drug application where the pharmaceutical is inextricably linked to a behavioral intervention creates profound paradigmatic friction 35. Regulators are forced to determine how much of the clinical improvement is attributable to the drug versus the intensive, specialized therapy accompanying it. In the Lykos MDMA trials, the FDA explicitly cited the inability to isolate the contribution of the psychotherapy from the overall treatment effect as a major complication in their review 28.
Safety Profiles and Clinical Exclusions
While clinical trials repeatedly report that psychedelics are generally well-tolerated when administered under close medical supervision, they possess unique safety profiles and strict contraindications that will likely limit their applicability to the broader psychiatric population.
Psychiatric Contraindications
Psychedelic therapy is not universally applicable and carries distinct, severe risks for specific psychiatric phenotypes. The most widely applied exclusion criterion across virtually all psilocybin and MDMA trials is a personal or family history of psychotic disorders (e.g., schizophrenia) or bipolar disorder, particularly Bipolar I 38394041.
This exclusion is firmly rooted in risk management. First-degree relatives of individuals with bipolar disorder have an approximate 10-fold increased risk of developing the condition 41. Administration of a powerful 5-HT2A agonist like psilocybin carries a theoretical, and historically observed, risk of precipitating acute mania, unmasking latent bipolar disorder, or inducing prolonged psychotic episodes 4142. Furthermore, trial protocols routinely exclude individuals with recent severe suicide attempts, severe dissociative disorders, and acute, unstable medical conditions 3940. Consequently, the demographic represented in clinical trials is highly curated, raising concerns regarding the generalizability of safety data to real-world populations where complex psychiatric comorbidities are common 39.
Physiological Risks and Tolerability
While classical psychedelics like psilocybin display remarkably low physical toxicity and virtually no lethal overdose risk in humans, empathogens and dissociatives carry significant physiological liabilities 24.
MDMA acts as a potent sympathomimetic, significantly increasing cardiovascular demand. During the Phase 3 PTSD trials, 46% of participants treated with MDMA experienced an acute increase in systolic blood pressure of ≥20 mm Hg, and 45% experienced an increase in diastolic blood pressure of ≥10 mm Hg during the active dosing sessions 30. Cases of severe hypertension and tachycardia necessitate strict cardiovascular screening and continuous vital sign monitoring throughout the session 3035. Furthermore, the FDA raised concerns regarding potential hepatotoxicity associated with MDMA, noting that the relatively small sample sizes in the clinical trials were insufficient to definitively rule out rare but severe liver injury 1228.
The following table contrasts the primary safety considerations across the major therapeutic psychedelic agents:
| Drug Category | Primary Physiological Risks | Primary Psychiatric Contraindications / Risks | Monitoring Requirements |
|---|---|---|---|
| Psilocybin | Minimal physical toxicity. Acute nausea, transient headache, mild transient increases in blood pressure 242643. | Active psychosis, personal/family history of Schizophrenia or Bipolar Disorder (risk of mania/psychosis) 384142. | Supportive clinical environment, psychological supervision to manage anxiety/panic 24. |
| MDMA | Significant cardiovascular stimulation (hypertension, tachycardia). Bruxism, hyperthermia, potential hepatotoxicity 283035. | Risk of serotonin syndrome (especially if combined with SSRIs). High abuse potential 2635. | Continuous cardiovascular monitoring. Rigorous REMS (Risk Evaluation and Mitigation Strategy) 35. |
| Ketamine / Esketamine | Dissociation, dizziness, acute blood pressure elevation, nausea 726. | Arteriovenous malformation, history of intracerebral hemorrhage, severe cardiovascular disease 43. | Post-administration observation for at least 2 hours (REMS program mandate) 1529. |
Health Economics and Healthcare Integration
A primary structural barrier to the widespread adoption of psychedelic-assisted therapies is the extraordinary cost of clinical delivery. The economic model for psychedelics fundamentally differs from standard pharmacotherapy; health insurers are not merely reimbursing the cost of a pill, but a complex, highly labor-intensive bundle of behavioral health services 244.
Cost Structure of Psychedelic-Assisted Therapy
The cost of administering a full course of MDMA-assisted therapy for PTSD, according to the Phase 3 clinical trial protocol, involves approximately 42 hours of dedicated clinician time per patient. This rigorous protocol requires two trained therapists to be present during the three 8-hour dosing sessions, in addition to the multiple 90-minute preparation and integration sessions 2.
Estimates for the total cost of this treatment cycle vary widely based on the assumptions of the models. Independent academic health economists utilizing Phase 3 trial data estimated the base cost at $11,537 per patient 4546. However, more recent industry-sponsored analyses project costs ranging from $20,000 to as high as $48,376 per patient cycle in modern US healthcare settings. This higher figure incorporates an assumed $12,000 cost for the MDMA medication itself, alongside over $36,000 for all psychotherapy and facility components 364748. In current legal environments, such as Australia's Authorised Prescriber scheme, real-world costs for a three-session MDMA protocol hover around $30,000 AUD (approximately $20,000 USD), which is currently paid almost entirely out-of-pocket by patients due to a lack of broad insurance coverage 248.
Cost-Effectiveness and Quality-Adjusted Life Years
Despite the prohibitive upfront costs, rigorous health economic modeling indicates that psychedelic therapies are highly cost-effective, and potentially cost-saving, over long time horizons. Chronic, severe PTSD and TRD place an enormous, compounding financial burden on healthcare systems through decades of continuous medication management, psychiatric hospitalizations, and lost productivity.
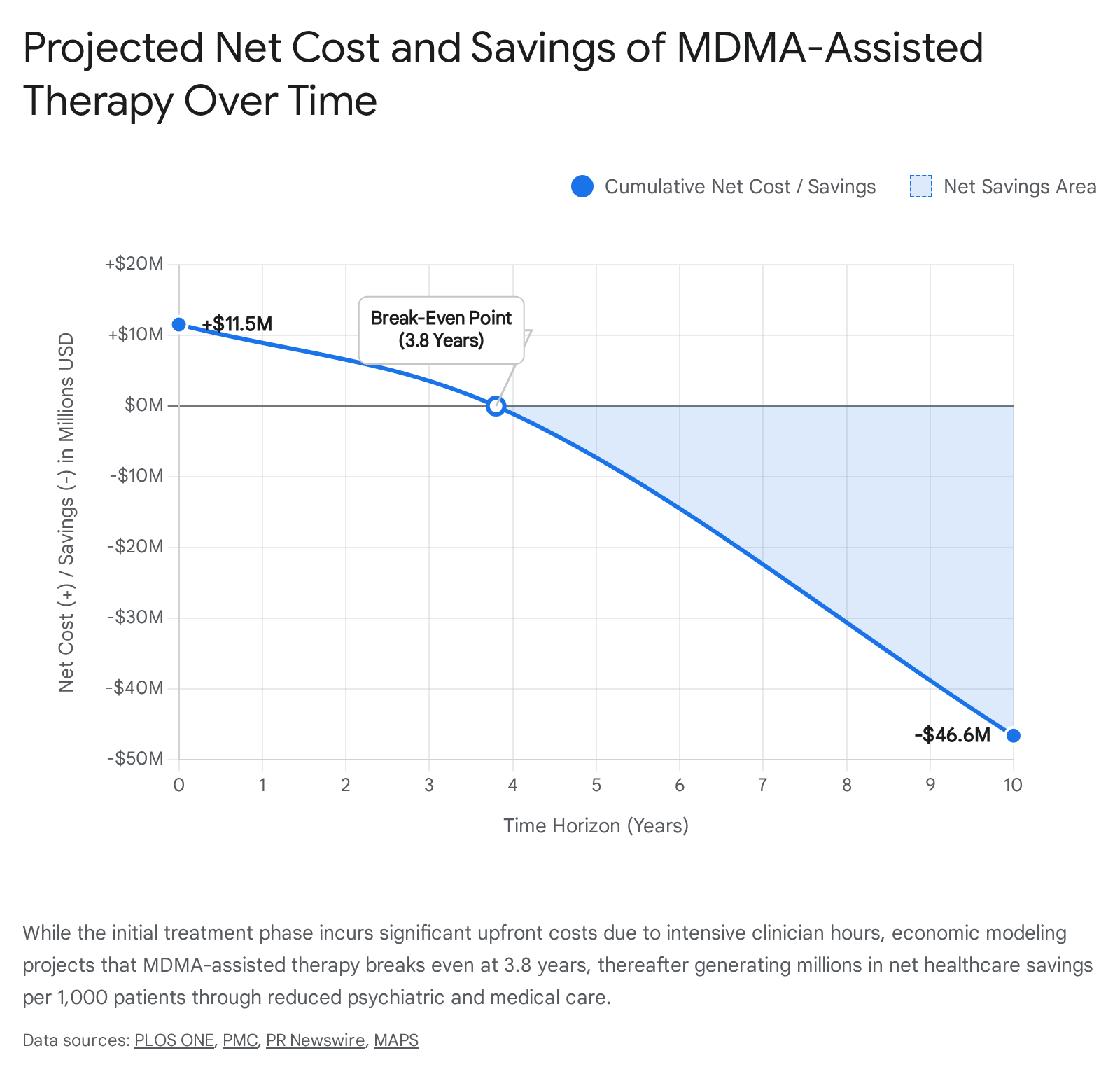
Using a Markov state-transition model based on US healthcare payer perspectives, researchers demonstrated that over a 30-year analytic horizon, treating a cohort of 1,000 patients with MDMA-assisted therapy generates discounted net healthcare savings of $132.9 million, accruing 4,856 Quality-Adjusted Life Years (QALYs), and averting 61.4 premature deaths compared to standard care 4546. Importantly, the therapy breaks even on costs at approximately 3.8 years 4546. Even in more conservative 5-year models that account for higher drug pricing (e.g., $12,000 for the medication component), MDMA-AT still yielded an Incremental Cost-Effectiveness Ratio (ICER) of $83,845 per QALY. This falls well below the standard $150,000 willingness-to-pay threshold in the US, generating a 0.377 QALY increment over the placebo arm 3647.
Similarly, psilocybin-assisted therapy for TRD has been modeled as an economically dominant strategy (more effective and less costly) over standard care in the long term. A recent 30-year scenario analysis predicted cumulative societal savings of $215,900 and gains of 9.87 QALYs per patient 49. In the UK, health economic models found the expected healthcare cost of psilocybin therapy varied from £6132 to £7652, and concluded it is a cost-effective alternative to conventional medication when the cost of therapist support is optimized 56.
Scalability and Group Therapy Models
To mitigate these severe economic and workforce barriers, researchers are actively investigating group therapy models. By treating patients in coordinated cohorts, clinics can optimize the high clinician-to-patient ratio without sacrificing safety. Economic analyses demonstrate that group therapy protocols can significantly reduce clinician costs - achieving a 34.7% reduction for psilocybin treatment of MDD, and a 50.9% reduction for MDMA treatment of PTSD 250. Group models effectively double the capacity of the highly specialized psychedelic clinician workforce, which is currently identified as a primary bottleneck preventing scalable, equitable access 50.
Regulatory Landscape and Recent Developments
The timeline for integrating psychedelics into mainstream medicine has experienced intense volatility over recent years, defined by unexpected regulatory rejections, targeted executive interventions, and varying international approaches to controlled access.
The FDA Rejection of MDMA
The psychiatric community widely anticipated that MDMA would become the first FDA-approved psychedelic therapeutic. However, in June 2024, the FDA's Psychopharmacologic Drugs Advisory Committee (PDAC) delivered a severe and unexpected setback, voting 10 - 1 against the proposition that the benefits of Lykos Therapeutics' MDMA-assisted therapy outweighed its risks for the treatment of PTSD 125152.
In August 2024, the FDA formally rejected the New Drug Application, issuing a Complete Response Letter that mandated an additional, large-scale Phase 3 clinical trial to further evaluate safety and efficacy 1122760. The regulatory rejection was driven by a confluence of methodological and safety factors: the profound functional unblinding that compromised the interpretability of the efficacy data, inadequate cardiovascular and hepatotoxicity safety monitoring, a lack of demographic diversity in the trial cohorts, and the failure of the sponsor to systematically collect data on positive subjective effects (which the FDA strictly requires to evaluate a compound's abuse potential) 1128303561.
The rejection was heavily compounded by emerging ethical controversies. Just days after the FDA's decision, the medical journal Psychopharmacology formally retracted three MDMA papers associated with the Lykos trials. The retraction note cited "protocol violations amounting to unethical conduct" and failures by researchers to disclose severe ethical breaches and potential competing interests at a specific clinical trial site 5153. Furthermore, an independent report by the Institute for Clinical and Economic Review (ICER) highlighted selection bias, noting patients were pulled heavily from communities with strong prior beliefs in psychedelics 51.
The Anticipated Approval of Psilocybin
Following the high-profile MDMA rejection, the industry's focus and capital shifted decisively toward psilocybin. The synthetic psilocybin formulation COMP360, developed by Compass Pathways, completed two highly anticipated Phase 3 trials for TRD: COMP005 in June 2025, and COMP006 in February 2026, both hitting primary endpoints with high statistical significance 3.
The regulatory pathway for psilocybin was dramatically accelerated by a targeted executive order signed by President Trump on April 18, 2026, explicitly aimed at accelerating mental health treatments for veterans and civilians 35455. In direct response to the order, on April 24, 2026, the FDA issued three Commissioner's National Priority Vouchers (CNPVs) to psychedelic programs holding existing Breakthrough Therapy designations. These exclusive vouchers - which truncate the standard New Drug Application review process from 10 - 12 months down to just one or two months - were awarded to Compass Pathways for COMP360 (psilocybin for TRD), Usona Institute for their non-profit psilocybin program (for MDD), and Otsuka Therapeutics (which acquired Transcend Therapeutics) for their methylone program for PTSD 60545556. With a rolling NDA submission targeted for the fourth quarter of 2026, Compass Pathways is currently positioned to potentially secure FDA approval for psilocybin by late 2026 or early 2027 3.
The Australian Prescribing Model
While the US navigates traditional clinical trial approvals, Australia has pioneered a real-world regulatory experiment. Effective July 1, 2023, the Therapeutic Goods Administration (TGA) rescheduled MDMA and psilocybin from prohibited substances (Schedule 9) to controlled drugs (Schedule 8), exclusively for authorized psychiatrists treating PTSD and TRD, respectively 666869.
The rollout has been deliberately cautious and highly bureaucratic. Psychiatrists must become "Authorised Prescribers" by obtaining stringent approval from a Human Research Ethics Committee (HREC) and the TGA, leading to significant workforce bottlenecks 57. By late 2024, only about ten psychiatrists nationwide had secured authorization for psilocybin 57. Uptake slowly accelerated through 2025; by September 2025, TGA data indicated that 87 unique patients had received MDMA and 47 had received psilocybin, with zero serious adverse events reported 66. By May 2026, the total number of patients treated eclipsed 180, supported by extensive philanthropic funding to offset the severe out-of-pocket costs 268.
The Australian model underscores the logistical realities of psychedelic medicine: even with legal rescheduling, the absolute requirement for highly specialized psychiatric infrastructure, rigorous ethical oversight, and expensive clinical supervision strictly throttles widespread patient access 4857. The Royal Australian and New Zealand College of Psychiatrists (RANZCP) continues to update comprehensive training frameworks, advocating strongly that these therapies be reserved strictly for treatment-resistant populations who have exhausted all established evidence-based interventions 69585960.
American Psychiatric Association Posture
The broader psychiatric establishment is proceeding with cautious optimism. In a consensus statement following the April 2026 executive order, the American Psychiatric Association (APA) commended federal investment but firmly stated that there is currently inadequate scientific evidence to endorse the use of psychedelics to treat any psychiatric disorder outside the context of approved investigational studies 61. The APA emphasized that the FDA's rejection of MDMA was a necessary enforcement of scientific rigor, noting that psychedelics must meet the same regulatory, blinding, and ethical standards applied to all emerging treatments in medicine 6161. Recognizing the inevitability of future approvals, the APA announced a 2025 initiative to roll out a comprehensive toolkit and training framework for psychologists and psychiatrists to ensure ethical, standardized models of care are in place prior to widespread commercialization 62.
Conclusion
The psychiatric field stands at a highly complex therapeutic turning point. The amassed neurobiological evidence unequivocally demonstrates that psychoplastogens - including psilocybin, MDMA, and ketamine - possess distinct, rapid mechanisms of action capable of breaking rigid, pathological neural circuits. These compounds represent the most significant pharmacological innovation in mental health in decades, offering the possibility of durable remission for patients suffering from devastating, treatment-resistant disorders.
However, asserting that psychedelics represent an immediate, flawless panacea ignores profound systemic and methodological challenges. The stark regulatory rejection of MDMA in 2024 exposed the vulnerabilities inherent in psychedelic research: trials are fundamentally difficult to blind, subjective expectation biases inflate perceived effect sizes, and the absolute requirement for synergistic psychotherapy confounds traditional biological drug evaluations. Furthermore, the prohibitive cost structure of delivering 40+ hours of specialized clinical care ensures that, without radical shifts in insurance coverage or the widespread adoption of group therapy models, equitable access will remain a severe limitation.
With psilocybin advancing rapidly toward potential FDA approval in late 2026, aided by national priority vouchers, and real-world prescribing models maturing in Australia, the coming years will shift the challenge from establishing basic efficacy to achieving sustainable healthcare integration. The ultimate success of psychedelic medicine relies not solely on the pharmacology of the molecules, but on the ability of healthcare systems, regulators, and the psychiatric establishment to implement ethically rigorous, economically viable, and clinically standardized frameworks for their administration.