Neural mechanisms of time perception and aging
Chronoception, the subjective experience of the passage of time, is a fundamental dimension of human cognition that enables the brain to organize sequential events, anticipate future outcomes, and construct a cohesive narrative of the self. Unlike other perceptual domains, such as vision or audition, the brain lacks a dedicated sensory organ for time. Instead, temporal perception is an emergent property derived from highly distributed neural networks that process intervals across diverse scales, ranging from milliseconds to decades 123. This structural reliance on internal neural states and cognitive resources renders psychological time highly malleable. This malleability is particularly evident across the human lifespan, where the subjective speed of time appears to accelerate continuously as individuals age 456. Resolving the mechanisms behind temporal processing requires synthesizing computational models of interval timing, the neuroanatomical substrates of memory and interoception, and the progressive biological regression of neural networks.
Foundational Models of Interval Timing
The dominant frameworks in cognitive neuroscience categorize the brain's timing mechanisms into dedicated central clock models and intrinsic, state-dependent population models 567. These theoretical models seek to explain how the brain computes duration without an external chronometer, highlighting distinct computational strategies depending on the duration of the interval and the sensory modality involved.
Scalar Expectancy Theory and Centralized Clocks
For decades, the standard computational framework for time perception has been the Scalar Expectancy Theory (SET), which models the brain as possessing a centralized, dedicated internal clock 68. The SET framework is built upon a pacemaker-accumulator model consisting of three distinct cognitive stages: a clock stage, a memory stage, and a decision stage 8910. In this model, an internal pacemaker emits pulses at a regular frequency, governed by an idealized Poisson process. When a temporal interval begins, a neural switch closes, allowing these pulses to flow into an accumulator 810. The total pulse count within the accumulator provides a linear metric of elapsed time, which is then transferred to working memory and compared against a reference duration stored in long-term memory 810.
A critical feature of SET is its adherence to the scalar property, an application of Weber's law to time perception. This property dictates that the standard deviation of time estimates grows linearly with the mean estimated duration, meaning the coefficient of variation (the ratio of variability to time) remains constant across scales 68. While SET provides a robust explanation for the cognitive evaluation of prolonged durations, psychophysical evidence indicates that the scalar property breaks down at specific temporal thresholds, suggesting that SET alone cannot account for all temporal processing 6. Specifically, the variability-to-time ratio increases significantly when intervals exceed the 1.2- to 1.5-second range, indicating a transition in the underlying neural mechanisms 6. Furthermore, SET's reliance on a singular, modality-independent accumulator has been challenged by findings that temporal processing heavily recruits modality-specific sensory networks depending on the stimulus 613.
Population Clocks and Intrinsic Neural Trajectories
In contrast to the centralized pacemaker model, state-dependent network models, frequently termed "population clocks," propose that time is an intrinsic, emergent property of the dynamic patterns within recurrent neural circuits 81115. The distinction between these two architectures is profound: whereas SET relies on the linear accumulation of discrete pulses through abstract cognitive switches, the population clock model encodes time through the reproducible geometry of high-dimensional neural manifolds 51213. According to this framework, temporal information is encoded in the evolving state of a neural population rather than accumulated as a scalar value 1214. As neural activity propagates through a highly interconnected network, the instantaneous firing rates of the population trace a distinct path, or "neural trajectory," through a state space 121415.
Because the network's dynamics are reproducible, the specific position of the neural state along this trajectory at any given moment serves as a unique temporal signature 816. For example, in rhythm-based tasks such as predictive tapping, neural populations in the primate medial premotor cortex generate circular, regenerating trajectories. The geometric features of these trajectories, such as their amplitude and speed of rotation, directly encode the length of the produced interval 132117. Linear readouts from these curved trajectories allow downstream brain regions to extract precise temporal estimates without requiring a dedicated clock 1318. Recent analyses have demonstrated that these trajectories often occupy a low-dimensional manifold within a broader, high-dimensional state space, allowing the brain to store information robustly against the flow of time while simultaneously retaining the timing information necessary for coordinating behavior 1618.
Sensory Modality and the Processing Threshold
The neurobiological debate between central clocks and population models is increasingly resolved by segmenting time perception into distinct temporal domains: sub-second (short) and supra-second (long) intervals. Research demonstrates that the perception of brief intervals relies heavily on sensory-specific neural architecture, while longer intervals require widespread cognitive resources and working memory networks 613.
| Feature | Sub-Second Processing (< 1.2 seconds) | Supra-Second Processing (> 1.2 seconds) |
|---|---|---|
| Dominant Theoretical Model | Population Clocks / State-Dependent Models | Scalar Expectancy Theory (Central Clock) |
| Primary Neural Network | Modality-specific (e.g., Auditory, Somatosensory cortices) and Sensorimotor areas | Distributed Central-Executive Network, Prefrontal Cortex, Parietal regions |
| Cognitive Load | Automatic, highly perceptual | Attentionally demanding, memory-dependent |
| EEG Markers | P250 component (deviance detection) | N400 component (cognitive updating) |
| Modality Specificity | High (temporal information embedded in specific sensory circuitry) | Low (amodal, utilizing general executive resources) |
Electroencephalogram (EEG) analyses involving vibrotactile stimuli confirm that deviations in sub-second timing elicit the P250 component, a marker generated within modality-specific networks 613. In these instances, recurrent activity within the somatosensory cortex functions as a localized population clock 6. Conversely, supra-second deviations elicit the N400 component, indicative of higher-order cognitive processing and distributed non-sensory networks, including the inferior frontal gyrus and anterior prefrontal cortex 613. Notably, recent studies have questioned the role of the Contingent Negative Variation (CNV) as a universal marker of temporal accumulation, noting its absence during tactile timing tasks despite its presence in auditory paradigms, further emphasizing the modality-specific nature of short interval timing 613.
Neuroanatomical Substrates of Temporal Processing
Timing is not centralized to a single "master clock" in the brain. Even the suprachiasmatic nucleus (SCN) of the hypothalamus, traditionally viewed as the master circadian pacemaker, operates alongside autonomous, independent oscillators distributed throughout peripheral tissues and brain regions, such as the histaminergic neurons of the tuberomammillary nucleus 192021. The orchestration of interval timing on the scale of seconds and minutes depends on the coordinated activity of specific subcortical and cortical structures.
Hippocampal and Entorhinal Time Cells
The encoding of episodic memory requires a robust representation of sequential temporal context. The medial temporal lobe (MTL), specifically the hippocampus and entorhinal cortex, achieves this via populations of "time cells" 2223. Initially discovered in rodents and recently confirmed in human microelectrode recordings from epilepsy patients, time cells fire sequentially at specific, circumscribed moments during a delay interval 222425. By tiling an entire delay period, these cellular ensembles create an internal timeline that allows the brain to bridge temporally discontiguous events and maintain temporal order during memory retrieval 2425. Human time cells exhibit theta-phase precession during encoding, a mechanism where action potentials occur at progressively earlier phases of the underlying theta rhythm, directly correlating with successful temporal order recall 2223.
The medial entorhinal cortex (MEC), conventionally associated with spatial navigation via grid cells, also plays a profound role in generating these temporal sequences. Experimental paradigms employing chemogenetic inactivation reveal that MEC activity is strictly necessary for learning context-dependent interval timing behaviors 26272829. Rather than acting as rigid stopwatches, MEC time cells display context-dependent activity; their firing patterns change and adapt as an organism learns new temporal contingencies, suggesting the MEC generates flexible "timelines" tailored to specific experiences via continuous attractor networks 2629. Complementing the discrete firing of time cells, the lateral entorhinal cortex (LEC) contains "ramping cells" that exhibit slowly evolving, monotonic changes in firing rates across minutes, establishing a macro-scale temporal context 2223. When retrieving episodic sequences, the hippocampus operates allocentrically, maintaining a perspective-agnostic representation of temporal distance, whereas the posterior parietal cortex retrieves these sequences egocentrically, based on the optimal perspective for the current task context 30.
Striatal Dynamics and Dopaminergic Pathways
The basal ganglia, particularly the striatum, form a critical hub for processing interval timing, largely mediated by dopaminergic transmission 103137. Striatal medium spiny neurons (MSNs) encode time through ramping activity, accumulating temporal evidence toward a decision threshold. Optogenetic tracking demonstrates that the two primary striatal output pathways contribute to timing through opposing dynamics: D1-receptor expressing MSNs (the direct pathway) systematically ramp down their firing rates across an interval, whereas D2-receptor expressing MSNs (the indirect pathway) systematically ramp up 31.
Despite these diametrically opposed neural trajectories, the disruption of either pathway yields similar behavioral deficits. Pharmacological blockade or optogenetic inhibition of either D1 or D2 receptors reliably slows the internal estimation of time, expanding the duration of produced intervals and impairing temporal precision 31. This suggests that temporal control over action requires an interdependent balance between excitatory and inhibitory dopaminergic pathways, functioning as a four-parameter drift-diffusion model where both pathways continuously accumulate temporal evidence 31. The disruption of either pathway degrades the accuracy of trial-by-trial temporal decoding from these ensembles independently of general motor impairments 31.
Interoception and the Insular Cortex
Beyond event tracking and interval estimation, the conscious "feeling" of time passing - psychological duration - is deeply tethered to bodily states. The anterior insular cortex serves as the primary interoceptive center of the brain, continuously integrating afferent signals from the body, including respiratory rates, cardiac rhythms, and autonomic arousal 13832.
Theoretical models posit that subjective time perception is not an abstract cognitive computation but an embodied sensory experience 138. The insula tracks the ongoing physiological changes of the organism, generating a stream of "global emotional moments" that typically span processing windows of roughly three seconds 3832. Functional neuroimaging confirms that activity in the insular cortex is strongly correlated with subjective time estimation tasks; the insula activates at the onset of a timed event, sustains elevated perfusion throughout, and sharply drops off at the event's conclusion, effectively acting as an interoceptive stopwatch 133. Conscious regulation of these interoceptive rhythms, such as deliberate slow nasal breathing, can mechanically entrain oscillations in the olfactory bulb that propagate to limbic structures, stabilizing neural trajectories and altering the subjective perception of duration 15.
Cellular Energy Dynamics and Neurovascular Coupling
Underpinning these complex network dynamics is a strict reliance on cellular energy metabolism. Neurons demand high levels of energy to generate action potentials and maintain synaptic plasticity, primarily relying on oxidative phosphorylation and the tricarboxylic acid (TCA) cycle 3435. Conversely, surrounding astrocytes predominantly engage in glycolysis, providing lactate to neurons via the astrocyte-neuron lactate shuttle 343536. The glutamate/glutamine cycle, which is essential for excitatory neurotransmission, is tightly coupled to this nonoxidative glucose consumption 36. This intense metabolic demand forms the basis of functional magnetic resonance imaging (fMRI) studies of time perception, where neurovascular coupling triggers local elevations in cerebral blood flow in response to the metabolic requirements of active neural populations 4437. As organisms age, the efficiency of these metabolic pathways profoundly degrades, establishing the foundation for systemic shifts in temporal perception.
Biological Regression and the Acceleration of Time
One of the most robust psychological phenomena is the subjective acceleration of time with aging. Older adults consistently report that days, months, and years appear to pass significantly faster than they did in youth 46. This phenomenon cannot be explained by conscious clock-tracking; instead, it represents a secondary perceptual consequence of biological regression across multiple physiological systems.
The Metabolic Model of Age-Related Time Compression
The "metabolic model" of chronoperception provides a biophysiological explanation for age-related time acceleration, framing it as a direct downstream effect of cellular energy decline within the memory systems of the brain 456. Subjective time, when evaluated retrospectively over months or years, is heuristically inferred by the density and vividness of episodic memories encoded during that period 35.
According to the metabolic model, the process follows a distinct causal chain.
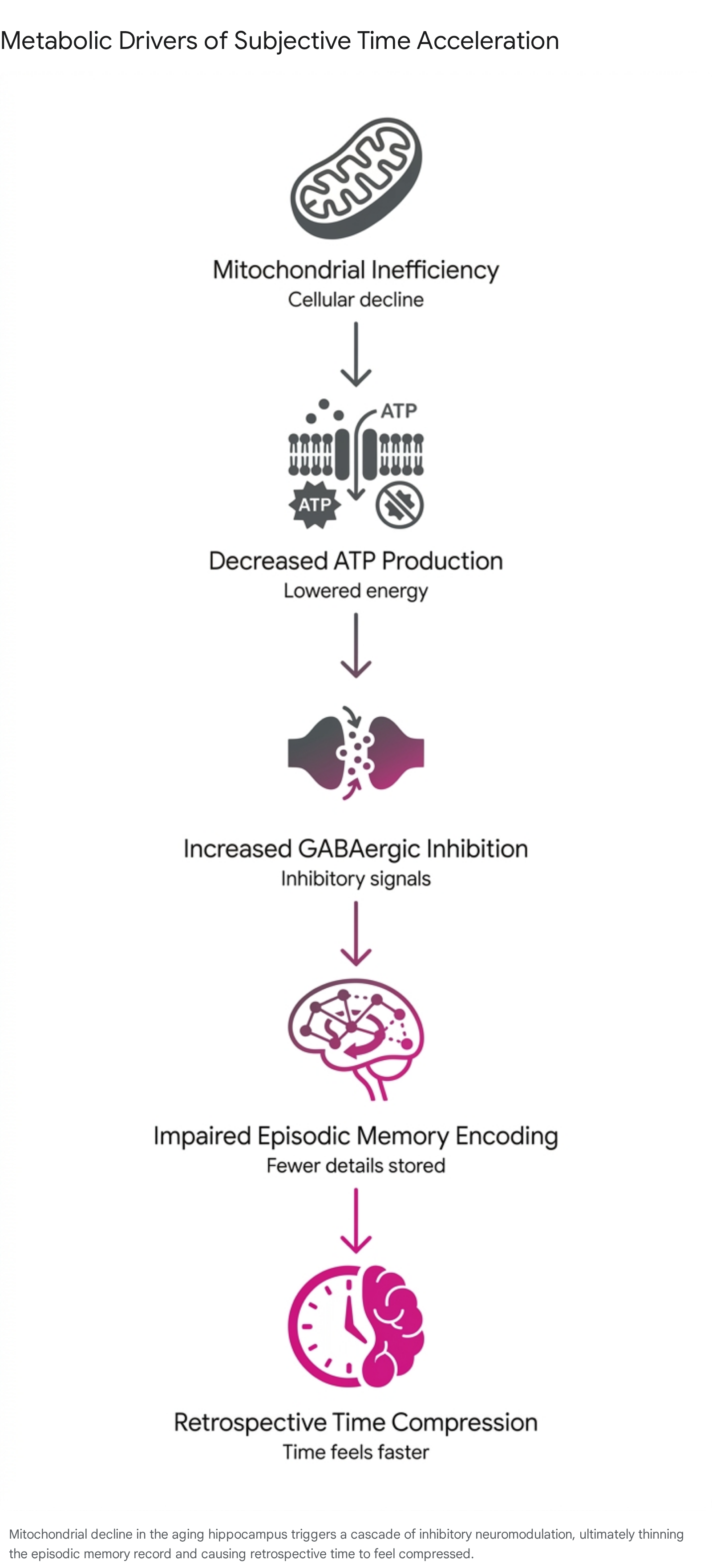
First, normal biological aging induces a progressive decline in mitochondrial efficiency, leading to a reduction in adenosine triphosphate (ATP) production within hippocampal neurons 5. Because synaptic plasticity and the consolidation of novel memory engrams are highly energy-dependent, the neurons face an energy deficit. In response to this metabolic stress, the brain upregulates the production of gamma-aminobutyric acid (GABA), the primary inhibitory neurotransmitter, to suppress overall neural excitability and protect against excitotoxic damage 45.
While this GABAergic shift preserves basic cellular integrity, it raises the threshold for robust episodic encoding 5. Consequently, the aging brain lays down fewer, less detailed episodic records per objective unit of time 5. When an older adult retrospectively evaluates a past epoch, the cognitive system accesses a sparse, thinly populated memory record. Because the brain uses event density as a proxy for elapsed duration, it infers that the period must have been brief, producing the unsettling psychological illusion that life is accelerating 5. Furthermore, contextual reconstruction becomes less precise as memories from different periods share indistinguishable features, weakening the distinctiveness necessary to establish prolonged chronological timelines 5.
Dopamine Degradation and Pacemaker Slowing
Paralleling changes in hippocampal metabolism, age-related degradation of dopaminergic pathways heavily impacts moment-to-moment interval timing. Neuroimaging evidence demonstrates that the availability of striatal dopamine transporters (DAT) declines linearly in healthy adults by approximately 6.6% per decade 38. This progressive loss of dopaminergic tone alters the speed of the internal pacemaker mechanism. Because dopaminergic activity via D1 and D2 receptors determines the rate at which temporal evidence is accumulated, a slower clock rate generates fewer internal neural signals during a standardized physical event 1031. Consequently, external durations are systematically underestimated by older individuals, contributing to the feeling that objective events pass more rapidly than expected 103137.
Nonlinear Aging Bursts
Recent longitudinal studies measuring systemic biological age confirm that aging is not a perfectly linear trajectory. Instead, it proceeds through non-linear bursts, with notable molecular shifts occurring roughly around ages 44 and 60 39. These inflection points involve systemic changes to carbohydrate and lipid metabolism, cardiovascular health, kidney function, and immune regulation 39. These dramatic metabolic shifts compound the energy deficits required for neural maintenance, potentially creating discrete eras where the acceleration of subjective time feels particularly acute 39. Furthermore, metrics of biological aging, such as phenotypic age acceleration, correlate significantly with systemic inflammation, oxidative stress, and increased monocyte activity, all of which drive cognitive decline and further impair the brain's capacity to maintain accurate temporal orientation 40.
Oculomotor Dynamics and Saccade-Induced Chronostasis
Changes in the way older adults physically sample the visual environment also heavily influence temporal phenomenology. The human visual system actively explores environments through rapid, ballistic eye movements known as saccades, occurring naturally multiple times per second 41. During a saccade, the brain suppresses visual processing to maintain stability, a phenomenon known as saccadic omission, which routinely disrupts the continuity of perception 50.
Visual Sampling and the Illusion of Chronostasis
To compensate for the interruption of visual input during a saccade, the brain employs a post-dictive perceptual illusion known as "saccadic chronostasis." Upon landing a saccade, the perception of the immediate post-saccadic fixation is subjectively extended backward in time, making the initial visual event feel longer than it physically was 4243. This illusion essentially bridges the gap caused by the eye movement, providing the observer with an uninterrupted sense of reality. Furthermore, research demonstrates that when a second event follows immediately after the initial post-saccadic fixation, its perceived duration is subjectively compressed 4243. This indicates that saccades induce a highly uneven temporal attentional gradient, systematically overestimating the immediate present while underestimating the adjacent sequence of events 4243.
Age-Related Declines in Saccadic Frequency
Crucially, the dynamics of saccadic exploration are profoundly altered by advanced age, cognitive load, and fatigue. The frequency of saccadic eye movements declines in older populations, driven by general motor slowing, reduced excitability of motor neurons, and the increased cognitive effort required to execute complex visual searches 44454647.
| Oculomotor Parameter | Age-Related Change | Impact on Temporal Processing |
|---|---|---|
| Saccade Frequency | Decreases | Fewer instances of chronostasis; less frequent micro-extensions of perceived time. |
| Saccade Latency | Increases | Slower reaction to stimuli; delayed updating of the internal temporal record. |
| Vertical Saccade Velocity | Decreases | Reduced speed of information integration; slower visual sampling rate. |
| Pupillary Dilation | Increases (higher effort cost) | Indicates increased cognitive load, which diverts resources away from temporal tracking. |
The reduction in saccade frequency holds direct implications for time perception. Because each saccade introduces a micro-distortion in time estimation - routinely expanding the perceived present via chronostasis - a lifetime reduction in saccade frequency means older adults experience significantly fewer of these temporal expansions 4244. The cumulative lack of these micro-extensions, combined with a generally reduced perceptual sampling rate, contributes to the overarching sensation that standard physical time passes uninterrupted and increasingly fast 4244. Moreover, the allocation of cognitive resources toward the increased effort of executing saccades (quantifiable via effort-linked pupil dilation) detracts from the central executive resources necessary for tracking elapsed supra-second intervals, leading to further temporal compression under heavy cognitive load 464849.
Cognitive and Cultural Frameworks of Time
While the neurobiological constraints of time perception are shared across the human species, the cognitive application of time is heavily shaped by cultural and linguistic environments. Time is inherently inferential; without a specialized sensory organ, societies depend on external structures, spatial metaphors, and communal narratives to segment events 350.
Event-Time Versus Clock-Time Orientations
A major division in temporal cognition occurs between "clock-time" and "event-time" cultures. In industrialized societies, standardized clock-time serves as a rigid external scaffold 505152. This system treats time as an autonomous, mathematical entity that proceeds independently of human activity. The mechanical clock dictates the initiation and cessation of tasks, demanding high levels of temporal coordination. However, this rigid dependence on arbitrary intervals obscures dependencies among events and generates phenomena like the "weekend illusion," where standardized routines erase episodic distinctiveness, making long-term retrospective time appear condensed 505152.
| Characteristic | Clock-Time Orientation | Event-Time Orientation |
|---|---|---|
| Pacing Mechanism | External standardized metrics (seconds, hours) | Internal task completion and natural transitions |
| Cultural Prevalence | Industrialized, Western societies | Southern European, Indigenous, and Agricultural societies |
| Cognitive Approach | Monochronic, rigid, linear sequencing | Polychronic, synchronous, contextual tracking |
| Neurological Parallel | Absolute interval tracking (amodal networks) | Episodic, sequential updating (time cells) |
Conversely, event-time cultures structure the passage of time according to the natural progression of activities and social interactions 515253. In such societies, including certain Amazonian tribes (e.g., Amondawa) and various agricultural communities, time is not an empty container in which things happen; rather, the event itself constitutes the time 35154. A new activity only begins when the current one reaches its natural, affective, or social conclusion. This reliance on event density directly parallels the brain's internal heuristics for duration estimation - where subjective time expands based on the volume of distinct memories formed - suggesting that event-time orientations are more closely aligned with the brain's native "mind time" than the arbitrary metrics of mechanical clocks 35055.
Mental Time Travel and Spatial Metaphors
The construction of time is ultimately intertwined with the evolutionary development of Mental Time Travel (MTT) - the cognitive capacity to project the self backward to relive autobiographical memories and forward to simulate possible futures 5055. Neuroimaging studies reveal a nearly identical overlap in the default mode network and medial temporal lobe when recalling the past and imagining the future, underscoring that memory is not a veridical recording device but an adaptive, constructive system geared toward prospective planning 50.
The directionality and organization of MTT are heavily influenced by language and cultural metaphors. While Western languages universally map the future to the space in front of the body and the past behind it, reflecting the physical experience of forward locomotion, alternative mappings exist. For instance, speakers of the Aymara language conceptualize the past as in front (because it is known and can be "seen") and the future as behind (because it is unknown) 50. These linguistic structures physically shape the mental timelines that individuals use to organize autobiographical memories 50. As people age and autobiographical narratives solidify into routine, the failure to generate distinctly novel episodes limits the raw material available for MTT. This reduction in narrative distinction further compresses the retrospective timeline, cementing the subjective acceleration of time 550.
Synthesis of Temporal Processing and Aging
The human perception of time is a fragile, inferential construct maintained by overlapping neurobiological mechanisms operating across multiple scales. In the immediate present, temporal reality is dictated by sensory-specific population clocks, the rotational dynamics of neural trajectories in premotor areas, and the ramping activity of dopaminergic pathways within the striatum. Concurrently, the insular cortex monitors interoceptive signals to ground these intervals into a conscious feeling of duration.
However, the long-term perception of time relies on the retrospective analysis of episodic memory, orchestrated by time cells within the hippocampus and entorhinal cortex. The pervasive feeling that time accelerates with age is the inevitable consequence of a biological system experiencing metabolic decline. As mitochondrial energy wanes and inhibitory GABAergic tone increases to compensate, the brain's ability to encode rich, distinctive episodic markers deteriorates. Paired with a lifelong degradation in dopamine transporter availability, reduced oculomotor sampling rates that eliminate micro-extensions of the present, and a societal reliance on repetitive, standardized clock-time, the aging brain is left to retrieve an increasingly sparse record of existence. When assessing a biologically impoverished memory record, the cognitive system naturally infers a shorter duration, resulting in the profound psychological illusion that life is speeding to its conclusion.