Neurobiology of Placebo and Nocebo Effects
Introduction to Psychobiological Context Effects
The study of placebo and nocebo phenomena has undergone a profound paradigm shift in recent decades, transitioning from a methodological consideration in clinical trials to a central subject of neuroscientific and psychobiological inquiry. Historically conceptualized primarily as an artifact of reporting bias, regression to the mean, or spontaneous disease remission, the placebo effect is now recognized as a genuine biological phenomenon mediated by the central nervous system 12. Placebo effects refer to the beneficial physiological or psychological changes that occur following the administration of an inert treatment or within the context of active care 34. These effects are driven by the psychosocial context, therapeutic rituals, and the patient's expectation of healing 345.
Conversely, the nocebo effect represents the inverse phenomenon: the induction or exacerbation of adverse symptoms resulting from negative expectations, the anticipation of harm, or warnings of potential side effects 367. In clinical trials and everyday medical practice, the "placebo response" encompasses the overall improvement observed in a control group, which inextricably combines the true psychobiological placebo effect with non-specific factors such as the natural history of the disease 89. The true placebo effect, however, is isolated as the specific neurobiological cascade triggered by belief and therapeutic context.
Advances in functional neuroimaging and neurochemistry have successfully mapped these subjective expectations to objective alterations in central nervous system function 169. The distinction between the psychological experience of a treatment and its biological realization has become increasingly blurred. Emerging evidence demonstrates that the human brain relies on complex, predictive architectures to modulate physiological states based on contextual information, directly impacting systems governing pain, autonomic function, immune response, and motor control 2910.
Neuroanatomical Circuitry of Expectation
The realization that psychosocial contexts alter objective biological states relies on highly conserved neuroanatomical networks. Placebo and nocebo responses hijack the brain's endogenous regulatory systems, demonstrating that expectations are translated into physiological outcomes through specific cortical and subcortical pathways.
The Descending Pain Modulatory System
The most comprehensively mapped anatomical circuitry involved in placebo analgesia and nocebo hyperalgesia is the descending pain modulatory system (DPMS). This network exerts top-down control over ascending nociceptive signals entering the spinal dorsal horn, dictating the ultimate conscious experience of pain 1112. The DPMS integrates cortical inputs representing cognition, prior experience, and expectation, routing them through brainstem structures to either inhibit or facilitate nociceptive processing 111314.
The core nodes of the DPMS include the prefrontal cortex, the rostral anterior cingulate cortex (rACC), the midbrain periaqueductal gray (PAG), and the rostroventromedial medulla (RVM) 111213.
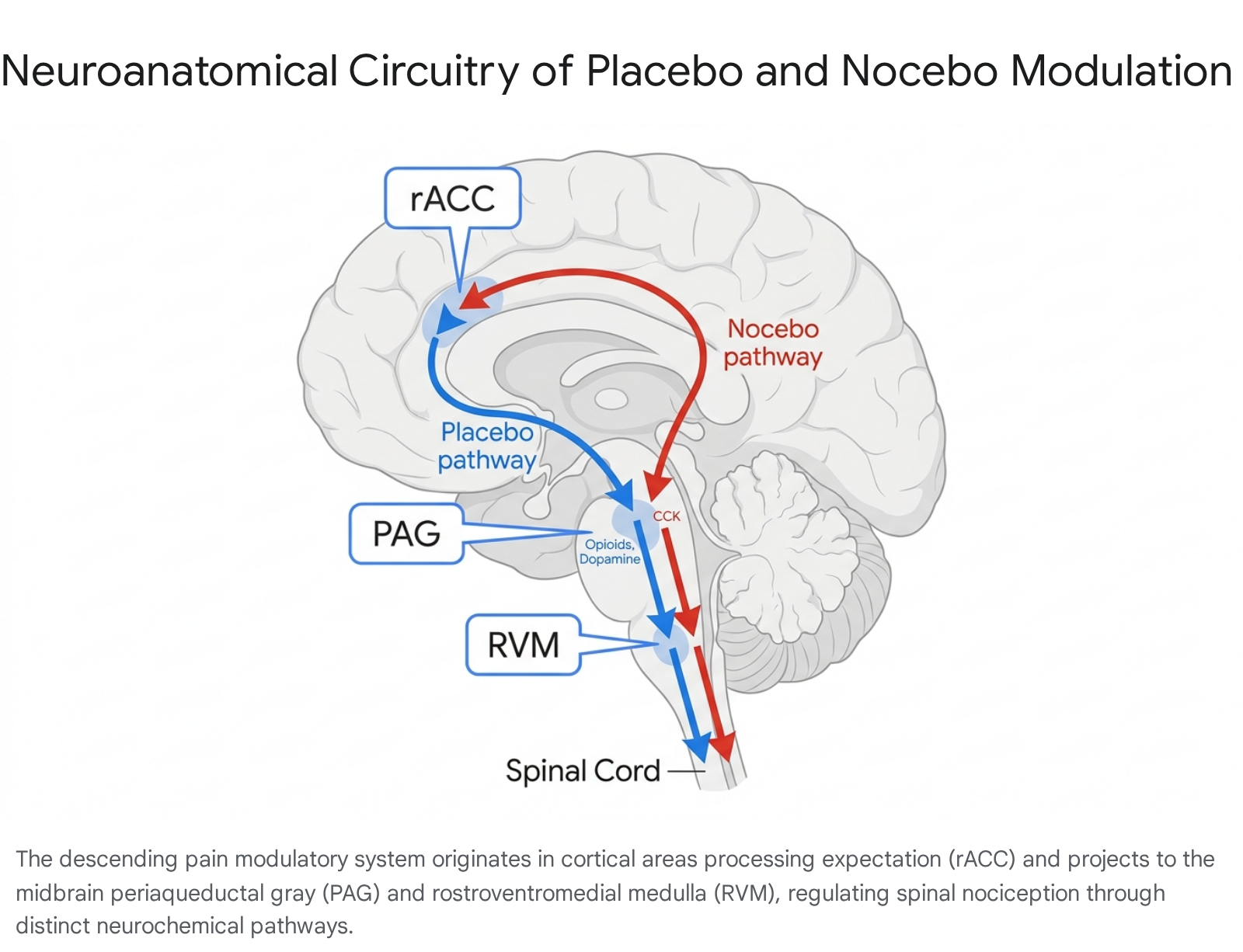
Functional neuroimaging reveals that coupling between the rACC and the PAG is essential for both opioid-induced analgesia and placebo-mediated analgesia 12. The PAG serves as the critical midbrain relay, receiving inputs from the amygdala, hypothalamus, and frontal cortices, and projecting directly to the RVM 1213. The RVM then sends descending projections via the dorsolateral funiculus to the spinal or medullary dorsal horns 12.
Bidirectional Control in the Rostroventromedial Medulla
The RVM maintains a bidirectional degree of control over nociception, governed by distinct, functionally opposing neuronal populations. "On-cells" within the RVM increase their firing rates prior to a nociceptive reflex, facilitating the experience of pain and enhancing ascending signal transmission 13. Conversely, "off-cells" cease firing immediately prior to a painful stimulus and are associated with powerful pain inhibition 13. The administration of exogenous opioids, as well as the endogenous opioid release triggered by placebo expectations, inhibits on-cells and excites off-cells, a condition considered necessary and sufficient to produce analgesia 13.
Placebo and nocebo effects dynamically and oppositely modulate this circuit. High-resolution functional magnetic resonance imaging (fMRI) reveals that the strength of placebo analgesia correlates with increased activity in the RVM and decreased functional activity in the PAG 15. The nocebo effect induces the exact opposite neurovascular changes, increasing PAG activity while decreasing RVM output 15. Cortical control of this descending pathway during placebo responses is gated by the rapid release of endogenous opioids within the ventrolateral PAG (vlPAG), which subsequently alters the processing of incoming noxious sensory information in the spinal cord on the timescale of seconds 11.
Neurochemical Signatures of Contextual Healing
The translation of belief into biological alteration relies on specific neurotransmitter systems. The biochemical foundation of the placebo effect is primarily linked to the activation of the endogenous opioid, dopaminergic, and endocannabinoid systems, whereas nocebo responses are characterized by cholecystokinin transmission.
Endogenous Opioids and Dopaminergic Pathways
Placebo administration in the context of anticipated pain relief triggers the robust release of endogenous opioids. Positron emission tomography (PET) imaging utilizing radioligands such as [11C]-carfentanil reveals that placebo analgesia is directly related to the activation of μ-opioid receptors in the rACC, the pregenual cingulate cortex, the dorsolateral prefrontal cortex, and the anterior insular cortex 1216.
Concurrently, neurochemical evidence demonstrates that the expectation of therapeutic benefit stimulates dopamine transmission. This is particularly evident in conditions characterized by dopaminergic deficits, such as Parkinson's disease. In these patients, the administration of a placebo under the expectation of receiving active dopaminergic medication triggers endogenous dopamine release within the nigrostriatal pathway and the ventromedial striatum, contributing to measurable improvements in motor velocity and a reduction in rigidity 6. The dopaminergic system's involvement extends beyond motor control; it plays a crucial role in reward expectation and the encoding of prediction errors across various clinical conditions, including depression and addiction 617.
Endocannabinoid System Mobilization
Beyond opioids and dopamine, the endocannabinoid (eCB) system has emerged as a crucial mediator of expectation-induced pain relief. Recent pharmacological and neuroimaging studies indicate that the endocannabinoid and opioid systems interact in a complex, state-dependent manner during placebo analgesia 18. This interaction appears to be governed by a gating mechanism linked to opioid peptides, specifically β-endorphin 18.
When β-endorphin elevations are comparatively low, increases in circulating Fatty Acid Amide Hydrolase (FAAH) substrates - such as anandamide (AEA), N-palmitoylethanolamide (PEA), and N-oleoylethanolamide (OEA) - strongly correlate with increased placebo analgesia 18. Interestingly, while these individual FAAH substrates may not independently predict analgesia with high statistical significance, a composite measure of their shared variance strongly predicts the magnitude of the placebo response 18. This demonstrates that the mobilization of the endocannabinoid system contributes to the relief of pain driven by psychosocial expectations 18. However, when β-endorphin elevations are highly robust, the relationship between FAAH substrates and analgesia is absent 18. This dynamic suggests that if profound opioid activation saturates the descending pain modulatory circuits, it limits the physiological capacity for additional eCB-mediated modulation, pointing to highly convergent neuromodulatory networks within the PAG and RVM 18. Notably, unlike FAAH substrates, the eCB ligand 2-arachidonoylglycerol (2-AG) is not significantly associated with placebo analgesia 18.
Cholecystokinin and the Neurobiology of Nocebo
While positive expectations trigger opioid, dopaminergic, and endocannabinoid release, negative expectations mobilize an entirely different neurochemical cascade. Nocebo hyperalgesia - the exacerbation of pain due to anticipated harm - is largely driven by the neuropeptide cholecystokinin (CCK) 619.
Research has identified a specific neural circuit mediating nocebo behaviors, characterized by the release of CCK from neurons projecting from the anterior cingulate cortex directly to the lateral periaqueductal gray 19. This ACC-to-lateral PAG CCK circuit is activated by both environmental conditioning and socially transmitted expectations of pain 19. The operational role of this pathway is confirmed by pharmacological blockade; the administration of proglumide, a CCK receptor antagonist, effectively blocks nocebo hyperalgesia in human subjects 19. The onset of the nocebo effect also involves a concurrent suppression of endogenous opioid and dopamine activity, illustrating a bidirectional neurochemical environment dictated by conscious or conditioned belief 6.
Furthermore, empirical studies frequently observe that nocebo effects manifest with greater intensity and persistence than placebo effects 72021. In experimental setups directly comparing the two, nocebo suggestions consistently exert a stronger influence on pain perception than placebo suggestions, and this heightened sensitivity persists across follow-up sessions weeks later 721. This asymmetry likely reflects an evolutionary adaptation that prioritizes the recognition of and response to potential environmental threats - a biological imperative where negative expectations carry more survival weight than positive ones 7.
| Feature | Placebo Effect | Nocebo Effect |
|---|---|---|
| Primary Trigger | Positive expectations and belief in therapeutic efficacy 6. | Negative expectations or belief in impending harm/side effects 67. |
| Key Neurotransmitters | Activation of dopamine, endogenous opioids, and endocannabinoids (FAAH substrates) 618. | Activation of cholecystokinin (CCK). Concomitant reduction in opioids and dopamine 619. |
| Brain Region Activity | Increased activity in prefrontal cortex, rACC, ventromedial striatum, anterior insula, and RVM. Decreased activity in PAG 615. | Activation of ACC-to-lateral PAG circuit. Decreased activity in RVM 61519. |
| Clinical Examples | Analgesia, improved motor velocity (Parkinson's), reduction in depressive symptoms 6. | Hyperalgesia, anticipatory nausea, reporting of expected adverse events (e.g., vaccine trials) 622. |
Predictive Coding and the Bayesian Brain Framework
The translation of psychosocial context into the profound neurochemical changes described above is most robustly understood through the theoretical lens of the "Bayesian brain" and predictive processing 10172324. Traditional biomedical models conceptualized the brain as a passive, stimulus-driven organ that absorbs bottom-up sensory signals from the periphery and converts them directly into conscious experience 2425. The predictive coding framework fundamentally inverts this view.
Hierarchical Inference and Prediction Errors
The Bayesian brain hypothesis postulates that the brain is an active inference engine, constantly generating a top-down cascade of hypotheses (or generative models) about the state of the body and the external environment 102627. Under this framework, perception is an integration of prior expectations (priors) and incoming sensory evidence (the likelihood) 282930.
When bottom-up sensory inputs do not match the brain's top-down internal model, a "prediction error" is generated, representing the mismatch or surprise 102831. The brain's fundamental operational goal, as articulated by theories such as the Free Energy Principle, is the continuous minimization of these prediction errors 2329. Error minimization occurs through two complementary strategies: perceptual inference, where the internal model is updated to better match incoming sensory information, and active inference, where the organism initiates physiological or motor actions to alter the sensory input so it aligns with the prediction 102932.
Precision-Weighting and Dopamine
Crucially, the resolution of conflicts between priors and sensory data depends on "precision weighting" 172733. Precision refers to the brain's estimate of the statistical reliability or inverse variance of a given signal 1027. Modulatory neurotransmitters, particularly dopamine, are hypothesized to control the precision of prediction errors within cortical networks 1729.
If a patient receives a placebo treatment accompanied by high clinical authority, elaborate medical ritual, and verbal assurance, the brain assigns immensely high precision to the prior expectation of healing 1025. Faced with ambiguous or noisy somatic sensory inputs - such as vague fluctuations in chronic back pain or irritable bowel syndrome - the highly precise top-down prediction overrides the imprecise bottom-up sensory data 1025. Consequently, the brain actively interprets minor interoceptive changes as evidence of recovery, culminating in the conscious perception of symptom relief - the computational manifestation of the placebo effect 102527. Re-analyses of fMRI data during associative learning tasks provide empirical support for this model, showing that the precision of unsigned prediction errors is coded in the superior frontal gyri and dorsal anterior cingulate cortex, and that this precision-weighting is directly perturbed by dopamine antagonists like sulpiride 17.
Threat Prediction Errors in Chronic Pain and Nocebo
The application of this computational model to chronic pain and nocebo responses yields the concept of the "Threat Prediction Error" (TPE) 34. While the dopaminergic system encodes reward prediction errors, defensive responses, allodynia, and pain amplification may be scaled to the magnitude of TPEs - the discrepancy between expected harm and actual sensory outcomes 34.
If negative expectations are deeply entrenched due to prior trauma or explicit warnings (e.g., severe anxiety regarding a medical intervention), the brain maintains a persistent state of high-precision threat prediction 2934. The nocebo effect occurs when the brain fulfills this highly precise negative prior, interpreting benign or mildly uncomfortable somatic signals as severely noxious events 1025. Recent findings suggest that midbrain dopamine neurons dynamically change their response to aversive stimuli, shifting from signaling novelty to signaling the predictive onset of a threat, guiding escape mechanisms via the periaqueductal gray 34. This persistent state of negative affective charge represents diminished confidence in one's own predictive model, explaining the high comorbidity between chronic pain states and anxiety disorders 34. Furthermore, predictive coding models suggest that nocebo effects may be less susceptible to prediction error correction than placebo effects, contributing to their remarkable persistence over time 2021.
The Modulatory Role of Attention
Attention serves as a critical mechanism for manipulating precision weighting. By directing selective or mindful attention toward or away from specific somatic sensations, individuals alter the perceived precision of the sensory likelihood 3035. Directing a patient's focus away from indicators of distress and toward signs of healing functionally alters the weight of prediction errors 3035. Neuroimaging data indicates that voluntary reduction in anticipatory anxiety via attention modulation monotonically reduces the magnitude of pain modulated by negative expectations 36. Thus, attention acts as an amplifier or silencer of sensory data, recalibrating maladaptive predictive coding states associated with central sensitization 3031.
Conditioning Versus Conscious Expectation
The precise psychological triggers that establish the predictive "priors" in the Bayesian brain have been the subject of a long-standing academic debate. Theoretical models have traditionally divided the genesis of placebo effects into two distinct mechanisms: explicit conscious expectation and classical Pavlovian conditioning 373839.
Divergence in Physiological Targets
Experimental evidence demonstrates that conditioning and conscious expectation govern different domains of the human physiological response 3840. Classical conditioning occurs when an initially neutral contextual cue (e.g., the taste of a uniquely flavored drink, the shape of a pill, the hospital environment) is repeatedly paired with an active biological agent (the unconditioned stimulus) 3841. In hallmark studies involving the immunosuppressive drug cyclosporin A, taste cues paired with the drug subsequently elicited suppression of T lymphocyte proliferation and a reduction in interleukin-2 and interferon-γ release entirely on their own 16. These autonomous, unconscious physiological processes - such as hormone secretion and immune function - are highly susceptible to conditioning but are remarkably resistant to verbally induced, conscious expectations 3840.
Conversely, conscious physiological processes - such as the subjective experience of pain or the voluntary modulation of motor performance - are heavily influenced by explicit verbal suggestions and conscious expectancies 3840. For instance, merely informing a patient that a powerful analgesic has been administered will produce a robust placebo hypoalgesic effect even in the absence of prior conditioning trials 40. Studies reveal that verbally induced expectations fail to alter unconscious growth hormone secretion, yet profoundly affect pain perception and motor performance in the exact same subject cohort 38.
The Integrated Learning Continuum
Contemporary neurobiological learning models suggest that conditioning and expectation are not entirely discrete phenomena but exist on an integrated continuum 373941. Classical conditioning procedures shape conscious expectancies, which in turn mediate the downstream placebo response 39. In neuroimaging studies assessing the processing of prediction errors, the magnitude of placebo-induced activation of μ-opioid receptor-mediated neurotransmission in error-detection regions (like the ACC) was predicted by the individual's expectation-effectiveness comparisons, rather than raw expectations alone 37. This underscores that prediction error processing serves as the critical cognitive bridge between associative learning and conscious expectation 37.
Furthermore, the pharmacological reversibility of these effects highlights their distinct but overlapping natures. Placebo analgesia induced purely by expectation can be antagonized by the opioid antagonist naloxone 40. However, conditioned placebo effects are antagonized by naloxone only when the initial pharmacological conditioning utilized an opioid; if the conditioning utilized a nonsteroidal anti-inflammatory drug (NSAID) like ketorolac, the resulting placebo effect is blocked by cannabinoid receptor antagonists instead 40. This indicates that the brain utilizes distinct neurochemical memory traces depending on the pathway of induction.
Boundaries of the Placebo Response
While the neurobiological reality of the placebo effect is undeniable, rigorous scientific inquiry necessitates defining its clinical boundaries. A persistent popular misconception is that placebo effects can halt or reverse autonomous organic disease progression. However, systematic reviews and meta-analyses demarcate a clear boundary between the modulation of subjective symptomatology and objective disease modification 3424344.
Symptom Palliation Versus Disease Modification
The placebo effect primarily influences self-appraised symptoms rooted in sensory, autonomic, and affective pathways - such as pain, nausea, fatigue, depression, sleep disorders, and functional motor limitations 51644. In these domains, the brain's descending modulatory networks have direct access to the neural representations of the symptom, allowing top-down expectations to alter the biological experience.
However, in conditions governed by autonomous, peripheral cellular pathologies - such as the proliferation of neoplasms in oncology or the replication of viral loads in infectious diseases - placebo treatments do not alter the fundamental disease course 3. A comprehensive meta-analysis of 45 phase 3 randomized controlled trials involving 5,684 patients with advanced solid tumors assessed the objective tumor response to placebo administration 45. The pooled overall response rate (ORR) in the placebo arms was exactly 1%, consisting almost entirely of partial responses, while the complete response (CR) rate was 0% 45. Previous independent analyses corroborate this, showing tumor response assessed by World Health Organization criteria in a mere 2.7% of placebo-treated cancer patients 46. While placebos in oncology provide vital palliation for side effects like pain and appetite loss, substantial improvements in objective organic pathology are negligible and expectations of complete spontaneous remission without active treatment are unsupported by data 34546.
Disentangling Neurobiology from Reporting Bias
Within clinical trials, separating true neurobiological placebo effects from statistical and reporting biases remains a severe methodological challenge 4348. Outcomes dependent on continuous, self-reported scales (e.g., a 1-10 pain scale) consistently exhibit much larger placebo responses than binary or objective outcomes 342. This suggests that a significant portion of the observed placebo response in trials is related to compliance bias, the desire to please the investigator, or altered symptom reporting rather than true physiological change 348. Furthermore, the lack of untreated control arms in standard two-arm trials leads to the conflation of the placebo effect with the natural remission of the disease and statistical regression to the mean 848. When clinical trials include a third, no-treatment arm, the isolated true placebo effect is often smaller than the overall placebo response initially suggests, highlighting the necessity of rigorous trial design 4248.
Geographic and Socioeconomic Variations in Clinical Trials
The global landscape of clinical trials has highlighted geographic and socioeconomic variables that profoundly influence the magnitude of the placebo response. Over the past few decades, researchers have observed a steady inflation of placebo response rates, creating significant hurdles for proving drug efficacy, particularly in psychiatric, gastrointestinal, and pain trials 4748495051.
The "United States Placebo Anomaly"
A unique geographic phenomenon has been identified within the United States. An analysis of 84 clinical drug trials for chronic pain medications conducted between 1990 and 2013 revealed that pain inhibition within placebo arms increased dramatically over the 23-year period 52. By 2013, American patients receiving placebos experienced an average 30% reduction in pain levels 52. Crucially, this exploding placebo response was exclusive to trials conducted entirely in the U.S.; comparable trials in Europe and Asia showed no such escalation in placebo efficacy over the same timeframe 52.
Researchers hypothesize that this U.S.-specific inflation is driven by multiple converging factors: the increasing size and duration of American clinical trials, and the unique presence of pervasive direct-to-consumer pharmaceutical advertising 4952. The constant bombardment of media illustrating the profound efficacy of medical interventions systematically raises the societal baseline of patient expectations, feeding into the Bayesian predictive models of trial participants and resulting in stronger top-down placebo responses 52.
Global Relocation of Clinical Trials
Simultaneously, the pharmaceutical industry is experiencing a massive geographic shift in trial execution. A comprehensive 2024 report by IQVIA for the European Federation of Pharmaceutical Industries and Associations (EFPIA) revealed that between 2013 and 2023, the European Economic Area's (EEA) share of global commercial trials plummeted from 22% to 12% 5356. This translates to approximately 60,000 fewer patients accessing trials within the EEA 56. In sharp contrast, China's share of commercial trials more than tripled, rising from 8% to 29% in the same period, driven by favorable regulatory environments and massive patient populations 535657. To mitigate slow recruitment times and lower costs, trial sponsors are increasingly relocating studies away from Western Europe and toward Central and Eastern Europe, Asia, and other less affluent regions 545556.
| Metric | European Economic Area (EEA) | China | United States |
|---|---|---|---|
| Global Commercial Trial Share (2013) | 22% 56 | 8% 53 | Remained dominant / stable 5655 |
| Global Commercial Trial Share (2023) | 12% 56 | 29% 53 | Remained dominant / stable 5655 |
| Trend in Trial Volume (10-Year) | Absolute and relative decline 56 | Exponential growth (Doubled since 2018) 56 | Moderate growth (3% increase in recent starts) 55 |
| Identified Impact on Placebo Response | Stable placebo response rates 52 | Increasing due to shifting socioeconomic metrics 5457 | "Placebo Anomaly" (Rapidly rising response rates) 52 |
Socioeconomic Correlates of the Placebo Response
This geographic shift has directly and measurably impacted the data extracted from placebo control groups. Extensive analyses of randomized controlled trials for psoriatic arthritis and plaque psoriasis demonstrate a statistically significant inverse relationship between a country's socioeconomic status - specifically per capita Gross National Income (GNI) - and the magnitude of the placebo response 5457.
In psoriatic arthritis trials, researchers found that for every $10,000 decrease in GNI, the placebo response rate for primary clinical endpoints (such as the ACR20) increased by 5.7% 5457.
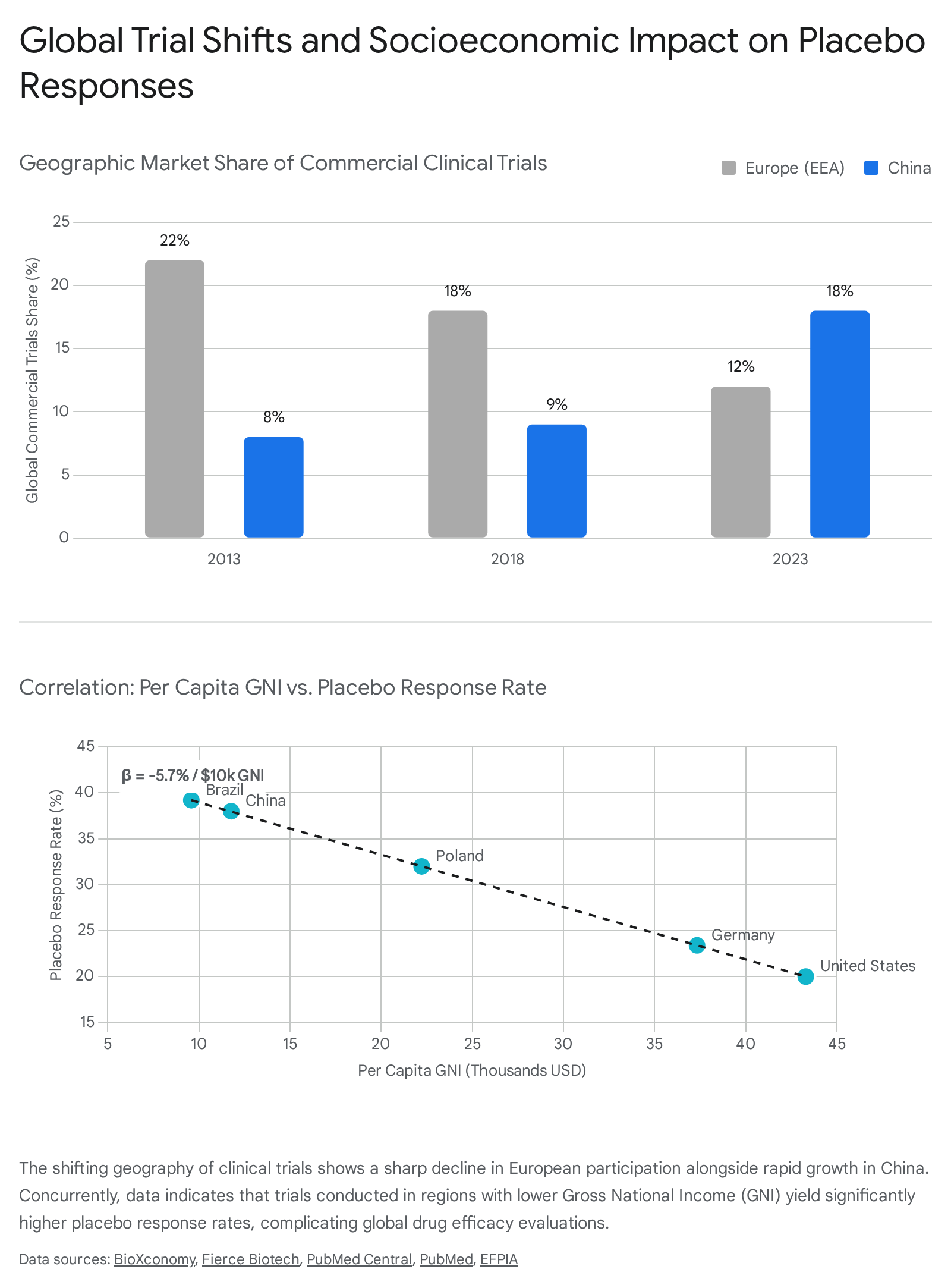
This negative association was consistently replicated across more stringent endpoints like the ACR50 and ACR70 54. In less affluent regions with limited access to innovative healthcare and standard therapeutics, enrollment in a major international clinical trial provides a high perceived benefit and intense clinical monitoring that patients otherwise lack. This heightened expectation of receiving world-class care, combined with the natural regression to the mean after successful enrollment, artificially inflates the placebo response 545758. The global expansion of trial recruitment to less affluent countries actively complicates the assessment of drug efficacy and necessitates adjustments in sample size calculations and statistical models to account for these socioeconomic disparities 5457.
Open-Label Placebos and Clinical Translation
The traditional ethical and clinical assumption that deception is an absolute prerequisite for the placebo effect has been systematically dismantled by recent empirical research, opening new avenues for translational medicine 659. "Open-label placebos" (OLPs) refer to the administration of an inert substance where the patient is fully informed that the treatment contains no active pharmacological ingredients 6. Crucially, this administration is accompanied by a scientifically grounded explanation of how positive expectations, conditioned medical rituals, and the brain's predictive coding systems can nevertheless trigger endogenous healing mechanisms 660.
Systematic reviews and meta-analyses comparing open-label placebos to no-treatment controls reveal statistically significant clinical benefits for a variety of conditions. These conditions are typically those heavily influenced by central sensitization, affective processing, and descending modulation, including irritable bowel syndrome (IBS), chronic low back pain, depression, allergic rhinitis, and attention deficit hyperactivity disorder (ADHD) 6065. The proven efficacy of OLPs demonstrates that the brain's predictive processing networks do not require cognitive deception to be activated; rather, transparent communication, a supportive therapeutic context, and a credible rationale for the intervention are sufficient to adjust predictive priors and instantiate the placebo response 661.
Conclusion
The neuroscience of placebo and nocebo establishes unequivocally that human belief, expectation, and contextual learning exert powerful, top-down control over objective biological networks. Modulated anatomically by the descending pain modulatory system and biochemically by endogenous opioids, dopamine, endocannabinoids, and cholecystokinin, these phenomena alter physiological states in strict accordance with the brain's Bayesian predictive models of the environment.
While the boundaries of these effects are generally limited to the subjective perception and autonomic modulation of symptoms - rather than the reversal of severe organic pathologies like advanced neoplasms - their impact on clinical trial validity and everyday patient care is monumental. As global socioeconomic shifts continue to influence how medical expectations are formed across different populations, recognizing, quantifying, and ethically managing the predictive algorithms of the human brain will be critical. The integration of placebo research into standard clinical practice promises to amplify the therapeutic efficacy of conventional treatments, minimize nocebo-induced adverse events, and utilize the brain's endogenous healing capacity without necessitating deception.