Neurological and Cognitive Effects of Walking
Walking is conventionally defined as a biomechanical mechanism of human locomotion. However, contemporary neuroimaging, cognitive psychology, and clinical neurology reframe walking as an immensely complex cognitive task demanding continuous multisensory integration, spatial navigation, and executive control. As a non-pharmacological behavioral intervention, walking induces profound acute and chronic adaptations within the central nervous system. These neurobiological adaptations span the dynamic modulation of large-scale functional brain networks, the preservation of macrostructural cortical and subcortical volumes, the upregulation of neurotrophic biochemical markers, and the delay of neurodegenerative pathologies.
The following analysis synthesizes current neuroscientific research to evaluate how walking alters the brain, examining its structural, functional, and biochemical mechanisms. Furthermore, it details how environmental contexts - specifically exposure to green and blue spaces - and varying intensity thresholds mediate these neurocognitive benefits.
Structural Brain Changes and Neuroplasticity
Beyond transient cognitive arousal, regular walking induces enduring macrostructural and microstructural adaptations within the brain. Longitudinal studies consistently indicate that structured aerobic activity can attenuate the global brain atrophy typically associated with advanced aging 12.
Macrostructural Preservation of Gray and White Matter
The preservation of total brain volume, gray matter, and white matter integrity is central to maintaining cognitive longevity. In populations with mild cognitive impairment (MCI), targeted exercise interventions utilizing walking and aerobic routines result in significant preservation - and occasionally increases - in gray matter volumes across the frontal, parietal, and posterior cingulate cortices 3. These are regions that typically demonstrate severe cortical thinning in the progression toward Alzheimer's disease and related dementias 3. Furthermore, deprivation from active environments and green spaces is identified as a direct risk factor for worsening white matter grade 2.
Subregional Hippocampal Adaptations
The hippocampus, a medial temporal lobe structure critical for episodic memory and spatial navigation, is highly vulnerable to age-related neurodegeneration. Aerobic walking promotes adaptive hippocampal plasticity, with interventions demonstrating significant increases in overall hippocampal volume in older adults 45.
Recent high-resolution magnetic resonance imaging (MRI) analyses reveal that the structural benefits of walking are both subregion-specific and dose-dependent. Low-intensity walking and passive exposure to natural environments preferentially increase the volume of the subiculum, a hippocampal subfield deeply involved in stress response inhibition 25. Conversely, vigorous walking (defined as exceeding 120 steps per minute) and walking paradigms that require active spatial navigation are associated with volume increases in the parahippocampal gyrus and the right hippocampus, regions integral to spatial memory processing and environmental mapping 5.
Amyloid-Beta and Tau Pathology Mitigation
Walking acts as an active biological barrier against the specific neuropathologies underlying Alzheimer's disease. A four-year longitudinal study utilizing positron emission tomography (PET) and MRI demonstrated that older adults engaging in long-duration (greater than 360 minutes per week) or high-intensity walking exhibited significantly reduced accumulation of beta-amyloid (Aβ) plaques, a primary pathological hallmark of Alzheimer's disease 6. This attenuation of Aβ deposition was most pronounced when high physical activity was initiated prior to late-life stages, suggesting a critical window for structural neuroprotection 6.
Simultaneously, active older adults exhibit a slower buildup of tau protein tangles, another critical driver of neurodegeneration that disrupts communication between brain cells 78. Sedentary individuals, by contrast, show substantially faster accumulation of these toxic proteins alongside more rapid declines in daily cognitive functioning 89.
Functional Connectivity and Brain Network Dynamics
Historically, neuroscience separated motor execution circuits from higher-order cognitive networks. However, modern functional neuroimaging - particularly the integration of functional magnetic resonance imaging (fMRI) with functional near-infrared spectroscopy (fNIRS) - demonstrates that walking heavily engages and modulates associative cognitive networks 1011.
Default Mode and Executive Control Networks
The Default Mode Network (DMN), comprising the medial prefrontal cortex (MPFC), posterior cingulate cortex (PCC), precuneus, and angular gyrus, dominates brain activity during rest, internal mentation, and self-referential thought 1213. Advanced aging is typically characterized by a decoupling or dysfunction within the DMN, correlating with cognitive decline 14.
Aerobic walking interventions selectively combat this decoupling. In healthy adults, a regimen of moderate-intensity walking (e.g., 40 minutes, three times a week for a year) enhances the functional coordination between the DMN and the fronto-executive network, facilitating improved planning, prioritizing, and multitasking 12. Acute bouts of walking also dynamically reconfigure these networks; connectivity from the PCC to the MPFC and the right angular gyrus significantly increases following exercise, optimizing the transition between internal thought and external arousal 13. The synchronization between the DMN and the Executive Control Network (ECN) during walking can also induce "flow states," marked by diminished self-referential anxiety, reduced amygdala activity, and heightened goal-directed processing 15.
Dual-Task Walking and Prefrontal Cortex Activation
The cognitive bandwidth required for walking becomes explicitly visible during dual-task paradigms, wherein participants walk while simultaneously executing a secondary cognitive task. Systematic reviews of fNIRS data reveal that dual-task walking requires substantially higher oxygenated hemoglobin (HbO) concentrations in the prefrontal cortex (PFC) compared to sitting or single-task walking 161718.
The magnitude of PFC activation scales with the complexity of the secondary task. Tasks such as letter generation yield the largest brain activation effect sizes, followed by serial subtraction and obstacle negotiation 18. In older adults and populations with neurological conditions, the PFC exhibits compensatory hyperactivation during walking, indicating that greater attentional resources are consumed to maintain motor control and prevent falls 18. Mobile Brain/Body Imaging (MoBI) utilizing electroencephalogram (EEG) technology reveals that individuals who effectively maintain or improve cognitive performance while walking exhibit distinct neural signatures in frontal brain function, indicative of superior neural flexibility 19. Conversely, highly sedentary behaviors negatively impact resting-state cortical network strength, a deficit that can be mitigated through regular walking routines 20.
Precision Mapping of Individual Networks
While group-level fMRI provides generalized functional maps, recent advancements in precision functional mapping reveal that walking and clinical interventions impact individual network topographies uniquely. Algorithms such as the "Krakencoder" have been developed to align an individual's structural connectome (the physical wiring) with their functional connectome (activity patterns) with remarkable precision 21.
This high-resolution mapping highlights that variations in functional connectivity can track alongside specific behavioral and clinical states. For instance, in individuals with major depression, the frontostriatal salience network exhibits state-dependent fluctuations that correlate precisely with the severity of anhedonia and anxiety 22. Exercise interventions reshape these functional connections; a six-month multicomponent physical activity program in early Alzheimer's disease patients resulted in increased resting-state functional connectivity in frontal, limbic, and cerebellar regions, strengthening brain-behavior coupling even before structural differences stabilized 1.
Biochemical Mechanisms and Brain-Derived Neurotrophic Factor
The structural and functional neuroplasticity driven by walking is largely mediated by molecular cascades, foremost among them being Brain-Derived Neurotrophic Factor (BDNF). BDNF is a highly active neurotrophin that supports the survival of existing neurons and promotes the neurogenesis and differentiation of new neurons and synapses, predominantly within the hippocampus 42324.
The Role of BDNF in Neurogenesis
Elevated circulating BDNF levels are consistently associated with improved spatial, episodic, recognition, and verbal memory, as well as enhanced overall hippocampal functioning 24. Meta-analyses of human trials demonstrate a reliable, moderate effect size for acute increases in BDNF following a single session of exercise, an effect that is intensified by establishing a regular, chronic walking routine 24.
Intensity Thresholds and Metabolic Equivalents
The precise relationship between walking intensity, metabolic equivalents (METs), and BDNF release is complex and varies by demographic. Standard public health guidelines define casual walking as light-intensity (approximately 2.0 to 2.9 METs), moderate brisk walking as 3.0 to 5.9 METs, and vigorous walking or stair ascending as high-intensity (6.0 to 9.0 METs) 2526.
Considerable evidence supports a dose-response relationship in healthy young adults, indicating that circulating BDNF levels elevate more significantly after acute high-intensity exercise compared to light-intensity walking 232728. The physiological mechanisms driving this high-intensity spike include exercise-induced thrombocytosis (wherein circulating platelets release stored BDNF) and increased cerebral blood flow accompanied by lactate crossing the blood-brain barrier 2327. Vertical architectural navigation, such as stair climbing, is modeled as a highly efficient daily booster of BDNF due to the high MET threshold achieved in short durations 2526.
However, network meta-analyses present a distinct optimal threshold for older adults and individuals with mild cognitive impairment. Data indicate that low-intensity short-duration walking (WLS) and moderate-intensity short-duration walking (WMS) protocols are highly effective, and occasionally superior, for elevating sustained resting BDNF levels in populations over the age of 55 429. This suggests an inverted U-shaped physiological response: moderate physiological arousal optimizes neurotrophic release and cognitive facilitation, whereas excessive intensity in older adults may trigger stress responses that inhibit cognitive gains 2830.
| Walking Intensity Level | Metabolic Equivalent (MET) Range | Primary Neurological and Biochemical Adaptations |
|---|---|---|
| Light Walking (Casual/Leisure) | ~2.0 - 2.9 METs | Associated with increased volume in the subiculum; optimized for stress reduction and environmental interaction 525. |
| Moderate Brisk Walking | 3.0 - 5.9 METs | Optimal for elevating baseline BDNF in older adults; enhances DMN functional connectivity 132529. |
| Vigorous Walking / Stair Ascent | > 6.0 METs | Triggers acute spikes in circulating BDNF via lactate and thrombocytosis; increases parahippocampal gyrus volume 52527. |
Longitudinal Impacts on Cognitive Decline
The synthesis of structural preservation, functional network flexibility, and biochemical enhancement positions walking as a primary frontline intervention against age-related cognitive decline.
Dose-Response and Step Thresholds
Quantifying the exact dose of walking required for neuroprotection is a major focus of preventative neurology. A 2025 study published in Nature Medicine analyzed data from the Harvard Aging Brain Study, utilizing PET scans and pedometers to track older adults over an average of 9.3 years 731.
The researchers discovered that in individuals with elevated baseline levels of amyloid-beta, daily walking volumes dramatically altered the clinical trajectory of cognitive decline. Participants who walked between 3,000 and 5,000 steps per day experienced an average delay in cognitive decline of three years compared to sedentary baselines 7831. Those who increased their volume to between 5,000 and 7,500 steps per day delayed significant decline by seven years 7831.
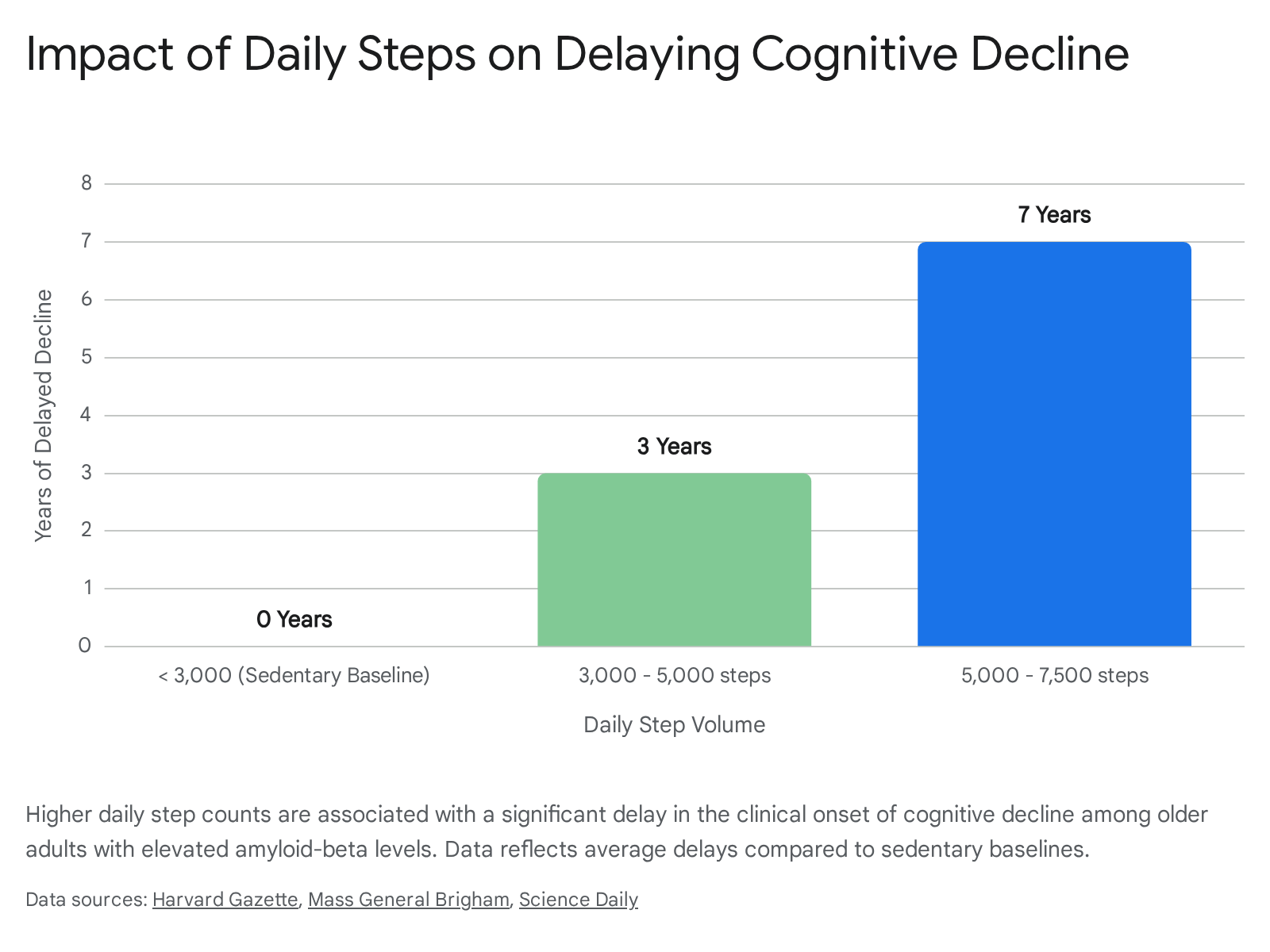
This protection was largely driven by a slower accumulation of tau proteins, suggesting that walking acts as an active physical decelerator of neurodegenerative pathology 731.
Energy Expenditure and Cognitive Trajectories
Data from the China Health and Retirement Longitudinal Study (CHARLS) corroborate the impact of continuous walking routines on cognitive preservation. The CHARLS data reveals an inverted U-shaped relationship where energy expenditures between 900 and 2,100 MET-minutes per week represent the optimal protective threshold against mild cognitive impairment, with the lowest absolute risk observed at approximately 1,600 MET-minutes per week 32.
Further analysis indicates that physical activity volumes greater than 600 MET-minutes per week reduce the risk of cognitive impairment by 14%, while volumes between 1,800 and 2,999 MET-minutes per week yield up to a 38% reduction in risk 30. Individuals maintaining a consistently high or moderate light physical activity trajectory (such as casual walking, which allows for cognitive interaction and environmental engagement) exhibited significantly slower declines in episodic memory and mental intactness compared to strictly sedentary peers 3033.
Multidomain Lifestyle Interventions
While walking is highly effective in isolation, its cognitive benefits are exponentially amplified when integrated into multidomain lifestyle matrices. The Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER) pioneered this paradigm, demonstrating that combining physical exercise with nutritional counseling, cardiovascular risk management, and cognitive training significantly improves processing speed and executive function in at-risk populations 3435. Notably, the FINGER intervention yielded cognitive benefits even for individuals with the APOE ε4 allele, a strong genetic risk factor for Alzheimer's disease 34.
The Japan-Multimodal Intervention Trial for Prevention of Dementia (J-MINT), an 18-month randomized controlled trial building upon the FINGER methodology, confirmed these synergistic effects 3637. In the J-MINT trial, the intervention group exhibited a 41.0% higher improvement in a global cognitive composite score than the control group, with particularly robust gains in memory and executive processing 37. A key component driving these memory improvements was "Cognicise" - a simultaneous dual-task training model requiring participants to perform complex cognitive tasks while walking or engaging in physical movement, effectively forcing the brain to adapt to high-demand resource allocation 37.
Environmental Context of Walking
The neurological benefits of walking are not derived solely from biomechanics; the physical environment in which the walking occurs serves as a profound physiological modifier. Environmental neuroscience demonstrates that natural settings - categorized broadly as "green spaces" (forests, parks, grasslands) and "blue spaces" (oceans, rivers, lakes) - exert distinct, quantifiable effects on brain states 383942.
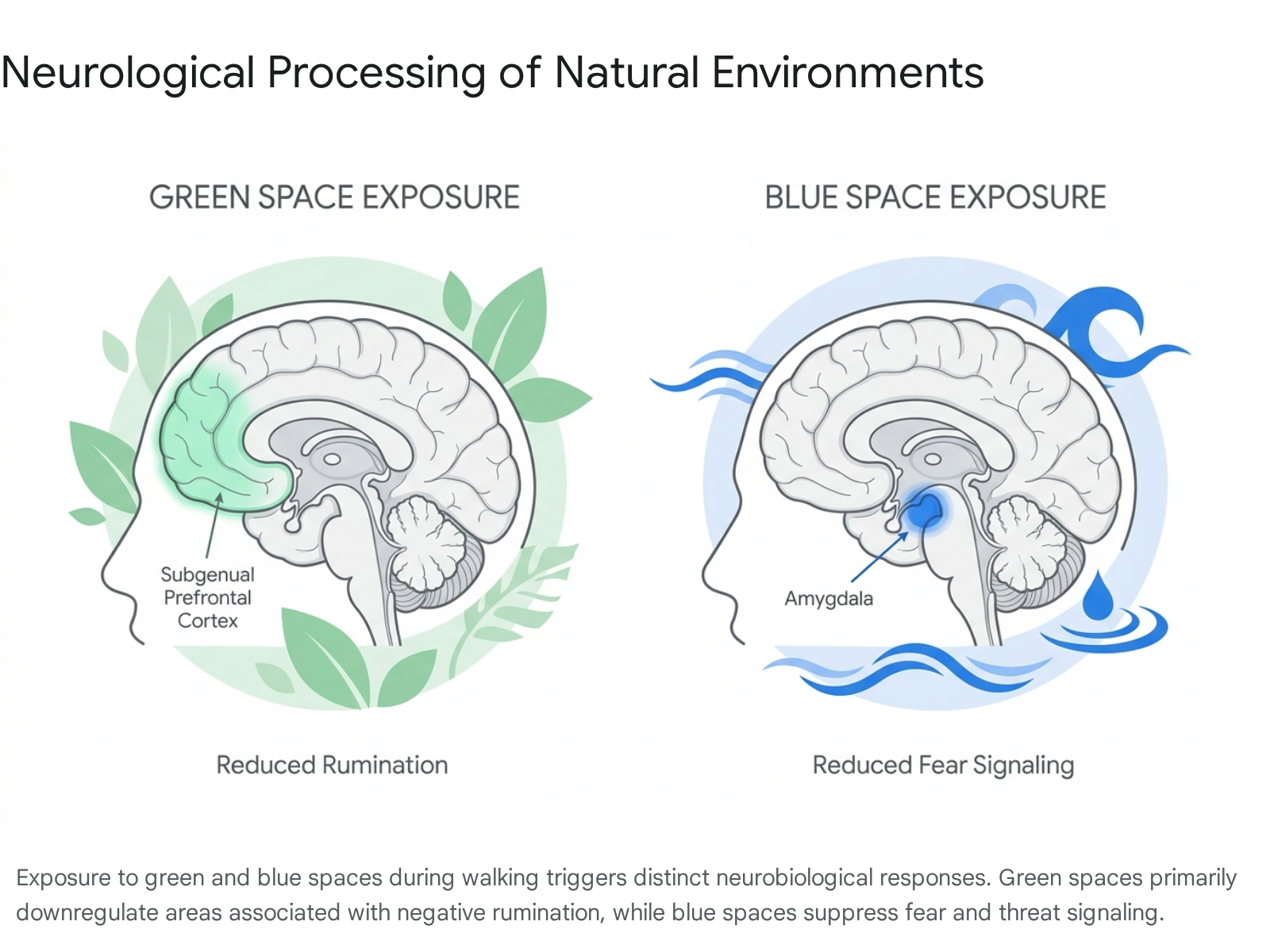
Neurological Processing of Green Spaces
Immersion in green spaces, a practice formalized in Japan as Shinrin-yoku (forest bathing), operates on the principles of Attention Restoration Theory (ART) and Stress Reduction Theory (SRT) 4041. ART posits that modern urban environments demand effortful, directed attention, leading to severe cognitive fatigue. Natural environments, conversely, offer "soft fascination," allowing the executive attention networks of the brain to rest and recover 4142.
Neuroimaging directly validates these theoretical frameworks. A 90-minute walk in a natural green environment measurably decreases neural activity in the subgenual prefrontal cortex (sgPFC) - a region that becomes markedly hyperactive during depressive rumination and the repetitive processing of negative thoughts 404143. A comparable walk of the same duration in a built urban setting yields no such reduction 4041. Furthermore, EEG studies demonstrate that walking in nature results in lower frontal midline theta (FMθ) activity compared to urban walking, serving as a direct neural biomarker of reduced executive attentional strain 42.
Neurological Processing of Blue Spaces
While green spaces are thoroughly documented, "blue space" research is a rapidly expanding domain in public health and neuroscience. Environments featuring water tend to be highly dynamic visually and aurally, capturing involuntary attention and further suppressing intrusive, self-referential thoughts 42.
Functional MRI studies indicate that walking near blue spaces lowers activation in the amygdala, the central brain structure responsible for processing fear and threat detection 4244. Blue spaces are also associated with enhanced parasympathetic nervous system activation ("rest and digest" states) and altered sensory processing, characterized by increased alpha and theta brain wave synchronization. These EEG patterns closely mirror the neurological states achieved during deep meditation 4245.
Comparative Health Outcomes in Natural Environments
The cumulative effect of walking in these restorative environments translates to profound population-level health outcomes. Longitudinal data drawn from the Secure Anonymised Information Linkage (SAIL) databank in Wales, analyzing over two million adults, indicates that routine exposure to blue and green spaces correlates with up to a 20% reduction in the likelihood of seeking medical help for anxiety or depression 4647. Furthermore, for every additional 360 meters an individual lives away from the nearest green or blue space, their risk for common mental health disorders increases by approximately 5% to 7% 4748.
Interestingly, the benefits of natural environments persist regardless of the time of day. Research indicates that individuals engaging in nighttime outdoor physical activity in green spaces report significantly lower anxiety levels and higher subjective happiness compared to those walking in non-green urban spaces at night, underscoring the intrinsic therapeutic value of the natural environment 49.
| Environmental Feature | Green Space (Forests, Parks, Trails) | Blue Space (Coasts, Rivers, Lakes) |
|---|---|---|
| Primary Brain Region Modulated | Subgenual Prefrontal Cortex (sgPFC) 4041 | Amygdala 4244 |
| Key Cognitive/Emotional Outcome | Decreased depressive rumination; restoration of executive attention 4042 | Decreased fear/threat signaling; heightened meditation-like states 4245 |
| Primary Neural Biomarker | Decreased Frontal Midline Theta (FMθ) activity 42 | Increased Alpha and Theta wave synchronization 45 |
| Structural Plasticity Benefit | Increased volume of the subiculum 25 | Associated with preserved whole-brain fractional anisotropy 50 |