Neuroplasticity and brain changes in chronic pain
Evolution of Pain Taxonomy and Clinical Definitions
From Cartesian Dualism to the Biopsychosocial Paradigm
The conceptualization of pain within medical science has undergone a profound transformation, evolving from a simplistic biomechanical symptom into a complex, standalone neurological disease. Historically, the biomedical understanding of pain was rooted in Cartesian dualism, established by René Descartes in 1644, which posited that pain was a direct, linear consequence of physical tissue injury, entirely separate from psychological or central nervous system influences 1. Under this framework, known as the specificity theory, the intensity of pain was assumed to be perfectly proportional to the extent of peripheral tissue damage 12. Within this paradigm, psychological and social factors were only considered when no identifiable physical pathology was present, often resulting in the dismissive and stigmatizing label of "psychogenic" pain 12.
This reductionist view dominated clinical practice until 1977, when George Engel introduced the biopsychosocial (BPS) model, arguing that illness cannot be understood solely through cellular or biochemical deviations 34. The BPS model posits that the experience of pain is a multidimensional, dynamic integration of physiological nociception, psychological state, and social environment 25. However, modern neurobiological research demonstrates that pain and nociception are entirely distinct phenomena. Nociception refers to the neural process of encoding noxious stimuli via high-threshold sensory receptors in the peripheral somatosensory nervous system, which can result in autonomic responses without the conscious sensation of pain 67. Conversely, nociceptor activation does not reliably predict the onset of conscious pain, nor does the firing rate of these receptors correlate perfectly with subjective pain intensity 6.
The International Association for the Study of Pain Revisions
Acknowledging the decoupling of tissue damage from the perception of pain, the International Association for the Study of Pain (IASP) revised its core definition of pain in 2020. The updated definition characterizes pain as "an unpleasant sensory and emotional experience associated with, or resembling that associated with, actual or potential tissue damage" 187. This revision codifies the understanding that pain is an individualized experience heavily modulated by biological, psychological, and social factors, and emphasizes that a patient's self-report of pain is valid regardless of the presence of visible structural damage 17.
The evolution of clinical definitions culminated in 2017 when the IASP formally adopted "nociplastic pain" as a third mechanistic descriptor, existing alongside the traditional categories of nociceptive and neuropathic pain 789. Nociplastic pain addresses the vast cohort of patients suffering from conditions such as fibromyalgia, complex regional pain syndrome (CRPS), irritable bowel syndrome, and chronic primary back pain, where persistent suffering occurs without ongoing tissue damage or somatosensory system lesions 8910.
Emergence and Controversy of Nociplastic Pain
The formal adoption of the nociplastic pain descriptor has generated ongoing debate within the clinical neurology community. Critics of the terminology argue that the word "plastic" in biology typically refers to adaptation, whereas the persistence of chronic pain represents a profound maladaptation 1011. Furthermore, some researchers assert that nociplastic pain should be viewed as an underlying mechanism rather than a standalone diagnosis, cautioning that applying the label too broadly may weaken diagnostic alertness toward occult, undetectable conditions that cause genuine neural damage 1011.
Additionally, controversies exist regarding the requirement to explicitly exclude nociceptive and neuropathic mechanisms before diagnosing nociplastic pain, as clinical observations indicate that nociplasticity can exist on a continuum or concurrently with structural pain conditions 1215. Despite these debates, the categorization provides a necessary framework for understanding why traditional biomedical treatments frequently fail for a large subset of chronic pain patients. Understanding the neuroplastic rewiring that characterizes this pain phenotype provides the foundational rationale for why treating the peripheral body is frequently insufficient to resolve chronic pain syndromes.
Neurobiological Mechanisms of Centralized Pain
The transition from acute, protective pain to chronic, pathological pain is mediated by profound structural and functional changes within the central nervous system. While acute pain relies on peripheral mechanisms to signal immediate physical threat, the persistence of chronic pain is driven by central sensitization and maladaptive neural plasticity 1314.
Peripheral Sensitization and Cellular Signaling
Peripheral pain is initiated when nociceptors detect mechanical, thermal, or chemical insults in the surrounding tissue. In the presence of acute tissue injury, an inflammatory cascade releases mediators such as prostaglandins, bradykinin, and specialized cytokines 141516. These substances lower the activation threshold of the nociceptors, creating a state of peripheral sensitization 1415. When the peripheral injury resolves and inflammation subsides, peripheral sensitization typically extinguishes, and the pain ceases.
However, in cases of severe trauma or sustained peripheral input, the nervous system undergoes an adaptive neuroplastic response that can fail to normalize during convalescence. Preclinical studies indicate that sustained electrical discharge from damaged or highly sensitized peripheral circuitry can lead to fiber hyperexcitability, ultimately triggering a cascade of maladaptive changes in the central nervous system 131718.
Central Sensitization and Maladaptive Neuroplasticity
Central sensitization is the primary neurophysiological process underlying nociplastic pain. It involves the hyperexcitability of neurons within the spinal cord and brain, resulting in enhanced pain signal amplification and the concurrent impairment of endogenous pain inhibition pathways 61415. In this state, a new balance is established between neural excitation and inhibition, heavily influenced by brain stem circuits that modulate spinal nociceptive processing 15.
Recent molecular research underscores the role of microglia-neuron crosstalk and the mapping of novel inhibitory circuitry in sustaining these abnormal pain signals 1317. Molecular factors, including the dysregulation of voltage-gated sodium channels (such as Nav1.7), neurotransmitter receptors, and complex neuron-glial cell interactions, perpetuate abnormal pain processing independent of peripheral input 1619. In central sensitization, the brain's internal alarm system remains perpetually engaged, interpreting normal or subthreshold afferent input as dangerous. Clinically, this manifests as hyperalgesia, where painful stimuli are perceived with exaggerated intensity, and allodynia, where normally innocuous stimuli, such as a light touch or a change in temperature, elicit severe pain 202122.
It is necessary to distinguish central sensitization from nociplastic pain; while central sensitization is the primary driving mechanism of nociplastic pain, it is also a feature frequently observed in chronic neuropathic pain conditions, making central sensitization a broader neurophysiological phenomenon rather than a distinct diagnostic category 22.
Functional Reorganization of Brain Networks
Advances in neuroimaging, particularly resting-state functional magnetic resonance imaging (rs-fMRI), reveal that chronic pain fundamentally reorganizes global brain topology. Research highlights that chronic pain shifts the brain's activity away from classical nociceptive regions, such as the primary and secondary somatosensory cortices, and heavily recruits emotion-related circuitry 6.
The persistence of chronic pain correlates with altered functional connectivity within key cortical networks, primarily the default mode network (DMN), the salience network, and the central executive network 92324. The anterior insula, a primary node in the salience network responsible for integrating sensory information with cognitive and emotional valuation, frequently exhibits pathological hyperactivity in nociplastic pain states. This hyperactivity amplifies both painful and non-painful stimuli, which explains the heightened sensitivity to light, sound, and odors commonly observed in centralized pain patients 915.
A comprehensive 2025 systematic review and meta-analysis of graph-based functional connectivity metrics further clarified these alterations. The analysis revealed that patients with chronic pain exhibit a significant decrease in the local efficiency of whole-brain topology, indicating an impairment in the brain's capacity for specialized processing within local cortical clusters 25. Consequently, the functional architecture shifts toward greater global integration, reallocating cognitive and emotional resources to continuous pain processing and reinforcing maladaptive feedback loops 2325.
Structural Brain Alterations and Gray Matter Volume
In addition to functional rewiring, chronic pain induces measurable, macroscopic structural modifications in the brain. Voxel-based morphometry (VBM) and individual participant data (IPD) meta-analyses evaluating structural MRI data have documented localized gray matter volume (GMV) reductions and cortical thinning associated with prolonged pain states 2627. These morphological reductions are predominantly observed in areas associated with sensory and interoceptive processing, including the somatosensory cortex, motor cortex, dorsal anterior insula, posterior cingulate, and medial prefrontal cortex 232627.
Conversely, certain regions exhibit structural expansions. IPD meta-analyses have identified significant GMV increases in the hippocampus, the parahippocampal gyrus, and bilaterally in the entorhinal cortex 2628. The diversity of these structural findings, which often exhibit high inter-study heterogeneity, suggests that structural brain alterations in chronic pain may be heavily influenced by condition-specific variables and co-occurring psychological distress 2629. For instance, alterations in the volume of the amygdala often correlate more strongly with comorbid depression and anxiety than with the pain phenotype itself 2829.
Despite these localized microstructural changes in gray matter, the 2025 graph-based topology meta-analysis concluded that the macroscale global organization of the structural connectome remains preserved 25. This preservation differentiates the pathophysiology of chronic pain from neurodegenerative diseases, indicating that while the brain undergoes significant regional volume changes, the overarching structural network integrity is maintained, presenting theoretical targets for therapeutic reversal 25.
Clinical Categorization and Phenotypic Diagnosis
The successful management of chronic pain requires precise diagnostic categorization to ensure that therapeutic interventions align with the underlying pathophysiology. Treating nociplastic pain with pharmacological or interventional therapies designed strictly for nociceptive pain routinely leads to clinical failure and potential iatrogenic harm.
Diagnostic Criteria for Nociplastic Pain
Diagnosing nociplastic pain presents significant clinical challenges due to its subjective nature and the inherent absence of visible peripheral pathology on standard imaging. To address this, clinical criteria developed by Kosek et al. and formally endorsed by the IASP provide a structured framework for identifying the nociplastic phenotype 930.
The criteria require a minimum of four conditions: the pain must endure for more than three months; it must present in a regional, multifocal, or widespread distribution rather than a discrete anatomical location; it cannot be entirely explained by concurrent nociceptive or neuropathic mechanisms; and there must be clinical signs of pain hypersensitivity, such as allodynia or hyperalgesia, in the region of pain 31. Furthermore, probable nociplastic pain is heavily associated with a cluster of non-pain comorbidities, most notably severe fatigue, unrefreshing sleep, cognitive difficulties (often referred to as "brain fog"), and hypersensitivity of special senses to environmental stimuli 921.
To aid in clinical decision-making, the distinctions between the three primary pain mechanisms are detailed below.
| Pain Mechanism | Etiology and Origin | Pathophysiology | Clinical Presentation and Characteristics | Common Clinical Examples |
|---|---|---|---|---|
| Nociceptive | Actual or threatened damage to non-neural tissue. | Activation of peripheral nociceptors by mechanical, thermal, or chemical stimuli. Intact neural pathways. | Highly localized, sharp, throbbing, or aching. Usually acute and proportional to the injury. Responds well to NSAIDs. | Post-surgical incisions, osteoarthritis, fractures, burns, gout 7820. |
| Neuropathic | Lesion or disease directly affecting the somatosensory nervous system. | Nerve damage resulting in abnormal sensory processing. Often accompanied by Wallerian degeneration or demyelination. | Burning, shooting, electric-shock sensations, numbness, or tingling. Can manifest far from the lesion site. | Diabetic neuropathy, postherpetic neuralgia, trigeminal neuralgia, radiculopathy 7820. |
| Nociplastic | Altered nociception without clear evidence of tissue damage or nervous system lesions. | Central sensitization, impaired descending inhibition, altered cortical connectivity, and maladaptive neuroplasticity. | Widespread, diffuse pain. Allodynia, hyperalgesia. Co-occurring fatigue, brain fog, and sensitivity to non-painful stimuli. | Fibromyalgia, non-specific chronic low back pain, irritable bowel syndrome, tension headaches 891522. |
Psychosomatic Distinctions and the Validity of Pain
A critical distinction must be made between nociplastic pain and psychosomatic or psychogenic classifications. Due to the limitations of historical biomedical models, patients lacking clear tissue damage have frequently had their pain dismissed as imagined or purely psychiatric in origin 4. Nociplastic pain is not fabricated; it represents a tangible, biological alteration in central nervous system processing 632.
While cognitive and emotional factors, such as fear, hypervigilance, and anxiety, play a major role in the amplification of pain via associative learning, they do so by physically altering neural connectivity 622. The principle that "neurons that fire together wire together" explains how an initial acute injury can subconsciously link pain with fear. Over time, the emotion-related circuitry sustains the pain independently of tissue injury 6. Recognizing that nociplastic pain is driven by objective alterations in neural signaling validates the patient's experience and redirects clinical focus from peripheral structural repair to central nervous system rehabilitation 6.
Limitations of Biomechanical and Structural Interventions
The widespread failure of modern medicine to effectively cure chronic pain stems largely from an overreliance on the traditional biomedical model, which assumes that identifying and correcting a structural defect in the periphery will reliably extinguish the corresponding pain signal 333. When pain is centrally generated or maintained, treatments aimed exclusively at peripheral tissues - such as surgery, steroidal injections, and purely structural physical therapy - frequently fail and can exacerbate the patient's physical and psychological distress 1433.
The Decoupling of Tissue Damage and Nociception
The fundamental flaw in strictly structural approaches to chronic pain management is the poor correlation between actual tissue damage and the perception of pain 332.
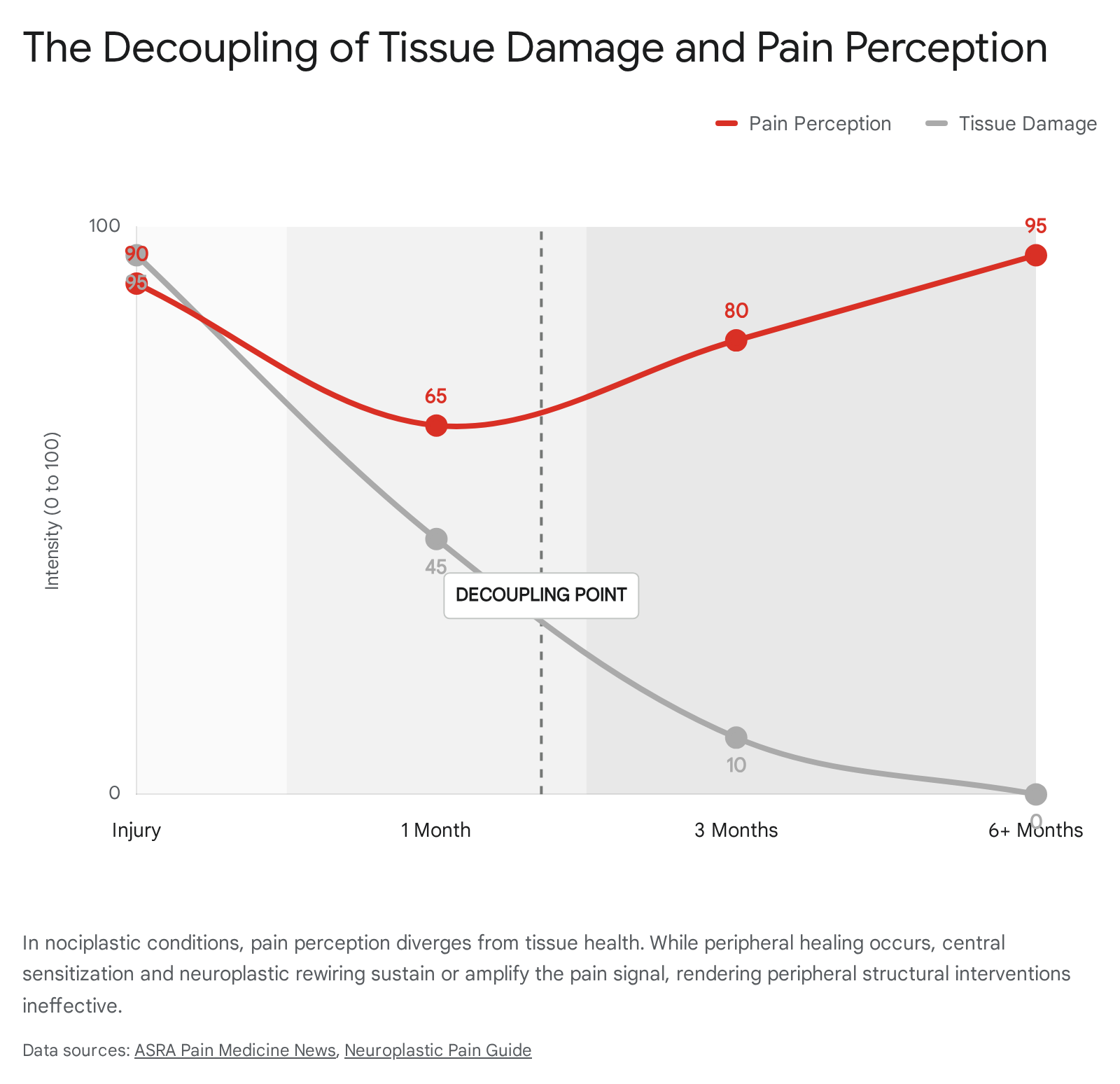
Routine diagnostic imaging frequently reveals "normal abnormalities," such as disc degeneration, mild herniations, or joint wear, that do not strictly correlate with the presence or intensity of a patient's pain 132. Advanced clinical observations demonstrate that nociceptor activation and subjective pain are entirely separable phenomena. For instance, classic medical literature notes cases where individuals sustain severe visible injuries with minimal pain in high-adrenaline situations, or conversely, experience agonizing pain from harmless stimuli, such as a nail visibly piercing a boot but passing safely between the toes without breaking the skin 6.
Surgical Failures and Post-Surgical Chronic Pain
When surgery is performed based on incidental imaging findings in patients who actually suffer from centralized, nociplastic pain, the clinical outcomes are predictably poor. Failed Back Surgery Syndrome (FBSS) represents a profound limitation of structural pain management. A robust 2023 systematic review and meta-analysis of 16 studies encompassing over 85,643 patients undergoing primary lumbar discectomy and fusion surgeries investigated the prevalence of chronic pain following spinal surgery 3435.
The analysis found a pooled prevalence of persistent, chronic pain following surgery of 14.97%, with specific procedural failure rates ranging from 10% to over 40% depending on the complexity of the operation 333435. In these instances, the surgical intervention successfully corrects the perceived structural defect - such as removing a herniated disc or fusing an unstable joint - yet the pain persists or even intensifies. This failure occurs because the primary driver of the pain was maladaptive brain neuroplasticity and central sensitization, rather than the isolated structural variance targeted by the scalpel 33.
Evolving Clinical Guidelines on Spinal Injections
Recognizing the limitations of biomechanical interventions for chronic, centralized pain, major clinical guidelines have begun a paradigm shift away from invasive, localized procedures. In 2025, an international panel of clinicians and methodologists published rapid recommendations in The BMJ based on an exhaustive systematic review and network meta-analysis evaluating 13 common interventional procedures for chronic, non-cancer spine pain 3642.
The procedures evaluated included epidural steroid injections, intramuscular injections, nerve blocks, and radiofrequency ablation 3642. After analyzing dozens of randomized trials, the panel concluded that there is no high-certainty evidence supporting the efficacy of any of these procedures over sham (placebo) interventions for meaningful, long-term pain relief 3642. All available low- and moderate-certainty evidence suggested that these injections provide little to no benefit for either axial or radicular chronic spine pain compared to sham procedures 3637.
Given the lack of sustained efficacy, the substantial out-of-pocket costs, and the inherent risks of invasive procedures - which include infections, prolonged stiffness, accidental dural punctures, and rare catastrophic nerve damage - the panel issued a strong recommendation against the use of these interventional procedures for chronic back pain 3642. This evolving clinical consensus reinforces the neurobiological understanding that when the central nervous system has rewired to perpetuate pain, local anesthetic and steroidal blockade in the periphery is mechanistically insufficient.
Emerging Neuroplastic and Psychological Interventions
Because nociplastic pain is maintained by learned neural pathways, central sensitization, and hyperactive emotion-related circuitry, emerging treatment modalities focus directly on retraining the brain. These therapies operate on the principles of neuroplasticity, aiming to decouple harmless sensory input from the brain's maladaptive threat-detection responses 638.
Pain Reprocessing Therapy
Pain Reprocessing Therapy (PRT) is a novel psychological treatment designed specifically for primary chronic pain. PRT utilizes a combination of cognitive, somatic, and exposure-based techniques to help patients fundamentally reconceptualize their relationship with pain 39. Rather than viewing pain as a dangerous indicator of ongoing tissue damage, patients are guided to appraise the pain as a "false alarm" - a neutral, uncomfortable sensation generated by reversible brain pathways 40. By systematically reducing the fear, urgency, and anxiety associated with the pain, PRT aims to break the self-reinforcing associative learning loop that sustains central sensitization 63941.
The efficacy of PRT was highlighted in a highly publicized 2022 randomized controlled trial published in JAMA Psychiatry. The study evaluated 151 adults with low-to-moderate severity chronic back pain 3940. After four weeks of treatment consisting of eight one-hour sessions, 66% of patients receiving PRT reported being pain-free or nearly pain-free, compared to only 20% of patients receiving a placebo and 10% receiving usual care 639. Longitudinal functional MRI evidence from the trial supported the clinical outcomes, demonstrating that PRT resulted in reduced prefrontal responses to evoked pain, decreased activity in emotion-related neural circuitry, and increased prefrontal-somatosensory functional connectivity compared to control conditions 642.
Clinical Efficacy and Methodological Critiques of PRT
Despite initial promising results, PRT faces substantial clinical scrutiny and methodological criticism. Analysts have noted that the evidence base currently relies heavily on a single, relatively small clinical trial, warranting calibrated uncertainty regarding its status as a definitive cure 3943.
Methodological critiques of the JAMA Psychiatry study have highlighted the use of flawed control groups. Specifically, the placebo group received open-label subcutaneous saline injections in the back; because patients were aware they were receiving a placebo (even if told placebos are effective), the comparative baseline is considered compromised by standard clinical trial metrics 39. Furthermore, critics caution that the results may not be broadly generalizable to patient populations with severe, multi-site pain syndromes, deep-rooted structural neuropathies, or diverse socioeconomic backgrounds that lack access to intensive, specialized psychotherapy 3943. Until independent, large-scale replications across diverse pain conditions are completed, the efficacy of PRT as a standalone intervention remains debated.
Graded Motor Imagery Protocols
Graded Motor Imagery (GMI) is a progressive, neuroplasticity-focused rehabilitation protocol designed to treat complex chronic pain conditions, including CRPS, phantom limb pain, and chronic musculoskeletal pain 4445. The protocol operates on the premise that chronic pain involves disrupted cortical reorganization within the somatosensory cortex, and it aims to sequentially activate motor networks without triggering a protective pain response 454647.
The classical GMI framework consists of three mandatory, sequential stages: 1. Left/Right Discrimination (Laterality Recognition): Patients view images of affected body parts and must quickly identify them as left or right. This task implicitly activates premotor cortices without engaging the primary motor cortex, allowing the brain to process body mapping without provoking pain 444849. 2. Explicit Motor Imagery: Patients visualize performing specific movements with the affected limb, such as bending or stretching, without physically executing the movement. This step progressively increases cortical activation in a safe, controlled mental environment 4849. 3. Mirror Therapy: Patients perform physical movements with their unaffected limb while watching its reflection in a mirror placed along the body's midline. This creates a powerful visual illusion that the affected limb is moving pain-free, driving structural cortical reorganization and helping to normalize maladaptive sensory signaling 484950.
Clinical Limitations and Translation Failures in GMI
While GMI shows efficacy in tightly controlled experimental environments, systematic scoping reviews published between 2024 and 2026 expose significant clinical limitations and translation failures 4445. A major issue is the disparity between strict clinical trial protocols and real-world therapeutic application. In successful randomized controlled trials, GMI is often administered with intense frequency, requiring patients to practice the exercises every waking hour under daily professional supervision 4651.
In routine clinical practice, where patients may only consult a physical therapist every two to four weeks, compliance drops drastically, leading to high treatment failure rates. In one clinical audit evaluating GMI for CRPS outside of trial conditions, pain intensities worsened or remained stable for more than half of the patients 51. Furthermore, recent meta-analyses emphasize that the overall quality of evidence supporting GMI remains low to very low. This is largely due to small sample sizes, a high risk of bias, and extreme heterogeneity in how the intervention is delivered, dosed, and sequenced across different studies 464752. Evidence suggests that individual components of GMI, such as left/right judgment training or motor imagery used as stand-alone techniques, frequently show no significant independent effect on pain reduction 4752.
Systemic Integration and Global Pain Management Models
The recognition that chronic pain involves complex neuroplastic, psychological, and systemic biological mechanisms has prompted a global shift toward multi-modal, integrated pain management models. However, the manifestation of these models varies drastically based on cultural paradigms, economic resources, and regulatory environments 53.
Critiques and Expansion of the Biopsychosocial Model
The biopsychosocial model fundamentally changed pain medicine by integrating the biological aspects of nociception with psychological factors (such as pain catastrophizing and self-efficacy) and social factors (such as interpersonal support and socioeconomic status) 25. Despite its widespread theoretical adoption, the practical application of the BPS model has drawn significant recent critique. In practice, the model is often reduced to a dualistic "bio versus psycho" approach. If a physician cannot locate structural tissue damage, the diagnosis defaults entirely to the psychological domain, inadvertently fostering stigma and signaling to the patient that the pain is purely psychosomatic 454. This reductionism fails to appreciate that nociplastic changes represent legitimate, biological alterations of the central nervous system 454.
Furthermore, researchers in 2025 have highlighted the limits of the traditional BPS paradigm for ignoring broader systemic biology 3. Emerging literature advocates for a transition toward a "bio-psycho-functional and social" model 3. This expanded framework acknowledges that chronic pain is deeply influenced by the biological "terrain" of the patient, encompassing metabolic health, neuroinflammation, the state of the mesenchymal matrix, nutritional deficiencies, and environmental factors - elements that the standard BPS model frequently overlooks in its rush to prioritize cognitive-behavioral interventions 316.
Advanced Regenerative and Neuromodulatory Interventions
As the understanding of centralized pain mechanisms improves, North American clinical frameworks are increasingly incorporating advanced, non-opioid interventions. The American Society of Pain and Neuroscience (ASPN) has recently issued guidelines supporting the use of injectable biologics for regenerative medicine in chronic pain management 55. These biologics, which include platelet-rich plasma (PRP), mesenchymal stem cells (MSCs), and autologous-conditioned serum, focus on modulating neuroinflammation, promoting tissue repair, and altering the biological milieu that contributes to peripheral and central sensitization 1655.
Additionally, neuromodulation techniques are gaining traction as direct interventions for neuroplastic pain. Noninvasive methods such as transcranial direct current stimulation (tDCS) and repetitive transcranial magnetic stimulation (rTMS) apply low-intensity electrical or magnetic currents to the scalp. These techniques modulate cortical excitability, alter thalamocortical pathways, and enhance advantageous neuroplasticity to override maladaptive pain signals 16.
European and North American Clinical Frameworks
In North America, the approach to chronic pain has been deeply colored by the opioid epidemic. The historical reliance on pharmacological management has necessitated a rapid, systemic pivot toward interdisciplinary models incorporating cognitive-behavioral therapy, non-opioid medications, and physical rehabilitation 53. However, access to these comprehensive care models remains fragmented and heavily dependent on socioeconomic status and commercial insurance coverage 53.
Conversely, Western European nations broadly embrace the biopsychosocial model within publicly funded, universal healthcare systems. Countries such as Germany, France, and the UK utilize multi-disciplinary pain clinics where care is inherently team-based, integrating physiatrists, psychologists, and physical therapists in co-located environments 53. In these systems, regulatory frameworks limit reliance on opioids and heavily prioritize functional rehabilitation, patient education, and psychotherapeutic support 53.
Traditional Medicine Integration in Non-Western Regions
In non-Western regions, the transition toward multi-modal pain care integrates contemporary neuroscience with deep-rooted cultural practices, forming highly effective, scalable hybrid models. Traditional, Complementary, and Integrative Medicine (TCIM) is heavily utilized to address both the physical and emotional dimensions of chronic pain 5657.
In China, Traditional Chinese Medicine (TCM) is fully integrated into modern hospital systems alongside conventional biomedical care. Treatments such as acupuncture, Tuina (massage), and Qigong are routinely prescribed for their roles in modulating neuroinflammation and downregulating central sensitization 5356. Scientific validation mechanisms have shown that acupuncture and TCM effectively alter cortical excitability, validating traditional concepts of physiological balance through the modern lens of neuroplasticity 5356.
In India, integrated care is operationalized under the national AYUSH framework (Ayurveda, Yoga and Naturopathy, Unani, Siddha, and Homeopathy). Public hospitals combine lifestyle management, yoga, and meditation with conventional pharmacological treatments 56. Yoga and mindfulness practices act directly on the central nervous system, enhancing parasympathetic tone, addressing the emotional circuitry involved in nociplastic pain, and improving overall functional recovery 385658.
In Brazil, through the National Policy for Integrative and Complementary Practices (PNPIC) within the Unified Health System (SUS), the government provides citizens free access to practices like acupuncture, reflexology, meditation, and phytotherapy 5758. Recent clinical evidence mapping by the Brazilian Academic Consortium for Integrative Health has validated these therapies as cost-effective strategies for managing widespread nociplastic conditions like fibromyalgia and lower back pain. Specifically, herbal medicines utilizing Curcuma longa, Zingiber officinale (ginger), and Salix daphnoides are supported by moderate to high-quality evidence for their efficacy in managing osteoarthritic and centralized pain, significantly reducing the burden on an overextended public health infrastructure 5758.
The success of these global models underscores a fundamental truth in contemporary chronic pain management: addressing neuroplastic pain requires comprehensive care pathways that treat the central nervous system, honor the patient's psychological reality, and utilize multi-disciplinary tools well beyond the scope of traditional biomechanical interventions.