Physiology of sleep architecture and microstructure
Sleep is a highly dynamic, biologically active state characterized by precise neurophysiological and neurochemical fluctuations. Far from a uniform period of physiological dormancy, sleep consists of an orchestrated progression of distinct stages, each serving specialized homeostatic, metabolic, and cognitive functions. Understanding sleep requires a dual perspective: analyzing its macrostructure - the cyclical progression of sleep stages through the night - and its microstructure, which comprises transient, high-frequency neuro-oscillatory events such as sleep spindles, K-complexes, and sharp-wave ripples. This report provides a comprehensive examination of sleep architecture, detailing the stage-by-stage physiological mechanisms, the neurobiological purpose of transient micro-events, the dynamics of cerebral waste clearance, the clinical manifestations of microstructural decay, and the evolutionary context of human sleep behavior.
Methodological Foundations of Sleep Measurement
The rigorous scientific evaluation of sleep relies on capturing both macrostructural and microstructural phenomena. The clinical and research gold standard for this assessment is polysomnography (PSG) 12. PSG relies on a multifaceted array of physiological sensors, primarily utilizing electroencephalography (EEG) to track cortical brain wave activity, electrooculography (EOG) to capture eye movements, and electromyography (EMG) to measure skeletal muscle tone 23.
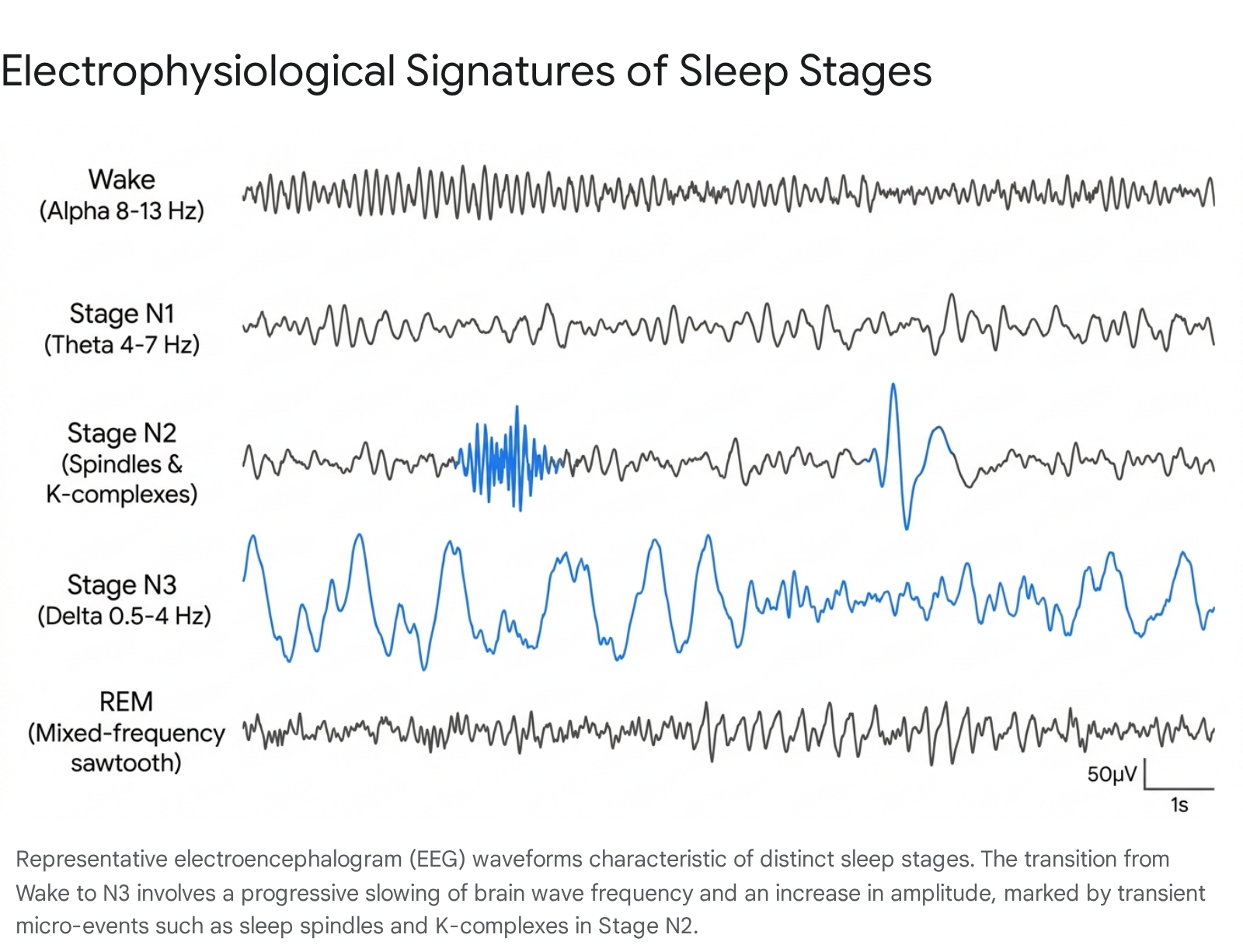
The continuous data streams from these modalities are traditionally segmented into 30-second epochs. Professional sleep technicians visually inspect each epoch, identifying specific phasic (transient) micro-events like sleep spindles or K-complexes, alongside tonic (continuous) features like the overall percentage of slow-wave delta activity 14. Based on the guidelines established by the American Academy of Sleep Medicine (AASM), each epoch is scored as Wakefulness, Non-Rapid Eye Movement Stage 1 (N1), Stage 2 (N2), Stage 3 (N3), or Rapid Eye Movement (REM) sleep 14.
Because visual scoring introduces subjectivity, inter-rater reliability is often quantified using Fleiss' kappa statistic ($\kappa$). A $\kappa$ value above 0.80 generally indicates strong agreement among clinical raters, ensuring that the manual interpretation of ambiguous micro-events remains statistically robust before proceeding to clinical diagnosis 4.
Beyond standard scalp EEG, advanced research methodologies frequently utilize intracranial electroencephalography (iEEG) or depth electrodes. Often employed in pre-surgical evaluations for patients with drug-resistant epilepsy, iEEG allows researchers to record deep-brain structures, such as the hippocampus and amygdala, with high spatial and temporal resolution 56. This invasive monitoring is critical for observing highly localized micro-events, such as sharp-wave ripples, which cannot be reliably detected using non-invasive surface electrodes due to signal attenuation through the skull 7.
The Technological Shift: Wearables and Ambulatory Monitoring
The emergence of consumer wearable technologies - such as smartwatches, fitness rings, and actigraphy bands - has decentralized sleep tracking, offering longitudinal data collection outside the artificial environment of a sleep laboratory 2. However, these devices introduce significant methodological limitations. Wearables predominantly rely on photoplethysmography (PPG) to measure heart rate and heart rate variability (HRV), combined with triaxial accelerometers to monitor actigraphy (motor quiescence) 13. While advanced signal processing and machine learning algorithms attempt to infer sleep stages from autonomic nervous system fluctuations, respiratory sinus arrhythmia, and movement, these devices lack the direct cortical electrophysiological data required to observe true sleep microstructure 23.
Consequently, wearables exhibit variable accuracy. Comparative studies benchmarked against PSG demonstrate that while consumer devices reliably detect sleep onset and total sleep period time, they frequently struggle to accurately distinguish between N1, N2, N3, and REM stages 248. Wearables often overestimate light sleep at the expense of deep sleep and REM, and they generally exhibit high specificity but low sensitivity for detecting brief nocturnal awakenings, leading to an overestimation of sleep efficiency 124.
To bridge the gap between expensive, intrusive PSG and imprecise consumer wearables, recent biomedical engineering efforts have developed ambulatory clinical tools, such as the flexible electrode sleep patch (FESP). The FESP integrates lightweight, self-applicable sensors capable of capturing fundamental EEG rhythms at home. Early validation studies indicate that FESP systems achieve a strong correlation with PSG for sleep parameters (Pearson coefficients of 0.80 - 0.96) and demonstrate an impressive accuracy range of 84% to 93% for basic sleep stage classification, providing a viable middle ground for continuous microstructural tracking 8.
| Modality | Primary Sensors | Microstructure Detection | Stage Classification | Limitations |
|---|---|---|---|---|
| Polysomnography (PSG) | EEG, EOG, EMG, ECG | Direct observation (spindles, ripples, K-complexes) | Precise AASM scoring (Wake, N1, N2, N3, REM) | High cost, clinical setting required, intrusive |
| Intracranial EEG (iEEG) | Depth electrodes | High-resolution deep brain events (hippocampal SWRs) | Highly localized, subcortical precision | Highly invasive, limited to specific patient populations |
| Flexible Patch (FESP) | Portable scalp electrodes | Moderate (detects primary wave bands and major events) | Accurate automated scoring | Limited channel montage compared to full PSG |
| Wearables (Actigraphy/PPG) | Accelerometer, PPG, Skin Temp | None (infers stages via autonomic proxy data) | Simplified (Wake, Light, Deep, REM) | Overestimates sleep efficiency; misclassifies specific NREM/REM boundaries |
Non-Rapid Eye Movement Sleep Microstructure
Non-Rapid Eye Movement (NREM) sleep accounts for the majority of the human sleep cycle, representing approximately 75% to 80% of total sleep time in healthy adults. It is subdivided into three stages - N1, N2, and N3 - representing a continuum of increasing depth, decreasing cortical arousal, and distinct oscillatory signatures 29.
Stage N1: The Wake-to-Sleep Transition
Stage N1 acts as the gateway between wakefulness and definitive sleep, typically lasting only one to five minutes and comprising roughly 5% of total sleep time 310. In a state of relaxed wakefulness with closed eyes, the EEG is dominated by the alpha rhythm - synchronized waveforms oscillating at 8 to 13 Hz, predominantly over the occipital regions 39.
As the brain transitions into N1, the alpha rhythm begins to dissipate and is replaced by lower-amplitude, mixed-frequency activity, predominantly in the theta band (4 to 7 Hz) 9. During this stage, EOG channels detect slow, rolling, conjugated eye movements (SEMs), a hallmark of early sleep onset 9. While skeletal muscle tone (EMG) decreases compared to active wakefulness, it remains relatively high. From a microstructural standpoint, N1 is often characterized by the appearance of vertex sharp waves - brief, high-amplitude negative deflections over the central cortical regions lasting up to 0.5 seconds 9. Because N1 is a highly superficial stage of sleep, environmental stimuli or minor physiological shifts can easily prompt an awakening, occasionally accompanied by hypnic jerks or a sensation of falling 11.
Stage N2: Sensory Gating and Early Consolidation
Stage N2 constitutes the bulk of human sleep, representing approximately 50% of the total nocturnal sleep period 3. As the individual descends into N2, physical responsiveness to external stimuli decreases significantly. Heart rate slows, and core body temperature begins to drop. The onset of N2 is formally defined in clinical PSG by the emergence of two critical microstructural elements: sleep spindles and K-complexes, occurring against a background of ongoing theta activity 39.
Sleep Spindles Sleep spindles are brief bursts of highly synchronized oscillatory activity, typically ranging from 12 to 15 Hz and lasting between 0.5 and 2 seconds 111312. These waxing and waning waveforms are generated by a complex feedback loop between the reticular nucleus of the thalamus and the neocortex 13. Spindles serve multiple physiological functions. First, they act as a sensory gating mechanism; by inhibiting the transmission of auditory and tactile stimuli from the thalamus to the cortex, spindles protect the sleeping brain from being roused by non-threatening environmental noise 12. Second, spindles play a critical role in memory consolidation. They frequently couple with cortical slow oscillations and hippocampal ripples, facilitating the transfer of newly encoded declarative memories from temporary hippocampal storage to long-term neocortical networks 1213.
K-Complexes A K-complex is a massive biphasic waveform featuring a large negative (upward) deflection followed immediately by a slower positive (downward) deflection, lasting at least 0.5 seconds. K-complexes can occur spontaneously or can be evoked by external stimuli (such as a sudden noise) or internal stimuli (such as a minor respiratory event). They are hypothesized to function as a homeostatic "decision-making" mechanism. When a stimulus occurs, the K-complex reflects rapid cortical excitation followed immediately by massive cortical inhibition (a "downstate"), effectively suppressing the arousal and maintaining sleep continuity if the brain deems the stimulus non-threatening 412.
Stage N3: Slow-Wave Activity and Cellular Homeostasis
Stage N3, commonly referred to as slow-wave sleep (SWS) or deep sleep, is the most restorative phase of the human sleep cycle, accounting for 15% to 25% of total nocturnal sleep 39. During N3, the EEG shifts dramatically and is dominated by delta waves - high-amplitude, low-frequency oscillations ranging from 0.5 to 2 Hz 39.
These slow oscillations represent a state of massive neuronal synchronization across the cerebral cortex. During the "up-state" of a slow oscillation, large populations of cortical neurons depolarize and fire synchronously; during the "down-state," these same neuronal networks enter a phase of profound hyperpolarization and electrical silence 712. Physiologically, N3 is characterized by maximal skeletal muscle relaxation, lowered core body temperature, heavily regularized respiration, and the lowest heart rate of the sleep period 310.
N3 sleep is indispensable for systemic physiological recovery. It is the primary phase for the secretion of human growth hormone, which facilitates cellular repair, bone and muscle growth, and immune system regulation 1014. Because arousal and sensory thresholds are at their absolute highest during N3, waking an individual from this stage requires vigorous stimulation. Arousal from deep sleep frequently results in "sleep inertia" - a state of profound grogginess, disorientation, and impaired cognitive performance that can last for up to an hour 101114. Deep sleep duration diminishes significantly with age; while it is robust in children and young adults, healthy individuals in their seventh decade of life may experience a drastic reduction in N3, which correlates strongly with age-related memory deficits 1415.
Rapid Eye Movement Sleep Architecture
Rapid Eye Movement (REM) sleep presents a neurophysiological paradox: it pairs a highly active, "awake-like" brain with a completely paralyzed physical body. First occurring roughly 90 minutes after sleep onset, REM periods elongate as the night progresses, concentrating heavily in the final third of the sleep period 10.
Cortical Activation and Muscular Atonia
The EEG during REM sleep shifts completely away from the highly synchronized, high-amplitude slow waves of N3. Instead, the cortex reverts to desynchronized, low-amplitude, mixed-frequency patterns that are heavily dominated by theta activity, similar to Stage N1 or active wakefulness 39. This intense cortical activation, particularly in limbic and visual association areas, correlates directly with vivid, emotionally charged, and narrative-driven dreaming 9. During this state, the prefrontal cortex - responsible for logical reasoning and impulse control - is relatively deactivated, which accounts for the bizarre and uncritical nature of dream content.
Despite this profound cerebral hyperactivity, the EMG reveals complete skeletal muscle atonia (paralysis). This is a protective, active mechanism originating in the pontine regions of the brainstem, which send inhibitory signals down the spinal cord to hyperpolarize alpha motor neurons. This paralysis prevents individuals from physically acting out their dreams 3. A failure of this specific mechanism results in REM Sleep Behavior Disorder (RBD), a dangerous condition strongly linked to the future development of synucleinopathies like Parkinson's disease 1619.
Ponto-Geniculo-Occipital Waves
A defining microstructural feature of REM sleep is the presence of ponto-geniculo-occipital (PGO) waves, frequently referred to simply as P-waves. These are distinct, phasic local field potentials that originate in the pons, travel upward through the lateral geniculate nucleus of the thalamus, and terminate in the occipital cortex. P-waves are tightly temporally correlated with the rapid, jerking eye movements that give REM sleep its name 17.
Interestingly, recent high-resolution electrophysiological research has discovered that P-waves are not entirely exclusive to REM sleep. They can occur in a modified form during NREM sleep. During REM sleep, P-waves are strongly coupled to hippocampal theta rhythms; however, when P-waves occur during NREM sleep, they are accompanied by a rapid reduction in hippocampal ripple power 17. This interaction indicates that P-waves function to modulate hippocampal ensemble activity in a highly state-dependent manner, actively suppressing certain micro-events during specific sleep phases to optimize overall neural coordination 17.
Hippocampal Sharp-Wave Ripples and Memory Consolidation
Perhaps the most significant breakthrough in understanding the physiological basis of memory formation has been the characterization of hippocampal sharp-wave ripples (SPW-Rs). These represent some of the most highly synchronized and fastest network oscillations observed in the mammalian brain, playing a mandatory, causal role in systems-level memory consolidation 181920.
Electrophysiological Genesis of the Ripple
SPW-Rs are transient local field potential events that occur primarily during NREM sleep and during states of quiet, immobile wakefulness 1819. Structurally, they consist of two intimately intertwined electrophysiological phenomena. The "sharp wave" is a large-amplitude, low-frequency negative deflection reflecting massive excitatory synaptic input from the CA3 region of the hippocampus to the apical dendrites of the CA1 region 1820. This profound depolarization triggers a robust response from fast-spiking parvalbumin-positive interneurons in the CA1 layer. These interneurons impose rapid, rhythmic inhibition on the local pyramidal cell population, effectively forcing them to fire in highly synchronized, microscopic bursts. The result is the "ripple" - a brief (30 to 100 milliseconds) high-frequency oscillation superimposed precisely on the sharp wave 1821.
In rodent models, where much of the foundational SPW-R research has been conducted, ripples oscillate at extremely high frequencies, typically between 150 and 250 Hz 1821. In human intracranial EEG recordings (iEEG), hippocampal ripples present at slightly lower frequencies, predominantly in the 70 to 180 Hz range, yet they maintain the same fundamental morphology, temporal brevity, and neuroanatomical origins as their rodent counterparts 62021.
The Two-Stage Model of Memory Reactivation
The mechanism by which SPW-Rs facilitate long-term memory is conceptualized in the "two-stage" hypothesis of memory consolidation 19. During active waking behavior (Stage 1), novel information and experiences are rapidly, but weakly, encoded by the hippocampus. During subsequent NREM sleep (Stage 2), the hippocampus spontaneously reactivates the specific spatiotemporal firing patterns of neurons that were engaged during the waking experience - a phenomenon known as "replay" 1920.
This replay occurs precisely within the temporal window of the SPW-R. In both rodents and humans, specific neurons tuned to newly acquired information (such as a navigational path or a set of learned images) fire synchronously during a ripple 2223. This massive, high-frequency burst of hippocampal output is transmitted outward to the prefrontal cortex and other distributed neocortical networks.
Crucially, human electrophysiological studies have demonstrated that hippocampal SPW-Rs do not occur in neurobiological isolation. They are highly correlated with neocortical graphoelements (NC-GEs), specifically sleep spindles and the downstate-to-upstate transitions of slow-wave delta activity 724. Cortical theta bursts often precede the hippocampal ripple, potentially acting as a top-down signal triggering the hippocampus 7. The hippocampus then fires the ripple precisely into the cortical upstate, a period of heightened neocortical excitability 712. Concurrently, sleep spindles occur, further facilitating synaptic plasticity within the cortex. This highly coordinated sequence effectively embeds the short-term hippocampal memory trace into stable, long-term neocortical storage networks 712. Experimental data demonstrates that neurons coding for items that are subsequently remembered fire significantly more strongly during sleep ripples than neurons coding for forgotten items, providing direct cellular evidence that ripples drive episodic memory consolidation 2223.
Amygdala Ripples and Optogenetic Interventions
Recent human intracranial recordings have expanded the scope of ripple research beyond the hippocampus, identifying distinct ripple events within the human amygdala during NREM sleep. Like their hippocampal counterparts, amygdala ripples are associated with sharp waves, are temporally linked to sleep spindles, and tend to co-occur with hippocampal ripples 6. This synchronous AMY-HPC communication during NREM sleep provides a physiological substrate for the consolidation of emotional memories and the offline regulation of affective reactivity 619.
The causal necessity of SWRs in memory consolidation has been definitively proven in rodent models using closed-loop optogenetics. When researchers selectively disrupt SWRs using light-activated neural inhibition, memory consolidation is severely impaired 2225. Conversely, optogenetic boosting or prolonging of SWRs during post-learning sleep selectively enhances ensemble memory reactivation in the hippocampus and prefrontal cortex, directly improving subsequent memory retrieval during wakefulness 2526.
The Glymphatic System and Sleep-Dependent Brain Clearance
One of the most consequential discoveries in modern sleep science is the identification of the glymphatic system - a macroscopic, brain-wide perivascular network responsible for clearing metabolic waste and neurotoxic proteins from the central nervous system 272829. Unlike peripheral tissues, the brain lacks a conventional lymphatic system; instead, it relies on the continuous, highly regulated exchange of cerebrospinal fluid (CSF) and interstitial fluid (ISF) to maintain metabolic homeostasis 2930.
Mechanisms of Perivascular Transport
The glymphatic pathway utilizes the perivascular spaces surrounding penetrating cerebral arteries and veins 30. CSF from the subarachnoid space is driven into the deep brain parenchyma along periarterial channels. This fluid influx is highly dependent on aquaporin-4 (AQP4) water channels, which are densely concentrated on the endfeet of astrocytes that form the boundary of the perivascular space 3031. The CSF mixes with the ISF within the neuropil, dissolving and washing away metabolic byproducts, and is subsequently driven out of the brain along perivenous drainage pathways and meningeal lymphatic vessels toward the deep cervical lymph nodes 2930.
The efficiency of this fluid transport is profoundly state-dependent, increasing dramatically during NREM sleep and becoming heavily suppressed during wakefulness or periods of sleep deprivation 2829. During slow-wave sleep (Stage N3), the extracellular interstitial space volume expands by up to 60%, drastically reducing parenchymal resistance and allowing a massive surge of CSF to flush through the neural tissue 2829. Recent mechanistic studies indicate that this clearance is driven not only by arterial pulsation but also by slow, sleep-associated vasomotion, which is mediated by a drop in central norepinephrine levels during NREM sleep 2930.
Pathological Implications of Glymphatic Dysfunction
The glymphatic system plays a central role in the clearance of amyloid-beta (A$\beta$) and tau - two hallmark neurotoxic proteins implicated in the pathogenesis of Alzheimer's disease (AD) 2831. Disruption of sleep microstructure, particularly a reduction in NREM slow-wave activity and sleep spindle density, is directly correlated with impaired glymphatic clearance, leading to the rapid accumulation of these neurotoxic proteins 3132.
In humans, glymphatic function can be evaluated indirectly using advanced neuroimaging techniques, such as the diffusion tensor imaging analysis along the perivascular space (DTI-ALPS) index 1327. Decreased DTI-ALPS indices have been consistently observed in patients suffering from diverse pathologies, including psychophysiological insomnia, post-COVID sleep disturbances, Parkinson's disease, and Alzheimer's disease, establishing a clear link between poor sleep architecture and glymphatic compromise 132731.
Experimental therapeutic interventions are currently targeting the glymphatic system to mitigate cognitive decline. For instance, low-frequency repetitive transcranial magnetic stimulation (LF-rTMS) applied to patients with chronic insomnia has been shown to not only improve objective sleep metrics but also significantly increase the DTI-ALPS index, suggesting that artificially stabilizing cortical slow waves can effectively restore glymphatic clearance function 33.
Microstructural Biomarkers of Neurodegenerative Disorders
The deterioration of specific sleep microstructures serves as a highly sensitive, early biomarker for distinct neurodegenerative trajectories. The order in which sleep architecture decays provides clinical insight into whether a pathology is originating in cortical or subcortical networks 34.
In Alzheimer's disease, which is primarily characterized by cortical thinning and hippocampal atrophy, EEG microstructure disturbances appear exceptionally early in the disease process. Patients exhibit a profound reduction in NREM slow oscillations and sleep spindle density years before the onset of overt clinical memory deficits 3134. Because spindles and slow waves are generated by thalamocortical networks, their degradation serves as a direct proxy for cortical amyloid and tau accumulation 3134.
Conversely, Parkinson's disease and Lewy body dementias initiate in subcortical and brainstem regions. Consequently, these patients often first present with REM sleep abnormalities, notably the loss of muscle atonia leading to REM Sleep Behavior Disorder (RBD) 1634. However, recent research demonstrates that PD also affects NREM microstructure. The degeneration of the locus coeruleus - a primary source of brain norepinephrine - in PD patients correlates strongly with reduced sleep spindle density, linking noradrenergic dysfunction to broader microstructural decay and subsequent cognitive decline 19.
Furthermore, primary sleep disorders such as psychophysiological insomnia exhibit distinct microstructural signatures. When compared to good sleepers, insomnia patients demonstrate significantly elevated rates of Cyclic Alternating Patterns (CAP) and increased high-frequency micro-arousals during NREM sleep, despite normal total sleep times. This indicates a state of persistent central hyperarousal that fragments the restorative quality of the sleep period 3536.
Evolutionary Perspectives on Sleep Ecology
The assertion that modern, industrialized humans are suffering from an unprecedented epidemic of sleep deprivation due to artificial lighting, electronics, and demanding work schedules is widespread in popular literature. However, evolutionary anthropologists have challenged the premise that ancestral humans routinely slept eight to nine uninterrupted hours a night 3738.
Extensive actigraphy and physiological research conducted on three distinct pre-industrial societies - the Hadza hunter-gatherers of Tanzania, the Tsimane horticulturalists of Bolivia, and the San hunter-gatherers of Namibia - reveals striking similarities in ancestral sleep ecology 373940.
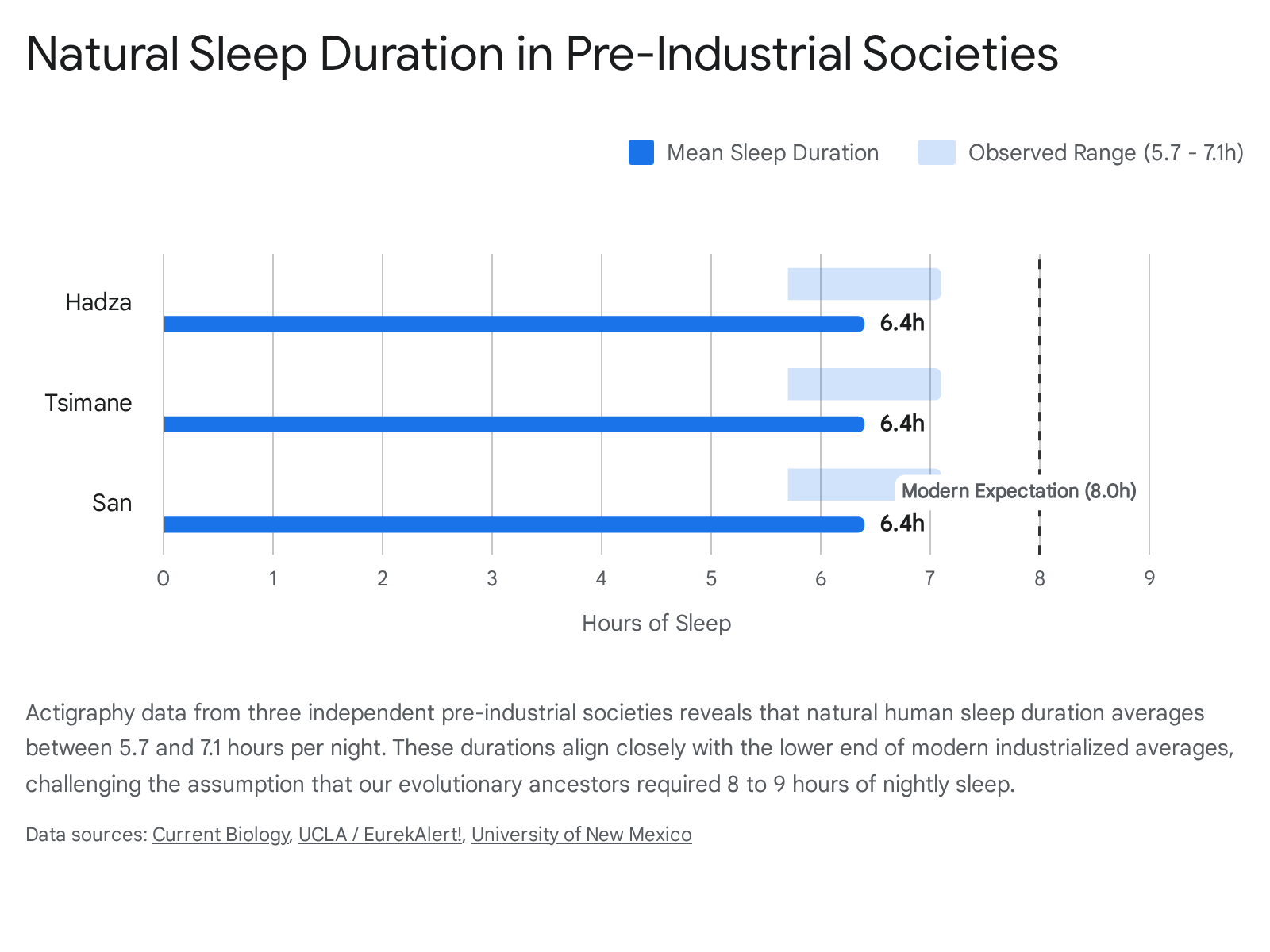
Despite lacking electricity, climate control, or modern entertainment, individuals in these populations do not exhibit the prolonged sleep durations commonly idealized in Western medicine.
Data indicates that these groups average between 5.7 and 7.1 hours of sleep per night, with total sleep periods (the time between sleep onset and offset) ranging from 6.9 to 8.5 hours 3739. Notably, daily napping is relatively rare in these populations 3738. Rather than going to sleep immediately at sunset as commonly presumed, these groups remain awake for several hours in the darkness, engaging in social activities, opportunistic hunting, or localized chores 3741.
Temperature as a Primary Circadian Zeitgeber
The strongest environmental determinant of sleep timing in these groups is not ambient light, but rather ambient temperature. Sleep onset consistently aligns with the period of falling environmental temperature in the evening. Conversely, awakening almost invariably coincides with the nadir (the lowest point) of the daily temperature rhythm, which generally occurs just before or exactly at sunrise 373839.
This pronounced physiological tethering to temperature suggests that the human sleep architecture evolved to utilize nocturnal cooling as a primary circadian zeitgeber (time-giver) 4042. The impact of temperature is further demonstrated by seasonal variations; the Tsimane sleep an average of 56 minutes longer during the colder winter months, while the San sleep 53 minutes longer 37. At awakening, researchers observed a strong systemic vasoconstriction in these subjects, indicating that peripheral heat loss signals the end of the sleep maintenance phase 39.
Despite sleeping less than the frequently recommended eight hours, individuals in these societies display robust cardiovascular health, high physical fitness, and essentially no clinical complaints of insomnia. In fact, insomnia is so virtually absent from their ecology that the San and Tsimane languages lack a specific word for the disorder 3738. Night-to-night variation in sleep duration among the Tsimane is driven largely by variable bedtimes resulting from social or opportunistic activities, rather than rigid work schedules, suggesting that high sleep variability is a natural adaptation to changing environmental opportunity costs 41.
Reconciling Modern Sleep Claims and Epidemiological Data
The science of sleep has periodically been subject to hyperbolic interpretation in popular literature. A prominent example is the bestselling book Why We Sleep by neuroscientist Matthew Walker, which catalyzed widespread public anxiety regarding the health impacts of sleep duration 4344.
Critical Evaluation of Popular Sleep Paradigms
Independent researchers, notably data scientist Alexey Guzey, have compiled extensive critiques pointing out methodological and statistical misrepresentations in Walker's assertions 4344. Several high-profile claims require strict scientific recalibration based on peer-reviewed epidemiological evidence: 1. The U-Shaped Mortality Curve: Walker asserted that "the shorter your sleep, the shorter your life span." However, broad epidemiological data consistently demonstrates a U-shaped relationship between sleep duration and mortality 4445. Both short sleep (e.g., under 5 hours) and long sleep (e.g., over 8.5 hours) are associated with higher all-cause mortality, with 7 hours routinely emerging as the statistical optimum for longevity and cardiovascular health 4446. 2. Cancer and the WHO Claim: The assertion that a lack of sleep universally doubles cancer risk and that the World Health Organization (WHO) declared a global sleep loss epidemic is inaccurate. The WHO did not declare a sleep loss epidemic. Furthermore, the International Agency for Research on Cancer (IARC) classified nighttime shift work - which severely disrupts circadian rhythmicity and light exposure - as a probable carcinogen, which is fundamentally distinct from simply sleeping fewer hours during a normal, stable nocturnal window 444748. 3. Immune System Suppression: The alarming claim that sleeping less than six hours a night results in a 70% reduction in natural killer (NK) cell activity is based on a specific, acute study where participants were severely restricted to just four hours of sleep for a single night 4748. Applying this severe, acute drop to populations who chronically sleep six hours a night is a severe extrapolation unsupported by the primary data 4449. 4. Fatal Familial Insomnia (FFI): The book posits that FFI proves pure sleep deprivation can kill a human. However, FFI is a progressive neurodegenerative prion disease that causes massive, systemic damage to the thalamus and autonomic nervous system. The patients die from widespread neurodegeneration and systemic failure, not merely from a lack of sleep 44.
The Limitations of Compensatory Sleep
Conversely, the notion that individuals can accrue massive "sleep debt" during the workweek and simply repay it by oversleeping on weekends is equally flawed 115350. Studies conducted by researchers such as Kenneth Wright at the University of Colorado demonstrate that weekend "catch-up" sleep offers only transient subjective relief and fails to reverse the systemic physiological damage of chronic weekday sleep restriction 5355.
Specifically, attempting to recover sleep debt on weekends does not adequately restore metabolic homeostasis, fails to correct insulin resistance, and further misaligns the body's circadian clock. This misalignment leads to prolonged sleep onset latency and reduced deep sleep efficiency when the individual attempts to return to a standard schedule on Monday 115355. Sleep architecture, governed by the precise timing of micro-oscillations and temperature rhythms, relies heavily on rhythmic consistency; it operates more like a metabolic cycle than a financial bank account where hours can be arbitrarily deposited and withdrawn 4950.
Conclusion
The science of sleep has progressed far beyond the simple quantification of hours spent in bed. Sleep is an intricately structured, multi-stage physiological process governed by a symphony of transient micro-events. Stage N2 sleep spindles and K-complexes act as the brain's sensory gatekeepers, actively protecting sleep continuity while initiating the early stages of memory transfer. The massive, slow delta waves of Stage N3 orchestrate the physical expansion of the brain's perivascular spaces, allowing the glymphatic system to utilize CSF to flush out neurotoxic proteins like amyloid-beta and tau, safeguarding against neurodegeneration. Concurrently, high-frequency hippocampal sharp-wave ripples fire in tightly coupled sequences with cortical upstates, engaging the neocortex in a rapid dialogue that translates ephemeral daily experiences into permanent biological memory.
While wearable technologies have popularized general sleep tracking, they currently lack the electrocortical resolution required to capture this delicate, high-frequency microstructure. Consequently, modern sleep anxiety is occasionally fueled by extrapolated epidemiological data and oversimplified metrics. Evidence from ancestral human populations indicates that a natural sleep architecture is remarkably flexible, averaging closer to six and a half hours, and is deeply tethered to environmental temperature declines rather than artificial light or rigid hourly quotas. Ultimately, optimizing sleep requires respecting its biological rhythmicity, recognizing the profound restorative power of its deep-wave microarchitecture, and understanding that consistency and microstructural quality vastly outweigh mere duration.