Neuroscience of social rejection and physical pain
The premise that social rejection causes a sensation akin to physical injury is a ubiquitous linguistic metaphor across human cultures. Over the past two decades, affective and social neuroscience has sought to determine the extent to which this metaphor reflects biological reality. Early neuroimaging research generated the "shared representation" theory, positing that the human brain co-opted primitive physical pain circuitry to process threats to social inclusion. However, recent advances in multivariate pattern analysis (MVPA), high-powered meta-analyses, and cross-cultural functional magnetic resonance imaging (fMRI) have significantly refined this model. The contemporary neuroscience of social pain reveals a highly complex landscape: while social and physical pain share gross anatomical substrates and neurochemical modulators, they are driven by distinct neural population codes, divergent cognitive networks, and specialized evolutionary adaptations.
Evolutionary Biology of Social Attachment
The biological necessity of social connection is rooted deep within the evolutionary history of vertebrate life. For mammals, which are born altricial - developmentally immature and wholly dependent - survival is entirely contingent upon maintaining close physical proximity to a primary caregiver 12. Because threats to social bonds directly imperiled survival, natural selection favored organisms equipped with internal neurological alarm systems that actively signaled social separation and motivated reunification.
Acoustic Communication and the Origins of Separation Distress
The earliest behavioral manifestations of social pain evolved in the form of separation distress vocalizations. Phylogenetic reconstructions of terrestrial vertebrates (tetrapods) suggest that acoustic communication has an ancient evolutionary origin dating back approximately 400 million years 345. This capability evolved independently across multiple clades, including mammals, birds, and crocodilians, largely in association with nocturnal lifestyles where visual signals were rendered ineffective 67.
While it was previously theorized that acoustic communication drove species diversification, macroevolutionary analyses reveal that acoustic signaling and speciation rates are largely uncoupled 56. Instead, the primary adaptive function of these vocalizations in mammalian and avian species is immediate behavioral response to maternal separation or social isolation, acting to solicit social support, caregiving, and defense from predators 89.
Mammalian Brain Development and the Social Neocortex
The evolution of mammalian social complexity required significant neuroanatomical adaptations, most notably the expansion of the neocortex. Comparative neurobiology models, such as those tracking 95 distinct neural development milestones across nine mammalian species, reveal an evolutionarily conserved pattern of sequence and timing in brain maturation 1011. As mammalian lineages diverged, species transitioning from solitary living to pair-living and complex group-living exhibited corresponding increases in relative brain size and longevity 1213.
The expansion of the prefrontal, posterior parietal, and insular cortices in primates facilitated a shift from purely olfactory-driven individual recognition to complex, multimodal social integration 1415. This cortical expansion allowed for the development of the "social brain," capable of managing polyadic interactions, evaluating conditional helping strategies, and processing the nuanced social signaling required to maintain group cohesion 1617.
Neuropharmacology of Social Bonding
The neurochemical foundation for social pain is most comprehensively explained by the Brain Opioid Theory of Social Attachment (BOTSA), originally pioneered by affective neuroscientist Jaak Panksepp 2818. BOTSA posits that the neural mechanisms that evolved to modulate physical pain via endogenous opioids were phylogenetically adapted to mediate social bonding 192021.
The Brain Opioid Theory of Social Attachment
Endogenous opioids - natural brain neuropeptides involved in pleasure, reward, and analgesia - play a critical role in reinforcing positive social interactions 22. Under the BOTSA framework, social isolation reduces endogenous opioid release, precipitating a state of neurochemical distress akin to physical opiate withdrawal 222. Exogenous manipulation of this system confirms the opioid-social pain link: the administration of low-dose morphine (an opioid agonist) substantially decreases distress vocalizations in socially isolated mammalian pups, whereas the administration of naltrexone (a mu-opioid receptor antagonist) exacerbates separation distress and vocalizations 192223.
In human subjects, double-blind, placebo-controlled studies reveal that naltrexone administration significantly reduces feelings of social connection both in laboratory settings and in daily diary reports 23. Furthermore, individuals with chronic opiate addictions exhibit profound deficits in adaptive attachment profiles, supporting the hypothesis that exogenous opiates hijack the neuro-peptide endogenous opioid system that normally modulates social bonds 1921.
Genetic Variation in the Mu-Opioid Receptor
The human opioid system's sensitivity to social pain is highly modulated by genetic variation. Research into the OPRM1 gene, which encodes the mu-opioid receptor, demonstrates that the A118G polymorphism influences individual differences in attachment style and social sensitivity 1824.
Carriers of the minor 118G allele exhibit heightened interpersonal sensitivity. Compared to A/A homozygotes, G-allele carriers report lower levels of avoidant attachment, experience greater pleasure in positive social situations, but conversely show stronger emotional and neural reactivity to maternal care deficits and acute social rejection 1824. Positron emission tomography (PET) imaging confirms that variations in avoidant attachment correlate negatively with mu-opioid receptor availability in the thalamus, anterior cingulate cortex, amygdala, and insula, firmly linking endogenous opioid tone to psychosocial well-being 18.
The Univariate Overlap Hypothesis
The hypothesis that social pain and physical pain share functional neural circuitry was initially solidified by functional neuroimaging studies utilizing the "Cyberball" paradigm 252627. Cyberball is a virtual ball-tossing game designed to experimentally manipulate social inclusion and ostracism. During the exclusion phase, participants unexpectedly cease receiving the ball from computer-generated co-players. Despite the artificiality of the setting, this manipulation reliably threatens fundamental psychological needs - belonging, self-esteem, control, and meaningful existence - and elicits self-reported social distress 252829.
The Cyberball Paradigm and Affective Pain Circuitry
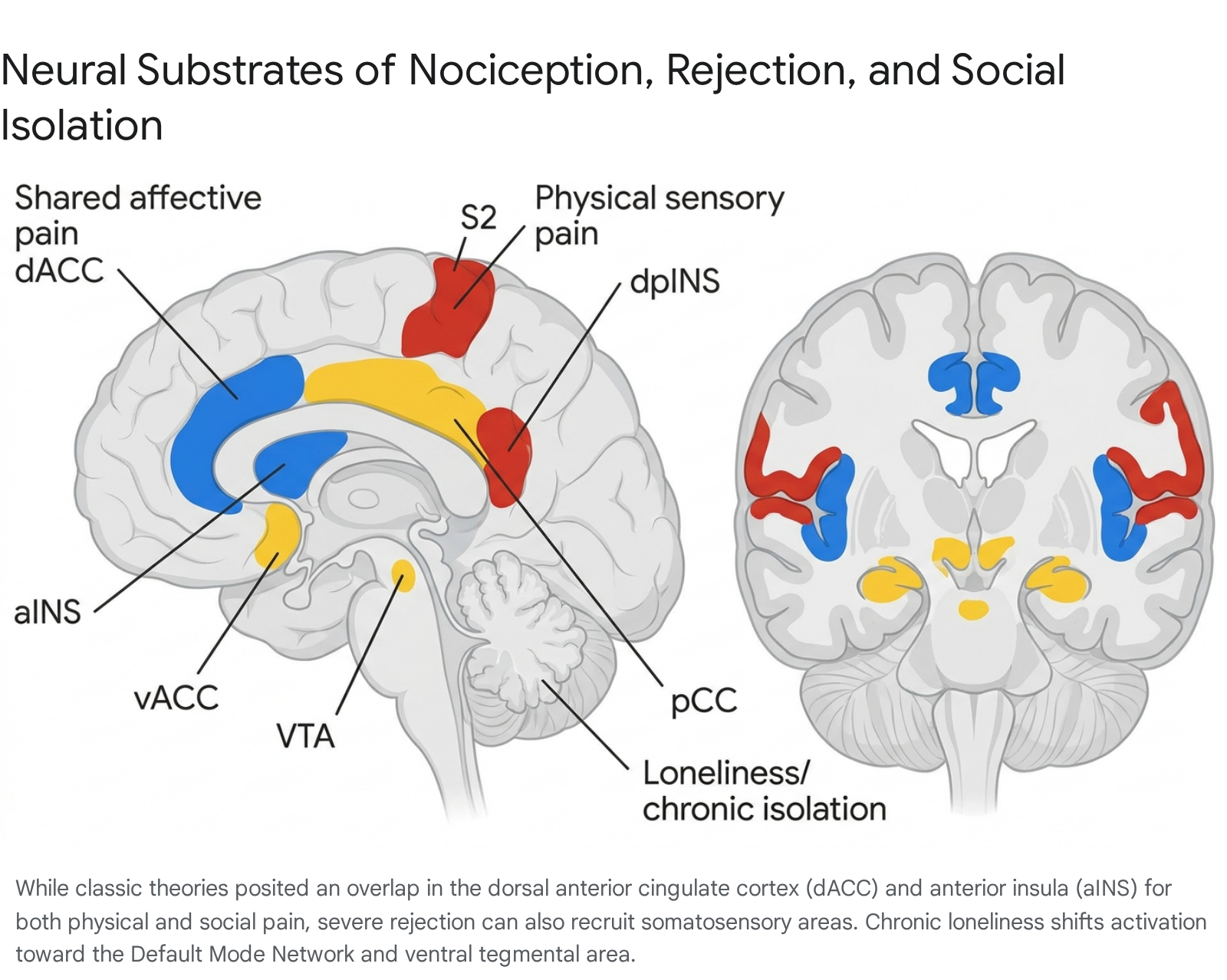
Early fMRI investigations into Cyberball-induced rejection, utilizing univariate general linear models, identified the dorsal anterior cingulate cortex (dACC) and the anterior insula (aINS) as the core neuroanatomical hubs of social pain 12630. These regions are classically recognized as the "affective" pain network - the circuitry responsible for processing the unpleasantness and distress of physical injury, distinct from the sensory-discriminative aspects of pain (such as the exact location and physical intensity of a somatic stimulus) 13132.
Subsequent research demonstrated that individual differences in physical pain sensitivity correlate with social pain sensitivity 1. Furthermore, neuropharmacological interventions typically utilized for somatic nociception (such as acetaminophen) have been shown to attenuate neural responses to social rejection in these specific affective regions 133.
Somatosensory Recruitment in Severe Rejection
While initial theories maintained that social pain solely recruited the affective components of the physical pain matrix, later studies suggested that intensely powerful social rejection might also recruit sensory-discriminative regions. In a highly cited study by Kross et al. (2011), participants who had recently experienced an unwanted romantic breakup were scanned while viewing photographs of their ex-partners 313435.
This intensely salient emotional stimulus activated not only the dACC and aINS, but also the secondary somatosensory cortex (S2) and the dorsal posterior insula (dpINS) - regions historically believed to be exclusive to the sensory processing of somatic injury 313435. These findings were interpreted as evidence that the overlap between physical and social pain was more extensive than previously theorized, bridging both affective and somatosensory domains, thereby providing a literal neurological basis for the sensation of a "broken heart" 3135.
Methodological Critiques and Meta-Analytic Revisions
Despite the widespread acceptance of the shared representation theory, recent high-powered meta-analyses and methodological critiques have challenged the primacy of the dACC in processing social exclusion. Skeptics argue that univariate fMRI analyses lack the anatomical specificity required to declare that two distinct psychological events are processed by identical neural mechanisms 3637.
The Expectancy Violation Confound
One primary critique of the Cyberball paradigm concerns the confounding factor of "expectancy violation." Because participants are included in the game before being abruptly excluded, the dACC activation observed in standard fMRI contrasts may reflect cognitive conflict monitoring, error detection, or generalized expectancy violation, rather than "social pain" per se 272838.
Electroencephalography (EEG) studies demonstrate that Cyberball exclusion elicits distinct event-related potentials (ERPs). Specifically, unexpected exclusion triggers a frontal P300 (P3a) component at 240 - 300 ms associated with novelty and threat detection, followed by a centroparietal P3b at 300 - 400 ms associated with context updating and subjective probability 3940. The magnitude of these P3 responses correlates directly with the participant's prior expectation of receiving the ball. This implies that the initial neural alarm during Cyberball is largely processing the violation of a predictive model of social interaction, challenging the interpretation that the dACC activation is exclusively a marker of pain 2540.
The Default Mode Network Shift
A comprehensive activation likelihood estimation (ALE) meta-analysis by Mwilambwe-Tshilobo and Spreng (2021), examining 53 Cyberball fMRI studies comprising 1,817 participants, found minimal reliable evidence for dACC involvement 414243. Using a strict probabilistic atlas, fewer than 15% of the included studies reported peak coordinates within the true anatomical bounds of the dACC 4243.
Instead, the meta-analysis revealed that social exclusion reliably engages nodes of the Default Mode Network (DMN). Consistent recruitment was observed in the ventral anterior cingulate cortex (vACC), subgenual ACC (sgACC), posterior cingulate cortex (pCC), inferior frontal gyrus (IFG), and superior frontal gyri 4243. The vACC and pCC are highly implicated in self-referential processing, autobiographical memory, and the regulation of social distress 2744. These findings indicate that rather than simply triggering an acute nociceptive reflex, social exclusion initiates complex mentalizing, rumination, and self-evaluation processes mediated by the DMN 2743.
Multivariate Pattern Analysis and Distinct Neural Signatures
The most definitive challenge to the physical-social pain overlap hypothesis has come from the application of Multivariate Pattern Analysis (MVPA). Univariate fMRI analyses average blood-oxygen-level-dependent (BOLD) signals across millions of neurons within a spatial voxel to determine if a gross anatomical region activates. However, overlapping univariate activity does not guarantee that the same underlying neural populations are driving the response 3637.
Deconstructing the Shared Representation Theory
In a landmark study, Woo et al. (2014) utilized machine-learning algorithms (linear Support Vector Machines) to analyze whole-brain fMRI patterns of participants subjected to both physical pain (noxious thermal heat) and social pain (viewing photos of ex-partners following a romantic breakup) 364546. The researchers trained MVPA classifiers using a "one-against-all" approach to identify spatially distributed, fine-grained neural signatures specific to each experience.
The findings fundamentally dismantled the idea of identical shared representation. The physical pain classifier discriminated thermal pain from non-painful warmth with 92% accuracy, and discriminated physical pain from social rejection with 100% accuracy 4546.
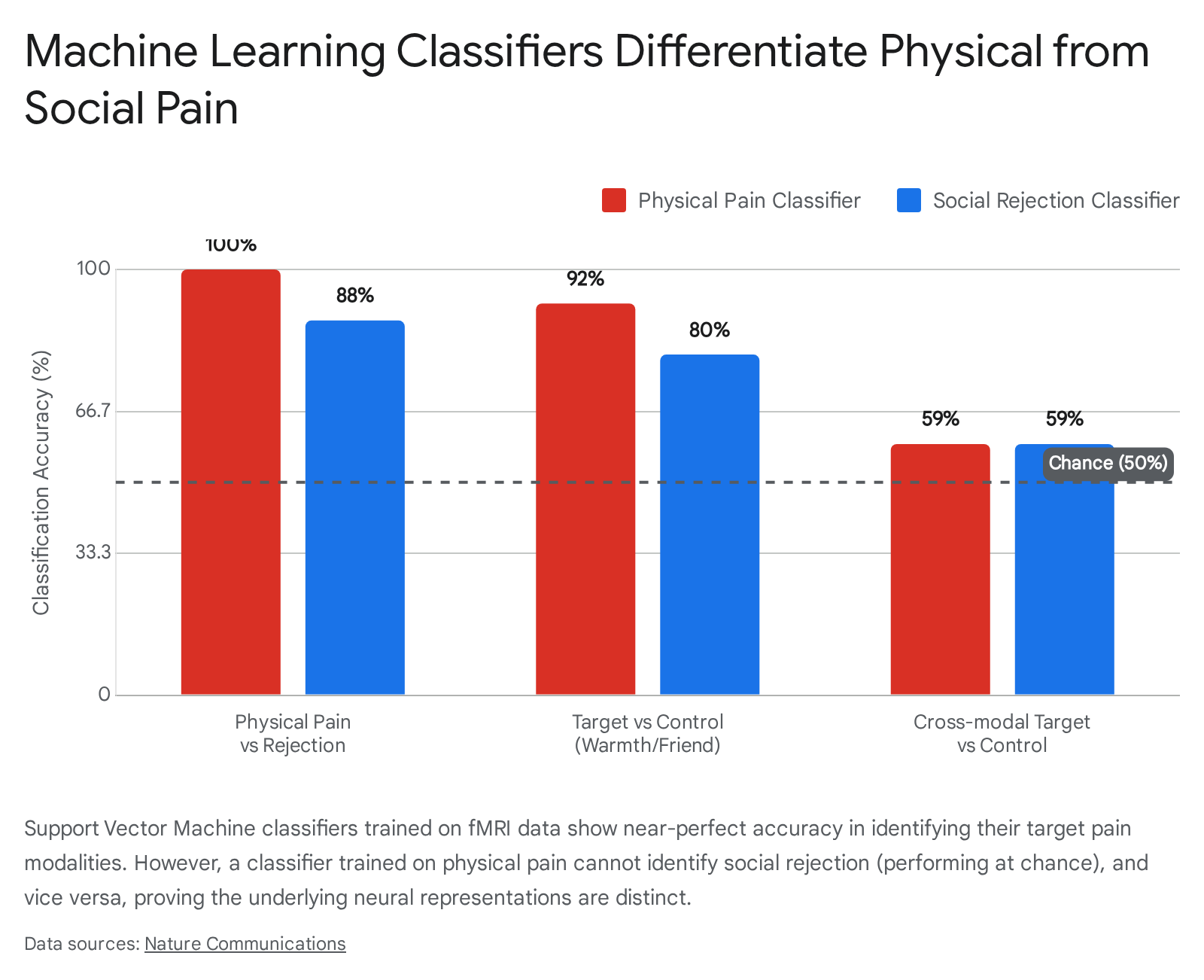
Conversely, the physical pain classifier performed at chance levels (59%) when attempting to discriminate social rejection from neutral social stimuli, demonstrating it was blind to social pain 45. The social rejection classifier was similarly blind to physical pain 3745.
Divergent Functional Connectivity
Crucially, even within the specific gross anatomical regions traditionally believed to process both pains uniformly (the dACC and aINS), the multivariate patterns for physical pain and social rejection were highly distinct and functionally uncorrelated (r = -0.04) 4546.
Using a separate resting-state dataset, researchers found that the specific dACC voxels encoding the physical pain pattern were functionally connected to the thalamus and posterior insula 45. In contrast, the dACC voxels encoding the social rejection pattern exhibited functional connectivity to the lateral prefrontal and parietal cortices 45. These data conclusively demonstrate that while physical pain and social rejection engage the same macroscopic brain regions, they rely on entirely distinct, separately modifiable neural population codes and functional subnetworks 374546.
Phenomenological Distinctions in Reliving Pain
The phenomenological differences between social and physical pain further support distinct neural mechanisms. Humans can vividly "relive" or re-experience the affective distress of a past social rejection merely through memory, whereas recalling a past physical injury rarely resurrects acute somatic pain 3247.
Functional MRI studies assessing internally generated pain show that reliving social pain heavily recruits the dorsomedial prefrontal cortex (dmPFC), a node of the mentalizing network, which functionally correlates with affective pain system responses 47. Reliving physical pain, however, recruits the inferior frontal gyrus (IFG), which is associated with body-state processing. This demonstrates that top-down cognitive pathways construct these pain states differently, relying on mental state processing for social pain and body state processing for somatic pain 47.
Empathy for Physical versus Social Pain
The evolutionary divergence of social and physical pain systems is similarly reflected in the neural mechanics of human empathy. Empathy - the capacity to vicariously experience and understand the affective states of others - relies on differing neural architectures depending on whether the observed pain is somatic or social 3348.
Mirror Neuron versus Mentalizing Networks
A massive activation likelihood estimation (ALE) meta-analysis of 35 fMRI studies demonstrated strict neurofunctional segregation between the two empathy subtypes, with virtually no shared activated brain areas detected in differential analyses 4148.
- Physical Pain Empathy: Engaging with another's physical injury primarily recruits core regions of the Mirror Neuron System (MNS). This includes significant left-lateralized activation in the anterior insula (aINS) for affective-cognitive integration, the anterior midcingulate cortex (aMCC) for emotional resonance, and the inferior parietal lobule (IPL) and inferior frontal gyrus (IFG) for action comprehension 334148.
- Social Pain Empathy: Empathizing with someone experiencing ostracism or social loss almost entirely bypasses the sensorimotor mirror network. Instead, it selectively activates the mentalizing network, specifically the dorsomedial and medial prefrontal cortices (dmPFC/mPFC) 4148.
Neuropeptide Modulation of Social Empathy
Recent neuropharmacological research underscores the specialized nature of social empathy. A 2024 double-blind, placebo-controlled study investigated the roles of oxytocin (OXT) and arginine vasopressin (AVP) in modulating empathy for social pain. Both neuropeptides significantly enhanced subjective empathy ratings for negative social scenarios (e.g., breakups, exclusion) without altering general arousal or valence 49.
However, their mechanistic profiles diverged substantially. OXT preferentially targeted perceptual-visual integration and cognitive empathy, interacting extensively with opioid signaling pathways and subcortical visual processing via the red nucleus 49. Conversely, AVP exerted stronger modulation over perceptual-execution circuits mapped onto the amygdala, driving the motivational and executive dimensions of social empathy 49.
| Feature | Physical Pain (Somatic) | Social Pain (Exclusion/Loss) | Empathy for Physical Pain | Empathy for Social Pain |
|---|---|---|---|---|
| Primary Evolutionary Function | Tissue protection, withdrawal reflexes | Maintenance of social bonds, soliciting care | Immediate threat detection, motor mirroring | Theory of Mind, complex group cohesion |
| Key Univariate Hubs | S1, S2, dpINS, dACC, aINS | vACC, sgACC, dACC, aINS, pCC | aINS, aMCC, IPL, IFG (Mirror Network) | dmPFC, mPFC (Mentalizing Network) |
| MVPA Signature Connectivity | Connects to thalamus / posterior insula | Connects to lateral prefrontal / parietal cortices | High somatosensory desynchronization | High Default Mode Network engagement |
| Recall / "Reliving" Pathways | Recruits IFG (body state processing) | Recruits dmPFC (mental state processing) | N/A | N/A |
Acute Social Isolation and Chronic Loneliness
While acute social rejection triggers an immediate pain-like alarm state, chronic social isolation - experienced subjectively as loneliness - results in profound structural and functional neuroadaptations that fundamentally alter baseline neural processing 5051.
Dopaminergic Shifts and Social Craving
Acute social isolation effectively starves the brain of social reward. Human fMRI studies reveal that acute isolation induces robust activation in the ventral tegmental area (VTA) and the substantia nigra, mimicking the neurological signatures of "craving" observed during fasting from food or during drug withdrawal 51.
However, as acute isolation transitions into chronic loneliness, this adaptive craving mechanism becomes blunted. Chronically lonely individuals exhibit diminished VTA activity and reduced activation in the ventral striatum (the primary reward center) when viewing positive social stimuli, reflecting a maladaptive reduction in the motivation to seek out social connection 50515253.
Idiosyncratic Neural Processing and the Anna Karenina Principle
Simultaneously, chronic loneliness shifts the brain into a state of hypervigilance for social threats. The amygdala and anterior cingulate cortex become hypersensitive to negative social cues, creating a self-perpetuating cycle where lonely individuals anticipate rejection, interpret ambiguous social interactions defensively, and ultimately withdraw further 505152. Structural imaging confirms that chronic loneliness correlates with reduced gray matter volume in the left dorsolateral prefrontal cortex (dlPFC), amygdala, and hippocampus, suggesting accelerated neural aging and impaired top-down emotional regulation 54.
Recent investigations into inter-subject neural synchronization have introduced the "Anna Karenina Principle" of lonely brains. When healthy, non-lonely adults watch naturalistic video stimuli, their fMRI brain processing patterns are highly synchronized, indicating a shared, normative understanding of social reality 5557. Conversely, lonely individuals not only process the world differently from non-lonely individuals, but they also process it differently from each other 5557. This idiosyncratic processing is particularly pronounced within the Default Mode Network and reward systems. The lack of shared neural perspective severely impairs the ability of lonely individuals to establish mutual understanding, exacerbating their isolation regardless of the absolute number of their social contacts 5557.
Developmental and Cross-Cultural Modulators
The neural processing of social pain is not entirely hardwired; it is subject to extensive developmental plasticity and cultural calibration.
Maturation of Executive Control and Aggression Regulation
Social rejection frequently precipitates aggressive behavior, but the capability to modulate this response develops significantly during childhood. Longitudinal fMRI studies using peer-evaluation paradigms demonstrate that behavioral aggression following social rejection decreases as children mature from middle childhood (ages 7-9) to late childhood (ages 9-11) 3256.
This behavioral improvement is mediated by increased activation in the dorsolateral prefrontal cortex (dlPFC), the brain's cognitive control center. Children who exhibit the greatest maturational increases in dlPFC recruitment during negative social feedback demonstrate the most significant reductions in retaliatory aggression 325657. Conversely, individuals with lower executive functioning exhibit a positive association between dACC/insula alarm activation and behavioral aggression, highlighting the importance of top-down executive functioning in buffering the raw signal of the social pain network 57.
Individualism, Collectivism, and Neural Coping Strategies
Cultural neuroscience has established that macro-level societal values significantly modulate the micro-level neural processing of social exclusion. Individuals from individualistic cultures (e.g., Western societies), which emphasize uniqueness, autonomy, and independence, demonstrate significantly higher physiological stress responses and greater negative psychological affect following social exclusion compared to individuals from collectivistic cultures 3258.
Neuroimaging reveals that these cultural differences correspond to dissociable neural pathways. Cross-cultural fMRI studies assessing social evaluation, exclusion, and financial social comparison indicate that collectivistic individuals - whose self-construal is heavily interdependent - rely more heavily on implicit socioemotional regulation regions, such as the ventromedial prefrontal cortex (vmPFC) and medial temporal gyrus 596061. Because the interdependent self is not primarily defined by absolute individual success, collectivist brains exhibit different modulatory patterns in the ventral striatum when processing relative social comparisons 5860.
Conversely, individuals primed with individualistic values recruit explicit, top-down cognitive control regions, specifically the dlPFC, to cope with exclusion 6162. When the individual self is viewed as an autonomous entity, social exclusion represents a more direct threat to self-integrity, necessitating higher-order cognitive regulation to manage the resultant distress 5861.
| Cultural Construct | Self-Construal Focus | Primary Neural Regulation Region | Behavioral Response to Exclusion |
|---|---|---|---|
| Individualism | Autonomy, Independence | dlPFC (Explicit cognitive control) | Higher physiological stress, direct threat to self-integrity |
| Collectivism | Interdependence, Group Harmony | vmPFC (Implicit socioemotional regulation) | Lower physiological stress, buffered self-integrity |
The neuroscience of social pain has evolved from a compelling metaphor into a sophisticated neurobiological framework. Early univariate fMRI studies revolutionized the field by proving that the pain of social rejection shares gross anatomical real estate with physical injury. However, advanced methodologies like Multivariate Pattern Analysis have fundamentally refined this view, revealing that social and physical pain are distinct neural population codes engaging divergent large-scale networks. From the evolutionary origins of mammalian separation distress vocalizations mediated by endogenous opioids, to the profound structural idiosyncrasies found in chronically lonely brains, the social pain system is a highly specialized adaptation. It operates as a biological imperative designed to maintain the interpersonal bonds necessary for human survival, intricately modulated by our cultural environments, developmental stages, and cognitive regulatory capacities.