Neuroscience of Addiction Recovery and Relapse
Theoretical Frameworks of Addiction
The scientific conceptualization of addiction has undergone a profound evolution over the past several decades, transitioning from archaic models of moral failing to rigorous neurobiological frameworks. It is universally recognized that repeated exposure to addictive substances or behaviors induces enduring neuroplastic changes within the central nervous system. These adaptations compromise associative learning, heighten physiological reactivity to drug-related cues, impair emotional regulation, and severely weaken top-down executive control 1. However, within contemporary neuroscience, a significant theoretical debate exists regarding the precise classification and interpretation of these neural adaptations.
The predominant paradigm in modern addiction psychiatry is the Brain Disease Model of Addiction (BDMA). Championed by prominent neuroscientists and institutions such as the National Institute on Drug Abuse (NIDA), the BDMA posits that addiction is a chronic, relapsing brain disease 2. According to this model, addictive substances essentially hijack the brain's natural reward circuitry, causing pathological tissue damage, structural loss in prefrontal cortical regions, and functional impairments 345. Proponents of the BDMA argue that these drug-induced alterations physically impair response inhibition and generate an insidious compulsion that justifies the classification of addiction as a neurological disease, thus validating medical intervention and reducing the stigma associated with the condition 245.
Conversely, the developmental-learning or "deep habit" model, articulated by researchers within the Addiction Theory Network, argues that the brain changes observed in addiction are not pathological malfunctions or disease states 26. In this view, addiction represents the expected outcome of the brain's normal learning mechanisms operating under conditions of extreme motivational salience 8. This model suggests that addiction is a form of deep learning and habit formation driven by highly potent rewards, resulting in neuroplastic changes akin to those seen in normative compulsive behaviors, intense athletic training, or romantic attachment 3. From this perspective, the brain is not "broken" or diseased; rather, it has efficiently and predictably adapted to the environmental input of intense reward.
Regardless of the semantic classification, the physiological reality of addiction is often understood through a widely accepted three-stage neurobiological cycle: binge and intoxication, withdrawal and negative affect, and preoccupation and anticipation 578910. The initial phase is driven by positive reinforcement, where the individual consumes the substance for its euphoric effects. Over time, neuroadaptation leads to tolerance, and the individual enters the withdrawal and negative affect stage, characterized by profound dysphoria and anhedonia. At this juncture, the motivation for substance use shifts from positive reinforcement (seeking pleasure) to negative reinforcement (seeking relief from distress) - a phenomenon known as allostasis or the opponent-process 5811. The final stage, preoccupation and anticipation, involves executive dysfunction and heightened craving, driving the chronic relapsing nature of the condition 910.
Neuroanatomical Circuitry of Substance Use and Relapse
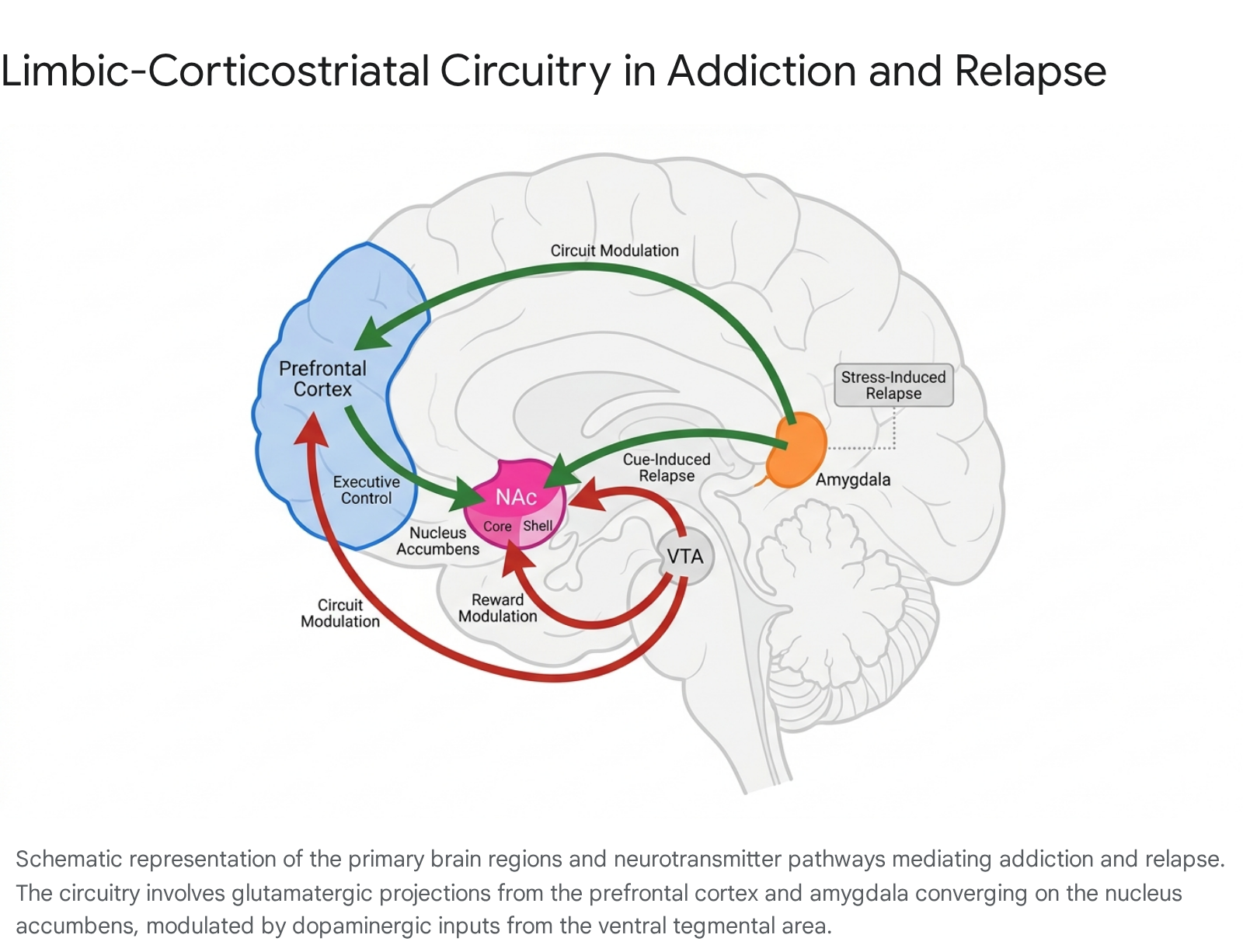
The transition from impulsive, voluntary substance use to compulsive addiction is mediated by a highly distributed limbic-corticostriatal circuitry. The triad of the prefrontal cortex, the nucleus accumbens, and the amygdala forms the core architecture of both active addiction and the physiological processes of recovery and relapse 891213.
Basal Ganglia and Reward Processing
The basal ganglia, particularly the nucleus accumbens (NAc) located in the ventral striatum, serves as the primary anatomical interface between motivation and motor action 89. The NAc receives dense dopaminergic projections from the ventral tegmental area (VTA) and glutamatergic inputs from the prefrontal cortex, amygdala, and hippocampus 91415. Addictive substances universally cause a rapid, supraphysiological surge of dopamine within the NAc, which artificially signals the presence of an overwhelmingly significant survival reward, thereby deeply reinforcing drug-seeking behavior 81617.
The NAc is anatomically and functionally compartmentalized into two distinct subregions: the shell and the core. These regions play divergent roles in the addiction cycle. The NAc shell is primarily involved in the initial reinforcing effects of novel drugs and the integration of hippocampal-dependent spatial and contextual information regarding where a drug was acquired 1819. The NAc core, however, is heavily implicated in the Pavlovian, conditioned influences on drug-seeking and is the critical locus for cue-induced craving and relapse 18. Preclinical neuropharmacological disconnection studies have demonstrated that functional inactivation of the NAc core - but not the shell - drastically reduces cue-induced drug-seeking behavior after a period of abstinence 1820.
At the cellular level, repeated substance use alters the expression of transcription factors within the medium spiny neurons of the NAc. The accumulation of $\Delta$FosB, an unusually stable transcription factor, acts as a molecular switch that initiates and sustains the addicted state by increasing neurocellular sensitivity to addictive substances and driving long-term compulsive behavior 15. Additionally, distinct populations of neurons expressing either Dopamine D1 (direct pathway) or D2 (indirect pathway) receptors play cooperative but distinct roles in mediating reward approach and aversion during the addiction cycle 91821.
The Extended Amygdala and Stress Response
While the basal ganglia mediate the rewarding effects of substances, the amygdala is heavily involved in the learning of drug-cue associations and the neurobiology of negative reinforcement. Like the NAc, the amygdala contains functionally distinct subregions with specialized roles in relapse 915.
The basolateral amygdala (BLA) is essential for the consolidation and reconsolidation of cue-drug emotional memories 91520. Glutamatergic projections from the BLA to the NAc core physically mediate the reinstatement of drug-seeking behavior when an individual is exposed to environmental cues associated with past substance use 915. The mammalian target of rapamycin complex 1 (mTORC1) pathway within the amygdala is activated by these cues; blocking this molecular pathway can prevent the reconsolidation of drug memories, presenting a highly targeted avenue for relapse prevention 9.
Conversely, the central nucleus of the amygdala (CeA) and the bed nucleus of the stria terminalis (BNST) - together known as the extended amygdala - are the primary drivers of stress-induced relapse 8914. During the withdrawal and negative affect stage of addiction, the extended amygdala is excessively recruited. This leads to the heightened release of stress-related neurotransmitters, including corticotropin-releasing factor (CRF), norepinephrine, and dynorphin 914. This hyperactive stress signaling generates a state of allostatic overload, rendering the individual highly susceptible to relapse when faced with minor environmental stressors 8911.
Prefrontal Cortex and Executive Control
The prefrontal cortex (PFC) is responsible for higher-order executive functions, including decision-making, impulse control, emotional regulation, and delay discounting 589. In a healthy neurological state, the PFC exerts top-down inhibitory control over the lower limbic structures. Chronic substance exposure disrupts this delicate equilibrium, resulting in "hypofrontality" - a measurable reduction in baseline prefrontal activity and gray matter volume 7922.
This prefrontal dysfunction manifests clinically as intense craving, emotional volatility, and a loss of self-control. The PFC is broadly understood to operate via two opposing functional networks. The "Go" system, which includes the medial prefrontal cortex (mPFC) and the anterior cingulate cortex (ACC), drives craving and habit execution via dense connectivity with the striatum 79. The "Stop" system, involving the ventromedial prefrontal cortex (vmPFC) and right inferior frontal gyrus, is tasked with inhibiting the craving and stress systems. In addiction, drug-induced neuroplasticity causes an overactivation of the "Go" circuitry and a profound underactivation of the "Stop" circuitry 79. During cue-induced relapse, the prelimbic region of the PFC sends a powerful, uninhibited glutamatergic projection to the nucleus accumbens, serving as the final common pathway for the initiation of drug-seeking behavior 913.
Mechanisms of Neural Repair During Abstinence
The neurobiology of recovery relies entirely on the brain's inherent neuroplasticity. Just as synaptic plasticity allows the brain to wire itself for compulsive drug-seeking, it provides the physiological mechanisms necessary for sustained healing 232425. When an individual maintains abstinence, the brain initiates a cascade of complex biological processes to repair structural damage, clear cellular debris, and restore neurochemical homeostasis 1.
Dopaminergic System Recalibration
One of the most significant physiological consequences of chronic substance use is the profound downregulation of dopamine D2 receptors in the striatum 2428. The brain deliberately reduces the density of these receptors to defend itself against the artificial, massive floods of dopamine caused by drugs. However, when substance use ceases, this receptor deficit leaves the individual in a state of clinical anhedonia, unable to derive pleasure from natural rewards like food, social interaction, or exercise 2426.
During sustained abstinence, the dopaminergic system gradually recalibrates. Longitudinal positron emission tomography (PET) imaging indicates that dopamine D2 receptor availability slowly increases and begins to normalize. This functional recovery requires substantial time, with measurable improvements continuing for 14 months or longer into the recovery process 243027. The gradual return of these receptors correlates directly with the subjective clinical experience of restored mood, increased motivation, and the renewed capacity for pleasure 2426.
Synaptic Remodeling and Neurotrophic Signaling
At the synaptic level, recovery entails reversing maladaptive structural changes, such as pathological variations in dendritic spine density and the abnormal redistribution of AMPA receptors 1. A central molecular axis for this synaptic restoration is the Brain-Derived Neurotrophic Factor (BDNF) and its high-affinity receptor, Tropomyosin receptor kinase B (TrkB) 1.
BDNF is a highly active protein that promotes neurogenesis, drives dendritic spine growth, and facilitates the stabilization of new, healthy synaptic connections 128. Interventions that naturally or pharmacologically enhance BDNF signaling - including physical exercise, environmental enrichment, and the administration of novel psychoplastogens - support the active regeneration of prefrontal circuits that were previously disrupted by chronic substance exposure or stress 133. Furthermore, brain healing involves complex epigenetic regulation. The transcription of maladaptive genes driven by chronic substance exposure (such as those regulated by $\Delta$FosB, MeCP2, and NF-$\kappa$B) is gradually suppressed, restoring molecular homeostasis and reducing compulsive behavioral drives 15.
Epigenetic Regulation and Resolution of Neuroinflammation
Emerging evidence highlights the critical role of neuroinflammation in the pathophysiology of addiction and the subsequent recovery process. Chronic exposure to drugs of abuse triggers a sustained neuroimmune response within the central nervous system, characterized by the activation of microglia and astrocytes, and the significant upregulation of Toll-like receptors (such as TLR4) 1529.
This chronic neuroinflammation exacerbates synaptic dysfunction, impairs healthy neurotransmission, and heightens the individual's vulnerability to craving and relapse 529. During prolonged abstinence, evidence indicates that microglial function is gradually restored, and inflammatory markers recede from the striatum and prefrontal regions 129. Antioxidant and anti-inflammatory mechanisms support neuroplastic recovery by clearing cellular debris, thereby facilitating a healthier, less toxic environment for synaptic repair and the establishment of new neural networks 1529.
Circadian Rhythms and Sleep Architecture
The neurobiology of addiction and recovery is deeply intertwined with circadian rhythms and sleep architecture. Addictive substances fundamentally alter the expression of core circadian clock genes - including CLOCK, NPAS2, and Bmal1 - which directly govern dopamine regulation, mood stabilization, and reward processing 303132.
Sleep is not merely a period of passive rest; it is an active, highly regulated neurobiological state essential for synaptic pruning, cellular repair, and memory reconsolidation. During sleep, the brain actively resets synaptic connectivity, a necessary neuroplastic process mediated by molecules such as BDNF 2833. Chronic substance use causes severe sleep fragmentation and insomnia, which effectively blocks this natural neural reset mechanism, perpetuating cognitive deficits and elevating the risk of relapse 2833. Consequently, interventions that prioritize sleep hygiene and restore circadian homeostasis are absolutely critical for facilitating the brain's long-term neuroplastic recovery and solidifying new, healthy habits 283031.
Longitudinal Trajectories of Brain Recovery
Determining a precise, universal timeline for neurobiological recovery remains a complex challenge. Recovery trajectories are highly individualized, influenced heavily by the specific substance of abuse, the chronicity and severity of use, genetic predispositions, and the presence of co-occurring psychiatric conditions 2425. However, recent longitudinal neuroimaging studies utilizing functional Magnetic Resonance Imaging (fMRI), Tract-Based Spatial Statistics (TBSS), and Voxel-Based Morphometry (VBM) have identified reliable, objective markers of structural and functional healing during sustained abstinence 34353637.
Early Abstinence and Network Recalibration
The initial phase of recovery, spanning roughly the first 1 to 4 weeks, is characterized by intense neurochemical turbulence. As the exogenous substance is abruptly removed, the brain is left with a severe deficit of dopamine and a hyperactive stress system (the extended amygdala) 3338. This period manifests clinically as acute withdrawal, featuring profound anhedonia, emotional lability, and autonomic dysregulation 2633.
Functional MRI studies evaluating the early abstinence period have observed highly dynamic adaptations in large-scale brain networks. For example, during the first four weeks of abstinence from alcohol, patients exhibit rapid, fluctuating changes in resting-state functional connectivity, particularly within the default mode network (DMN) and the frontoparietal network (FPN) 3940. The FPN is critical for top-down cognitive control over impulsivity and anxiety. These early network recalibrations are a delicate biological pivot point; failure of the brain to establish a new functional equilibrium during this turbulent period frequently presages early relapse 3940.
Protracted Abstinence and Structural Repair
As abstinence extends from months into years, longitudinal neuroimaging reveals significant structural and functional repair. A systematic review of within-subject longitudinal neuroimaging studies indicates that structural recovery generally precedes comprehensive neurochemical and functional recovery 3437.
Volumetric increases in gray matter and measurable improvements in white matter tract integrity (measured via fractional anisotropy) are consistently documented across various substance use disorders. Cortical thickness, particularly in the prefrontal cortex, insular cortex, and hippocampus, begins to normalize within 6 to 12 months 3738414243. A recent study evaluating individuals with alcohol use disorder noted highly significant improvements in cortical thickness after an average of 7.3 months of sustained abstinence 42.
Similarly, in cohorts recovering from severe heroin use disorder, longitudinal fMRI data demonstrated that structural impairments in the anterior prefrontal cortex (aPFC) and dorsolateral prefrontal cortex (dlPFC) showed normalization after 15 weeks of inpatient treatment. Crucially, this biological recovery correlated directly with measurable behavioral improvements in inhibitory control and target detection sensitivity 3644. While functional improvements are measurable within the first year, complex cognitive recovery and the complete structural recalibration of deeply entrenched reward pathways often extend well beyond 12 to 24 months, particularly for severe stimulant or opioid dependencies 2426.

Comparative Neurobiology of Behavioral and Substance Addictions
The clinical and scientific definition of addiction has expanded significantly to encompass not only the ingestion of chemical substances but also behavioral addictions (e.g., gambling disorder, internet gaming disorder, compulsive food consumption). Neurobiological research, particularly utilizing fMRI, confirms that behavioral addictions share profound mechanistic similarities with substance use disorders, specifically involving the severe dysregulation of the fronto-striatal circuits 45464748.
Convergence in Fronto-Striatal Circuitry
A comprehensive meta-analysis of functional magnetic resonance imaging studies involving behavioral addictions demonstrated highly specific neural activation patterns during reward-seeking, self-control, and decision-making stages 464950. Individuals with behavioral addictions consistently exhibited significant hyperactivation in the right inferior frontal gyrus (IFG), bilateral caudate, and left middle frontal gyrus (MFG) when exposed to addiction-related cues 464950.
These findings provide robust neurobiological support for the Interaction of Person-Affect-Cognition-Execution (I-PACE) model, which emphasizes the interplay between heightened emotional responses to cues and impaired cognitive inhibitory control 46. In both substance and behavioral addictions, the brain's reward system is conditioned to prioritize a specific stimulus over natural survival rewards, leading to a loss of control despite severe negative consequences 4648.
Divergence in Neurotoxicity and Physiological Withdrawal
Despite the overlap in craving and impaired executive control, structural and physiological differences exist between the two classes of addiction. The primary divergence lies in the direct neurotoxicity, neuroinflammatory responses, and severe physiological withdrawal syndromes associated with certain chemical substances, which are largely absent in behavioral addictions 52946.
| Neurobiological Domain | Substance Use Disorders (SUD) | Behavioral Addictions (BA) |
|---|---|---|
| Primary Reward Mechanism | Exogenous chemicals directly force massive, supraphysiological dopamine release in the NAc, bypassing natural sensory inputs 616. | Endogenous dopamine release triggered by anticipatory environmental cues and the execution of the behavior itself 5651. |
| Structural Brain Changes | Generalized gray and white matter volume loss; direct chemical neurotoxicity specific to the substance (e.g., alcohol-induced cerebellar atrophy, stimulant neurotoxicity) 295253. | Alterations in functional connectivity and some gray matter volume loss (e.g., PFC shrinkage due to disuse), but fundamentally devoid of exogenous chemical toxicity 2246. |
| Circuitry Dysregulation | Severe hyperactivation of reward circuits (ventral striatum) and profound hypoactivation of inhibitory control (PFC) 7944. | Pronounced hyperactivation in the right inferior frontal gyrus (IFG), bilateral caudate, and left middle frontal gyrus (MFG) during active reward seeking 464950. |
| Relapse Vulnerability | Driven by intense cue-reactivity, stress (extended amygdala activation), and severe physiological withdrawal symptoms 89. | Driven primarily by cue-reactivity, psychological stress, and impaired decision-making networks, without severe physiological withdrawal 46. |
Clinical Interventions Modulating Neuroplasticity
Modern addiction treatment increasingly utilizes advanced interventions that directly target and modulate the underlying neuroplasticity of the brain. Rather than relying solely on cognitive behavioral therapies, these interventions aim to accelerate the brain's natural structural and chemical healing trajectories 105455.
Pharmacotherapy and Medication-Assisted Treatment
Pharmacological interventions aim to stabilize turbulent neurochemistry, manage severe withdrawal symptoms, and suppress craving, providing the biological stability necessary for structural brain repair. Medication-Assisted Treatment (MAT) utilizing opioid agonists like methadone and buprenorphine is considered the gold standard for treating severe opioid use disorder 565758.
While both medications stabilize the $\mu$-opioid receptors to prevent physiological withdrawal, they possess distinct neuropharmacological profiles that impact brain recovery differently 5657. Recent preclinical studies utilizing human cortical organoids (hCOs) have demonstrated that buprenorphine exhibits a significantly more favorable neurodevelopmental profile compared to methadone. In these organoid models, methadone administration restricted neural growth and severely suppressed neural network communications, an effect driven by its action as an NMDA receptor antagonist 575960.
Buprenorphine did not induce this growth restriction. This protective, pro-recovery effect is attributed to buprenorphine's unique mechanism as a $\kappa$-opioid receptor antagonist, combined with its distinct lack of NMDA antagonism 575960. Furthermore, longitudinal clinical assessments suggest that strict adherence to buprenorphine treatment is directly associated with improvements in cognitive domains such as learning and memory, indicating that this specific MAT actively facilitates neurocognitive recovery rather than merely substituting one dependency for another 58.
Other pharmacological agents target the glutamatergic system to prevent relapse. Acamprosate, a medication utilized for alcohol use disorder, is hypothesized to exert its therapeutic effects by attenuating the hyperglutamatergic signaling that occurs during repeated cycles of bingeing and withdrawal. By reducing extracellular glutamate levels, acamprosate normalizes synaptic tone and reduces the neurotoxicity associated with alcohol withdrawal 30.
Neuromodulation and Transcranial Magnetic Stimulation
Transcranial Magnetic Stimulation (TMS) is a non-invasive neuromodulation technique that utilizes targeted, painless magnetic fields to stimulate specific neural populations. In the context of addiction recovery, TMS primarily targets the dorsolateral prefrontal cortex (dlPFC) 2854616263.
By delivering high-frequency magnetic pulses, TMS effectively "wakes up" underactive nerve cells in the hypofrontal PFC, promoting localized neuroplasticity and actively restoring top-down executive control 5461. Clinical trials combined with fMRI monitoring have shown that when patients are presented with drug cues, TMS interventions calm the overactive "hot spots" in the reward circuits (like the medial prefrontal cortex), rebalance disrupted dopamine signaling, and significantly reduce cue-induced cravings for substances including alcohol, opioids, and nicotine 28565463.
The clinical efficacy of this approach is highly supported by regulatory bodies; in 2020, the FDA cleared a specific TMS device (developed by BrainsWay) for smoking cessation, formally validating the efficacy of circuit-based neuromodulation in treating addictive disorders 62. As technology advances, the field is shifting toward smaller, portable neuromodulation devices (such as transcranial direct current stimulation, tDCS) that patients can use at home to manage localized cravings in real-time, effectively bridging the gap between clinical intervention and daily relapse prevention 62.
The Neurobiology of Relapse and Setbacks
Despite extensive periods of abstinence and comprehensive clinical treatment, the biological vulnerability to relapse persists. This vulnerability is driven by latent, deeply entrenched neuroadaptations that lie dormant until triggered by specific environmental cues, severe stress, or re-exposure to the substance 91418.
The Incubation of Craving
A paradoxical and highly challenging phenomenon in addiction recovery is the "incubation of craving" - a time-dependent increase in cue-induced drug-seeking behavior that actually grows stronger, rather than weaker, during the initial weeks and months of abstinence 91864.
Preclinical research utilizing animal models has demonstrated that this incubation is mediated by highly specific, progressive synaptic changes within the nucleus accumbens core 1864. During protracted abstinence (e.g., measured at days 1, 15, and 60), the brain recruits distinct, calcium-permeable AMPA-type glutamate receptors into the synapses of the NAc core. The NAc shell, conversely, does not exhibit this progressive functional alteration 18.
When an individual in recovery is exposed to a previously drug-associated environmental cue, the prelimbic prefrontal cortex sends a glutamatergic signal to the NAc. The high concentration of these newly recruited calcium-permeable AMPA receptors in the core results in an exaggerated, hyper-reactive cellular response, driving an intense and overwhelmingly powerful subjective experience of craving 918. Targeted pharmacological blockade of these specific receptors, or the anatomical pathways connecting them, can prevent cue-induced reinstatement, representing a significant target for future anti-relapse medications 918.
Stress, Cues, and the Extinction of Memory
Relapse is rarely a random event; it is almost universally precipitated by either exposure to conditioned cues or acute psychological stress. Cue-induced relapse is tightly linked to the basolateral amygdala (BLA) and its glutamatergic projections to the NAc core. When a cue is encountered, the BLA activates the mTORC1 signaling pathway, which initiates the reconsolidation of the emotional memory linking the cue to the drug reward 915.
Conversely, stress-induced relapse is governed by the extended amygdala (the CeA and BNST). Acute stress triggers the release of corticotropin-releasing factor (CRF) and norepinephrine in these regions, rapidly reinstating drug-seeking behavior even in the total absence of drug cues 914. Overcoming these deeply wired pathways requires active inhibitory learning - often facilitated through targeted cognitive behavioral therapies - where the prefrontal cortex is trained to consistently override and extinguish the reactive signals originating from the amygdala 5565.
Reframing Relapse as a Neurochemical Setback
A pervasive clinical, societal, and psychological misconception is the belief that a single relapse event completely erases the structural and neurochemical progress achieved during a period of abstinence. Neurobiological evidence firmly and consistently refutes this "reset" fallacy 242566.
The neural pathways that are actively strengthened during sustained recovery - such as the measurable increases in prefrontal cortex gray matter volume, the restoration of white matter tract integrity, and the gradual normalization of dopamine D2 receptor density - are cumulative, structural adaptations. They do not instantly vanish or dissolve following a singular return to substance use 242566.
While a relapse undoubtedly introduces sudden neurochemical turbulence, temporarily reactivating dormant addiction pathways and flooding the system with exogenous dopamine, the brain retains the broader architecture of its neuroplastic recovery 2466. Each severe cycle of heavy substance use and subsequent withdrawal does re-disrupt receptor systems, which may incrementally extend the overall recovery timeline, but the underlying foundation of healing remains intact 242567.
The first 72 hours following a relapse represent a critical neurobiological window of extreme vulnerability, where the brain's stress systems are highly reactive 66. Recognizing a relapse as a transient neurochemical setback - rather than a complete biological reset to the day the addiction began - is absolutely vital for adjusting clinical treatment protocols, mitigating patient shame, and continuing the trajectory of recovery without abandoning the physiological gains that have already been accumulated 256667.
Conclusion
The neuroscience of addiction recovery reveals a highly dynamic, bidirectional process of lifelong neuroplasticity. Just as chronic exposure to addictive stimuli structurally and functionally hijacks the brain's reward, emotional, and executive control networks, sustained abstinence provides the requisite biological environment for profound, measurable neural repair. While precise timelines fluctuate based on individual physiology and substance type, empirical evidence demonstrates that dopamine receptors successfully recalibrate, prefrontal cortical volume reliably increases, and large-scale neural networks normalize over months and years of dedicated recovery.
Emerging, evidence-based treatments - including targeted transcranial magnetic stimulation, optimized medication-assisted therapies like buprenorphine, and interventions supporting circadian homeostasis - actively accelerate these natural healing processes. Understanding that relapse is driven by measurable, physical mechanisms - such as the progressive incubation of craving and stress-induced limbic activation - and that long-term structural gains in the brain are not erased by a single setback, provides a scientifically grounded, highly optimistic framework for treating addiction as a manageable and physiologically reversible condition.