Neurological construction of pain perception
Evolution of Pain Frameworks
Cartesian Dualism and Specificity Theory
The scientific conceptualization of pain has undergone a profound transformation, moving from a rigidly mechanistic paradigm to a highly complex, multidimensional model of human perception. In 1644, the philosopher René Descartes introduced the dualistic theory of pain, which formally separated the mind from the body 123. The Cartesian model posited that pain was an exclusive, direct process operating entirely within the sensory nervous system, creating a one-to-one relationship between the severity of tissue damage and the subjective experience of pain 14. Descartes likened the nervous system to a mechanical bell: pulling a string in the foot directly rang an alarm in the brain 45.
This specificity theory dominated medical science for centuries, embedding the assumption that injuries were purely objective biological events 34. Consequently, the Cartesian model dictated that identifying and repairing the damaged tissue would universally eliminate the pain 4. However, the limitations of this framework became apparent in clinical practice. The model failed to account for severe trauma that yields no immediate pain, or chronic pain syndromes that persist long after tissues have healed or in the complete absence of identifiable pathology 45. Modern magnetic resonance imaging (MRI) studies routinely identify significant structural abnormalities - such as major disc herniations, meniscal tears, and rotator cuff injuries - in completely asymptomatic individuals, demonstrating that structural pathology is neither necessary nor sufficient to produce a pain state 67.
The Biopsychosocial Model
The failure of the Cartesian and strictly biomedical models to explain the diverse presentations of pain led to the adoption of the biopsychosocial framework 138. Originally conceptualized by George Engel, this model asserts that pain is an illness - a subjective experience - rather than merely a disease, which describes an objective biological disruption 1. The biopsychosocial model proposes that pain emerges from a dynamic, non-linear interplay of biological factors (tissue injury, genetics, neurochemistry), psychological factors (anxiety, depression, expectations, fear-avoidance), and social variables (socioeconomic status, cultural norms, social support) 910.
By treating the organism as a unified system, the biopsychosocial model recognizes that psychological distress and social disconnection can directly amplify the central nervous system's sensitivity, thereby increasing pain perception without any corresponding increase in peripheral tissue damage 19. This paradigm shift has established interdisciplinary pain management as the most therapeutic and cost-effective approach, emphasizing function and systemic modulation over isolated anatomical fixes 13.
Nociception Versus Pain Perception
Definitions and Distinctions
Modern neuroscience requires a strict distinction between nociception and pain. The International Association for the Study of Pain (IASP) defines nociception as the neural process of encoding noxious stimuli 11. Nociception is a purely physiological event triggered by actual or threatened tissue damage, which activates specialized peripheral sensory neurons known as nociceptors 111213. The transmission of nociceptive signals may result in autonomic responses, such as elevated blood pressure, or reflexive motor actions, such as withdrawing a hand from a hot surface 11. Crucially, nociception does not inherently imply consciousness or sensation; a patient under general anesthesia exhibits robust nociceptive responses to surgical incisions without experiencing any pain 11.
Pain, conversely, was redefined by the IASP in 2020 as an unpleasant sensory and emotional experience associated with, or resembling that associated with, actual or potential tissue damage 1114. Pain is an emergent property constructed entirely within the central nervous system, representing a highly processed output rather than a raw input 1115. While nociception typically triggers pain perception, the two are entirely dissociable: pain can occur without nociception (as in phantom limb pain or central post-stroke pain), and nociception can occur without pain 81116.
Mechanistic Classifications
Clinically, pain is classified into three primary mechanistic categories: nociceptive, neuropathic, and nociplastic 13. Nociceptive pain arises from actual or threatened damage to non-neural tissue and is characterized by aching, localized sensations that respond predictably to physical triggers and anti-inflammatory treatments 1317. Neuropathic pain results from a lesion or disease of the somatosensory nervous system itself - a defect in the "wiring" - and presents with distinct symptoms such as burning, tingling, or electric-shock sensations along a nerve pathway 1317.
Nociplastic pain, formally recognized by the IASP in 2017, involves altered nociception and central sensitization without clear evidence of actual or threatened tissue damage, nor any demonstrable lesion of the somatosensory system 13. Conditions such as fibromyalgia, irritable bowel syndrome, and tension-type headaches fall into this category. Nociplastic pain is marked by allodynia (pain generated by non-noxious stimuli, such as light touch) and hyperalgesia (an exaggerated response to mildly painful stimuli), signaling a central nervous system operating in a hyper-reactive state 13.
| Clinical Feature | Acute Nociceptive Pain | Chronic Nociplastic / Neuropathic Pain |
|---|---|---|
| Biological Function | Adaptive; signals potential tissue damage and triggers protective withdrawal reflexes 1718. | Maladaptive; persists beyond healing, taking on a self-sustaining pathology 171819. |
| Duration | Short-lived; typically resolves within the standard healing timeframe of 6 weeks 13. | Persistent; lasts well beyond 3 months, often decoupling from the original trigger 132021. |
| Primary Drivers | Peripheral nociceptor activation (A-delta and C fibers) via mechanical, thermal, or chemical stimuli 1121. | Central sensitization, maladaptive neuroplasticity, and continuous top-down amplification 171820. |
| Cellular Mechanisms | Pro-inflammatory mediators (TNF-α, IL-1β) modulating peripheral ion channels (TRPV1, Nav1.8) 2022. | Glial activation, BDNF-TrkB-NMDAR synaptic plasticity, and altered brainstem inhibition 20. |
| Symptom Profile | Localized, predictable, aching; proportional to the physical trigger 1321. | Diffuse, unpredictable, characterized by allodynia, hyperalgesia, and sensory hypersensitivity 13. |
Neurobiology of Ascending Pathways
Peripheral Transduction and Spinal Integration
The physiological journey of a pain signal begins in the periphery. Noxious thermal, mechanical, or chemical stimuli are detected by free nerve endings called nociceptors 1112. When tissue damage occurs, an inflammatory "soup" is generated - comprising arachidonic acid, prostaglandins synthesized via cyclooxygenase (COX) pathways, and cytokines - which sensitizes these nociceptors, lowering their activation threshold 2022.
The signal is transmitted toward the central nervous system via two primary types of afferent fibers: thinly myelinated A-delta fibers, which conduct rapidly and convey sharp, localized "first pain," and unmyelinated C fibers, which conduct slowly and convey dull, diffuse "second pain" 111221. These primary afferent neurons project their central branches into the dorsal horn of the spinal cord, specifically terminating in the superficial Rexed laminae (Laminae I and II) 2123.
Within the dorsal horn, the transmission of the nociceptive impulse to second-order neurons involves the release of excitatory neurotransmitters, primarily glutamate and Substance P 222425. Substance P acts as a potent neuromodulator; binding to neurokinin-1 (NK1) receptors, it yields prolonged postsynaptic excitation, effectively sensitizing the dorsal horn neurons to subsequent inputs 23.
Brainstem and Thalamic Relays
Following synaptic transmission in the dorsal horn, second-order projection neurons decussate (cross the midline) through the anterior white commissure and ascend the spinal cord 2123. This upward transit splits into major tracts. The spinothalamic tract carries impulses directly to the ventral posterolateral (VPL) nucleus of the thalamus, routing signals to the somatosensory cortex to process the location and intensity of the stimulus 2126. Conversely, the spinoreticular tract and related projections route signals through the reticular formation and parabrachial nucleus in the brainstem, interfacing with the hypothalamus and amygdala 2325. These divergent pathways ensure that nociceptive data simultaneously informs the brain's sensory-discriminative mapping and its autonomic, arousal, and affective-motivational systems 2627.
The Pain Neuromatrix
Conceptual Foundations
Historically, researchers sought a singular "pain center" in the brain. This concept was dismantled by Ronald Melzack's Neuromatrix Theory, first proposed in 1990 and expanded in 2001 82829. Melzack theorized that the brain possesses a widespread, genetically determined neural network - the "body-self neuromatrix" - which integrates multiple sensory, cognitive, and emotional inputs to produce the perception of pain 2728. In this framework, pain is the output of the brain, not a mandatory response to sensory input 828.
The neuromatrix theory successfully explains phenomena such as phantom limb pain. When the active neuromatrix is deprived of modulating peripheral inputs following an amputation, the existing neural loops can independently generate an abnormal "neurosignature" that subserves the conscious qualities of burning or cramping pain, demonstrating that the brain can construct the pain experience without any somatic trigger 816. Researchers such as Lorimer Moseley advanced this concept, articulating that "pain is an emergent property" and "the brain's opinion of what's going on in the body" 1531. Pain is fundamentally an assessment of threat; it is modulated by any evidence - biological or psychological - that implies danger 1530.
Cortical Hierarchy of Pain Integration
Advanced functional magnetic resonance imaging (fMRI) has mapped the neuroanatomy of the pain neuromatrix, revealing a coordinated network that is functionally divided into three hierarchical levels:
- The Primary Cortical Pain Matrix: This level comprises the primary and secondary somatosensory cortices (S1 and S2), the parietal operculum, and the posterior insula (PI) 31. It is responsible for the sensory-discriminative dimensions of pain, extracting data regarding the location, quality, and raw intensity of the stimulus 31.
- The Secondary Cortical Pain Matrix: Comprising the anterior insula (AI), anterior cingulate cortex (ACC), hippocampus, and amygdala, this network governs the affective-motivational experience 31. The AI transforms the sensory data from the PI into emotional feeling states, mediating anticipatory arousal and pain-related anxiety 31.
- The Third Cortical Pain Matrix: This higher-order network involves the prefrontal cortex (PFC), orbitofrontal cortex, and the medial and posterior cingulate cortices (MCC/PCC) 31. It provides evaluative-cognitive integration, contextualizing the pain, generating conscious meaning, and driving complex behavioral and motor avoidance responses 31.
Through extensive reciprocal connections, the brain seamlessly unifies the somatosensory awareness of tissue disruption with the emotional distress of the event, rendering pain a holistic perceptual experience rather than a mere reflex 262731.
Descending Modulation Systems
Midbrain and Medullary Control Centers
The brain does not passively endure ascending nociceptive traffic; it actively modulates the volume of these signals via a robust top-down network known as the descending pain modulatory pathway 112432. Cortical structures, particularly the frontal lobe, insula, and amygdala, project downward to the midbrain's periaqueductal gray (PAG) 222433. The PAG acts as a central integration hub for emotional and cognitive pain modulation, projecting subsequently to the rostral ventromedial medulla (RVM) in the brainstem 222433.
The RVM serves as the final common relay for descending control, sending direct projections down the dorsolateral funiculus to the spinal dorsal horn 3234. Critically, the RVM is bidirectional: it contains distinct neuronal populations capable of either inhibiting or facilitating pain 32. "ON-cells" in the RVM burst into activity immediately prior to a nociceptive reflex, acting to facilitate or amplify pain transmission. Conversely, "OFF-cells" cease firing prior to a nociceptive reflex; their continuous baseline activity exerts a suppressive, antinociceptive tone over the spinal cord 33343536. Exogenous opioids, as well as endogenous endorphins, produce profound analgesia by directly inhibiting pronociceptive ON-cells and disinhibiting antinociceptive OFF-cells 243436.
Neurotransmitter Dynamics
At the level of the dorsal horn, descending modulation relies on a complex interplay of specific neurotransmitters: * Serotonin (5-HT): Released by projections from the raphe nuclei within the RVM, serotonin exerts paradoxical, receptor-dependent effects. Activation of spinal 5-HT1A receptors inhibits the excitability of spinothalamic neurons, yielding antinociception. However, activation of 5-HT2 and 5-HT3 receptors strongly facilitates nociceptive transmission 3637. * Norepinephrine: Noradrenergic projections originating primarily from the locus coeruleus provide a robust inhibitory tone. By binding to alpha-2 adrenergic receptors in the spinal cord, norepinephrine hyperpolarizes second-order neurons and decreases excitatory transmitter release, producing significant analgesia 233237. The efficacy of Serotonin-Norepinephrine Reuptake Inhibitors (SNRIs) in treating chronic pain leverages this precise descending pathway 3237. * GABA and Glycine: Within the dorsal horn, descending inputs activate local inhibitory interneurons that release gamma-aminobutyric acid (GABA) and glycine, directly dampening the excitability of ascending projection neurons 2338.
In chronic pain states, central sensitization is heavily driven by a dysfunction of this modulatory system. The endogenous balance shifts toward facilitation: inhibitory GABAergic and glycinergic tone is lost, ON-cells become hyperactive, and descending serotonergic facilitation dominates, amplifying non-noxious sensory input into severe pain perceptions 353639.
Predictive Coding and Active Inference
Bayesian Models of Pain Perception
Over the last decade, computational neuroscience has formalized the brain's construction of pain through the mathematical framework of predictive coding and active inference 404142. This paradigm fundamentally challenges the classical view of the brain as a passive receiver. Instead, the brain is modeled as a Bayesian inference engine that continually generates internal representations of the world and the body 414344.
Within the predictive coding architecture, higher cortical regions (such as the PFC and ACC) generate "priors" - top-down predictions about incoming sensory data based on past experiences, context, memory, and emotional state 4345. These top-down predictions cascade down the neural hierarchy to meet bottom-up sensory data ascending from the spinal cord 4346. When the actual sensory input diverges from the predicted input, a "prediction error" (PE) is generated 4345. To maintain homeostasis and minimize free energy, the brain must resolve this PE. It does so either by updating its internal model (perceptual inference) or by executing a motor action to change the sensory input to match the prediction (active inference) 4547.
Neurophysiological data, particularly from EEG studies, supports this hierarchical structure by revealing distinct spectral asymmetries. Top-down predictions are signaled via slower alpha and beta frequency oscillations, while bottom-up prediction errors are encoded by faster gamma band oscillations flowing from the primary somatosensory cortex toward higher regions like the ACC 424348. The anterior insula plays a critical intersectional role in this process, matching the expected cue against the actual somatic event to compute the prediction error 42.
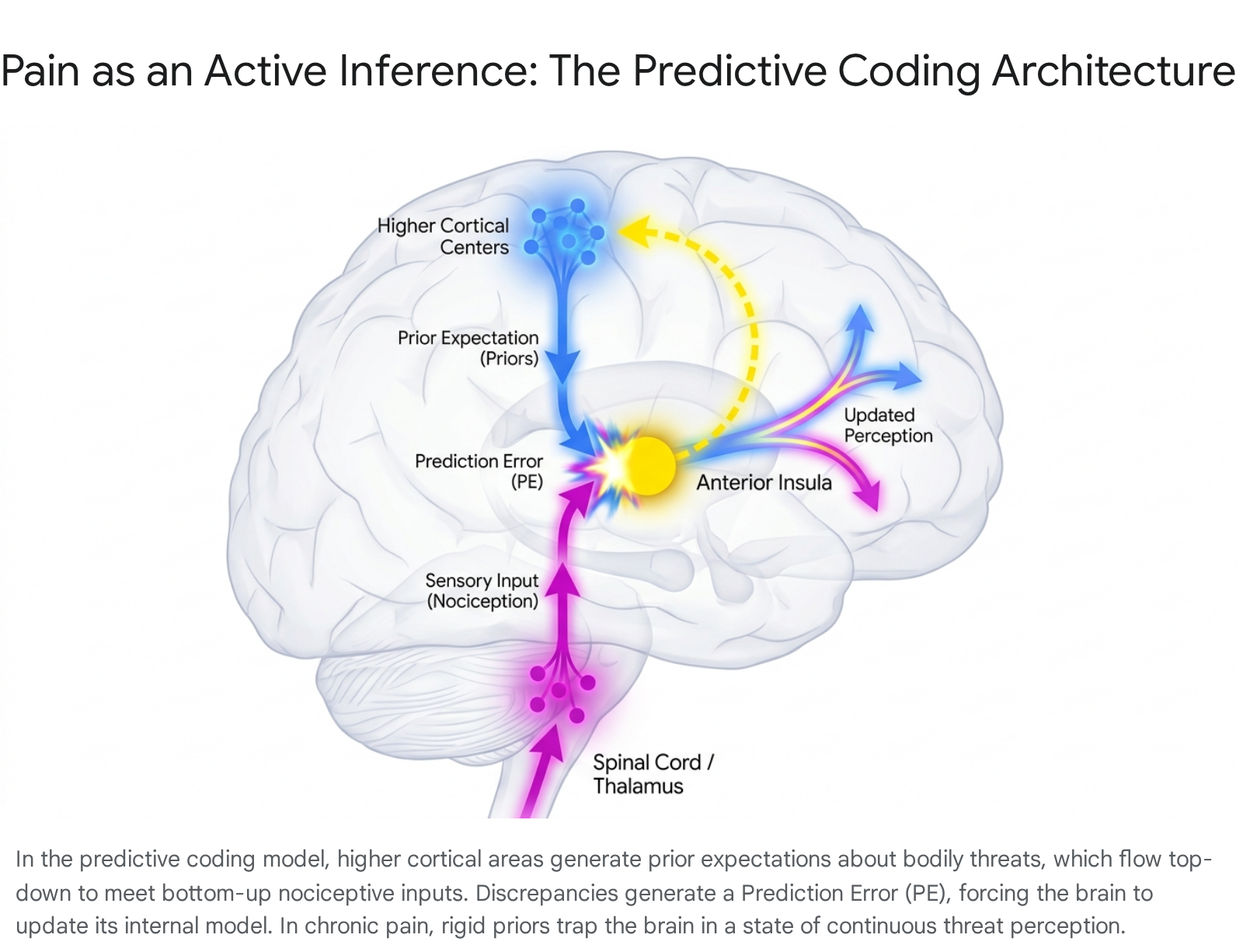
Central Sensitization as Maladaptive Inference
The predictive coding framework elegantly explains the chronification of pain. The resolution of prediction errors depends entirely on the "precision" (confidence) assigned to the top-down prior versus the bottom-up sensory data 434647. In healthy systems, attention modulates this precision appropriately 46. However, prolonged exposure to severe acute pain, compounded by high anxiety, hypervigilance, and fear-avoidance behaviors, causes the brain to formulate overly precise, rigid priors regarding bodily threat 4749.
Through Bayesian updating - often modeled computationally via hierarchical Kalman filtering - the internal model becomes heavily biased to expect pain 5250. If the brain assigns extreme precision to the expectation of danger, it effectively ignores or misinterprets subsequent safe sensory inputs 4647. Normal interoceptive signals or minor tactile stimuli are forced to conform to the rigid prior, generating a massive prediction error that the brain resolves by fabricating the perception of pain 4750. Chronic pain, therefore, represents a state of maladaptive forward model updating, wherein the perceptual system loses the capacity to adapt to changing, benign sensory environments 4551.
Clinical Limitations and the Concept of Metastability
While predictive coding provides profound mechanistic insights, its integration into clinical practice faces debate. The primary operational limitation lies in the difficulty of objectively measuring the strength of subjective Bayesian priors and prediction errors in human patients, though emerging virtual reality and local-global oddball auditory paradigms are attempting to quantify these domain-general predictive dysfunctions 455152.
Crucially, the predictive coding model refutes culturally pervasive, albeit biologically inaccurate, metaphors suggesting that "the body keeps the score" - the idea that trauma or pain memories are stored directly in fascia or muscle tissue 4753. Systems neuroscience argues that non-innervated tissue does not store experience; rather, the brain dynamically reenacts the trauma through circular inference 47. The pathology of chronic pain and trauma is not a persistent somatic memory, but a collapse of metastability - the healthy brain's cognitive flexibility to fluidly transition among semi-stable neural network states 4753.
In a traumatized or chronic pain state, the nervous system becomes trapped in steep local minima within its free energy landscape, bound by overly rigid threat priors 47. Interventions designed to treat chronic pain must therefore target the brain's predictive machinery to restore cognitive flexibility. Treatments such as Pain Neuroscience Education (PNE) - which aims to cognitively downgrade the brain's threat appraisal by teaching the patient that "hurt does not equal harm" - and flow-state therapies have shown efficacy in altering the internal predictive models and reducing perceived disability 30495354.
Sociocultural and Epidemiological Dimensions
Cultural Variability in Pain Perception
Because pain is an evaluative output constructed by the brain's predictive models, it is inherently shaped by the sociocultural context in which an individual operates 555657. Cultural beliefs, language, and social norms continuously feed into the neuromatrix, dictating not only how pain is behaviorally expressed but altering the actual neurobiological threshold of nociceptive perception 5558.
Cross-cultural and cross-ethnic research reveals profound variations in pain expression and sensitivity. Certain cultures view stoicism as a virtue, prompting individuals to suppress outward signs of distress, which can lower self-reported pain scores but severely complicate accurate clinical assessment 5559. Coping strategies diverge significantly along cultural lines; studies comparing cohorts note that African American individuals frequently employ distraction or faith-based coping mechanisms more often than white cohorts, while also exhibiting statistically higher experimental pain sensitivities 5859. However, researchers emphasize that these differences are rarely innate biological variances; rather, they are heavily mediated by systemic environmental factors, including chronic psychosocial stress, perceived racialized discrimination, and a diminished trust in medical providers 5859.
These cultural nuances routinely lead to systemic clinical disparities. Medical gaslighting - where a patient's self-reported pain is dismissed as exaggerated or entirely psychological - has a well-documented history of disproportionately affecting women and minority ethnic groups 1460. Observational data demonstrates that Black, Hispanic, and Asian patients consistently receive inadequate analgesic treatment and fewer pain medication prescriptions compared to white patients in acute settings 14. Addressing these inequities requires the deployment of culturally sensitive pain assessment tools, appropriate translational language, and systemic cultural competency training for healthcare providers to ensure the equitable interpretation of diverse pain expressions 5561.
Global Epidemiological Trends
The global burden of chronic pain has escalated into a primary public health crisis, shaped by biological aging, psychosocial stress, and disparate socioeconomic development. Data aggregated from the Global Burden of Disease (GBD) study across 204 countries from 1990 to 2021 illustrates a steady, significant rise in both the prevalence and the Years Lived with Disability (YLDs) associated with chronic pain 6263.
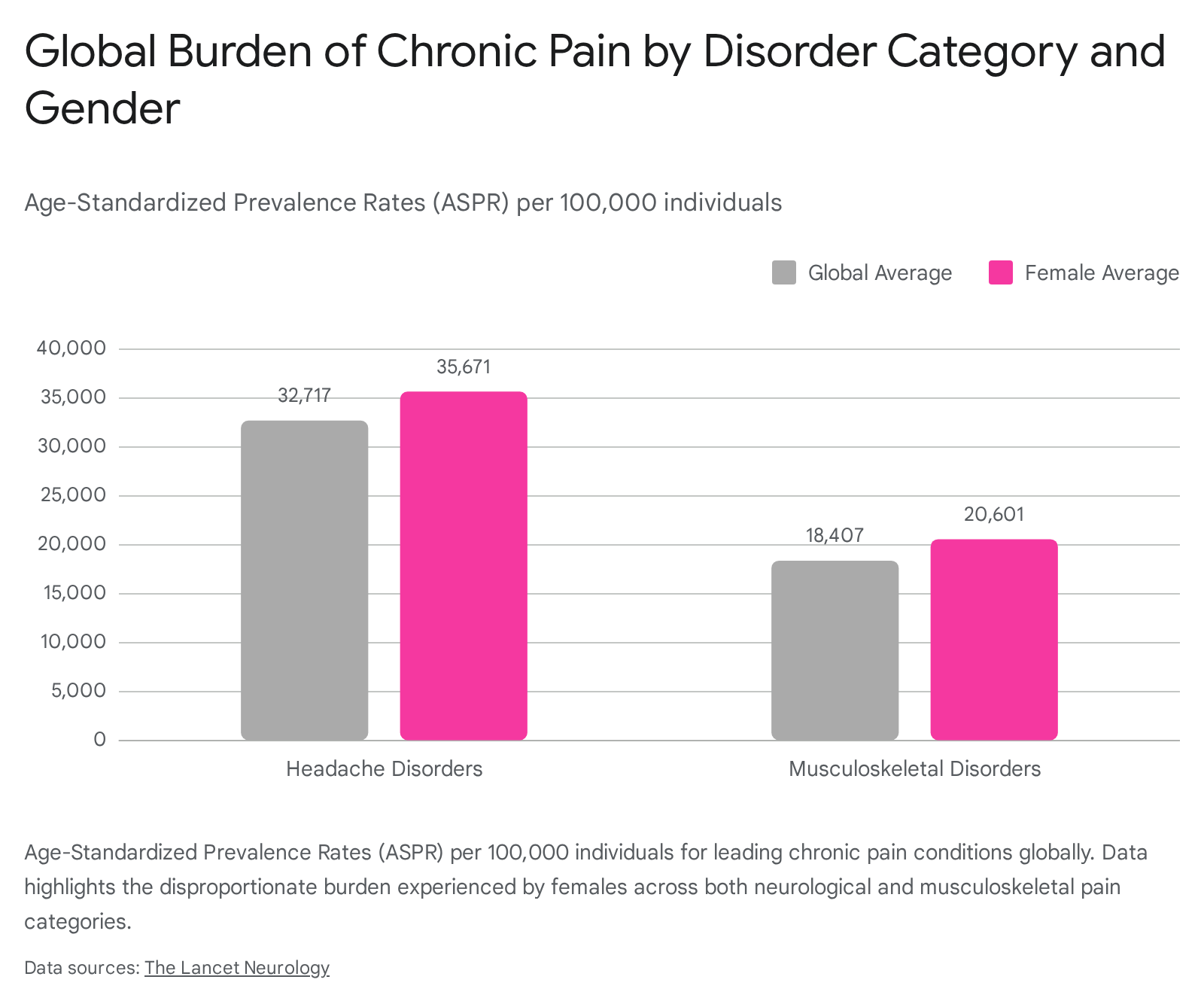
Headache disorders and musculoskeletal conditions dominate the global epidemiological landscape. In 2019, headache disorders affected an estimated 2.60 billion individuals worldwide, with tension-type headaches (TTH) functioning as the predominant driver, particularly among adolescents and younger adults 62. Concurrently, musculoskeletal disorders impacted approximately 1.52 billion people, with low back pain (LBP) representing the single greatest contributor to global disability 6264. Projections indicate that the raw number of individuals suffering from low back pain alone will swell from 619 million in 2020 to 843 million by 2050, driven heavily by population growth and advancing age 6465.
The distribution of chronic pain reveals pronounced demographic and economic disparities. Across virtually all categories and geographic regions, females exhibit higher age-standardized prevalence rates (ASPR) and absolute morbidity burdens compared to males 626366.
Furthermore, the burden of pain correlates distinctly with socioeconomic development. Nations with a high Socio-demographic Index (SDI) - such as high-income regions in North America and Europe - report higher overall ASPRs for musculoskeletal and cancer-related pain 6263. However, deeper demographic analysis within national populations reveals that individuals in the lowest wealth quartiles suffer drastically higher rates of high-impact chronic pain (pain that restricts daily life activities), underscoring the compounding effects of unstable employment, lack of healthcare access, and overlapping psychological distress on the neurological maintenance of pain 63666768.
| Diagnostic Category | Global Estimated Cases (2019) | Global ASPR (per 100,000) | Female ASPR (per 100,000) | Primary Sub-Condition ASPR |
|---|---|---|---|---|
| Headache Disorders | 2.60 Billion 62 | 32,717 62 | 35,671 62 | Tension-Type Headache (25,113) 62 |
| Musculoskeletal Disorders | 1.52 Billion 62 | 18,407 62 | 20,601 62 | Low Back Pain (6,973) 62 |
Conclusions
The scientific understanding of pain has transitioned irrevocably from a structural, tissue-centric alarm system to a highly sophisticated, emergent perception governed entirely by the central nervous system. Pain is not a passive signal ascending from a damaged limb; it is a complex, biopsychosocial output constructed by the brain's neuromatrix to protect the organism from perceived existential threat 11528.
By framing pain through the computational lens of predictive coding, chronification can be understood as an algorithmic failure - a pathological state where Bayesian priors become excessively rigid, predicting danger and subsequently overriding safe, objective sensory evidence 414753. This paradigm shift highlights exactly why traditional, mechanically focused biomedical interventions frequently fail to resolve chronic nociplastic pain 4. Recognizing the vast neurochemical pathways involved in descending modulation, alongside the powerful sociocultural and psychological variables that govern threat processing, is critical for modern medicine 3655. Future clinical strategies must prioritize restoring the nervous system's cognitive flexibility through multidisciplinary approaches, integrating targeted neuropharmacology, pain neuroscience education, and comprehensive lifestyle medicine to effectively dismantle the rigid neurosignatures of chronic pain 94749.