Neurobiology of trauma memory and healing mechanisms
Psychological trauma profoundly alters the human central nervous system, shifting the brain from a state of homeostatic regulation to a state of chronic threat detection. For decades, clinical paradigms conceptualized trauma primarily through a psychological lens, treating it as a disorder of memory and fear conditioning. Advancements in neuroimaging, computational psychiatry, and molecular epigenetics have since redefined post-traumatic stress disorder (PTSD) and trauma-related conditions as complex, systemic alterations in neurobiology 11. This research report synthesizes the contemporary understanding of how traumatic experiences are encoded in neural networks, the neurochemical mechanisms driving these adaptations, the current empirical debates surrounding somatic memory, cross-cultural neural variances, and the biological changes induced by evidence-based therapies.
Neural Circuitry and Memory Encoding
The neurobiological foundation of trauma is characterized by a fundamental disruption in the corticolimbic circuitry. This distributed neural network, responsible for evaluating environmental stimuli, orchestrating emotional responses, and contextualizing memory, undergoes maladaptive restructuring following severe psychological distress.
Corticolimbic Disruption
In individuals exposed to severe trauma, the brain's primary threat detection center - the amygdala - often becomes hyperreactive 235. The amygdala operates within a regulatory feedback loop with the ventromedial prefrontal cortex (vmPFC) and the dorsal anterior cingulate cortex (dACC), regions responsible for executive control, emotional regulation, and fear extinction 345. In a healthy neural architecture, the vmPFC exerts top-down inhibition on the amygdala once a threat has passed, allowing the autonomic nervous system to return to baseline arousal levels.
In trauma-adapted brains, this top-down inhibition is severely impaired.
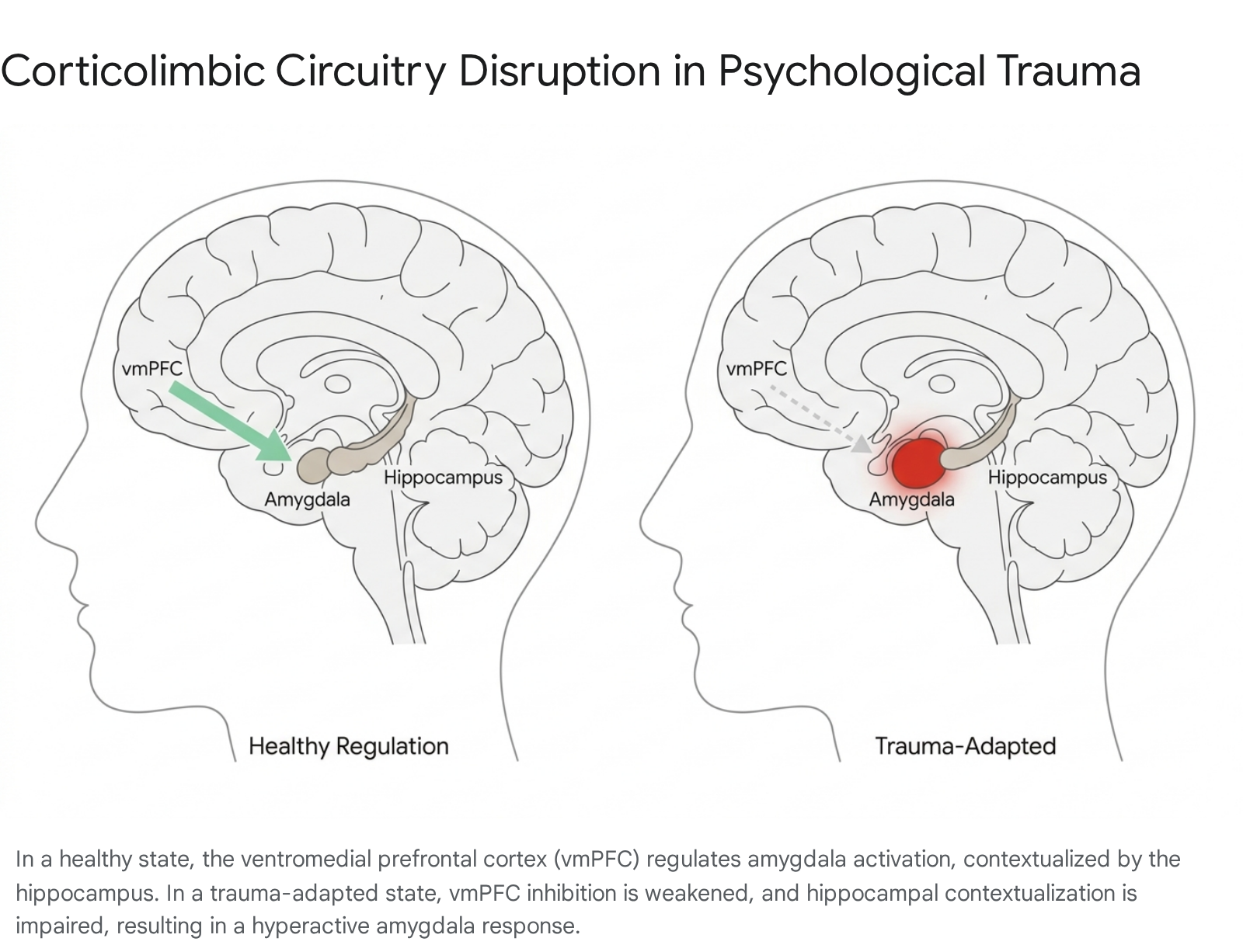
Functional magnetic resonance imaging (fMRI) studies consistently reveal diminished vmPFC activation during fear extinction learning paradigms, indicating that the brain fails to integrate "safety" signals effectively 356. Concurrently, the dACC, which plays a critical role in conflict monitoring and fear appraisal, often exhibits abnormal activation 7. This neurobiological profile contributes to hyperarousal and a diminished capacity to distinguish between past trauma and present reality.
The hippocampus, a structure critical for episodic memory consolidation and spatial contextualization, is also profoundly affected. Structural imaging frequently identifies reduced hippocampal volume in individuals with PTSD 138. While early hypotheses suggested that neurotoxic stress caused this localized shrinkage, recent prospective longitudinal studies - which assess neural structures before and after trauma exposure - suggest that smaller hippocampal volume may predominantly act as a predisposing vulnerability factor (a diathesis) that increases the likelihood of developing PTSD following a traumatic event, rather than being solely a consequence of the trauma itself 19.
Differentiation of Traumatic and Autobiographical Memories
Recent neuroimaging research has elucidated a biological explanation for why traumatic memories manifest as intrusive, fragmented flashbacks rather than coherent autobiographical narratives. A 2023 analysis published in Nature Neuroscience by researchers at the Icahn School of Medicine at Mount Sinai and Yale University demonstrated that the brain processes traumatic memories fundamentally differently than standard negative or sad memories 10.
When individuals recall standard sad autobiographical memories, the hippocampus is highly active, integrating the event into a chronological, contextualized narrative 10. However, when individuals with PTSD recall traumatic experiences, typical hippocampal activation is notably absent. Instead, the posterior cingulate cortex (PCC) - a primary hub of the brain's default mode network associated with internal focus and self-referential processing - shows distinct semantic-to-neural engagement 10.
This neural divergence indicates that traumatic memories function as an alternate cognitive entity, bypassing traditional episodic encoding. They are experienced not as past events being deliberately remembered, but as unintegrated sensory intrusions occurring in the present 10. This finding offers a biological rationale for therapeutic goals aimed at "returning" the traumatic memory representation into a typical hippocampal framework.
Structural and Functional Alterations in Brain Networks
Beyond localized structures, trauma disrupts broad functional connectivity across the brain's macroscopic networks. Resting-state fMRI (rs-fMRI) studies consistently identify aberrations in the salience network and the default mode network (DMN). The salience network, comprising the dACC, insula, and amygdala, typically shows heightened within-network functional connectivity in PTSD patients, correlating with persistent hyperarousal and threat monitoring 15.
Conversely, the default mode network, which includes the precuneus, posterior cingulate cortex, and vmPFC, frequently demonstrates decreased connectivity, which is associated with dissociation and an impaired sense of self 111. Additional studies indicate that visual network processing - encompassing the visual cortex and lingual gyrus - is elevated during rest in PTSD patients, potentially predisposing them to intrusive visual flashbacks 5.
Variations in Direct Versus Indirect Trauma Exposure
Research also suggests that the neurobiological mechanisms of trauma storage may differ depending on how the trauma was acquired. A 2025 study published in PLOS ONE investigated the molecular differences between directly acquired PTSD and bystander PTSD (resulting from witnessing trauma) 12. By analyzing protein degradation patterns in the amygdala, anterior cingulate cortex, and retrosplenial cortex, researchers identified distinct molecular profiles based on whether the trauma was experienced firsthand or observed indirectly 12. This indicates that the neural pathways mediating direct survival threats and indirect vicarious trauma are biologically distinct, raising the possibility that they may eventually require differentiated pharmacological or psychotherapeutic interventions.
Neurochemical Pathways of Trauma
The structural and functional anomalies observed in the traumatized brain are driven by profound neurochemical dysregulation. This dysregulation primarily involves the hypothalamic-pituitary-adrenal (HPA) axis, glutamatergic excitatory systems, GABAergic inhibitory systems, and neurotrophic factors that govern brain plasticity.
Glutamatergic Excitotoxicity
During a traumatic event, the HPA axis triggers a massive release of stress hormones, which in turn causes a surge of glutamate - the brain's primary excitatory neurotransmitter 313. Under normal physiological conditions, glutamate is essential for learning and memory formation through a process called long-term potentiation. However, severe traumatic stress induces a flood of glutamate that overwhelms the brain's regulatory mechanisms.
This overactivation of N-methyl-D-aspartate (NMDA) receptors triggers stress-activated kinases, such as ERK and MSK, leading to the rapid overconsolidation of fear memories 3514. Furthermore, the resulting excitotoxicity causes dendritic retraction in the prefrontal cortex; the branching structures that allow neurons to communicate physically shrink, reducing synaptic density 5. Consequently, the brain's physical architecture is altered, diminishing the connectivity required for higher-order emotional regulation.
GABAergic and Neurotrophic Deficits
Concurrently, trauma survivors frequently exhibit diminished gamma-aminobutyric acid (GABA) function. Because GABA serves as the central nervous system's primary inhibitory neurotransmitter, its depletion effectively removes the "brakes" on the nervous system 31415. The resulting imbalance between excitatory glutamate and inhibitory GABA sustains chronic hyperarousal, severe anxiety, and sleep disturbances, while preventing the successful extinction of conditioned fear responses 1415.
Furthermore, chronic stress suppresses the expression of brain-derived neurotrophic factor (BDNF), a critical protein responsible for neurogenesis, synaptic repair, and cellular resilience 1314. Reduced BDNF/TrkB signaling limits the brain's neuroplastic capacity, impeding its ability to adapt to new environments and form novel, non-threatening associations 1415. This reduction in plasticity effectively locks the neural architecture into a rigid trauma response.
| Neurochemical Agent | Primary Role in Healthy Brain Function | Dysregulation in PTSD and Trauma Pathology |
|---|---|---|
| Glutamate | Excitatory neurotransmitter essential for learning, memory, and long-term potentiation. | Excitotoxic surge over-consolidates fear memories; causes dendritic retraction in the prefrontal cortex 3514. |
| GABA | Inhibitory neurotransmitter responsible for downregulating arousal and promoting calm. | Diminished levels prevent the nervous system from inhibiting fear responses, sustaining hypervigilance and insomnia 31415. |
| BDNF | Protein supporting neurogenesis, synaptic plasticity, and cellular repair. | Chronic stress suppresses BDNF expression, limiting neuroplasticity and preventing the brain from forming new, adaptive associations 1314. |
| Cortisol / HPA Axis | Modulates the acute stress response and regulates metabolism. | Dysregulated feedback loop (often hypocortisolism with heightened receptor sensitivity) maintains chronic stress reactivity and systemic inflammation 315. |
Debates on Somatic Storage and Evolutionary Models
As public and clinical interest in trauma has expanded, several simplified paradigms regarding how trauma is stored have permeated discourse. Chief among these are the "triune brain" model and the concept of somatic memory - the literal storage of trauma in peripheral bodily tissues. In recent years, rigorous evaluations within peer-reviewed neuroscience literature have heavily critiqued both frameworks.
Reassessment of the Triune Brain Theory
The "triune brain" theory, proposed by Paul D. MacLean in the 1960s, posits that the human brain evolved sequentially in three distinct layers: a "reptilian" brainstem controlling survival instincts, a "paleomammalian" limbic system managing emotions, and a "neomammalian" neocortex handling rational thought 161718. The corresponding clinical metaphor suggests that during trauma, the higher cognitive functions shut down, leaving the primitive "lizard brain" to govern behavior.
Contemporary comparative neuroanatomy and evolutionary biology have definitively discredited the triune brain model 16182219. The human brain did not evolve by adding independent anatomical layers like geological strata. Core neuron types are distributed across the entire brain, and evolution acts by modifying existing networks rather than stacking new, independent systems 18. Furthermore, structures like the amygdala, central to the supposedly primitive limbic system, are significantly more developed in higher primates than in other mammals 17.
Modern neuroscience replaces the triune model with the "adaptive brain" framework 22. This paradigm emphasizes interdependent, distributed neural networks that utilize interoception (internal bodily signals) and exteroception (external sensory input) to predictively balance metabolic and survival needs 22. Trauma does not cause a physiological reversion to a reptilian state; rather, it represents a complex adaptation of highly integrated networks prioritizing immediate survival over deliberate cognitive reflection.
Critiques of Somatic Memory Frameworks
A closely related and highly influential narrative is that trauma is stored physiologically in the body's peripheral tissues, fascia, and organs. Popularized largely by Bessel van der Kolk's 2014 book The Body Keeps the Score, this concept posits that trauma induces permanent brain damage and bypasses cognitive memory to lodge directly in the somatic system 20.
Recent systematic reviews in peer-reviewed psychiatric literature have rigorously challenged these assertions. A comprehensive 2025 review published in the BJPsych Bulletin evaluated 122 discrete neurobiological and treatment claims from the book, concluding that the core assertions regarding trauma-induced brain damage and the unique necessity of body-based treatments lack robust empirical support 212622.
The critique highlights severe issues with causal inference. While neuroimaging reliably shows structural and functional differences between traumatized and non-traumatized brains, the vast majority of these studies are cross-sectional and hold no causal explanatory power 921. As noted earlier, prospective longitudinal studies frequently indicate that structural differences, such as reduced hippocampal volume, precede trauma exposure, aligning with diathesis-stress models rather than trauma-induced permanent damage 9.
Furthermore, claims that trauma directly and mechanically causes physical conditions - such as autoimmune diseases or obesity - by remaining physically "trapped" in peripheral body tissues conflate the systemic, downstream effects of chronic stress (such as sustained inflammatory cytokine circulation and HPA axis dysregulation) with localized cellular memory 921. Critics, including Harvard psychologist Richard McNally, have long argued that the therapeutic methods relying on the concept of repressed somatic memory are often based on incorrect theories of human memory processing, and run the risk of generating memory illusions 2329.
Predictive Processing as an Alternative Model
Computational and systems neuroscience offer a biologically accurate explanation for somatic trauma symptoms that does not rely on peripheral cellular storage. The physical sensations of trauma being "in the body" are profoundly real to the patient, but the peripheral tissues themselves do not maintain a memory of the event 24.
Rather, these somatic symptoms arise from "maladaptive inference" within the brain's predictive processing networks 1124. Following trauma, the central nervous system experiences a collapse in metastability; executive networks lose the capacity to properly weight and control sensory precision 1124. Consequently, the brain chronically misinterprets benign interoceptive signals - such as an elevated heart rate from climbing stairs or muscle tension from poor posture - as evidence of an ongoing external threat, instantly triggering a systemic autonomic defense response. The "score," therefore, is kept by the brain's predictive models, rather than inscribed in the flesh 24.
Intergenerational Transmission and Epigenetics
A critical frontier in psychotraumatology is the investigation of intergenerational trauma - the phenomenon where the physiological and psychological effects of severe stress appear in the offspring of traumatized individuals who did not experience the original event directly 2526. The primary biological mechanism hypothesized to mediate this transmission is epigenetics.
Epigenetic Mechanisms in Trauma Inheritance
Epigenetics refers to chemical modifications that regulate gene expression without altering the underlying DNA sequence. The most extensively studied epigenetic mechanism is DNA methylation (DNAm), in which methyl groups attach to specific cytosine bases, acting as biochemical dials that upregulate or downregulate gene transcription 262728.
Severe traumatic stress triggers physiological responses that can alter DNA methylation patterns, particularly on genes associated with the HPA axis, such as the glucocorticoid receptor gene (NR3C1) and the FKBP5 gene, which regulates stress hormone sensitivity 1429. In animal models, these epigenetic tags have been observed to pass through the germline to subsequent generations. However, establishing causal intergenerational epigenetic inheritance in humans has been historically difficult due to confounding environmental variables, shared psychosocial environments, and the ethical limitations of experimental human research 29.
Evidence from Syrian Refugee Cohorts
In 2025, a landmark study published in Scientific Reports provided first-of-its-kind evidence regarding the intergenerational epigenetic signatures of war-related violence in human populations 303132333435. Led by an international team including researchers from the University of Florida and Hashemite University, the study analyzed 131 participants across 48 intergenerational Syrian refugee families in Jordan 323336.
The researchers assessed families who experienced varying exposures to extreme violence, including the 1982 Hama massacre and the post-2011 civil war. The study compared direct exposure, prenatal exposure (in utero), and germline exposure (grandmothers exposed, affecting the fetal germ cells that would eventually become the grandchildren) by scanning 850,000 sites of DNA methylation 30323536.
The analysis identified 21 specific differentially methylated positions (DMPs) associated with direct exposure to violence, and 14 distinct DMPs associated with germline exposure in the grandchildren 32333537.
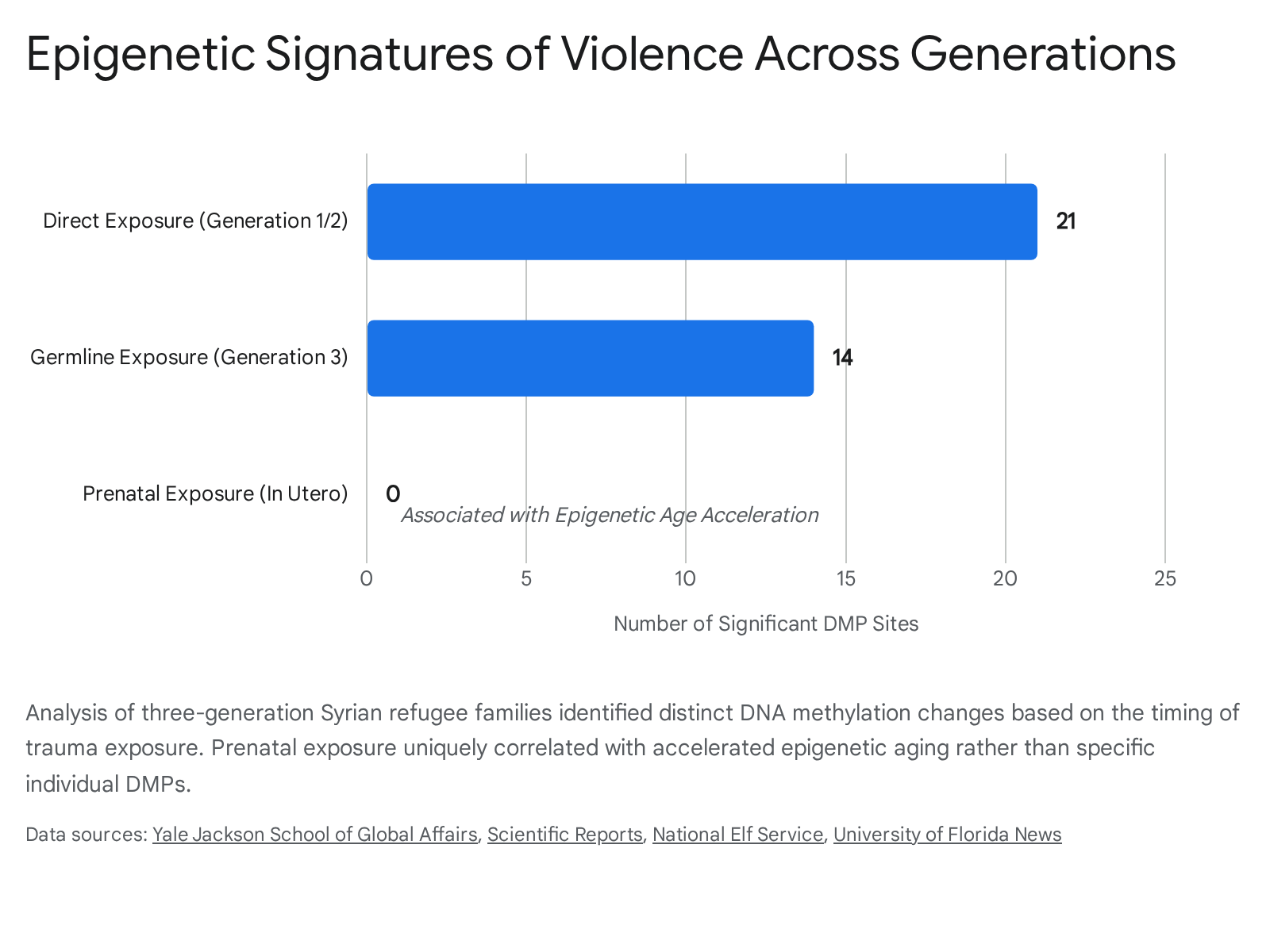
Crucially, across the 35 identified sites, the directionality of the epigenetic change (whether methylation increased or decreased) was overwhelmingly identical regardless of the type of exposure, suggesting a common, persistent biological signature of violence across generations 283032.
Epigenetic Aging and Adaptive Wisdom
Furthermore, the study found that children exposed to violence prenatally (in the womb) exhibited significant epigenetic age acceleration, indicating that their DNAm-predicted age was biologically older than their chronological age 303234. This finding highlights in utero development as a uniquely sensitive period for the biological embedding of stress.
Importantly, researchers noted that the inherited epigenetic marks in the grandchildren were not linked to known disease pathways (such as stress or immunity) 34. Rather than viewing these epigenetic marks solely as biological damage or pathology, researchers hypothesize that they may serve higher regulatory functions. This intergenerational epigenetic tuning may function as an evolutionary mechanism - referred to conceptually as "grandmother's wisdom" - meant to confer adaptation, resilience, and survival advantages to future generations born into harsh or unpredictable environments 3437. Similar adaptive epigenetic mechanisms are currently being documented in Indigenous populations subjected to historical trauma, where trauma-related gene modifications alter baseline homeostasis and immune thresholds 443839.
Cross-Cultural Variations in Trauma Processing
The neurobiology and psychological manifestation of trauma are not uniform across the human species. While biological threat responses are universal, the affective and behavioral expressions of trauma, and potentially the neural circuits mediating them, are heavily influenced by cultural frameworks. Western psychiatric models, which prioritize the diagnostic criteria of PTSD, frequently fail to capture the full breadth of trauma reactions in global populations 14041.
Cultural Idioms of Distress
The concept of "idioms of distress" refers to the culturally specific ways individuals experience, conceptualize, and communicate suffering 404243. Because emotional expression is culturally socialized, behaviors that are considered maladaptive or indicative of avoidance in one context may be normative or highly communicative in another 40.
For example, among Cambodian refugees who survived the Khmer Rouge genocide, trauma frequently manifests as khyâl (wind) attacks 434445. Khyâl attacks are characterized by panic-like autonomic arousal accompanied by an intense cultural fear that a wind-like substance is rising through the body, which the sufferer believes may cause bodily dysfunction and death 45. Similarly, "thinking a lot" is a prominent idiom of distress in this population, functioning as a cultural marker for trauma rumination, while baksbat (broken courage) serves to describe a post-genocidal state of systemic fear and submission 43444647. If clinicians apply strictly etic (outsider) diagnostic frameworks, they risk severely misdiagnosing these patients by ignoring prominent somatic complaints and focusing solely on psychological avoidance, a symptom cluster that varies considerably across cultures 4044.
Neural Correlates in Non-Western Populations
Emerging cross-cultural neuroimaging research suggests that cultural and experiential lenses do not merely alter the psychological expression of trauma; they may influence the underlying neural circuitry itself 48.
Studies on refugee populations indicate distinct neurobiological signatures compared to standard PTSD models derived from Western civilian or military cohorts. For instance, refugees who have survived systemic torture exhibit neural hypersensitivity and a severely impaired ability to downregulate sensory responses compared to healthy controls 48. Specifically, torture severity strongly correlates with distinct activation patterns in the left interior insula (related to avoidance behaviors) and increased activity in the bilateral hippocampus (related to emotional numbing) 48. This specific hippocampal engagement stands in contrast to the generalized hippocampal functional deficits typically observed in broader PTSD populations, suggesting that the precise nature of the trauma (e.g., systemic torture versus acute accidents) heavily modifies the brain's encoding parameters 48.
Neurobiological Mechanisms of Therapeutic Interventions
While trauma induces profound alterations in the brain, inherent neuroplasticity ensures that these changes are not entirely permanent. Evidence-based treatments do not merely manage psychological symptoms; they physically alter brain activity, network connectivity, and autonomic regulation, restoring homeostatic balance. The current landscape of trauma treatment relies on several distinct modalities with varying levels of empirical support.
Cognitive Restructuring and Extinction Learning
Prolonged Exposure (PE) and Trauma-Focused Cognitive Behavioral Therapy (TF-CBT) remain the most rigorously researched and highly recommended first-line treatments for PTSD across major clinical guidelines, including the American Psychological Association (APA) and the VA/DoD 4957505160.
These cognitive, top-down therapies operate on the principle of habituation and extinction learning. By having patients methodically and safely recount traumatic memories (imaginal exposure) or confront safe but triggering real-world environments (in vivo exposure), the brain gradually learns that the memory no longer represents an active threat 5051.
Neurobiologically, successful completion of PE facilitates increased functional coherence between the vmPFC, the amygdala, and the hippocampus 4. Post-treatment fMRI scans reveal that PE restores the inhibitory control of the vmPFC over the amygdala, reducing hyperreactivity 452. Exposure therapy also leads to decreased activation in the rostral anterior cingulate cortex and the insula, correlating directly with symptom remission 452. Despite high efficacy and large effect sizes, top-down therapies are cognitively demanding and can induce severe distress, leading to dropout rates approaching 50% in real-world settings, particularly for patients with highly complex or dissociative trauma presentations 60.
Bilateral Stimulation and Memory Reconsolidation
Eye Movement Desensitization and Reprocessing (EMDR) is another Tier 1, heavily supported trauma therapy 495760. EMDR involves recalling distressing images while engaging in bilateral sensory stimulation, typically guided eye movements.
Extensive meta-analyses demonstrate that EMDR is highly effective in reducing PTSD symptoms, often requiring fewer sessions to achieve remission than traditional exposure therapies 4957516053. Furthermore, EMDR demonstrates notably lower dropout rates than PE, largely because it does not require extensive verbal narrative exposure or distressing out-of-session homework, making it more tolerable for severely dysregulated patients 5160.
At the neural level, successful EMDR treatment has been shown to reduce hyperactivity in the amygdala and normalize processing within the visual cortex and right fusiform gyrus 849. While the exact mechanism of action remains debated - whether the efficacy is derived primarily from the bilateral stimulation taxing working memory, or simply from the structured imaginal exposure itself - the resulting neurobiological shifts effectively integrate the traumatic memory into standard autobiographical storage 4950.
Somatic and Sensorimotor Methodologies
Recognizing the limitations and high dropout rates of cognitively demanding top-down therapies, bottom-up modalities like Somatic Experiencing (SE) and Sensorimotor Psychotherapy (SP) have gained significant clinical traction 605363. These therapies posit that to achieve cognitive integration, therapy must first regulate the autonomic nervous system.
Developed by Peter Levine, Somatic Experiencing focuses on interoception, tracking bodily sensations, and discharging pent-up survival energy through techniques like titration and pendulation (moving gently between states of stress and calm) 545566. Rather than analyzing the trauma narrative cognitively, the therapist guides the patient to safely process implicit, physical states of fight, flight, or freeze 6366. Similarly, Sensorimotor Psychotherapy combines somatic awareness with cognitive attachment themes to address developmental trauma 665657.
Historically, somatic therapies lacked the rigorous randomized controlled trials (RCTs) required for top-tier clinical endorsements. However, the evidence base is shifting. A landmark 2017 RCT by Brom et al. demonstrated significant reductions in PTSD severity (Cohen's d = 0.94 to 1.26) following SE treatment 605456. Further systematic reviews support SE's ability to positively impact affective and somatic symptoms, particularly for patients who have disengaged from exposure-based treatments 5658. While the overall evidence base remains smaller than that of CBT, somatic therapies are considered highly valuable for facilitating subcortical integration before cognitive processing begins 53635456.
| Treatment Modality | Primary Mechanism of Action | Evidence Level & Guideline Status | Key Neurobiological Targets | Clinical Tolerability Profile |
|---|---|---|---|---|
| Prolonged Exposure (PE) / TF-CBT | Top-down cognitive restructuring and extinction learning via systemic exposure. | High: Gold standard. Strongly recommended by APA, VA/DoD. Extensive RCT support 575060. | Strengthens vmPFC inhibition; reduces amygdala/insula hyperreactivity 452. | Can be emotionally taxing; real-world dropout rates can reach up to 50% 60. |
| EMDR | Memory reconsolidation aided by bilateral dual-attention stimulation. | High: Tier 1 recommendation. Meta-analyses show equivalence to PE/CBT 49575153. | Normalizes visual cortex processing; reduces limbic hyperactivity 84960. | High tolerability; lower dropout than PE due to lack of extensive narrative homework 5160. |
| Somatic Experiencing (SE) | Bottom-up autonomic regulation, focusing on interoceptive tracking and somatic discharge. | Emerging/Moderate: Supported by recent RCTs and reviews, but lacks the vast literature of CBT 605458. | Autonomic nervous system regulation (sympathetic/parasympathetic balance) 605659. | Highly tolerable; gentle approach beneficial for severe dissociation and complex trauma 606356. |
Experimental Pharmacological Adjuncts
Alongside psychotherapy, clinical neuroscience is continuously exploring pharmacological adjuncts to accelerate trauma processing. While traditional SSRIs are commonly prescribed to manage depressive and anxious symptoms, recent attention has focused heavily on psychedelic-assisted therapies, most notably MDMA (midomafetamine).
MDMA acts as a potent monoamine releaser, flooding the synapse with serotonin, dopamine, and norepinephrine while temporarily suppressing amygdala fear responses. This unique neurochemical state theoretically provides an optimal window of tolerance, allowing patients to process severe traumatic memories without debilitating autonomic fear 3.
However, the clinical integration of MDMA has faced major regulatory hurdles. Following an advisory committee rejection in June 2024, the U.S. Food and Drug Administration (FDA) issued a Complete Response Letter (CRL) in August 2024 rejecting Lykos Therapeutics' New Drug Application for MDMA-assisted therapy for PTSD 6072. The FDA's CRL, publicly released in September 2025, cited significant methodological and safety concerns.
Key issues included functional unblinding (participants knowing they received the active drug due to its unmistakable psychoactive effects), selection bias (nearly 40% of participants had prior illicit MDMA use), a lack of long-term durability data (efficacy was only evaluated up to 18 weeks), and the failure of trials to systematically collect abuse-related adverse events 6073. Furthermore, follow-up studies evaluating long-term durability were deemed inadequate due to design flaws and low enrollment 60. While Lykos Therapeutics continues to negotiate trial designs with the FDA to address these safety components, MDMA currently remains an unapproved, experimental treatment 606162.
Conclusion
The neuroscience of trauma reveals a central nervous system profoundly altered by the biological imperative to survive. Through the dysregulation of the corticolimbic circuitry, excitotoxic neurochemical floods, and the disruption of episodic memory encoding, traumatic experiences override the brain's baseline regulatory systems. While popular frameworks often rely on metaphors of localized cellular memory or ancient "lizard brains," rigorous neurobiological research confirms that trauma is a complex, systemic dysfunction of predictive processing and executive control. Furthermore, emerging epigenetic studies suggest that the biological impact of severe violence may span generations, embedding adaptive biological instructions into the human genome. Ultimately, however, the brain remains highly plastic. Through targeted, culturally sensitive, and evidence-based therapeutic interventions - whether top-down cognitive restructuring or bottom-up autonomic regulation - the traumatized brain can reconsolidate fearful memories, restore inhibitory networks, and reclaim homeostatic balance.