Neurobiology of loneliness and social isolation
Demographic and Epidemiological Indicators
Social interactions represent a fundamental biological imperative required for the survival, somatic maintenance, and psychological stability of mammalian species. Analogous to primary physiological drives such as hunger and thirst, the motivation for social connection operates via highly conserved homeostatic neurobiological systems 12. Disruptions to these social structures - whether through objective social isolation or the subjective perception of isolation (loneliness) - trigger profound neurophysiological shifts that fundamentally restructure brain cytoarchitecture, neurotransmitter availability, and peripheral immune function 34.
Epidemiological surveys across diverse populations underscore the psychiatric and physical burden of social disconnection. Loneliness acts as a potent psychosocial stressor that substantially increases the risk of mortality, cardiovascular disease, stroke, and metabolic syndrome 56. Large-scale biobank data illuminate the scale of this phenomenon. Within the Association of Southeast Asian Nations (ASEAN), data from the Indonesia Family Life Survey (IFLS-5) encompassing 31,447 participants revealed a loneliness prevalence of 10.6%, distributed in a U-shaped curve that peaks during adolescence and late adulthood. In this cohort, chronic loneliness was independently associated with stroke (adjusted odds ratio of 1.58), accelerated cognitive decline, and generalized sleep impairment 678.
Simultaneously, proteomic analyses of blood samples from over 42,000 adults in the UK Biobank demonstrate that objective isolation and subjective loneliness consistently upregulate specific circulating stress proteins, such as adrenomedullin (ADM), which governs cardiovascular responses and regulates neurohypophysial hormones 8. The translation of these macroscopic demographic trends into cellular pathology relies on distinct, temporally segregated neurobiological adaptations. Research highlights a sharp mechanistic contrast between acute social deprivation, which motivates social seeking, and chronic isolation, which induces defensive territoriality, metabolic breakdown, and social aversion 910.
The Conserved Transcriptional Response to Adversity
The biological embedding of perceived social isolation begins in the periphery via profound shifts in systemic gene expression. Chronic psychological threats alter human genome function primarily through a phenomenon defined as the Conserved Transcriptional Response to Adversity (CTRA) 1211.
Genomic Architecture of the CTRA
The CTRA represents an evolved leukocyte gene regulatory program that prepares the immune system for anticipated physical injury. In ancestral environments, transient isolation signaled heightened vulnerability to wounding or bacterial infection, thereby favoring innate immune defense over acquired viral immunity 12. Under conditions of chronic modern social disconnection, however, this pattern promotes maladaptive, low-grade chronic inflammation conducive to neurodegenerative and cardiovascular diseases 12.
Transcriptomic analyses of circulating peripheral blood mononuclear cells in socially isolated populations continuously demonstrate an upregulation of pro-inflammatory transcription factors (e.g., NF-κB, AP-1) and their corresponding target genes (e.g., IL1B, IL6, IL8/CXCL8, PTGS2, and TNF) 121113. Concurrently, the CTRA dictates a marked downregulation of genes involved in Type I interferon-mediated innate antiviral responses (e.g., IFI, MX, and OAS family genes) alongside a reduction in antibody synthesis transcripts 111314. This molecular reconfiguration is orchestrated through beta-adrenergic signaling pathways that transduce sympathetic nervous system (SNS) hyperactivity into downstream alterations of the hematopoietic output of myeloid lineage cells 1115.
Investigations utilizing non-human primate models verify these mechanistic drivers. In rhesus macaques, chronic social isolation induces a specific expansion of the CD14++/CD16- classical monocyte pool, functional glucocorticoid receptor (GR) desensitization, and profoundly impaired physiological responses to viral infections such as the simian immunodeficiency virus (SIV) 1516. In human populations, including high-risk cohorts such as allogeneic hematopoietic cell transplant recipients, elevated pre-transplant CTRA profiles significantly correlate with lower social well-being and heightened risk of post-operative complications 17.
Psychosocial Buffering of Transcriptomic Shifts
The activation of the CTRA is highly sensitive to the subjective interpretation of social environments. Activation of the CTRA is most profoundly linked to subjective loneliness rather than physical isolation metrics 18. When comparing biologically connected versus isolated human groups, subjective social interactions can occasionally register as more stressful than physical isolation for individuals lacking social motivation, indicating that subjective perception governs transcription 18.
Psychological resilience factors - specifically eudaimonic well-being, characterized by a profound sense of purpose, meaning in life, and positive relations with others (PRWO) - exhibit a potent counter-regulatory effect on CTRA activation. Longitudinal studies, including data from 108 older adults in the US Health and Retirement Study, indicate that high eudaimonic well-being downregulates the pro-inflammatory CTRA profile, an effect that persists even when statistically adjusting for baseline loneliness, depressive symptoms, and demographic risks 142119. Furthermore, analyses of Korean adults reveal that PRWO significantly reduces CTRA profiles independent of loneliness, with this buffering effect heavily magnified in individuals possessing a highly collectivistic social orientation 2021. Hedonic well-being (happiness derived from immediate gratification) yields no such protective transcriptomic effect 1213.
| Biological/Psychological Profile | Hedonic Well-Being | Eudaimonic Well-Being (PRWO) | Perceived Social Isolation (Loneliness) |
|---|---|---|---|
| Primary Driver | Immediate self-gratification or positive affect | Purpose, meaning, and positive social relations | Subjective perception of lacking social connection |
| CTRA Gene Expression | Upregulated or neutral | Significantly Downregulated | Significantly Upregulated |
| Pro-inflammatory Genes | Elevated or unbuffered | Suppressed | Chronically Elevated (IL1B, IL6, TNF) |
| Antiviral Defense Genes | Suppressed or baseline | Upregulated | Suppressed (IFI, OAS family genes) |
Neuroimaging and Cognitive Processing Alterations
The subjective dimension of loneliness implies that affected individuals do not merely suffer a deficit of social stimuli; they structurally process identical social stimuli differently than their non-lonely counterparts. The isolated brain exhibits heightened vigilance networks, shifting the interpretation of standard social cues toward hostility and expected rejection 322.
Idiosyncratic Neural Processing
Human functional magnetic resonance imaging (fMRI) studies utilizing naturalistic paradigms (e.g., free viewing of social scenes) reveal that non-lonely individuals exhibit highly synchronized neural responses - measured via Inter-Subject Correlation (ISC) - indicating a shared, normative understanding of the environment 2324. In contrast, lonely individuals process identical stimuli idiosyncratically, displaying highly variable, discordant neural responses 2324.
Higher levels of loneliness correlate directly with lower ISC in regions critical for social cognition, specifically the dorsal medial prefrontal cortex (DMPFC), anterior cingulate cortex, and superior temporal sulcus (STS) 24. Simultaneously, loneliness correlates with augmented activation in regions governing negative affect, such as the insular cortex and amygdala, underscoring an increased sensitivity to social threat 2224.
Functional Connectivity and Gray Matter Volume
Large-scale resting-state functional connectivity analyses further map this hyper-vigilance. In a cohort of 1,336 young adults, greater loneliness scores significantly correlated with stronger functional connectivities between the inferior frontal gyrus (IFG) and the supplementary motor area (SMA), precentral gyrus, and superior parietal lobule - networks heavily implicated in heightened visual attention and environmental monitoring 25.
Structural neuroimaging confirms that chronic loneliness physically alters corticolimbic architecture. Voxel-based morphometry and diffusion tensor imaging (DTI) identify reduced gray matter volume and compromised white matter integrity across the left amygdala, anterior hippocampus, posterior parahippocampus, and specific cerebellar lobules (vermis VI and crus II) 26. Paradoxically, compensatory increases in gray matter volume have been documented in the dorsolateral prefrontal cortex (DLPFC), reflecting sustained cognitive exertion required to navigate perceived social threats 2627.
Cellular Mechanisms of Acute Social Craving
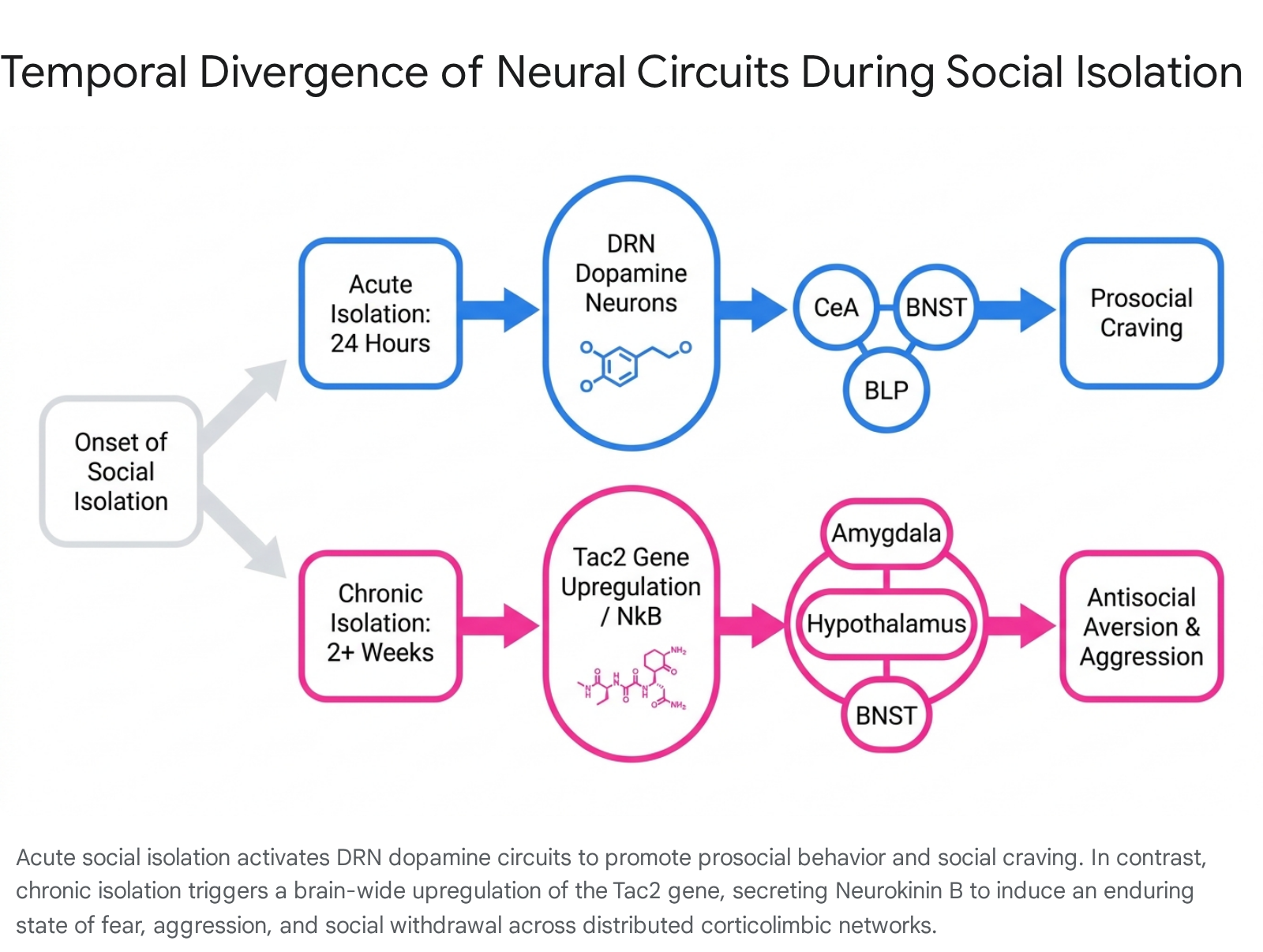
Acute isolation protocols (spanning 24 hours in rodent models) trigger a neurobiological state analogous to nutritional fasting. In human fMRI studies, acute enforced isolation activates midbrain reward centers in response to social cues with the same intensity that a 10-hour fast activates the brain in response to food cues 2829.
Dorsal Raphe Nucleus Dopaminergic Dynamics
At a cellular level, acute "social craving" is governed predominantly by discrete populations of dopamine neurons residing in the dorsal raphe nucleus (DRN). Under normal group-housed conditions, these specific dopamine neurons remain relatively quiescent. Following acute social isolation, they exhibit robust increases in overall neural activity and calcium signaling, coupled with enhanced synaptic strength 1030.
Optogenetic activation of DRN dopamine neurons promotes prosocial approach behaviors, while their chemogenetic inhibition strictly prevents the characteristic rebound of social interaction normally observed following an isolation period 10. These neurons exert their effects through highly collateralized projections to distinct forebrain structures: 1. Central Amygdala (CeA): Photostimulation of DRN projections to the CeA selectively promotes affiliative social behavior, recruiting specific ensembles of CeA neurons tuned to social stimuli 2830. 2. Bed Nucleus of the Stria Terminalis (BNST): DRN projections to the BNST drive general exploratory behavior and environmental sampling 2830. 3. Posterior Basolateral Amygdala (BLP): Projections to the BLP appear to encode the negative affective, aversive component of acute loneliness, driving place-avoidance behaviors and signaling the unpleasant state of social deficit 2830.
Insular Cortex Neuromodulation and Pain
Acute social isolation fundamentally alters the encoding of nociceptive stimuli, physically manifesting the sensation of "social pain." Utilizing Fear of Missing Out (FOMO) behavioral paradigms, studies demonstrate that socially excluded rodents display severely amplified responses to physical pain and disrupted valence encoding 228.
This hyperalgesia is mediated by convergent chemical signals in the insular cortex. Endocannabinoid and oxytocin signaling within the insula exhibit opposing neuromodulatory responses during social exclusion events 28. Intra-insular pharmacological blockade of oxytocin signaling directly exacerbates the organism's response to physical pain following social isolation, establishing the insular cortex as a primary substrate translating social omission into physical distress 228.
Neuropeptidergic Regulation in Chronic Isolation
When social isolation is sustained over a prolonged period (e.g., weeks in rodent models), the homeostatic drive for social reintegration is overridden by a persistent corticolimbic state prioritizing territorial self-preservation. This behavioral shift from prosocial craving to antisocial aggression is driven by distinct neuropeptide networks.
Tachykinin 2 and Neurokinin B Upregulation
The master regulatory switch for chronic social isolation stress is the brain-wide upregulation of the Tac2 gene, which encodes the neuropeptide tachykinin 2, also known as neurokinin B (NkB) 3132. Tac2 expression and subsequent NkB production increase significantly in subjects exposed to two weeks of isolation, but remain unaffected during short-term 24-hour isolation 933.
The NkB peptide acts via a distributed mode of action, binding to the neurokinin 3 receptor (Nk3R) across discrete limbic modules 3234. The specific behavioral dysfunctions induced by isolation depend strictly on the anatomical locus of Tac2 upregulation: * Amygdala: Local Tac2 upregulation drives stimulus-locked persistent fear responses and threat hypersensitivity, yet leaves aggressive behaviors unchanged 3536. * Dorsomedial Hypothalamus (DMH): Tac2 expression in the DMH fundamentally drives isolation-induced hyper-aggression toward unfamiliar conspecifics, without altering fear processing 3536. * Bed Nucleus of the Stria Terminalis (BNST): Tac2 coordinates systemic arousal and anxiety phenotypes. Variable dosages of shRNA-mediated knockdown within the BNST reveal that differing thresholds of Tac2 determine whether an organism exhibits acute versus enduring defensive responses to stress 32.
Validating this mechanism, systemic administration of an Nk3R antagonist fully prevents or rescues the behavioral effects of chronic isolation 3234. Conversely, artificially overexpressing Tac2 and releasing NkB in group-housed, unstressed subjects perfectly phenocopies the specific behavioral pathologies (aggression and persistent fear) of long-term isolation 936.
Prefrontal and Amygdalar Circuit Adaptations
Chronic isolation severely disrupts the balance of excitatory and inhibitory signaling within the prefrontal cortex (PFC) and the basolateral amygdala (BLA) 3738. Prolonged isolation induces profound hypoactivity in the medial PFC (mPFC), evidenced by decreased expression of immediate-early genes and neuronal activity markers such as Egr1, c-Fos, and Arc 3839. Because the mPFC exerts top-down, inhibitory control over the amygdala, its dampening results in the unchecked hyperactivity of the BLA 3840.
Recent optogenetic and circuit tracing studies reveal that the specific routing of these signals dictates the clinical phenotype. Socially isolated subjects rely heavily on an indirect neural pathway - the mPFC → BLA → BNST circuit - to encode isolation-induced affective distress. Chemogenetic inhibition of BLA neurons within this indirect relay rescues anxiety and depression-like behaviors without altering social motivation 41. Conversely, a direct projection - the mPFC → BNST pathway - governs the maintenance of social recognition. Inhibiting this direct pathway selectively restores a subject's ability to recognize conspecifics while leaving innate anxiety levels intact 41.
Sexual Dimorphism in Circuit Response
The molecular alterations within these circuits exhibit stark sexual dimorphism, governing why isolation precipitates varying psychiatric outcomes in males versus females 38.
| Circuit Feature / Gene Marker | Impact of Isolation in Males | Behavioral Outcome in Males | Impact of Isolation in Females | Behavioral Outcome in Females |
|---|---|---|---|---|
| PFC Neuronal Activity | Decreased (Egr1, c-Fos) | Loss of top-down emotional regulation | Decreased (Egr1, c-Fos) | Loss of top-down emotional regulation |
| Basolateral Amygdala (BLA) | Hyperactivity; profound upregulation of CREB-target genes | Heightened territorial aggression | No significant change in baseline neuronal activity | No primary aggression phenotype |
| Ventral Tegmental Area (VTA) | Baseline neuropeptide expression maintained | N/A | Downregulation of Hypocretin/Orexin (Hcrt) | Severe social withdrawal and isolation anhedonia |
In isolated males, increased aggression is causally linked to hyperactive mPFC-to-BLA signaling, primarily mediated by the hyperactivation of the transcription factor CREB within the amygdala 3840. In isolated females, profound social withdrawal is driven by widespread downregulation of the neuropeptide Hypocretin (Orexin) in the ventral tegmental area (VTA), and systemic administration of Orexin-A successfully restores social approach behaviors 3840.
Synaptic Plasticity in the Medial Habenula-Interpeduncular Nucleus Pathway
A critical relay affected by chronic social stress is the dorsal diencephalic conduction system, predominantly the pathway connecting the medial habenula (MHb) to the interpeduncular nucleus (IPN) 4243. Functioning as an evolutionarily conserved conduit between the limbic forebrain and midbrain neuromodulatory centers, the MHb-IPN circuit integrates emotional context to evaluate aversive states and withdrawal 4243.
The circuit operates via a strict topographical organization. The dorsal MHb (dMHb) utilizes Substance P (SP) to innervate the lateral IPN, while the ventral MHb (vMHb) primarily utilizes acetylcholine (ACh) and glutamate to innervate the medial IPN 4347. The IPN exhibits exceptional densities of nicotinic acetylcholine receptors (nAChRs), specifically α3β4 and α5 subtypes. Transgenic tracing of the Chrna5 gene (encoding the α5 subunit) reveals it is expressed exclusively in GABAergic interneurons within the IPN that receive monosynaptic cholinergic input from the vMHb, linking this circuit directly to downstream serotonergic networks 44.
Under conditions of chronic, unpredictable social stress, the entire MHb-IPN axis becomes metabolically hyperactive. In particular, excessive SP release from the dMHb excessively excites IPN neurons, generating profound anhedonia 45. Lesioning the MHb reverses this metabolic overload and behavioral despair 45. Concurrently, specific subregions, such as the superior part of the MHb (MHbS), become heavily activated in anxiogenic environments, utilizing dopamine receptor D3 (DRD3)-positive cell pathways to counteract excessive anxiety .
GABAB Receptor-Mediated Potentiation
At the synaptic level, the MHb-IPN pathway harbors an unprecedented mechanism of presynaptic plasticity intricately linked to stress processing. Unique to the mammalian brain, the activation of inhibitory GABAB receptors at the presynaptic terminals of the vMHb does not suppress transmission; rather, it induces a colossal potentiation of neurotransmitter release 46.
This mechanism involves a rapid transition from low-efficiency tonic release to high-capacity phasic vesicular release. GABAB activation precipitates a 4-fold increase in the size of the readily releasable pool (RRP) of synaptic vesicles and a mirrored physical accumulation of vesicles docked at the presynaptic active zone 46. This transition is orchestrated by two distinct vesicle-associated molecules: synaptoporin, which mediates the baseline augmentation of tonic release, and CAPS2, which is selectively recruited to the active zone to stabilize the massive RRP during high-frequency phasic bursts 46.
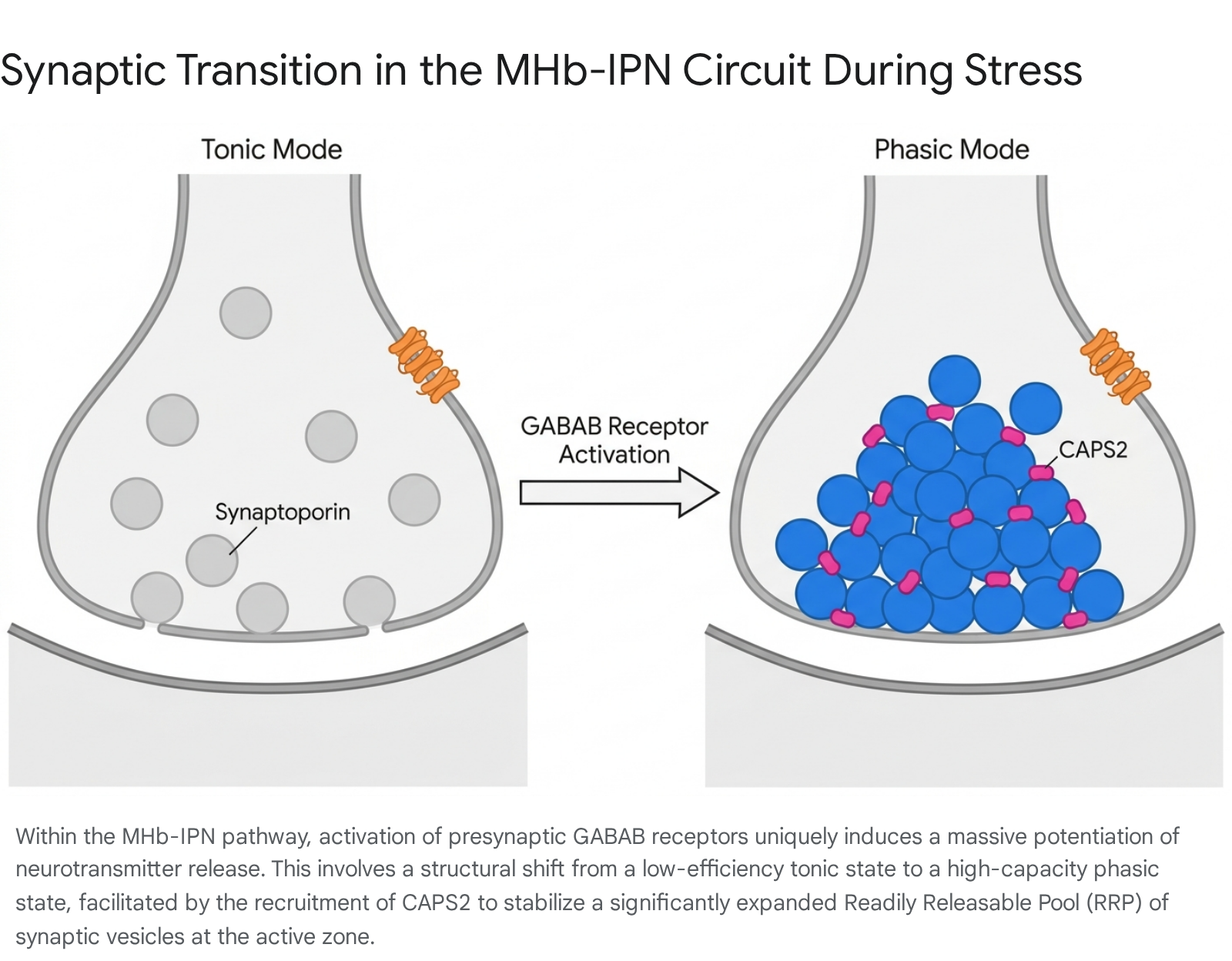
Sustained social isolation places profound strain on this unique synaptic architecture, physically expanding synaptic capacity to engrain aversive social memories.
Epigenetic Embedding of Chronic Social Defeat
To render the effects of chronic social isolation permanent, external psychosocial stressors physically alter the genome without changing the underlying DNA sequence. Epigenetic modifications - specifically DNA methylation and histone modifications - regulate how specific neural networks encode the protracted memory of adversity 4753.
DNA Methylation in Reward Circuitry
Whole-genome bisulfite sequencing in socially defeated or isolated animals reveals massive, genome-wide alterations in cytosine methylation patterns within the nucleus accumbens (NAc) 48. Chronic social stress dictates a highly specific epigenetic profile: an aggregate hypermethylation at CG dinucleotide sites (typically repressing gene transcription) combined with significant hypomethylation at non-CG sites 4849.
Differential methylation targets highly specific regulatory networks. The estrogen receptor alpha (Esr1) gene emerges as a central hub for differentially methylated sites in stress-susceptible subjects, correlating directly with an organism's vulnerability to depression 48. Other highly methylated genes include the deleted in colorectal cancer (Dcc) gene and Cacna1c 49. Bioinformatic network analyses further reveal that stress-induced DNA methylation heavily represses genes implicated in canonical WNT/beta-catenin signaling, a pathway integral to adult neurogenesis and structural synaptic plasticity 4849. The validity of these animal findings is strongly supported by human genomic data; the identical WNT/beta-catenin pathways hypermethylated in isolated mice overlap significantly with genome-wide association studies (GWAS) identifying the genetic risk architecture for high neuroticism in humans 4849.
Histone Modifications in the Prefrontal Cortex
In parallel to DNA methylation, chronic isolation drives robust histone modifications. In the PFC, extended isolation induces the profound methylation of histone H3 at lysine 9 (H3K9me2), an epigenetic mark definitively linked to chromatin condensation and targeted gene repression 39.
Isolation upregulates specific H3K9 methyltransferases, including SUV39H1 and SETDB1, physically tightening the chromatin structure around the enhancer regions of essential activity-dependent genes 39. Consequently, immediate-early genes critical for synaptic plasticity and memory formation - such as Arc, c-Fos, and Npas4 - are silenced, leaving the PFC structurally and functionally impaired 39. Experimental administration of specific H3K9 methylation inhibitors (e.g., UNC0642) re-opens these chromatin domains, rescues Arc transcription, and abolishes isolation-induced anxiety, confirming the causal role of this epigenetic blockade 39.
Additional data indicates that durations of stress dictate epigenetic permanence. Alterations to transcriptionally active histone marks, such as H3K4me3, only mature into highly correlated transcriptomic phenotypes when social isolation is sustained past 15 to 30 days. Short-term isolation (10 days) yields shallow epigenetic landscapes that fail to consistently dictate long-term systemic gene expression 50.
Transcriptomic Signatures in the Brain
In humans, chronic loneliness correlates directly with accelerated epigenetic aging. DNA samples assessed via established epigenetic clocks (e.g., Horvath, DunedinPACE, GrimAge) show that lonely older adults possess a significantly older biological age than their chronological age indicates, contributing substantially to cumulative health morbidities 5751.
Translating these molecular shifts directly to the human brain requires examining post-mortem tissues. Genome-wide RNA expression from the post-mortem nucleus accumbens of 26 deceased donors with documented antemortem loneliness phenotypes revealed 1,710 differentially expressed transcripts. These transcripts mapped heavily to systemic inflammation, disrupted upstream RNA regulators, and organismal injury pathways 52. Similarly, investigations utilizing the dorsolateral prefrontal cortex (DLPFC) of 181 participants tracked longitudinally prior to death uncovered 337 upregulated and 43 downregulated gene sets directly tied to objective loneliness 53. Strikingly, these isolated human transcriptomes demonstrate significant pleiotropy with the biological pathways driving Alzheimer's disease, severe psychiatric illness, and immune dysfunction 2753.
Rodent models corroborate these transcriptomic shifts utilizing high-resolution RNA sequencing. Spatial transcriptomics (Visium) of socially defeated subjects identifies massive dysregulation of synaptic organization genes primarily within the hippocampus, isocortex, and amygdala, with inhibitory interneurons showing the highest degree of transcriptional impairment 54. In the hippocampus specifically, isolation induces profound sexual dimorphism at the RNA level, resulting in 1,215 differentially expressed genes (DEGs) in females compared to 400 DEGs in males, with high convergence on common variant heritability linked to human neurodevelopmental disorders like autism and schizophrenia 555657. Notably, the sequencing of bulk prefrontal cortex tissue in isolation-reared models also points to substantial disruptions in nucleic acid binding, cellular communication, and basic metabolic processes 58.
Methodological Limitations of Tissue Analysis
While transcriptomic data is robust, translating animal models to human pathophysiology requires caution. Animal models often lack genetic heterogeneity and fail to replicate the complex etiology of human psychiatric conditions. Initiatives promoting the Framework for Internal Validity and Model Discrimination (FIMD) emphasize that while animal models accurately predict pharmacological ranges and behavioral directionality, they cannot simulate the subjective cognitive burden unique to human loneliness 59.
Furthermore, relying exclusively on post-mortem human brain tissue introduces analytical bias. Recent single-nucleus RNA sequencing (snRNA-seq) studies directly comparing living brain biopsies (LBP) to post-mortem samples demonstrate massive disparities. While identical cell types are identified in both, a massive proportion of gene expression signatures associated with mental illness and aging in post-mortem tissues are simply artifacts of cell death and tissue degradation, necessitating rigorous computational deconvolution to approximate the actual living brain state 6068.
Glial Cell Dysregulation and Subcellular Pathology
Beyond neuronal alterations, social isolation precipitates severe dysfunction within glial populations and intracellular organelles, leading to a generalized state of neurochemical exhaustion.
Oligodendrocyte and Astrocyte Alterations
Normal social development heavily relies on the synchronized myelination of corticolimbic tracts. Early-life social isolation profoundly depletes the pool of oligodendrocyte progenitor cells (OPCs) and forces their premature differentiation into mature oligodendrocytes 1. This truncated maturation compromises myelin sheath integrity, leading to impaired action potential propagation in tracts connecting the PFC to subcortical hubs. Concurrently, isolation induces pathological astrocyte activation within the locus coeruleus and severely diminishes the hippocampal microglia population, a deficit that can be experimentally reversed to ameliorate depressive-like behaviors 161. Furthermore, single-cell transcriptional profiling demonstrates that specific populations of hippocampal astrocytes are highly enriched for isolation-induced DEGs, proving that non-neuronal cells actively mediate the behavioral pathologies of loneliness 5556.
Mitochondrial Dysfunction and Impaired Autophagy
The perception of prolonged isolation forces the brain into a state of chronic allostatic load, rapidly depleting metabolic reserves. Cellular assays of the PFC, hippocampus, and nucleus accumbens in isolated subjects demonstrate a consistent decrease in general ATP production. This is heavily mediated by the diminished functional activity of mitochondrial respiratory complexes I, II, and IV 562. This metabolic deficiency triggers excess reactive oxygen species (ROS) production, overwhelming endogenous antioxidant systems and inflicting severe oxidative damage to synaptic proteins 562.
Compounding this mitochondrial failure is the suppression of cellular clearing mechanisms. Chronic isolation inherently inhibits neuronal autophagy, an essential quality-control process responsible for degrading misfolded proteins and damaged organelles 62. Molecularly, isolation correlates with a marked reduction in vital autophagic initiation factors (Beclin1 and LC3B-II) alongside a pathological accumulation of the autophagic receptor p62/SQSTM1 62. The cessation of proper autophagic clearance directly disrupts post-synaptic protein density, severely impairing hippocampal long-term potentiation.
Interventions and Translational Perspectives
Because chronic isolation permanently alters the structural interpretation of social value - via Tac2-induced fear, H3K9me2 transcriptional silencing, and mitochondrial depletion - the brain becomes locked into a defensive state that simple social exposure cannot easily reverse 3.
However, targeted molecular and environmental interventions demonstrate efficacy in reversing these specific biological pathologies. Pharmacological administration of mTOR inhibitors, such as rapamycin, artificially induces autophagy and has been shown to fully rescue the synaptic and cognitive deficits induced by social deprivation 62. Epigenetic interventions utilizing targeted methylation inhibitors successfully re-open silenced chromatin to restore behavioral equilibrium 39. From an environmental perspective, robust human-animal interaction (HAI) interventions, such as companion pet ownership, have been shown to significantly buffer the subjective perception of loneliness, with large-scale surveys indicating 80% of pet owners derive clinically measurable reductions in subjective social isolation 6364. Understanding these specific molecular and cellular architectures offers the distinct possibility of combining psychological interventions with targeted biological therapies to dismantle the deep physiological roots of social isolation.