Neurobiology of cognitive aging and dementia prevention
Introduction
The aging of the human brain encompasses a highly complex matrix of structural, cellular, and metabolic alterations. As global life expectancies increase and demographic shifts result in older populations worldwide, distinguishing between inevitable, nonpathological cognitive aging and preventable neurodegenerative disease has emerged as a central focus of neuroscience and public health 12. Cognitive aging is not a uniform process of deterioration. While certain domains, such as processing speed, fluid intelligence, and episodic memory, exhibit gradual natural declines, other cognitive domains like crystallized intelligence, vocabulary, and emotional regulation remain remarkably stable or even improve over the human life course 3456.
Simultaneously, the trajectory toward pathological cognitive decline - most notably Alzheimer's disease and related dementias - has been shown to be highly modifiable. Recent epidemiological models demonstrate that nearly half of all global dementia cases could theoretically be prevented or delayed by aggressively addressing specific lifestyle, environmental, and medical risk factors across the lifespan 78. This comprehensive analysis synthesizes current neurobiological research to delineate the mechanisms of normal brain aging, the profound neuroendocrine transition of the female brain during menopause, the biomarkers differentiating benign anatomical changes from pathological decline, and the clinical efficacy of preventative interventions.
Structural and Cellular Mechanisms of Normal Brain Aging
Morphological and Volumetric Trajectories
Cerebral atrophy is the most prominent macrostructural hallmark of the aging human brain. Normal aging is characterized by gradual volume loss, cortical thinning, degradation of white matter, loss of gyrification, and the widening of sulci alongside ventricular enlargement 12. The volume and weight of the human brain decrease at a steady rate of approximately 5% per decade after 40 years of age, with the rate of decline accelerating acutely after the age of 70 1.
During nonpathological aging, specific tissue compartments degrade at distinct rates. The gray matter volume fraction, which houses neuronal cell bodies and the brain's dense vascular networks, drops from approximately 52.35% in individuals in their 40s to 50.49% in those in their 80s 1. Gray matter atrophy manifests primarily as cortical thinning driven by microstructural changes at the molecular and cellular levels 2. However, it is critical to note that cognitive decline in normal aging is attributed primarily to the disruption of neural circuits, dendrite degeneration, and synaptic loss, rather than the massive, widespread neuronal death that characterizes Alzheimer's disease and other related dementias 29.
White matter, which occupies nearly 50% of total cerebral volume in humans and provides the structural foundation for efficient neurotransmission between cortical and subcortical areas, undergoes even more profound degradation. The white matter volume fraction drops from 47.63% in the 40s to 40.29% by the 80s 110. White matter aging is characterized by demyelination, axon degeneration, and the formation of white matter lesions 2. The degeneration of the myelin-producing oligodendrocytes and the myelin sheath reduces the packing density and structural anisotropy of white matter tracts 11. This degradation triggers inflammatory responses from microglia, an increase in extracellular fluid space, and small vessel disease, which is associated with microbleeds, lacunes, and localized hypoperfusion 21011. Consequently, to accommodate the loss of cortical and subcortical tissue, the ventricular volume fraction expands significantly from 3.22% to 5.66% during the same 40-year span 1.
Neurotransmitter System Alterations
Nonpathological aging is accompanied by highly selective quantitative changes in the pre-synaptic and post-synaptic elements of major neurotransmitter systems, specifically the monoamines (dopamine and serotonin) and acetylcholine 112. These neurochemical shifts correlate directly with observed changes in motor function, mood, and cognitive processing speed.
The dopaminergic system exhibits a significant, steady age-related decline. Dopamine levels in the human brain decrease by roughly 10% per decade from early adulthood 1. Presynaptically, both the absolute levels of dopamine and the number of midbrain dopamine-containing neurons decline by up to 50% at advanced ages in the absence of neurological disease 1213. Postsynaptically, the density of D-2 dopamine receptors declines by 25% to 50% in the striatum, a change that correlates robustly with declines in motor performance and executive cognitive function 121314. This progressive decline in D-2 receptors can be detected during midlife and continues steadily through the lifespan. Conversely, D-1 dopamine receptors in the human brain generally remain stable or exhibit slight increases, potentially as a compensatory mechanism 1213.
The serotonergic system operates differently. The overall concentration of serotonin remains largely stable during nonpathological aging 1213. However, the density of specific serotonin receptors declines significantly. S-1 and S-2 serotonin receptors decrease by 30% to 50% over the lifespan, with structural losses particularly concentrated in the frontal cortex and hippocampus 1213.
Cholinergic pathways, which control wakefulness, learning, short-term memory, and motivation, also experience localized functional degradation. According to the cholinergic hypothesis of memory deficits, dysfunction in this system plays a central role in normative memory decline 114. During normal aging, specific nicotinic acetylcholine receptor (nAChRs) subtypes are eliminated, and general nicotinic binding ability is reduced 1.
| Neurotransmitter System | Normal Aging Trajectory | Comparison to Pathological Aging (e.g., AD, Parkinson's) |
|---|---|---|
| Dopamine | Levels decline ~10% per decade. D-2 receptors decrease 25 - 50% in the striatum. D-1 receptors remain stable or increase 11213. | Parkinson's involves major (>90%) loss of basal ganglia dopamine. Alzheimer's shows smaller, inconsistent additional D-2 receptor loss 1214. |
| Serotonin | Absolute concentration remains stable. S-1 and S-2 receptors decrease 30 - 50% in frontal cortex and hippocampus 1213. | Alzheimer's involves severe loss of serotonin-containing raphe neurons and up to 80% loss of serotonin receptors 12. |
| Acetylcholine | Gradual reduction in nicotinic binding ability and loss of specific nAChR subtypes 1. | Alzheimer's involves highly variable but severe (25 - 90%) loss of choline acetyltransferase (ChAT) and other cholinergic markers 14. |
Synaptic Alterations Versus Neuronal Attrition
A critical distinction between normal aging and pathological neurodegeneration lies in the cellular integrity of the neuron itself. In normal aging, cognitive decline is fundamentally a result of synaptic disruption, altered neurotransmitter binding, and accumulated damage within the cellular environment 9. Neurons, which require massive energy expenditure to maintain electrical activity, become susceptible to mitochondrial impairment. Aging mitochondria exhibit morphological enlargement, fragmentation, membrane depolarization, and increased oxidative damage to mitochondrial DNA (mtDNA) 9.
However, normal aging is not characterized by the extensive, widespread neuronal death seen in Alzheimer's disease and related dementias 9. The normal aging brain maintains a baseline capacity for neuroplasticity and neural network remodeling. This plasticity allows older adults to compensate for regional structural losses by recruiting alternative neural pathways and forming new synaptic connections under conditions of learning or environmental compensation 1115.
Endocrine Aging and the Female Brain
The Menopause Transition as a Neurological Event
Research over the past decade has fundamentally redefined menopause, indicating that the transition is not merely a reproductive phase but a dynamic neuro-endocrinological event that heavily impacts the aging trajectory of the female brain 161718. Brain architecture and the ovaries are intimately linked through the neuroendocrine system; consequently, the sharp decline in ovarian estradiol production during perimenopause triggers profound, systemic neurological changes 19.
Estrogen functions as a primary regulatory brain hormone. It crosses the blood-brain barrier to bind to estrogen receptors distributed throughout the brain, where it triggers protein synthesis, promotes the production of brain-derived neurotrophic factor (BDNF), dials down pro-inflammatory cytokines, and critically regulates cellular glucose metabolism 18. When estradiol levels drop, neurons lose a primary biological signal that drives them to metabolize glucose for energy production 19.
Multi-modality neuroimaging studies - utilizing positron emission tomography (PET), magnetic resonance spectroscopy, and MRI - demonstrate substantial differences in brain structure, connectivity, and energy metabolism across the pre-menopausal, peri-menopausal, and post-menopausal stages 1620. Postmenopausal brains exhibit a reduction in glucose uptake of approximately 30% across large portions of the temporal lobes and regions commonly associated with higher-order cognitive processing 182021. This significant metabolic slowing is the physiological origin of many menopausal symptoms that are frequently misunderstood as purely systemic or psychological. For instance, hot flashes occur because the hypothalamus loses estrogen-mediated thermoregulatory activation, while insomnia, depression, anxiety, and memory lapses reflect temporary functional disruptions across various cortical networks 1719.
Compensatory Adaptations and Pathological Vulnerabilities
Despite the significant metabolic drop in glucose utilization, the female brain demonstrates robust and unexpected compensatory mechanisms. Clinical research highlights that rather than passively deteriorating, the brain increases cerebral blood flow and mitochondrial ATP production in the exact regions where glucose utilization declines 20.
Furthermore, utilizing specialized radioactive tracers, researchers have observed that estrogen receptor density does not decline alongside circulating estrogen. Instead, receptor density actively increases during perimenopause and continues to rise through at least age 65 18. The brain actively upregulates receptors to capture whatever residual circulating estrogen remains, reflecting an adaptive, biological distress response designed to maintain cognitive performance 1820.
For most women, brain biomarkers stabilize post-menopause, and gray matter volume frequently recovers in key regions necessary for long-term cognitive preservation 1620. However, for a vulnerable subset of women - particularly those carrying the APOE-4 genetic variant (the major genetic risk factor for late-onset Alzheimer's disease) - this midlife period of metabolic instability can accelerate the deposition of amyloid-beta plaques 1621. In these high-risk populations, the neuroendocrine transition unmasks underlying vulnerabilities, marking perimenopause as a critical inflection point where interventions such as hormone therapy may be necessary to support lifelong brain health 1718.
Differentiating Normal Aging from Pathological Decline
The Nuanced Role of Amyloid and Tau Pathologies
Accumulating neuropathological evidence indicates that the traditional hallmarks of Alzheimer's disease - extracellular amyloid-beta (Aβ) plaques and intracellular neurofibrillary tangles of hyperphosphorylated tau protein - are not exclusive to demented individuals 2223. Many older adults who remain cognitively intact exhibit high levels of these pathologies upon autopsy or advanced imaging 2223. Consequently, the mere presence of Aβ does not directly dictate cognitive function, precluding a simple binary definition of normal versus abnormal brain aging based solely on protein accumulation 22.
Instead, the distinction lies in the rate of regional tissue loss and the overall morphometric organization of the brain. In healthy individuals, hippocampal volume wanes naturally, but the annual rate of loss is generally less than 1% 24. However, once an individual passes from mild cognitive impairment (MCI) into active Alzheimer's disease, the rate of hippocampal loss accelerates rapidly to 3% to 4% annually, accompanied by severe atrophy in the entorhinal cortex and temporal lobes 2425.
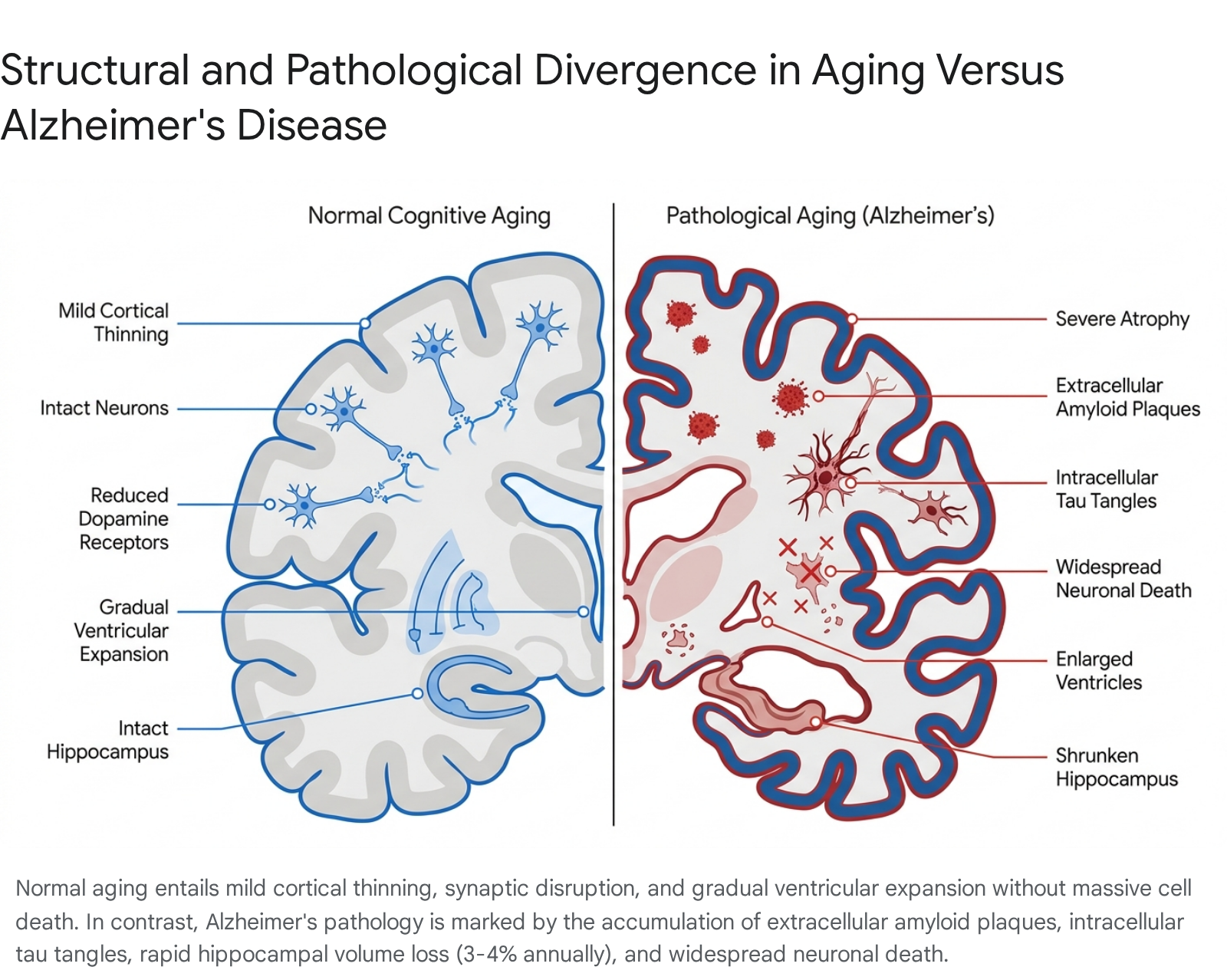
Furthermore, structural analyses reveal that the clinical expression of Alzheimer's disease is uniquely associated with the severe disruption of whole-brain morphometric organization. Normal aging, despite general cerebral atrophy, maintains a much more uniform, preserved global structural pattern 26.
Advancements in Blood Biomarkers for Early Detection
Historically, early detection of preclinical Alzheimer's pathology required expensive, highly invasive procedures such as amyloid PET scans or cerebrospinal fluid analysis via lumbar punctures 2728. Recent clinical breakthroughs have validated the use of plasma phosphorylated tau 217 (pTau217) as a highly sensitive blood biomarker for detecting early disease activity years before symptoms manifest 282930.
In cognitively healthy older adults, elevated baseline levels and rapid longitudinal increases in plasma pTau217 strongly predict a faster accumulation of amyloid and tau in the brain, as well as subsequent cognitive decline 2729. Crucially, prospective cohort studies - such as the Harvard Aging Brain Study following adults aged 50 to 90 over eight years - reveal that increases in plasma pTau217 frequently occur long before amyloid PET scans show clear positive abnormalities 2730.
Data from the AHEAD3 and AHEAD45 preclinical Alzheimer's disease trials, which screened over 15,000 cognitively healthy adults, demonstrated that the prevalence of a positive pTau217 test rises steadily with age, hitting roughly 12% in 55-to-59-year-olds and 35% in those 75 and older 31. As a standalone diagnostic, plasma pTau217 achieves an overall accuracy of 81% in predicting the presence of amyloid-beta pathology in cognitively unimpaired people, a figure that rises to 91% when followed up with a confirmatory PET scan 28. Individuals maintaining consistently low pTau217 levels are highly likely to remain free of significant amyloid accumulation over long-term follow-ups, establishing the biomarker as a highly effective tool for stratifying risk and differentiating benign aging from preclinical dementia 2730.
Cognitive Trajectories: Stability, Improvement, and Decline
While processing speed, executive function, and working memory reliably slow with advancing age, cognitive aging is not universally characterized by decline. The brain maintains specific neurocognitive strengths into late life, relying on accumulated experience and strategic neural recruitment.
Fluid Versus Crystallized Intelligence
Fluid intelligence - defined as the capacity for novel problem solving, pattern recognition, and abstract reasoning independent of acquired knowledge - deteriorates gradually throughout adulthood 3. Conversely, crystallized intelligence, which relies on the accumulation of life experiences, learned facts, and cultural knowledge, is highly resilient 3. Older adults frequently demonstrate superior vocabularies and a deeper semantic understanding of language compared to younger cohorts 34. Crystallized abilities typically remain stable or gradually improve at a rate of 0.02 to 0.003 standard deviations per year well into the sixth and seventh decades of life 3. Visual confrontation naming (the ability to see a common object and instantly name it) remains stable until approximately age 70 before showing gradual decline 3.
Attention, a complex cognitive domain, also exhibits divergent trajectories. Data from the Social Environment and Biomarkers of Aging Study in Taiwan, which tested hundreds of adults aged 58 to 98, revealed that while the "alerting" network (readiness to respond to sudden incoming stimuli) slows with age, the "orienting" network (the ability to efficiently move and direct attention to specific spatial locations) actively and steadily improves from middle age into older adulthood 531. The executive control aspect of attention - the ability to focus on relevant information while successfully ignoring distractions - also improves continually until the mid-to-late 70s before eventually declining 531.
Age-Related Advantages in Emotional Regulation and Wisdom
Normative aging is strongly associated with enhanced emotional stability and improved emotional regulation, a phenomenon heavily mediated by functional shifts in brain activation 63233. Neurological imaging reveals that as individuals age, they respond to negative stimuli with decreased activation of the amygdala (the primitive brain region implicated in automatic fear and stress responses) and increased activation of the medial prefrontal cortex (a region responsible for conscious thought, complex decision-making, and cognitive control) 6.
This neural reorganization allows older adults to exert greater top-down control over negative emotions. This structural shift supports the "positivity effect" - a psychological bias where older adults increasingly focus on and remember positive information over negative events, contributing to higher self-reported levels of affective well-being 633. The Value-Based Cognitive Control Model of Emotion Regulation in ADulthood (VBCC-MERiAD) posits that this emotional improvement relies heavily on the integrity of fronto-striatal circuitry 32. Furthermore, the maintenance of this positive mood is neurobiologically dependent on the preservation of white matter integrity and stable executive functioning, which together facilitate the rapid neural communication required for effective, subconscious emotional processing 34.
Preventable Cognitive Decline and Dementia Risk Factors
The Lancet Commission 2024 Framework
While chronological aging and genetic predispositions (such as the APOE-4 allele) represent inevitable, non-modifiable risk factors, a massive proportion of dementia risk is environmentally, behaviorally, and pathologically mediated. The highly influential 2024 update of the Lancet Commission on dementia prevention, intervention, and care identified 14 potentially modifiable risk factors across the life course. If these 14 factors were entirely eliminated globally, researchers estimate that the overall prevalence of dementia could be reduced by approximately 45% 78.
The 2024 report updated previous epidemiological models by incorporating compelling new clinical evidence, adding untreated vision loss and high levels of low-density lipoprotein (LDL) cholesterol to the established list of threats 78. Interventions targeting these variables are theorized to promote structural cognitive reserve, reduce progressive neurovascular damage, and mitigate chronic neuroinflammation 711. Because risks heavily cluster within individuals - such as socioeconomic deprivation concurrently increasing the likelihood of poor diet, limited education access, and higher exposure to air pollution - preventative clinical interventions and population-level policies must be multi-component and sustained throughout the lifespan 7.
Disparities in Risk Across Geographic and Socioeconomic Contexts
The global burden of modifiable dementia risk is not distributed equally. The potential for absolute risk reduction is significantly higher in low- and middle-income countries (LMICs) and among minoritized or lower socioeconomic groups within high-income countries (HICs) 78. For example, the estimated weighted Population Attributable Fraction (PAF) of preventable dementia cases reaches 42.16% in South Asia, compared to a global low of 23.25% in Western Europe 35.
LMICs suffer from a disproportionately high incidence of foundational risk factors such as low early-life educational attainment, untreated hearing and vision loss, and high exposure to household and ambient air pollution (such as solid fuel combustion) 736. Data comparing populations reveals striking disparities: in Australia, the overall national prevalence of modifiable risk factors sits at 38.2%, yet among First Nations communities, the incidence jumps to 44.9% due to higher rates of diabetes and smoking 38. Similarly, poor regions in Brazil exhibit a 54% potentially modifiable risk factor burden compared to 49.2% in wealthier regions, driven largely by midlife hypertension and lack of early education 38.
Recent epidemiological critiques of the 2024 Lancet report suggest that by failing to formally include specific socioeconomic variables - such as extreme poverty, severe wealth shocks, income inequality, and HIV infection rates - the current framework may underestimate the true modifiable burden in LMICs 37. Some researchers posit that incorporating these broader structural determinants could push the global proportion of preventable dementia cases closer to 65% 3738.
The following table summarizes the 14 modifiable risk factors identified by the 2024 Lancet Commission, categorized by life stage, alongside their respective global Population Attributable Fractions (PAF).
| Life Stage | Modifiable Risk Factor | Global Population Attributable Fraction (PAF) | Primary Neuropathological Mechanism 78353637 |
|---|---|---|---|
| Early Life | Less education | 5% | Limits peak structural and functional cognitive reserve. |
| Midlife (Ages 18 - 65) | Hearing loss | 7% | Increases cognitive load, reduces cortical stimulation, and drives social isolation. |
| High LDL cholesterol | 7% | Accelerates atherosclerotic plaque formation and cerebrovascular disease. | |
| Traumatic brain injury | 3% | Induces acute neuroinflammation and physical axon shearing. | |
| Depression | 3% | Alters cortisol/stress hormone profiles and limits cognitive engagement. | |
| Physical inactivity | 2% | Exacerbates metabolic dysfunction, insulin resistance, and vascular risks. | |
| Hypertension | 2% | Causes small vessel disease, white matter lesions, and hypoperfusion. | |
| Diabetes | 2% | Impairs insulin signaling directly in the brain and accelerates volume atrophy. | |
| Smoking | 2% | Promotes massive oxidative stress and accelerates cerebrovascular damage. | |
| Obesity | 1% | Linked to systemic, chronic inflammation and metabolic syndrome. | |
| Excessive alcohol | 1% | Direct neurotoxicity and accelerated cortical volume loss. | |
| Late Life (Age >65) | Social isolation | 5% | Accelerates cognitive decline through lack of complex neural stimulation. |
| Air pollution | 3% | Fine particulate matter induces severe neuroinflammation and vascular damage. | |
| Untreated vision loss | 2% | Reduces sensory input, compounding cognitive load and isolation. | |
| Total | ~ 45% | Adjusted mathematically to account for communality (overlap) between risks. |
Efficacy of Interventions: Nootropics and Cognitive Training
As public awareness of dementia risk grows, commercial markets have rapidly expanded, offering a plethora of digital tools and pharmaceutical supplements promising to halt cognitive decline. However, the scientific and medical consensus regarding digital brain training and over-the-counter supplements remains highly nuanced, often contradicting commercial claims.
Evaluating Commercial Brain Training Apps
The efficacy of commercial brain training applications (frequently marketed as "digital nootropics") is heavily debated within the neuropsychological community. Peer-reviewed literature generally concludes that while users readily improve at the specific games and puzzles they practice within the app, these improvements suffer from a profound lack of "far transfer" 413940. In practical terms, becoming highly proficient at a digital memory-matching game on a smartphone does not reliably translate to improved working memory in real-world scenarios, nor does it yield a clinically significant reduction in the incidence of dementia 414044. Consequently, several leading commercial brain training companies have faced regulatory penalties from agencies like the Federal Trade Commission for advertising unsubstantiated claims regarding dementia prevention and intelligence enhancement 4044.
However, highly structured, specific forms of clinical cognitive training have shown empirical validity. The landmark Advanced Cognitive Training for Independent and Vital Elderly (ACTIVE) trial - a rigorous 10-year longitudinal study tracking over 2,800 healthy older adults - demonstrated that targeted "speed of processing" training significantly reduced the hazard ratio for dementia risk (HR 0.71) compared to control groups 1541. Interestingly, training focused purely on memory and reasoning did not yield the same statistically significant long-term risk reduction 1541. Ultimately, while the World Health Organization suggests that staying mentally active and engaging in cognitive training is beneficial for maintaining baseline thinking ability and mood, there is no evidence that commercial digital brain games can reverse dementia pathology once it is established 4441.
Over-the-Counter Supplements and Nootropics
Nootropics - often colloquially referred to as "smart drugs" or "cognitive enhancers" - are substances intended to enhance mental performance, focus, and overall neural health 4243. The landscape of nootropics is bifurcated into strictly regulated prescription medications and loosely regulated over-the-counter (OTC) dietary supplements.
Prescription nootropics (such as Modafinil for narcolepsy or methylphenidate for ADHD) operate through validated pharmacological pathways, primarily acting as stimulants that modulate dopamine and norepinephrine 4248. While these can enhance acute alertness, their off-label use by healthy individuals poses significant risks of cardiovascular strain, insomnia, and addiction, and they are not indicated for age-related cognitive decline 48.
Conversely, the clinical efficacy of OTC dietary supplements marketed for brain health is poorly supported by rigorous data. Systematic reviews funded by health authorities conclude that there is insufficient evidence to recommend OTC supplements - including popular herbal extracts like Ginkgo biloba, Bacopa monnieri, ginseng, and ashwagandha - for the prevention of cognitive decline, mild cognitive impairment, or clinical Alzheimer's disease 424849. Most commercial brain supplements fail to address the fundamental biological root causes of cognitive decline 41.
Certain specific nutritional compounds do demonstrate utility, but usually only under highly specific physiological conditions. For example, Omega-3 fatty acids and creatine are backed by some literature for overall metabolic and cellular support when paired with a Mediterranean diet and regular cardiovascular exercise 4950. Furthermore, B-vitamin complexes (including folic acid and vitamin B12) have been shown to improve objective memory test scores exclusively in older adults suffering from preexisting nutritional deficiencies 4950. Nonetheless, authoritative medical consensus asserts that no dietary supplement can satisfy the rigorous criteria of a true neuroprotectant, nor can a pill substitute for the comprehensive lifestyle modifications required to mitigate the 14 vascular and environmental risk factors identified by the Lancet Commission 424850.
Conclusion
The human brain's response to chronological aging is neither uniform nor entirely predetermined by genetics. While structural cortical atrophy, the degradation of white matter tracts, and the down-regulation of dopaminergic and serotonergic receptors represent inevitable physiological mechanisms of the aging process, they do not universally mandate severe functional cognitive failure. Rather, the aging brain exhibits profound structural plasticity and adaptive resilience. This is clearly demonstrated by the compensatory shifts in cerebral blood flow and ATP production during the female neuroendocrine transition of menopause, the lifelong preservation and improvement of crystallized intelligence, and the neurobiological adaptations in the medial prefrontal cortex that actively enhance emotional regulation and wisdom in late life.
Crucially, the progression from benign cognitive slowing to pathological neurodegeneration is highly susceptible to medical and behavioral intervention. The accumulation of amyloid-beta and tau proteins does not operate in a vacuum; it is heavily influenced by systemic health. By aggressively recognizing and mitigating the 14 modifiable risk factors - ranging from hypertension, high LDL cholesterol, and diabetes in midlife to social isolation, air pollution, and untreated sensory loss in late life - nearly half of all global dementia cases could theoretically be delayed or prevented entirely. As advanced blood biomarkers like pTau217 make early, preclinical detection a reality, future public health frameworks and individual clinical strategies must pivot away from unsupported commercial supplements. Instead, the focus must shift decisively toward equitable, population-level risk reduction, addressing the socioeconomic disparities that currently drive the heaviest burdens of preventable cognitive decline.