Neuroscience of grief and complicated grief
Foundations of Bereavement Neuroscience
Grief is a universally experienced, profoundly stressful neurobiological response to the permanent loss of a significant attachment figure. Historically relegated to behavioral sciences and qualitative stage models, bereavement is increasingly understood through the lens of cognitive neuroscience as a fundamental problem of learning, memory updating, and systemic stress adaptation 123. When a deeply encoded social bond is severed by death, the brain must undertake the metabolically and cognitively demanding task of reconciling an intensely wired biological reality - the expectation of the loved one's presence - with the newly acquired semantic knowledge of their permanent absence 134.
For the majority of bereaved individuals, the acute distress following a loss gradually transitions into an integrated form of grief. The intensity of yearning softens, cognitive and emotional resources are reallocated, and the individual recalibrates their internal models to function within a changed environment 5. However, for approximately 5% to 10% of the bereaved population, this neurobiological recalibration stalls 678. The emotional pain does not ease, and the bereaved individual remains in a state of chronic, debilitating yearning. This failure of adaptation is formally recognized in the Diagnostic and Statistical Manual of Mental Disorders (DSM-5-TR) and the International Classification of Diseases (ICD-11) as Prolonged Grief Disorder (PGD) 579.
The formalization of PGD as a distinct clinical entity has accelerated research into the distinct circuit-level differences between normative bereavement processing and pathological mourning. Emerging evidence from neuroimaging, psychoneuroendocrinology, and molecular biology demonstrates that grief operates through specific, overlapping large-scale brain networks - primarily the physical pain matrix, the reward and attachment circuitry, and cognitive control networks 61011.
Physical and Social Pain Matrix Overlap
The human brain is fundamentally wired for social connection, as evolutionary pressures have tightly coupled the mechanisms ensuring mammalian survival with the neurobiology of attachment 41012. Consequently, the brain registers the loss of a vital social bond as a critical threat to survival, activating neural pathways that overlap substantially with those responsible for processing physical injury.
A growing body of literature indicates that the brain processes social rejection, isolation, and bereavement using highly conserved subcortical and neocortical networks. This network, sometimes categorized in evolutionary neuroscience as the PANIC/GRIEF system, involves structures such as the periaqueductal gray (PAG), dorsomedial thalamus, stria terminalis, and septal areas 1012. At the neocortical level, the experience of social pain is mediated primarily by the dorsal anterior cingulate cortex (dACC) and the anterior insula 101213. The dACC serves a dual function, integrating cognitive processing with conflict monitoring, while the anterior insula is deeply involved in interoception - the brain's perception of internal bodily states 12. During acute grief, the concurrent activation of the dACC and anterior insula generates the visceral, affective distress that characterizes the physical "ache" of loss 71415.
The profound overlap between physical and social pain networks is further supported by pharmacological interventions. Clinical trials have demonstrated that administering acetaminophen (a common physical pain reliever) can significantly reduce self-reported feelings of social pain and dampen corresponding neural responses in the dACC and anterior insula over time 14. Furthermore, the sensitivity of this social pain network is modulated by an individual's underlying attachment style. Neuroimaging results indicate that during episodes of social rejection or loss, individuals with highly anxious attachment styles exhibit heightened activity in the dACC and anterior insula, while those with highly avoidant attachment styles display dampened activity in these areas, reflecting a neural disengagement from social signals 1012.
| Brain Region | Primary Function in Grief Processing | Clinical Implication in Bereavement |
|---|---|---|
| Dorsal Anterior Cingulate Cortex (dACC) | Conflict monitoring; processing the distress component of physical and social pain. | Generates the visceral feeling of psychological pain and distress upon realizing the loved one's absence. |
| Anterior Insula | Interoception; mapping internal bodily states to emotional experiences. | Responsible for the somatic manifestations of grief (e.g., the physical "ache" or "heaviness" in the chest). |
| Periaqueductal Gray (PAG) | Autonomic regulation; part of the subcortical PANIC/GRIEF system. | Triggers vocalizations, crying, and the immediate physiological stress response to separation. |
| Nucleus Accumbens (NAc) | Reward processing; incentive motivation; expectation of social reward. | Drives the persistent yearning and craving for the deceased; heavily implicated in Prolonged Grief Disorder. |
Table 1: Key Neural Structures in the Processing of Acute Grief and Social Pain. 6101215
Reward Circuitry and Attachment Neurobiology
While the acute pain of loss is mediated by the dACC and insula, the persistent, repetitive yearning that defines the long-term timeline of grief is driven by the brain's reward and motivation circuitry. Bereavement represents a fundamental disruption to the neural networks that encode attachment and predict reward 479.
Dopaminergic and Opioidergic Pathways
The formation of a deep social bond wires the brain to associate the attachment figure with intense neurochemical rewards. This process is governed by the convergence of dopaminergic, oxytocinergic, and endogenous opioid receptors within the nucleus accumbens (NAc), a primary node of the basal ganglia reward system 616. Dopamine, projecting from the ventral tegmental area (VTA), drives the incentive motivation (the "wanting" or seeking behavior) to pursue the loved one, while endogenous opioids mediate the hedonic pleasure (the "liking") experienced upon reunion and physical proximity 161718.
Animal models have been critical in mapping these receptor pathways. Studies on socially monogamous animals, such as prairie voles and coppery titi monkeys, reveal that the formation and severing of pair bonds involve highly specific dopamine receptor (D1 and D2) and opioid receptor modulations 41920. For instance, Positron Emission Tomography (PET) imaging utilizing highly selective radiotracers (such as [11C]GR103545) has shown that Kappa Opioid Receptors (KOR) play a pivotal role in the stress and anxiety behaviors induced by social separation and isolation 20.
When a loved one dies, the neural circuitry that previously motivated the individual to seek out their partner remains fully intact and highly sensitized. Reminders of the deceased trigger the release of dopamine, which signals the brain to expect the reward of the person's presence 171921. Because the person is permanently gone, the expected reward is never delivered, resulting in a profound prediction error. The brain is essentially driven into a state of chronic craving, highly analogous to the neurobiology of substance withdrawal 212223. Recent high-resolution studies have demonstrated significant overlap between the neuronal populations in the NAc that respond to natural social rewards and those hijacked by addictive substances, further explaining the compulsive yearning seen in complicated grief 21.
The Predictive Coding of Attachment
To contextualize how the brain processes this absence, cognitive neuroscientists have proposed the "Gone-But-Also-Everlasting" theory of grieving 13. According to this neurocognitive predictive coding framework, the brain maintains dual, conflicting models of reality following a profound loss.
On one level, the semantic memory system (supported by the neocortex) rapidly acquires the factual knowledge that the person has died. However, the episodic memory and attachment systems (heavily reliant on the hippocampus, amygdala, and striatal reward networks) contain deeply entrenched, implicitly learned predictions that the loved one is still available and will provide comfort 134. Just as the brain constructs a virtual map of spatial environments, it constructs a virtual map of social attachments.
Grieving, therefore, is an active, neurologically demanding process of learning. The bereaved individual must repeatedly encounter situations where the brain expects the deceased to be present, experience the painful prediction error when they are not, and gradually update the deep-seated attachment map to reflect the new reality 134.
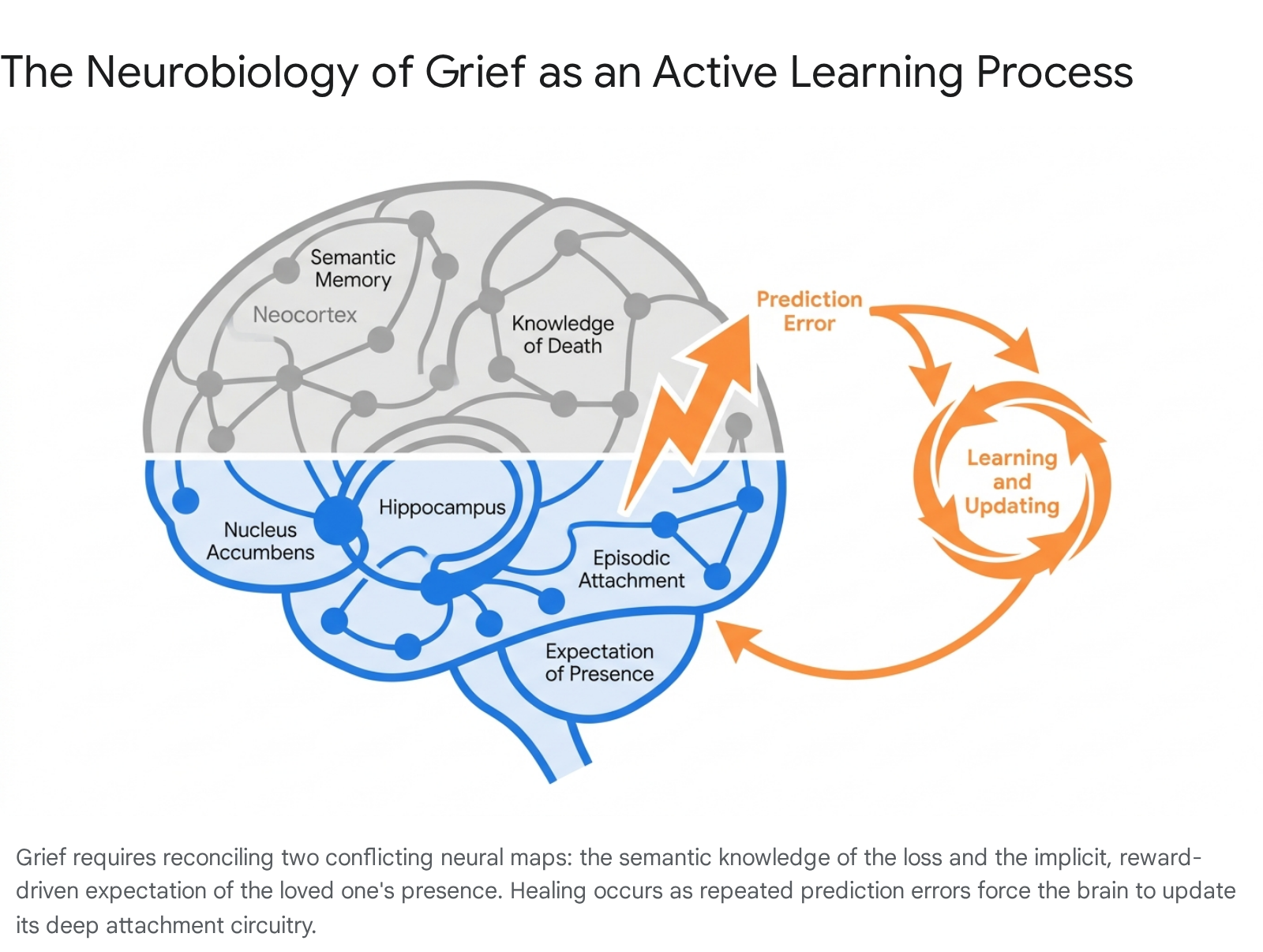
In typical grief, this continuous updating eventually reduces the dopaminergic craving signal, allowing the individual to integrate the loss.
Pathophysiology of Prolonged Grief Disorder
While most individuals successfully update their internal models and integrate the loss, a subset remains trapped in the acute phase of yearning and emotional pain. Prolonged Grief Disorder (PGD) is diagnosed when severe grief symptoms - including identity disruption, intense loneliness, emotional numbness, and persistent preoccupation with the deceased - extend beyond 12 months (or 6 months under some international guidelines) and cause significant functional impairment 792224.
Differentiating Pathological Grief from Depression
Historically, psychiatry struggled to differentiate pathological grief from Major Depressive Disorder (MDD) and Post-Traumatic Stress Disorder (PTSD). However, distinct clinical boundaries and neurobiological signatures have since been identified. While MDD is characterized by pervasive anhedonia, global loss of interest, and generalized sadness, PGD is highly specific to the loss event. The emotional pain in PGD is tethered tightly to the absence of the specific attachment figure 7925. Furthermore, whereas anxiety and PTSD are fundamentally driven by an aversion to threat (hyperactivation of the amygdala and fear circuits), PGD is driven by an appetitive, reward-oriented function - a relentless desire to be reunited with the deceased 5.
Functional Neuroimaging Signatures of PGD
Neuroimaging studies utilizing functional Magnetic Resonance Imaging (fMRI) have revealed that PGD is primarily a disorder of the reward and attachment systems rather than simply an emotional processing error. When exposed to cues related to the deceased (such as photographs or specific words), individuals with PGD exhibit sustained, heightened activation in the nucleus accumbens (NAc) compared to those experiencing normative grief 671822. This hyperactivation indicates that the brains of individuals with PGD continue to process the memory of the deceased as an expected, highly salient reward, trapping them in a cycle of chronic craving that impedes long-term adaptation 6726.
Simultaneously, PGD is characterized by deficits in top-down emotional regulation. In fMRI tasks such as the Emotional Counting Stroop (ecStroop) - where participants must identify the ink color of death-related words versus neutral words, thereby testing their ability to disengage attention from emotionally distressing stimuli - individuals with non-complicated grief demonstrate increased recruitment of the rostral anterior cingulate cortex (rACC) and orbitofrontal cortex (OFC) 62728. The rACC is heavily implicated in emotion regulation and conflict resolution. In contrast, individuals with PGD fail to recruit the rACC effectively when confronting grief-related stimuli, indicating a profound inability to disengage from emotional distress and regulate their psychological response 182729.
Additionally, PGD is associated with altered activity in the amygdala. While both normative grievers and individuals with PGD may show amygdala activation in response to loss cues, individuals with PGD display heightened reactivity specifically to death-related stimuli, which correlates directly with the intensity of self-reported sadness and an inability to process positive emotional stimuli 3031.
| Functional Metric | Normative / Integrated Grief | Prolonged Grief Disorder (PGD) | Major Depressive Disorder (MDD) |
|---|---|---|---|
| Nucleus Accumbens (Reward) | Initial activation that recalibrates as the brain learns the reality of absence. | Sustained hyperactivation upon exposure to loss cues, indicating chronic craving. | Blunted activation, reflecting broad deficits in anticipating or enjoying rewards (anhedonia). |
| rACC / OFC (Emotion Regulation) | Increased recruitment during exposure to loss reminders, indicating successful emotional regulation. | Diminished or absent recruitment, reflecting a failure of top-down emotional control. | Variable; often hypoactivation related to generalized emotional dysregulation. |
| Amygdala (Salience/Threat) | Transient activation correlated with acute distress, subsiding over time. | Heightened, sustained reactivity specifically to death-related stimuli. | Hyperactivity generally correlated with widespread negative affect and anxiety. |
Table 2: Distinct Neural Signatures of Normative Grief, PGD, and MDD based on fMRI findings. 6718222730
Desynchronization of Large-Scale Brain Networks
Beyond isolated regions, the pathophysiology of PGD involves the desynchronization of large-scale functional brain networks. Resting-state functional connectivity studies emphasize the roles of the Default Mode Network (DMN), the Salience Network (SN), and the Executive Control Network (ECN) 113233.
The DMN is active during internally directed cognition, such as autobiographical memory retrieval, self-referential thought, and prospection. The SN, anchored by the anterior insula and dACC, continuously monitors the environment and internal states for behaviorally relevant cues 33. The ECN governs working memory and directed attention. In a healthy brain, the SN acts as a switch, modulating the dynamic equilibrium between the internal focus of the DMN and the external focus of the ECN 3334.
In PGD, individuals frequently exhibit increased functional connectivity within the DMN, correlating with high levels of rumination, intrusive memories of the deceased, and an inability to project a meaningful future self without the lost partner 235. Furthermore, studies have identified aberrant functional connectivity between the amygdala, the DMN, and the SN 3435. Specifically, increased baseline amygdala functional connectivity with the dorsal frontal executive control and salience networks has been shown to predict worsening grief symptoms over time. This reflects an entrenched neural pattern where affective distress consistently hijacks cognitive control, preventing the individual from engaging with restorative environmental stimuli 3435.
Neuroendocrine and Immunological Correlates
The psychological distress of acute and prolonged grief triggers profound cascades within the body's physiological stress systems, extending the neurobiology of grief far beyond the central nervous system.
HPA Axis Dysregulation
The perception of loss activates the hypothalamic-pituitary-adrenal (HPA) axis. The hypothalamus secretes corticotropin-releasing hormone (CRH), stimulating the pituitary to release adrenocorticotropic hormone (ACTH), which prompts the adrenal glands to secrete cortisol 1624. In normal acute stress, this response mobilizes the organism. However, the permanent absence of the attachment figure means the stress response cannot be deactivated through reunion, leading to chronic HPA axis dysregulation 123.
In Prolonged Grief Disorder, cortisol rhythms are markedly altered. Systematic reviews note that individuals with PGD often display flattened diurnal cortisol slopes and lower morning cortisol levels compared to healthy controls, indicative of severe exhaustion of the HPA axis following prolonged hyperarousal 162236. This physiological state contributes to the severe fatigue, disrupted circadian rhythms, and sleep fragmentation commonly reported as "grief brain fog" 37.
Immunological Impairment and Epigenetic Shifts
Bereavement has been consistently associated with systemic immune system dysregulation. Neuro-psycho-endocrine studies show that bereaved individuals, particularly those with PGD, exhibit elevated levels of pro-inflammatory cytokines, including interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), and interferon-gamma (IFN-γ), alongside a reduction in natural killer (NK) cell activity 162437. This chronic inflammatory state essentially impairs the body's ability to combat infections and accelerates cellular aging. It provides a distinct biological mechanism for the increased cardiovascular morbidity and mortality rates observed in the initial months following the death of a spouse - a phenomenon historically termed the "widowhood effect" 373839.
Furthermore, recent reviews have highlighted epigenetic modifications of stress-related genes in grieving individuals 23. These modifications alter how the brain and body respond to future stressors, suggesting that severe grief does not merely produce transient hormonal fluctuations but structurally reprograms the organism's baseline stress reactivity and immune functioning 3.
Peripheral biomarkers also show distinct patterns regarding oxytocin. A neuropeptide associated with social bonding and trust, oxytocin levels might be expected to drop following the loss of a loved one. However, studies investigating peripheral bloodstream oxytocin have found that individuals with a primary diagnosis of PGD demonstrate significantly higher oxytocin levels than those with Major Depressive Disorder or non-bereaved controls 61636. This elevation is theorized to reflect a biological persistence of the attachment system continually attempting to signal and seek out the lost figure 616.
Structural Brain Alterations
The chronic stress, elevated inflammatory markers, and severe neuroendocrine disruption inherent in prolonged bereavement do not merely alter functional connectivity; they precipitate measurable structural changes within the brain. Long-term structural MRI studies demonstrate that the intense, unrelenting stress of PGD can lead to neurotoxicity and volume reduction in key limbic and cortical structures 1140.
Hippocampal and Amygdala Atrophy
The hippocampus, a critical hub for episodic memory consolidation and the regulation of the HPA axis, is highly dense with glucocorticoid receptors. Chronic elevation of cortisol - a hallmark of early severe grief - can exert neurotoxic effects on hippocampal neurons, leading to dendritic retraction and inhibited neurogenesis 311. Structural MRI studies have consistently revealed significant hippocampal atrophy in bereaved individuals suffering from prolonged grief, particularly among those who have experienced the traumatic loss of a child or older adults who have lost a spouse 1140.
This hippocampal volume loss directly correlates with the memory impairments and cognitive difficulties frequently reported by those with PGD. Additionally, volumetric reductions are observed in the amygdala 1141. The structural remodeling of the amygdala and hippocampus deeply impairs the brain's ability to extinguish the fear and distress responses associated with the loss, perpetuating the cycle of pathological mourning.
Cortical Thinning and White Matter Abnormalities
Grief's structural impact extends to the neocortex and white matter tracts. For example, targeted neuroimaging studies of grieving Chinese "Shidu" parents - parents who have lost their only child and are past reproductive age - identified significant reductions in gray matter volume in the left supramarginal gyrus and the right amygdala 1142. The atrophy in the supramarginal gyrus was found to directly mediate the association between intense grief severity and impaired cognitive inhibition 11.
Furthermore, diffusion tensor imaging (DTI) studies have noted white matter abnormalities in individuals with prolonged grief, indicating compromised structural integrity of the axonal tracts connecting emotion regulation centers (like the prefrontal cortex) to deeper limbic structures 11. Collectively, these structural alterations explain why PGD is closely correlated with broader public health risks, including accelerated cognitive decline, higher susceptibility to neurodegenerative diseases in later life, and cardiovascular comorbidities 1140.
Theoretical Models in Grief Processing
The advancing understanding of grief at the circuit level has forced a reevaluation of traditional psychological models. While popular culture heavily relies on Elisabeth Kübler-Ross's "Five Stages of Grief" (denial, anger, bargaining, depression, acceptance), modern clinical psychology and neuroscience consider this model empirically unsupported, overly rigid, and potentially harmful if it imposes prescriptive, linear expectations on the bereaved 374344. Instead, contemporary neuroscience strongly supports dynamic, non-linear models of grief processing that account for biological oscillation and cognitive coping.
The Dual Process Model of Coping
The most empirically supported framework in contemporary bereavement research is the Dual Process Model (DPM) of Coping with Bereavement, developed by Stroebe and Schut 184546. The DPM posits that adaptive grieving requires the individual to oscillate continuously between two states: loss-oriented coping (focusing on the emotional pain of the loss, yearning, and processing episodic memories) and restoration-oriented coping (focusing on secondary stressors, adapting to new life roles, forming new identities, and engaging with the external world) 374647.
Neuroimaging has provided a biological substrate for this behavioral oscillation. Loss-oriented coping maps closely onto the activation of the Default Mode Network (internal rumination, autobiographical memory retrieval) and the reward/salience networks (yearning, recognizing absence) 3347. Restoration-oriented coping maps onto the Executive Control Network, allowing the individual to suppress intrusive emotional distress temporarily to complete necessary daily tasks. PGD, through the lens of the DPM, represents a fundamental failure of oscillation. The individual becomes rigidly trapped in loss-oriented processing (hyperactive DMN and NAc), unable to recruit the ECN and rACC necessary to disengage and shift toward restoration 24748.
The Approach-Avoidance Processing Hypothesis
Building upon the DPM, clinical researchers such as Paul Boelen and Maarten Eisma have advanced an "approach-avoidance" framework for understanding complicated grief. This model observes that PGD is characterized by a paradoxical combination of behaviors: the mourner exhibits profound approach behaviors toward the deceased (persistent yearning, lingering attachment, and seeking out physical or psychological reminders) while simultaneously demonstrating intense avoidance of the reality of the loss and the difficult emotions associated with it (experiential avoidance) 225049.
Neurobiologically, this manifests as an intense conflict between distinct circuits. The approach behavior is driven by the dopaminergic and oxytocinergic reward signaling in the nucleus accumbens, which continues to code the deceased as an indispensable survival figure 192249. Concurrently, the avoidance behavior - often driven by the fear of profound emotional pain - recruits the amygdala and insular threat networks 6.
A recent study utilizing a novel free-viewing attention task tested this hypothesis. Bereaved participants were shown pictures of the deceased alongside combinations of loss-reality words. The study found that individuals with high lingering attachment and severe PGD symptoms exhibited a strong attentional bias toward pictures of the deceased (approach behavior), yet showed no such engagement with loss-reality reminders (avoidance behavior) 5049. This confirms that higher prolonged grief symptom levels are characterized by persisting, unbalanced approach tendencies toward the deceased, maintaining the neurobiological conflict.
Methodological Limitations and Cross-Cultural Research
Despite significant advances over the last two decades, the neuroscience of grief remains constrained by substantial methodological limitations that require cautious interpretation of current data.
Neuroimaging Limitations and Statistical Artifacts
The bulk of our understanding regarding the neurobiology of grief comes from functional neuroimaging studies utilizing tasks such as the Emotional Counting Stroop, the Grief Elicitation task (viewing photos of the deceased), or resting-state functional connectivity scans 82750. However, neuroimaging experts frequently note that these studies are hampered by extremely small sample sizes, heterogeneous clinical definitions prior to the formal DSM-5-TR PGD criteria, and cross-sectional designs 8242851.
Furthermore, the statistical methods used in fMRI analyses, particularly parametric statistical tests on clustered voxel data, have historically been prone to high false-positive rates 52. Because the neural circuits for reward, emotional regulation, and social pain overlap significantly with those for major depression and generalized anxiety, parsing out the unique neurobiological signature of PGD versus general affective distress remains a complex bioinformatic challenge 2651. To advance the field, researchers are advocating for longitudinal, multi-modal designs (integrating fMRI, PET receptor mapping, and psychoneuroimmunological blood panels) that track bereaved individuals over time to observe the exact neural changes as acute grief either integrates or becomes prolonged 112840.
The WEIRD Demographic Bias
A critical limitation in the current neuroscience of grief is the overwhelming reliance on data from Western, Educated, Industrialized, Rich, and Democratic (WEIRD) populations 535455. The vast majority of fMRI grief literature originates from North America and Western Europe, where grief is largely conceptualized as a private, individual psychological process centered on emotional closure and independence 535657.
In contrast, non-Western and collectivistic cultures often frame bereavement as a communal journey, heavily mediated by shared social rituals, ancestor veneration, and differing spiritual conceptualizations of the boundary between life and death 5658. The expression of grief, and theoretically its neural processing, is subject to intense cultural shaping.
Recent cross-cultural investigations are beginning to address this gap. For example, a study validating a Yearning Induction Task (YIT) among bereaved individuals from Switzerland and China found significant cultural divergence in emotional responses following the induction. Swiss participants reported declines in positive emotions and increases in sadness and anger, whereas Chinese participants experienced distinct shifts related to happiness and negative emotional regulation that did not align perfectly with Western norms 59.
Similarly, neuroimaging of Chinese Shidu parents reveals unique socio-cultural pressures - such as stigma, social exclusion, and cultural beliefs about the failure of lineage continuity - that map onto specific patterns of cortical atrophy and depressive symptomology not uniformly observed in Western cohorts 1156. Meta-analyses of Chinese populations indicate that PGD prevalence is exceptionally high following specific demographic tragedies, reaching up to 22.7% among those who lost an only child and 80.4% among earthquake survivors 6061. Establishing a universally applicable neurobiological definition of pathological grief will require equitable research infrastructure and expansive neuroimaging data from low- and middle-income countries 5357.
Conclusion
The neuroscience of grief fundamentally reframes bereavement from a passive period of emotional suffering to a highly active, neurobiologically demanding period of learning and systemic adaptation. Following a loss, the brain must undertake the monumental task of updating deep-seated predictive models encoded within the reward, attachment, and pain networks. It must reconcile the episodic expectation of the loved one's presence with the stark semantic reality of their death, navigating a barrage of neuroendocrine stressors and inflammatory responses in the process.
For those who develop Prolonged Grief Disorder, this learning process is neurologically arrested. The sustained hyperactivation of the nucleus accumbens traps the individual in a state of chronic craving, while diminished regulatory control from the anterior cingulate cortex leaves them unable to modulate their intense emotional pain. Concurrently, chronic cortisol elevation drives neurotoxic structural changes in the hippocampus and amygdala. By moving beyond rigid stage models and understanding grief at the circuit level, science is mapping the precise biological mechanisms that cause a specific subset of mourners to become dangerously stuck. Ultimately, these neurobiological insights hold the key to developing highly targeted psychotherapeutic and pharmacological interventions capable of restoring the brain's capacity to learn, adapt, and eventually find life meaningful after devastating loss.