Physiological effects of prolonged stress
The human stress response evolved as a highly conserved, adaptive physiological mechanism designed to maintain homeostasis in the face of acute environmental threats. However, when psychological or environmental stressors persist over months, years, or decades, this immediate survival mechanism degrades into a chronic state of biological dysregulation. Prolonged stress initiates an insidious physiological reshaping of the human body, fundamentally altering neuroendocrine signaling, immune system regulation, cardiovascular hemodynamics, cellular metabolic processes, and even the structural integrity of genomic DNA.
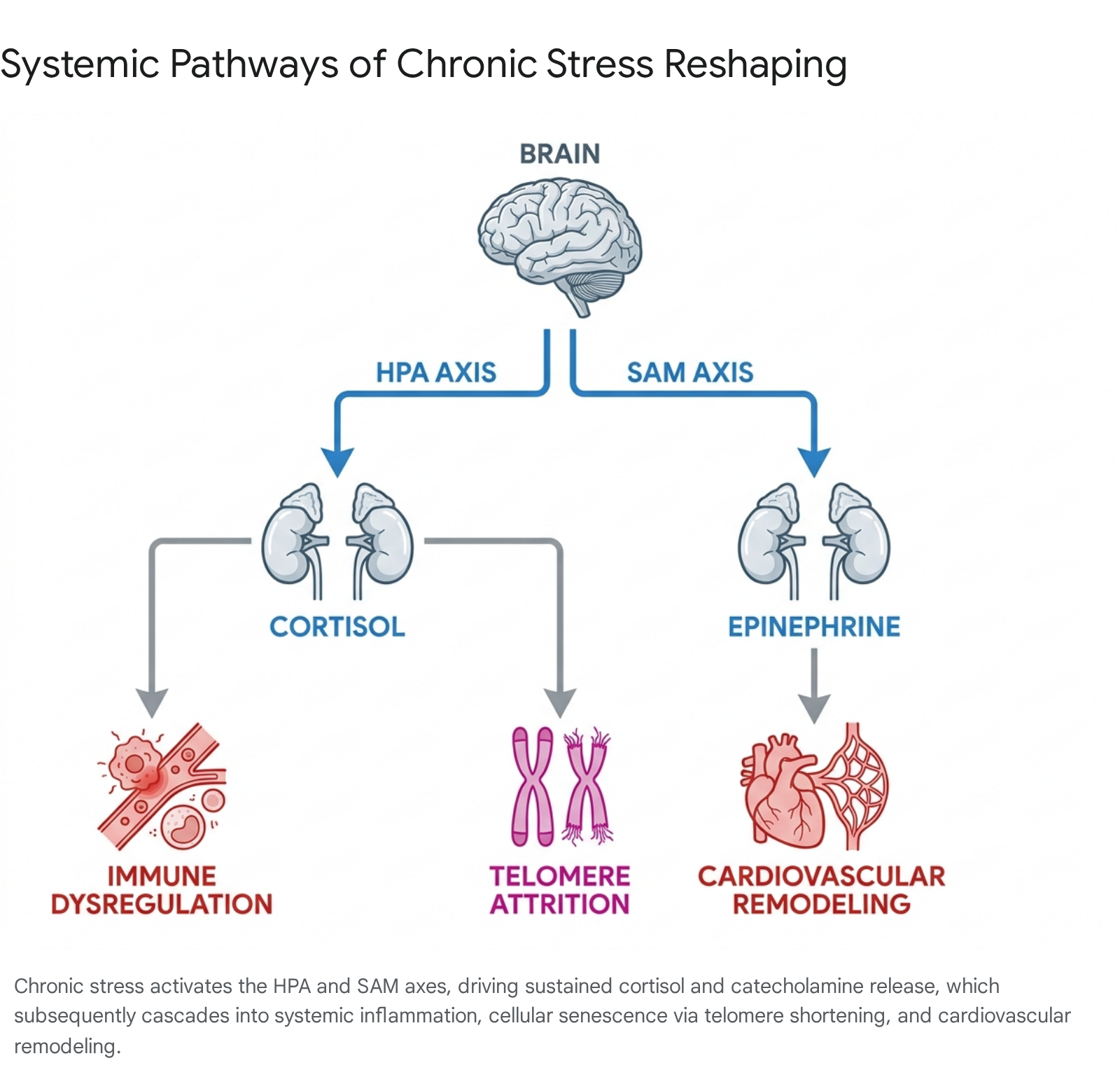
Rather than existing merely as a psychological or emotional phenomenon, chronic stress precipitates systemic, quantifiable physical deterioration that accelerates biological aging and drives the pathogenesis of numerous chronic diseases.
Neuroendocrine Dysregulation and the Hypothalamic-Pituitary-Adrenal Axis
The neuroendocrine response to stress is primarily governed by the interaction between the autonomic nervous system (ANS) and the hypothalamic-pituitary-adrenal (HPA) axis 12. In an acute stress scenario, corticolimbic regions of the brain, specifically the amygdala, detect a threat and signal the hypothalamus to secrete corticotropin-releasing hormone (CRH) 34. This action stimulates the anterior pituitary gland to release adrenocorticotropic hormone (ACTH), which subsequently signals the adrenal cortex to produce glucocorticoids, primarily cortisol 34. Concurrently, the sympathetic-adrenomedullary (SAM) axis releases catecholamines, such as epinephrine and norepinephrine, facilitating the immediate physiological changes associated with the fight-or-flight response 356.
Under normal physiological conditions, cortisol initiates an essential negative feedback loop, binding to receptors in the hypothalamus and pituitary to suppress further hormone production once the threat has passed, thereby restoring homeostasis 136. However, prolonged psychological stress disrupts this delicate equilibrium. Sustained exposure to stressors causes the HPA axis feedback mechanisms to falter, significantly altering basal cortisol levels and disrupting natural diurnal hormone rhythms 18.
Glucocorticoid Receptor Resistance and Hormonal Imbalance
A critical mechanism driving the physical reshaping of the body under chronic stress is the development of glucocorticoid receptor (GR) resistance 16. Due to continuous, elevated cortisol circulation, immune and somatic cells downregulate their glucocorticoid receptors, effectively becoming desensitized to cortisol's normal regulatory signals 1. Under healthy conditions, cortisol suppresses excessive inflammation by inhibiting the expression of pro-inflammatory genes 6. When GR resistance takes hold, this anti-inflammatory brake is removed, permitting unchecked systemic inflammation to proliferate 16.
The dysregulation of the HPA axis also alters how cortisol interacts with cellular receptors. In states of chronic excess, cortisol may increasingly bind to mineralocorticoid receptors (MRs) rather than saturated GRs 1. In immune cells such as macrophages, MR activation actively promotes the upregulation of pro-inflammatory cytokines like tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6), compounding the inflammatory shift 1. Furthermore, stress hormones act upon the bone marrow to remodel the hematopoietic system, driving the expansion of myeloid lineages at the expense of lymphoid progenitors 14. This structural shift creates a cellular microenvironment that favors the continuous release of inflammatory monocytes into the bloodstream 1.
Paradoxical Hypocortisolism and Adrenal Exhaustion
While chronic stress is traditionally associated with elevated cortisol, severe and prolonged stress can paradoxically transition into a state of profound hypocortisolism, sometimes referred to clinically as adrenal exhaustion 1. Under these conditions, the adrenal glands lose their capacity to produce adequate amounts of cortisol 1. This leaves the body devoid of its primary immune regulator, resulting in inadequate glucocorticoid-mediated immune suppression 1. This paradoxical deficiency increases the organism's vulnerability to overactive immune responses and serves as a mechanistic link to the onset and exacerbation of autoimmune conditions 1.
Laboratory Biomarkers of Acute Versus Chronic Stress
The transition from a healthy, acute stress adaptation to a pathogenic chronic stress state is measurable through specific laboratory biomarkers across multiple physiological systems 23. While acute stress triggers transient spikes in specific hormones that quickly return to baseline to facilitate adaptation, chronic stress establishes a persistent elevation and subsequent dysregulation of these parameters, leading to biological wear and tear 37.
| Biomarker Category | Acute Stress State | Chronic Stress State |
|---|---|---|
| Neuroendocrine | Short-term surge in serum cortisol and ACTH; rapid spike in catecholamines (epinephrine, norepinephrine) 37. | Sustained elevation or paradoxical blunting of cortisol; high hair cortisol levels. Altered dehydroepiandrosterone (DHEA) and aldosterone ratios 310. |
| Immunological | Temporary mobilization of leukocytes and natural killer (NK) cells; transient increase in IL-6 and C-reactive protein (CRP) to prepare for potential injury 3611. | Persistent elevation of pro-inflammatory cytokines (TNF-α, IL-1β, IL-6) and sustained high CRP. Reduced protective anti-inflammatory cytokines like IL-10 137. |
| Metabolic | Brief spike in blood glucose to meet immediate energetic demands; transient cholesterol elevation 37. | Insulin resistance, elevated glycosylated hemoglobin (HbA1c), chronic hyperglycemia, and sustained dyslipidemia (high triglycerides and cholesterol) 3710. |
| Oxidative Stress | Increased reactive oxygen species (ROS) countered by elevated reduced glutathione (GSH) for cellular adaptation 38. | Persistently high ROS with depleted antioxidant defenses (reduced GSH), resulting in oxidative cellular damage 3. |
Because serum and salivary cortisol levels fluctuate significantly throughout the diurnal cycle, modern neuroendocrinology increasingly relies on hair cortisol concentrations as an integrated, long-term biomarker of chronic HPA axis burden 23. Catecholamines, while effective markers of acute sympathetic activation, offer limited diagnostic value for chronic stress due to their rapid clearance rates 3.
Immune System Dysregulation and Systemic Inflammation
The physical reshaping driven by chronic stress is exceptionally profound within the immune system, resulting largely from the loss of glucocorticoid-mediated control and continuous sympathetic arousal. While short-term stress temporarily enhances immune surveillance to protect against acute injury or infection, chronic stress fundamentally remodels immune cell populations and impairs their functional capacities 611.
Pro-Inflammatory Cytokine Imbalance and Transcription Factors
The prolonged activation of the SAM axis results in the continuous release of norepinephrine and epinephrine, which interact directly with β-adrenergic receptors located on the surface of various immune cells 5. This interaction initiates complex intracellular signaling cascades, specifically elevating cyclic AMP (cAMP) levels, which subsequently activates protein kinase A (PKA) and exchange proteins directly activated by cAMP (EPAC) 5. These pathways stimulate crucial pro-inflammatory transcription factors, most notably nuclear factor kappa B (NF-κB) and signal transducer and activator of transcription 3 (STAT-3) 5.
Activated NF-κB and STAT-3 drive the sustained synthesis of pro-inflammatory cytokines, including IL-6, TNF-α, and interleukin-1 (IL-1) 511. Under chronic stress, the resulting cytokine storm initiates autocrine and paracrine positive feedback loops, fostering a state of chronic mild systemic inflammation 58. Furthermore, stress hormones shift the polarization of macrophages toward the highly inflammatory M1 phenotype, transforming the cellular landscape of the body's tissues to sustain long-term inflammatory responses rather than tissue repair 5.
Autoimmunity and Loss of Immune Tolerance
Chronic HPA axis dysfunction not only drives systemic inflammation but actively weakens protective immune mechanisms, predisposing the body to autoimmune conditions 16. Chronic stress blunts the glucocorticoid-mediated tolerogenic effects on dendritic cells, which are critical antigen-presenting cells 1. This impairment reduces the immune system's capacity to properly distinguish between self and non-self antigens 1.
This loss of immune tolerance is severely compounded by a reduction in both the numbers and functional capacity of T regulatory cells (Tregs), which are vital for suppressing autoreactive immune responses 1. By simultaneously driving pro-inflammatory cytokine production and dismantling regulatory T-cell function, prolonged stress shifts the immune environment toward autoimmunity, serving as a critical mechanistic link in the pathogenesis and exacerbation of systemic lupus erythematosus, rheumatoid arthritis, and multiple sclerosis 1.
Cardiovascular Remodeling and Endothelial Dysfunction
The cardiovascular system bears the mechanical and molecular brunt of chronic neuroendocrine and inflammatory dysregulation. Persistent psychological stress correlates with higher morbidity and mortality rates from cardiovascular diseases, driven by structural alterations in the heart and vasculature 91015. Evidence indicates that stress-reduction interventions might mitigate perceived stress levels and potentially reduce cardiovascular risk, though larger randomized trials are still required to firmly establish clinical protocols 4.
Corticolimbic Brain Regions and Cardiovascular Regulation
The systemic cardiovascular response to stress is regulated by the corticolimbic regions of the brain, specifically the medial prefrontal cortex, hippocampus, and amygdala 4. The amygdala functions as the center of the fear response, initiating the peripheral fight-or-flight mechanisms that activate the sympathetic nervous system 4. Studies utilizing advanced brain imaging have linked resting metabolic hyperactivity in the amygdala to downstream bone marrow activation, subsequent arterial inflammation, and an increased risk of severe cardiovascular events 4. The medial prefrontal cortex typically sends inhibitory inputs to the amygdala to suppress fear responses; however, under chronic stress, this top-down regulatory mechanism frequently becomes impaired 4.
Hemodynamic Volatility and Vascular Strain
Prolonged sympathetic nervous system hyperactivity leads to persistent hypertension, increasing the mechanical strain on blood vessel walls 910. This continuous physical stress contributes directly to endothelial dysfunction, a condition where the inner lining of the blood vessels loses its ability to properly regulate vascular tone, prevent pathological clotting, and modulate immune cell adhesion 910. Over time, stress-induced hemodynamic volatility manifests as a dysregulation of normal autonomic indices. Individuals suffering from chronic stress often display decreased heart rate variability (HRV), a primary index of vagal function indicating an inability of the parasympathetic nervous system to properly brake sympathetic arousal 410.
The systemic inflammatory environment detailed previously - particularly elevated levels of IL-6 and CRP - accelerates the progression of atherosclerosis 410. Inflammatory mechanisms weaken the fibrous caps of existing atherosclerotic plaques, significantly increasing plaque vulnerability and the risk of acute rupture, which is the primary pathophysiological event causing myocardial infarctions and ischemic strokes 4.
Microcirculation and Myocardial Ischemia
While macrovascular damage is well documented, chronic stress also profoundly reshapes microcirculation. During periods of mental stress, the smaller blood vessels responsible for feeding the heart muscle - the coronary microvasculature - can undergo paradoxical constriction, leading to a transitory increase in arterial stiffness 411.
Data from the Mental Stress Ischemia Prognosis Study (MIPS) indicates that this microvascular vulnerability is particularly pronounced in specific demographics. Women aged 50 or younger exhibit up to twice the reduction in blood flow to the heart muscle during psychological stress compared to male counterparts 11. This distinct physiological response indicates that stress-induced ischemia is not merely a passive byproduct of existing plaque buildup, but a dynamic, direct result of autonomic and vascular hyperreactivity triggered by emotional and psychological burden 411. To investigate these complex biological pathways further, the American Heart Association recently awarded significant research grants to dedicated networks to study how chronic psychosocial stressors drive cardiovascular risk 15.
Organ-Specific Pathologies of Chronic Stress
Beyond the cardiovascular and immune systems, the dysregulation of the HPA axis exerts profound organ-specific consequences across the entire body, demonstrating the truly systemic nature of stress-induced physical reshaping 1.
Neuroimmune and Cognitive Consequences
In the central nervous system, chronic psychological stress disrupts the HPA axis, leading to sustained cortisol exposure that alters glucocorticoid receptor sensitivity in the brain 1. This mechanism is deeply linked to depressive phenotypes, specifically melancholic depression, where HPA hyperactivity and systemic inflammation coexist 1. Stress-induced activation leads to increased central cytokine production, microglial activation, and oxidative stress, all of which impair neuroplasticity and promote neurodegeneration 1. In the context of aging and social isolation, HPA dysregulation creates a feedforward loop of immune senescence and neuroinflammation that accelerates cognitive decline, particularly in Alzheimer's disease 1. Furthermore, prenatal stress can program an offspring's developing HPA axis toward lifelong hypersensitivity, increasing the risk of neuroinflammatory responses to future stressors 1.
Gastrointestinal and Musculoskeletal Systems
In the gastrointestinal tract, HPA signaling abnormalities cause visceral hypersensitivity, motility changes, and gut barrier dysfunction 1. This breakdown contributes to functional disorders like Irritable Bowel Syndrome (IBS) and worsens intestinal inflammation in Inflammatory Bowel Disease (IBD) by intensifying mucosal immune activation 1. Microbial metabolites from the gut microbiota can further heighten HPA responses, creating a bidirectional feedback loop of stress and GI dysfunction 1.
The musculoskeletal system also undergoes physical degradation. Chronic glucocorticoid exposure leads to continuous muscle wasting and adverse changes in body composition 1. Central nervous system inflammation can activate the HPA axis to induce muscle atrophy through catabolic pathways, such as ubiquitin-proteasome degradation, a process that significantly speeds up muscle wasting in cachexia 1. Early-life stress also sensitizes the HPA axis to produce chronic muscle pain and tension, with female populations showing higher susceptibility 1.
In the integumentary system, localized dysregulation of a cutaneous HPA-like axis delays wound healing 1. Local activation of the enzyme 11β-HSD1 in keratinocytes raises tissue-level cortisol, impairing skin barrier repair, worsening conditions like atopic dermatitis, and degrading skin collagen 1.
Telomere Attrition and Cellular Senescence
Perhaps the most profound physical reshaping caused by chronic stress occurs at the genomic level. Telomeres are the protective, repetitive non-coding DNA-protein complexes (specifically TTAGGG repeats) capping the ends of linear chromosomes 81213. Due to the inherent "end-replication problem," DNA polymerases cannot fully replicate the lagging strand during cellular division, leading to the gradual loss of 50 to 200 base pairs per division 12. When telomeres reach a critical minimum length, they trigger a DNA damage response, forcing the cell into replicative senescence or apoptosis (programmed cell death) 1214.
While telomere attrition is a natural biomarker of biological aging, substantial clinical and experimental evidence indicates that chronic psychological stress significantly accelerates this process 81214. In a seminal study conducted by Epel and colleagues focusing on premenopausal women, those experiencing the highest levels of perceived psychological stress exhibited telomeres that were, on average, 550 base pairs shorter than those of low-stress women - a loss equivalent to roughly 9 to 17 years of additional chronological aging 812.
Telomere Dynamics and Oxidative Stress
The specific biological mechanisms connecting psychological stress to accelerated telomere shortening are deeply intertwined with the neuroendocrine and immune disruptions previously described 1415. Chronic exposure to glucocorticoids increases the production of reactive oxygen species (ROS) and drives mitochondrial dysfunction 81215.
Telomeric DNA is particularly vulnerable to oxidative damage because it is highly rich in guanine bases, which are exceptionally susceptible to oxidation 12. High levels of ROS cause single-strand DNA breaks specifically in the telomeric region. Furthermore, chronic stress and associated oxidative damage disrupt the structural integrity of the "shelterin" protein complex, which normally binds to and physically shields the telomere 1415. The displacement of shelterin rapidly exposes the chromosome end, prompting structural collapse and premature cellular senescence 1415. Stress also directly impairs the enzymatic function of telomerase - the reverse transcriptase enzyme responsible for replenishing telomeric DNA - further removing the cell's capacity for structural repair 812.
Intergenerational and Epigenetic Telomere Effects
The physical consequences of stress on telomere biology can cross generational lines. Research indicates that severe maternal stress during pregnancy is associated with shorter telomere length in newborn offspring 14. Elevated maternal cortisol (as assessed by hair cortisol concentrations) during pregnancy predicts shorter telomeres in infants, suggesting that prenatal programming via stress mediators profoundly impacts fetal genomic stability 1314. Epel and colleagues describe this as "telotype transmission" - the directly inherited transmission of short telomeres from parental germlines shaped by environmental adversity 15.
Exceptions and Calibrated Uncertainty in Telomere Science
While stress-induced telomere shortening has been robustly documented in numerous observational cohorts, contemporary molecular biology injects calibrated uncertainty into the universal application of telomere length as a simplistic, overarching "mitotic clock" 151622.
A highly precise April 2024 study mapped the telomeres of 147 individuals and revealed that different chromosomes possess unique, end-specific telomere-length distributions, rather than operating under a single uniform range 2217. In some subjects, lengths varied by up to 6,000 bases across different chromosomes, suggesting that specific chromosome ends, rather than average overall leukocyte telomere length, may be the primary trigger for stem-cell failure 22. Furthermore, investigations into premature aging (progeroid) syndromes show that while disorders like Werner Syndrome exhibit profound telomere shortening, others, such as Hutchinson-Gilford Progeria Syndrome, do not 16. These findings suggest that while stress undoubtedly accelerates cellular aging via oxidative damage to telomeres, the dynamic of telomere loss is highly complex, chromosome-specific, and heavily mediated by varying underlying genetic predispositions 1622.
Allostatic Load and the Weathering Hypothesis
To understand how systemic, micro-level damage - such as telomere attrition and cytokine imbalance - aggregates into macro-level physical decline, the scientific community relies on the concept of "Allostatic Load," pioneered by neuroendocrinologist Bruce McEwen 241819.
The Concept of Allostatic Load and Energy Dynamics
Allostasis is the organism's active process of maintaining stability through change by adjusting physiological parameters, primarily through the release of stress hormones 1920. Allostatic load refers to the cumulative, physiological "wear and tear" exacted upon the body's organs and tissues when these adaptive systems are chronically over-activated or fail to properly shut down following a stressor 241920.
Energy regulation sits at the heart of allostasis; the body constantly adjusts its energy expenditure in response to internal and external threats 24. However, under conditions of chronic stress, these continuous energy adjustments contribute to allostatic load, severely affecting metabolic processes and driving physical health issues 24. It acts as a transdiagnostic metric, tracking the sum of the immune, cardiovascular, metabolic, and neuroendocrine damage caused by a lifetime of stress 2021. Recent theoretical frameworks, such as the process model of "Stress Generation," further elucidate this dynamic by suggesting that individuals, influenced by their momentary personal characteristics, actively contribute to the occurrence of daily hassles. These short-term dynamics act as the building blocks for long-term stress generation and the subsequent accumulation of allostatic load 2223.
Cumulative Burden of Marginalization and Weathering
Building upon the biological plausibility of allostatic load, public health researcher Arline Geronimus conceptualized the "Weathering Hypothesis" in the early 1990s to explain stark health disparities affecting marginalized groups, particularly Black Americans 242533. Weathering suggests that the relentless, daily psychosocial stress of navigating systemic racism, economic exclusion, implicit bias, and political oppression acts as a continuous, toxic environmental stressor 242534.
Geronimus equated the process of weathering to the tabletop game Jenga; as more pieces are pulled from the tower due to daily stressors, the physiological structure can no longer stand and eventually collapses 26. Much like a physical building deteriorating under harsh weather, the human body undergoes premature, accelerated physiological aging when constantly subjected to marginalization 3334. Geronimus initially demonstrated this through maternal health outcomes. Unlike White women, whose pregnancy risks generally decrease from their teens into their mid-twenties, Black women were shown to have healthier pregnancies in their teens 2426. By their mid-twenties, the accumulated weathering from a decade of chronic, race-based stress substantially increased their biological age, subsequently increasing maternal and infant mortality risks 2426.
Data indicates that by age 45, half of Black women exhibit high allostatic load scores - a physiological deterioration that socioeconomic status alone cannot fully explain, as both poor and nonpoor Black women face the highest probabilities of bearing high allostatic load scores compared to their White counterparts 26.
Epigenetic and Transgenerational Implications
The physical reshaping of weathering extends beyond systemic inflammation and hormonal imbalance into the epigenome 2526. Chronic stress triggers DNA methylation, an epigenetic process that provides stable alterations to the expression of specific genes without changing the underlying genetic sequence 26. Epigenetic modifications can literally rewrite an individual's biological destiny by suppressing protective genes or permanently promoting inflammatory pathways in response to environmental adversity 26. This raises the significant possibility that the epigenetic changes resulting from a mother's cumulative life stress can be inherited by her children, physically linking the weathering hypothesis with transgenerational trauma 25.
Stress-Induced Tumor Microenvironment Alterations
The interplay between psychological stress and oncology was historically debated, yielding conflicting results in epidemiological studies. However, modern animal and molecular studies have firmly established that chronic stress profoundly reshapes the tumor microenvironment (TME), actively facilitating cancer growth, invasion, and metastasis 27282930. The biological mechanisms by which stress drives cancer progression are both direct - binding neuroendocrine signaling molecules to cancer cell receptors - and indirect, via severe immune system dysregulation 2730.
Neutrophil Extracellular Traps and Metastasis
A primary mechanism of stress-induced cancer metastasis is driven by the influence of glucocorticoids on specific innate immune cells. Under conditions of chronic stress, elevated glucocorticoids induce neutrophils to overproduce web-like DNA-protein structures known as neutrophil extracellular traps (NETs) 40. While normally deployed as a defense mechanism to physically trap pathogens, in the context of cancer, these sticky nets create highly hospitable biological niches in distant organs 40.
Experiments in mouse models demonstrate that chronic stress induces NET formation, which then physically captures circulating tumor cells and alters local tissue environments in organs like the lungs and spleen 40. This stress-induced architecture shields malignant cells from immune surveillance and dramatically increases the rate of metastasis 40. When researchers administered DNase I - a compound that breaks down NETs - to stressed mice with mammary tumors, the rate of lung metastasis was significantly reduced, confirming the mechanistic role of NETs in stress-driven cancer spread 40.
Oncogenic Transcription Pathways and Immunosuppression
Simultaneously, the continuous release of SAM axis catecholamines (norepinephrine and epinephrine) directly influences tumor biology 2728. These hormones bind to β-adrenergic receptors heavily expressed on the surface of many cancer cells, triggering intracellular signaling that activates the STAT-3 and NF-κB pathways 530.
Within the tumor itself, activated STAT-3 acts as a potent pro-oncogenic transcription factor, driving cancer cell proliferation, VEGF-mediated angiogenesis (the formation of new blood vessels to feed the tumor), and epithelial-mesenchymal transition (EMT), which allows tumor cells to detach from the primary mass and invade surrounding tissues 530. Concurrently, these stress pathways exert an indirect immunosuppressive effect on the tumor microenvironment by shifting the polarization of tumor-associated macrophages into the immunosuppressive M2 phenotype and recruiting myeloid-derived suppressor cells (MDSCs) 52930. These modified immune cells act as a defense shield for the tumor, actively suppressing the cytotoxic T-cells and natural killer cells that would otherwise target and destroy malignant tissue 2930. The result is a systematically reprogrammed microenvironment that promotes tumor stemness and accelerates resistance to chemotherapy, linking chronic psychological stress directly to poorer oncological prognoses 2830.
Reversibility and Lifestyle Interventions
While chronic stress inflicts pervasive, multi-system damage, modern geroscience and lifestyle medicine demonstrate that substantial elements of this biological reshaping are reversible 3132. Because the mechanisms of physical decline - HPA axis dysregulation, oxidative stress, telomere attrition, and epigenetic methylation - are highly dynamic, targeted behavioral and clinical interventions can effectively alter physiological trajectories 3133.
Comprehensive Lifestyle Medicine
Clinical trials reveal that comprehensive lifestyle modifications exert measurable, disease-modifying benefits at the molecular level. A landmark randomized controlled trial conducted by Dean Ornish evaluated patients with early-stage Alzheimer's disease 31. The results demonstrated that 20 weeks of intensive lifestyle change resulted in significant improvements in cognition and function, accompanied by a dose-response reduction in amyloid deposition (measured through the amyloid-beta 42/40 ratio) correlated to the degree of lifestyle change 31.
Similarly, behavioral habit programs emphasizing dietary shifts, sensory awareness, and daily movement have shown marked success in achieving sustained remission of metabolic syndrome (MetS). A 24-month clinical trial found that a habit-based lifestyle program physically reversed the abdominal obesity, dyslipidemia, and insulin resistance driven by long-term allostatic load, outperforming standard education and activity monitoring 33.
Telomerase Activation and Habit Modification
Protective physiological mechanisms can also be actively stimulated to combat cellular senescence and telomere attrition. Large-scale population data from the National Health and Nutrition Examination Survey (NHANES) indicates that regular strength training operates as a potent mitigator of biological aging 34. US adults engaging in just 90 minutes of strength training per week demonstrated significantly longer telomeres compared to sedentary peers, equating to an average of 3.9 fewer years of biological aging 34. Furthermore, participants with shorter telomeres who maintained high "Brain Care Scores" - indicative of healthy lifestyle choices including sleep, exercise, and diet - did not show a significantly increased risk for age-related brain diseases, suggesting that lifestyle can buffer the consequences of genetic or stress-induced cellular aging 1735.
Targeted stress-reduction techniques also directly influence genomic stability. Studies evaluating the Transcendental Meditation technique alongside health education demonstrated that stress reduction could directly increase telomerase gene expression (specifically hTERT and hTR mRNA levels) while significantly reducing systolic blood pressure in populations with high cardiovascular risk 36. Furthermore, researchers like Elissa Epel have demonstrated that structured resilience training and targeted breathing practices can rapidly improve executive function, emotional regulation, and biosensor profiles of daily stress within weeks 4737.
Ultimately, while the weathering effects of chronic stress drive the physical reshaping of the body toward disease and cellular senescence, the human organism retains a profound capacity for biological plasticity. Mitigating allostatic load through deliberate environmental, behavioral, and clinical interventions is fundamentally capable of shifting the body's physiological network out of pathological deterioration and back into homeostatic balance 12431.