Neuroscience of classic psychedelics
The study of serotonergic psychedelics has transitioned from the peripheries of psychopharmacology to the forefront of translational neuroscience. Following a decades-long regulatory moratorium, the biomedical evaluation of classic psychedelics - namely psilocybin, lysergic acid diethylamide (LSD), N,N-dimethyltryptamine (DMT), and mescaline - has resumed, driven by clinical trials demonstrating robust efficacy in treating major depressive disorder, post-traumatic stress disorder, and substance use disorders 12. Beyond clinical outcomes, neuroscientific inquiry focuses on the mechanisms through which these molecules systematically alter hierarchical brain dynamics, induce neuroplasticity, and reshape subjective consciousness.
Recent multi-modal investigations combining cellular electrophysiology, wide-field optical imaging, and high-resolution functional magnetic resonance imaging (fMRI) have fundamentally revised historical models of psychedelic action 34. The contemporary consensus indicates that psychedelics are not exclusively cell-surface serotonin 2A (5-HT2A) receptor agonists. Rather, they act as complex neuromodulators targeting intracellular receptor pools, driving enduring functional electrophysiological plasticity that outlasts structural synaptic growth, and inducing profound state-changes in global network connectivity 456. This report comprehensively analyzes the molecular, cellular, and systems-level neuroscience of classical psychedelics, while addressing novel methodological confounders related to neurovascular coupling.
Receptor Pharmacology and Binding Affinities
The foundational hypothesis of psychedelic neuroscience posits that acute subjective effects are primarily mediated by agonism or partial agonism at the 5-HT2A receptor 789. This G protein-coupled receptor (GPCR) is densely expressed on the apical dendrites of Layer V pyramidal neurons in the prefrontal cortex, posterior cingulate cortex, and claustrum 710. The necessity of 5-HT2A activation is well-documented; pretreatment with the highly selective 5-HT2A antagonist ketanserin reliably abolishes the core subjective effects of psychedelics in human subjects and blocks the head-twitch response in murine models 1112. However, focusing exclusively on 5-HT2A obscures the complex polypharmacology of these substances.
Comprehensive GPCR and Kinase Profiling
Psychedelics exhibit broad and highly variable binding profiles. A 2025 systematic pharmacological study profiled 41 classical psychedelics against a massive panel of 318 human GPCRs and over 450 human kinases 1314. The data revealed that classical psychedelics act potently across an unexpectedly wide array of serotonergic, dopaminergic, and adrenergic receptor subtypes 1314.
Ergolines, such as LSD, display highly promiscuous receptor binding. LSD interacts with high affinity not only at 5-HT1A, 5-HT2C, and 5-HT7 receptors but also at dopamine D1 through D4 receptors and alpha-2 adrenergic sites 1516. Tryptamines, including DMT and psilocin (the pharmacologically active metabolite of the prodrug psilocybin), exhibit a narrower pharmacological spectrum, primarily targeting serotonergic networks and trace amine-associated receptors 1516. Phenethylamines, such as mescaline, are highly selective for 5-HT2A and 5-HT2C receptors with negligible dopaminergic affinity 1516.
Table 1: Comparative Binding Affinities ($K_i$ in nM) of Classic Psychedelics Across Receptor Subtypes
| Receptor Target | Psilocybin | Psilocin | LSD | DMT | 5-MeO-DMT |
|---|---|---|---|---|---|
| 5-HT1A | >10,000 | 567.4 | 1.1 | 119.5 | 60 - 200* |
| 5-HT2A | >10,000 | 107.2 | 3.5 | 75.3 | 200* |
| 5-HT2C | >10,000 | 97.3 | 23.6 | ~100 | ~100* |
| Dopamine D1 | >10,000 | >10,000 | 27.0 | >10,000 | >10,000 |
| Alpha-1 Adrenergic | >10,000 | >10,000 | 670* | >10,000 | >10,000 |
Data derived from multi-receptor binding assays. Lower $K_i$ values denote stronger binding affinity. The table highlights the pharmacological inertness of psilocybin prior to hepatic dephosphorylation into psilocin, and the exceptionally high, pan-receptor potency of LSD 9151617. Asterisks denote values aggregated from specific animal models or varied radioligand contexts.
Dose Equivalence and Physiological Divergence
The diverse binding profiles of these compounds result in distinct physiological trajectories, despite generating remarkably similar states of altered consciousness. Controlled head-to-head clinical trials conducted by the Liechti Lab at the University Hospital Basel compared psychoactive-equivalent doses of LSD (100 - 200 μg), psilocybin (15 - 30 mg), and mescaline (300 - 500 mg) 1819.
Phenomenological assessments, including the Mystical Experience Questionnaire and ego dissolution inventories, indicated that the acute subjective experiences produced by these three distinct chemical classes are virtually indistinguishable at equivalent doses 1819. However, systemic physiological effects diverged in a manner consistent with their extended receptor profiles. LSD induced sustained increases in heart rate and circulating oxytocin, reflecting its dopaminergic and adrenergic agonism 1819. Conversely, psilocybin drove more pronounced elevations in diastolic blood pressure without significant alterations in oxytocin 1819. Effect duration also scaled precisely with pharmacokinetics: psilocybin effects dissipated within 4.9 hours, LSD averaged 8.2 hours, and mescaline persisted for 11.1 hours due to prolonged plasma elimination half-lives 19.
Intracellular Receptors and Molecular Signaling
Pharmacological models have historically presumed that classical psychedelics act upon receptors located on the external surface of the cellular membrane. Recent investigations into molecular localization have fundamentally overturned this assumption.
Lipophilicity and Membrane Permeability
Serotonin is a highly polar, hydrophilic molecule that cannot cross the neuronal lipid bilayer, strictly confining its action to cell-surface receptors 5. In contrast, classical psychedelics are highly lipophilic molecules. This chemical property allows them to passively diffuse across neuronal membranes and engage a distinct, intracellular pool of 5-HT2A receptors located on the endoplasmic reticulum and Golgi apparatus 516.
Activation of these intracellular populations triggers entirely different signal transduction cascades than surface activation. Intracellular 5-HT2A receptors associate with cytoskeletal microtubule-associated proteins (such as MAP1A), positioning them to directly modulate neuronal cytoarchitecture and dendritic reorganization 5. This spatial selectivity represents a critical mechanism for neuroplasticity. Membrane-impermeable serotonin analogs do not trigger cortical growth, whereas membrane-permeable psychedelics do, explaining why conventional selective serotonin reuptake inhibitors (SSRIs) fail to produce the rapid psychoplastogenic effects observed with psilocybin and DMT 516.
Neuroplasticity and Cellular Alterations
The therapeutic durability of psychedelics is widely attributed to their capacity to induce rapid neural plasticity in brain regions damaged by chronic stress, trauma, and depression. Initially, this plasticity was characterized strictly in morphological terms.
Acute Structural Plasticity: Dendritogenesis and Spinogenesis
Preclinical studies demonstrate that single exposures to classical psychedelics (including LSD, DMT, and DOI) induce rapid structural neuroplasticity. Within 24 hours of administration, cultured cortical neurons exhibit significantly increased dendritic arborization, elevated spine density, and larger spine head sizes 122021.
This structural growth is mediated by complex intracellular signaling networks rather than the 5-HT2A receptor acting in isolation. Psychedelic agonism stimulates the extracellular release of Brain-Derived Neurotrophic Factor (BDNF) 2022. The subsequent binding of BDNF to the tropomyosin receptor kinase B (TrkB) receptor activates the mammalian target of rapamycin (mTOR) signaling pathway, which synthesizes the proteins necessary for new synapse formation 1223. The necessity of this cascade has been empirically validated: pretreatment with rapamycin (an mTOR inhibitor), ANA-12 (a TrkB antagonist), or ketanserin completely aborts psychedelic-induced neuritogenesis 1221. Notably, psychedelics may also bind allosterically to the TrkB receptor, stabilizing it to enhance the efficacy of endogenous BDNF 7.
The Shift to Enduring Functional Plasticity
While structural plasticity provides an attractive mechanism for rapid antidepressant effects, the physical longevity of these new synapses has been questioned. In a paradigm-shifting 2025 study published in Neuropsychopharmacology, Kramer et al. evaluated the long-term cellular architecture of rodent brains months after a single administration of psilocybin 424.
The researchers discovered that the initial surge in structural plasticity is highly transient. By the three-month mark, dendritic spine density, synaptic architecture, and local gene expression in the medial prefrontal cortex had entirely reverted to baseline control levels 42425. Despite the disappearance of the physical scaffolding, the behavioral antidepressant effects (measured via the forced swim test) remained robustly intact 424.
Subsequent whole-cell patch-clamp electrophysiology revealed the actual mechanism sustaining these behavioral changes: enduring functional plasticity. Layer V excitatory pyramidal neurons exhibited permanent, significant alterations in their resting membrane potentials, baseline firing rates, and overall synaptic excitability 42425. This indicates that psychedelics achieve their months-long therapeutic effects not by maintaining new physical connections, but by rewriting the long-term electrophysiological transmission rules of the existing neural circuitry 425.
Macroscopic Brain Network Dynamics
At the systems level, normal waking consciousness is maintained by distinct, modular neural networks operating with high internal synchrony. Psychedelics temporarily abolish this modularity, inducing a state of global neural desegregation.
The Global Meta-Analysis of Functional Connectivity
In 2024 and 2026, researchers published the results of an international consortium study in Nature Medicine, constituting the largest mega-analysis of psychedelic functional neuroimaging ever conducted. By aggregating 11 independent datasets containing over 500 brain imaging sessions from 267 participants across five countries, the researchers established a definitive "neural fingerprint" common to psilocybin, LSD, mescaline, DMT, and ayahuasca 1226.
The meta-analysis isolated two universal shifts in resting-state functional connectivity across all tested compounds: 1. Intra-network Desegregation: The synchronized, highly structured communication within canonical brain networks systematically breaks down, rendering individual networks less rigid and less cohesive 16. 2. Inter-network Hyperconnectivity: Concurrently, global cross-talk radically increases between networks that normally operate independently. The most pronounced connectivity increases occurred between high-level executive systems (the frontoparietal and default mode networks) and lower-level sensory regions (the visual and somatomotor networks) 226.
This functional flattening of the brain's hierarchy enables raw sensory inputs to interface directly with higher-order cognitive processing centers, providing a direct biological correlate for the synesthesia, geometric hallucinations, and novel cognitive insights reported during acute psychedelic states 226.
Reevaluating the Default Mode Network (DMN)
Historically, psychedelic neuroimaging focused heavily on the Default Mode Network (DMN), a distributed circuit including the medial prefrontal cortex and posterior cingulate cortex that manages self-referential thought and autobiographical memory 272829. Early correlations between DMN disintegration and the subjective experience of "ego dissolution" led to widespread claims that the DMN was the biological seat of the ego, and that psychedelics functioned simply by disabling this network 282930.
Current neuroscience regards this narrative as a severe oversimplification. Recent analyses highlight that numerous non-psychedelic substances, including amphetamines and alcohol, significantly decrease DMN connectivity; however, these substances frequently increase egotistical behavior, directly contradicting the DMN-ego hypothesis 31. Furthermore, the DMN is critically involved in abstract social cognition and empathy. The suppression of the DMN under psychedelics is now understood not merely as the deletion of the "self," but as a blurring of the computational boundaries between the self and the external environment, facilitating the profound sense of interpersonal and ecological connectedness reported in clinical trials 2731.
Methodological Confounders: The Neurovascular Disconnect
The foundation of human psychedelic neuroimaging relies heavily on Blood Oxygen Level-Dependent (BOLD) fMRI. BOLD signals capture changes in blood flow and oxygenation, relying on the assumption of neurovascular coupling - the principle that local vascular dilation perfectly mirrors local neuronal firing 332. A critical 2025 study in Nature Neuroscience by Padawer-Curry et al. revealed that classical psychedelics systematically violate this assumption.
Because serotonin receptors are located not only on neurons but also on vascular smooth muscle cells and astrocytes, psychedelics exert direct, independent control over cerebral vasculature 3. Using wide-field optical imaging alongside 5-HT2A agonists in murine models, researchers demonstrated that psychedelics completely decouple neuronal activity from hemodynamic activity. The drug induces significant vasodilation or vasoconstriction depending on the brain region, regardless of whether the underlying neurons are firing 33334.
Consequently, the sweeping functional connectivity changes mapped over the past decade via fMRI are partially confounded by the drug's action on blood vessels 3236. While fMRI remains a vital tool, researchers must now apply advanced calibrated models to separate true neural desynchronization from chemically induced vascular artifacts, particularly when assessing DMN connectivity 336.
Theoretical Models of Brain Action
To synthesize molecular pharmacology with macroscopic network alterations, computational neuroscientists have advanced three dominant, competing circuit-level models of psychedelic action.
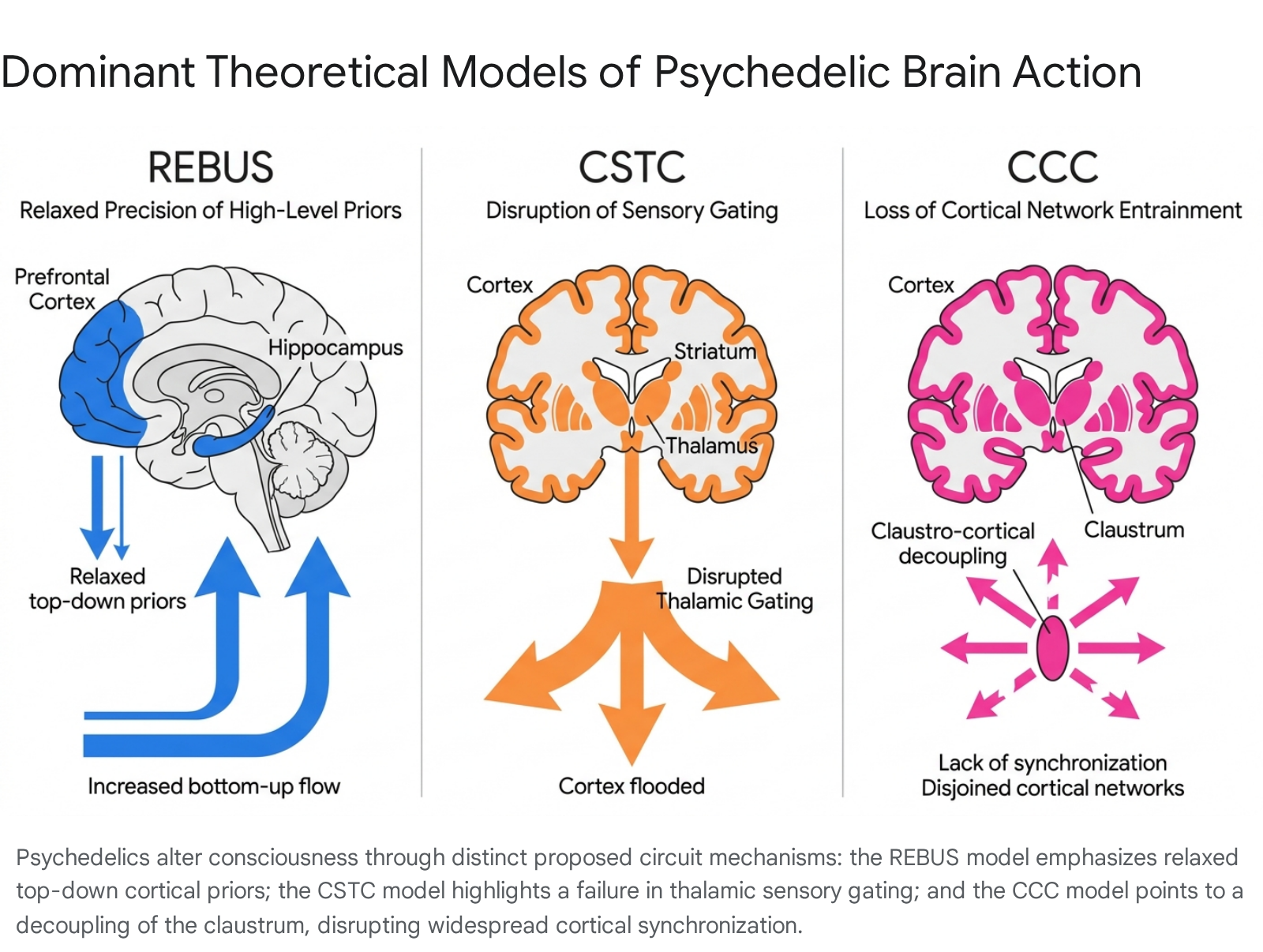
Relaxed Beliefs Under Psychedelics (REBUS)
The REBUS framework, developed by Carhart-Harris and Friston, interprets psychedelic action through the lens of hierarchical predictive coding and the Free-Energy Principle 83536. The brain operates as an inference engine, continuously generating top-down expectations ("priors") to interpret incoming, bottom-up sensory data 37. In psychiatric conditions such as depression or addiction, these high-level priors become pathologically rigid and overweighted, severely restricting cognitive flexibility 836.
REBUS asserts that 5-HT2A agonism flattens this hierarchical architecture by drastically reducing the "precision weighting" (confidence) of high-level priors 837. This relaxation increases overall brain entropy, permitting unfiltered bottom-up sensory and limbic information to flood the cortex. A 2025 causal evaluation of the REBUS model in mice performing perceptual decision-making tasks confirmed these computational mechanics 40. Under the influence of psilocybin, cellular-resolution calcium imaging of the orbitofrontal cortex revealed a reduction in neuronal population correlations - the precise signature of weakened top-down priors - forcing the animals to rely heavily on bottom-up sensory evidence rather than established behavioral routines 40.
The Cortico-Striato-Thalamo-Cortical (CSTC) Loops
The CSTC model represents a more strictly structural approach, positing that sensory gating is governed by feedback loops connecting the cortex, striatum, and thalamus 103538. The thalamus normally acts as an informational filter, restricting the volume of raw sensory data reaching consciousness. The CSTC model argues that psychedelic activation of the medial prefrontal cortex heavily modulates striatal output, effectively releasing the inhibitory brakes on the thalamus 353940. This disinhibition inundates the sensory cortices with unprocessed stimuli, directly driving visual distortions and sensorimotor alterations 3538.
The Cortico-Claustro-Cortical (CCC) Circuit
The CCC framework centralizes the role of the claustrum, a subcortical sheet of gray matter exhibiting an exceptionally high density of 5-HT2A receptors 103538. The claustrum acts as a global pacemaker, entraining distinct cortical regions into highly synchronized resting-state networks 104445.
The CCC model proposes that psychedelics aggressively decouple the claustrum from the prefrontal cortex 3544. Without claustral entrainment, canonical networks immediately desynchronize. In late 2025, dynamic causal modeling of resting-state fMRI provided the first in vivo confirmation that psilocybin disrupts claustral effective connectivity 45. Concurrently, electrophysiological studies on murine models revealed that 5-HT2A agonists reverse synaptic plasticity rules in claustro-cortical circuits - flipping baseline long-term depression (LTD) to long-term potentiation (LTP) - providing a direct synaptic mechanism for the disruption of global network states 41.
Dimethyltryptamine and Endogenous Neuroprotection
While psilocybin and LSD dominate therapeutic discourse, N,N-dimethyltryptamine (DMT) functions through unique neurobiological channels. DMT is the only classic psychedelic endogenously synthesized in the mammalian central nervous system, where it exists at concentrations comparable to serotonin and dopamine 4243.
The Sigma-1 Receptor Interface
DMT possesses a dual pharmacological identity. In addition to 5-HT2A agonism, DMT functions as an endogenous ligand for the Sigma-1 receptor 4243. Sigma-1 is an intracellular chaperone protein located at the mitochondria-associated endoplasmic reticulum membrane, where it regulates cellular survival, oxidative stress, and the misfolding of proteins 4243.
Stroke Models and Neuroimmune Regulation
Recent data indicate that DMT's interaction with the Sigma-1 receptor imparts powerful, non-hallucinogenic neuroprotective properties. A 2025 study by the HUN-REN Biological Research Centre utilized transient middle cerebral artery occlusion (ischemic stroke) in rat models to test DMT's neurorestorative capacity 4445.
Administration of DMT resulted in significant reductions in both stroke infarct volume and cerebral edema 4445. Crucially, DMT restored tight junction integrity, physically repairing the damaged blood-brain barrier 4445. Molecular analyses confirmed that these benefits were driven via the Sigma-1 receptor, which suppressed the release of pro-inflammatory cytokines and heavily restricted the activation of destructive microglial cells 4445. These findings elevate endogenous DMT from a mere hallucinogen to a potent neuroimmune regulator with substantial adjuvant potential for acute neurological injury 4244.
Global Clinical Infrastructure and Ritual Context
The acceleration of psychedelic neuroscience has been heavily supported by global research ecosystems, particularly in South America, where the legal status of the DMT-containing brew ayahuasca has facilitated robust clinical investigation 4647. Research institutions in Brazil, notably the Brain Institute at the Federal University of Rio Grande do Norte, have pioneered trials mapping ayahuasca's rapid antidepressant effects and its modulation of inflammatory biomarkers 474849.
In January 2026, researchers published a landmark randomized controlled trial evaluating ayahuasca in 429 participants - the largest psychedelic clinical trial to date 46. The trial utilized a unique methodology to unbundle the pharmacological effects of the psychedelic from the psychological impact of the group ritual setting 46. The data revealed that participation in the ritual alone (via placebo) occasionally induced a "mystical-type experience," which correlated with mild improvements in well-being 46. However, the administration of the active DMT compound within the ritual setting maximized the duration and magnitude of psychological relief, confirming that the optimal therapeutic outcome relies on the synergistic interaction between intense neuropharmacological disruption and highly structured psychosocial contexts 46.
Conclusion
The neuroscience of classic psychedelics has evolved dramatically from mid-century paradigms that viewed these compounds merely as simple surface-level serotonin agonists or psychotomimetics. Contemporary research outlines a highly sophisticated class of neuromodulators that interface with the brain across every biological scale. From their passive diffusion across lipid bilayers to activate intracellular receptors, to their induction of enduring electrophysiological plasticity that outlasts physical synaptic growth, psychedelics act as profound systemic disruptors. As complex network theories like REBUS and CCC are refined by new data addressing neurovascular confounders and massive receptor profiling, classical psychedelics stand not only as breakthrough interventions for psychiatric illness but as indispensable molecular probes for decoding the architecture of human consciousness.