Neural mechanisms of olfaction and its link to emotion and memory
The olfactory system represents one of the most ancient, structurally distinct, and highly plastic sensory networks in the mammalian central nervous system. Unlike visual, auditory, and somatosensory processing - which rely on the thalamus as an obligatory preliminary relay to the cerebral cortex - primary olfactory signals bypass the thalamus entirely to project directly into the limbic system. This divergent neuroanatomical architecture establishes the biological foundation for the uniquely profound relationship between odor, emotion, and autobiographical memory.
Peripheral Olfactory Transduction and Neurogenesis
The initial processing of olfactory information begins in the nasal cavity, where volatile chemical stimuli are transformed into electrical impulses. This sequence is initiated within the olfactory epithelium, a specialized neuroepithelial tissue containing primary olfactory sensory neurons (OSNs).
Odorant Binding and the Molecular Cascade
Odorants inhaled from the environment dissolve into the nasal mucus and bind to specific odorant receptors (ORs) located on the fine cilia of OSNs 123. In mammals, the OR gene family is exceptionally large and diverse; mice, for example, express over 1,000 distinct OR genes 13. Each OSN expresses only one specific type of OR gene, establishing a highly precise receptor-to-neuron functional mapping 1.
The binding of an odorant to a receptor activates a localized, rapidly amplifying molecular cascade. Odorant receptors are G protein-coupled receptors. Upon the binding of a volatile molecule, the associated G protein dissociates and activates adenylyl cyclase type III (ACIII) 14. This enzyme catalyzes the synthesis of cyclic adenosine monophosphate (cAMP), drastically elevating intracellular cAMP levels 14. The accumulation of cAMP subsequently opens cyclic nucleotide-gated (CNG) ion channels, allowing an influx of positive ions (such as calcium and sodium) that depolarizes the OSN cell membrane 124.
This amplification cascade is highly efficient, allowing the production of an electrical quantal event from the binding of a single odorant molecule 2. Furthermore, this level of the transduction process is where physiological adaptation occurs; the initial robust response of an OSN to an odorant stimulus is rapidly followed by a period of reduced responsiveness, filtering out constant background odors 2.
Embryonic Development of the Olfactory System
The basic circuit of the mammalian olfactory system is one of the most precocious sensory systems to develop during embryogenesis. The system relies on the protomap hypothesis, suggesting that the development of olfactory structures is heavily intrinsically regulated while also relying on the sequential arrival of projection fibers 5.
| Developmental Stage (Mouse Model) | Key Morphological and Neurological Events |
|---|---|
| Embryonic Day 9.5 (E9.5) | Formation of the olfactory placode; initiation of neurogenesis in the presumptive olfactory epithelium. |
| Embryonic Day 10.0 (E10) | Generation of the first neurons within the nascent olfactory cortex. |
| Embryonic Day 11.0 - 12.0 (E11-E12) | Olfactory sensory neuron (OSN) axons coalesce to form a distinct olfactory nerve; lateral olfactory tract (LOT) fibers begin to emerge. |
| Embryonic Day 13.0 - 13.5 (E13-E13.5) | LOT fibers cover the surface of the olfactory cortex; OSN axons enter the olfactory bulb to form initial synapses. |
During these earliest stages of primary olfactory pathway formation, the olfactory epithelium and the olfactory bulb undergo simultaneous, yet independent, developmental programs 6. However, as embryonic development progresses and axons from the epithelium innervate the nascent bulb, their developmental programs become functionally interrelated 6.
Neural Stem Cell Maturation and Postnatal Neurogenesis
The mammalian olfactory epithelium possesses the rare capacity for continuous neurogenesis during adulthood. Precursor cells continuously divide and differentiate into mature OSNs under physiological conditions throughout the organism's lifespan 7. This process is mirrored centrally in the subventricular zone (SVZ) and the subgranular zone (SGZ) of the dentate gyrus, where quiescent adult neural stem cells (NSCs) generate neurons that integrate into olfactory and hippocampal circuitry 89.
The transition of proliferating developmental NSCs to a quiescent adult state is not an abrupt cellular switch but a protracted, multi-step molecular cascade defined by specific metabolic milestones 810. Targeted single-cell RNA-sequencing of the early postnatal mouse dentate gyrus reveals that NSCs undergo a transition to quiescence followed by further functional maturation 810. A critical metabolic milestone preceding the acquisition of quiescence is a distinct burst in autophagy (cellular self-degradation and recycling) 810. If this autophagic burst is pharmacologically or genetically disrupted, the NSC transition to quiescence is impaired, jeopardizing the adult stem cell pool required for continuous neurogenesis 89. Subsequent maturation phases are marked by elevated levels of cellular reactive oxygen species (ROS), fatty acid oxidation, and mitochondrial pyruvate metabolism 89.
Presynaptic Maturation and Synaptogenesis
As newborn OSNs project their axons into the olfactory bulb to establish functional synapses, they follow a tightly regulated, intrinsic developmental program. The expression of presynaptic molecules occurs sequentially, establishing discrete steps in presynaptic development independent of actual physical contact with target dendrites in the bulb 7.
In situ hybridization studies demonstrate that scaffolding proteins are expressed first. Messenger RNAs for bassoon, piccolo, syntaxin 1A, and its interacting partner Munc18-1 show the earliest onset during the OSN lineage, co-localizing with MASH1 (a cellular marker for OSN progenitors) 7. Conversely, genes encoding proteins involved in the regulation of vesicle release and actual neurotransmission emerge much later. The signature molecule for glutamatergic neurons, Vesicular Glutamate Transporter 2 (VGLUT2), shows the latest onset of expression, marking the final stage of presynaptic maturation prior to functional signal transmission 7.
Anatomical Connectivity and Central Pathways
Following transduction, action potentials travel along the olfactory nerve to the first central relay station. The structural processing of these signals defines how broad chemical representations are narrowed into specific odor object identities.
Signal Convergence at the Olfactory Bulb
Inside the olfactory bulb (OB), OSN axons synapse onto the dendrites of second-order neurons - mitral and tufted cells - within spherical neuropil structures known as glomeruli 135. The organization of the OB exhibits precise spatial segregation; axons from all OSNs expressing the same specific OR converge onto a single, corresponding pair of glomeruli 13.
Because individual ORs can be stimulated by multiple odorants with varying affinities, and a single odorant can activate multiple OR types, odor information is topographically represented as a unique spatial pattern of activated glomeruli 13. This combinatorial coding allows the olfactory system to discriminate among thousands of distinct odorant molecules 3. Unlike the somatosensory cortex, which exhibits patchwork-like synchronized firing patterns (barrel maps) during development, spontaneous activity in the primary olfactory system generally lacks spatiotemporal correlation 1. Instead, cell-type-specific temporal activity patterns and heterogeneous lateral inhibition across different OR classes dictate the refinement of the olfactory map 111.
Direct Limbic Projections and the Primary Olfactory Cortex
The defining characteristic of the olfactory neural circuit is its direct monosynaptic projection to limbic and archeocortical structures. Mitral and tufted cells project their axons via the lateral olfactory tract (LOT) directly to the primary olfactory cortex, bypassing the thalamus 561213. The primary olfactory cortex is not a singular region but a collection of interconnected areas, primarily the anterior olfactory nucleus (AON), the olfactory tubercle, the piriform cortex, the amygdaloid complex, and the entorhinal cortex 5612.
Within these regions, functional differentiation is highly specific. Functional magnetic resonance imaging (fMRI) studies in humans and tracing studies in rodents indicate that the anterior piriform cortex (aPir) reflects sensitivity to odor valence (pleasantness or unpleasantness) and specific behavioral aversions 1314. For example, the aPir is robustly activated during retching-like behaviors triggered by aversive odorants, sending direct signals to the nucleus accumbens and lateral hypothalamus 14. Conversely, the temporal or posterior piriform cortex (pPir) primarily codes for odor quality, categorical perception, and similarity between complex odor mixtures 1314.
The direct routing of signals to the amygdala (central to emotion and threat detection) and the entorhinal cortex (the primary gateway to the hippocampus) allows the olfactory system to trigger affective and mnemonic responses rapidly, prior to conscious cognitive appraisal 15161718. Neural selectivity to specific odorants increases progressively from the broadly tuned AON through the entorhinal cortex and into the hippocampal subfields (CA1 and subiculum), transforming unrefined chemical representations into specific, experience-dependent associative memories 19.
Secondary Processing and the Mediodorsal Thalamus
While initial olfactory processing bypasses the thalamus, secondary pathways actively engage it, primarily through the mediodorsal thalamic nucleus (MDT) 12202122.
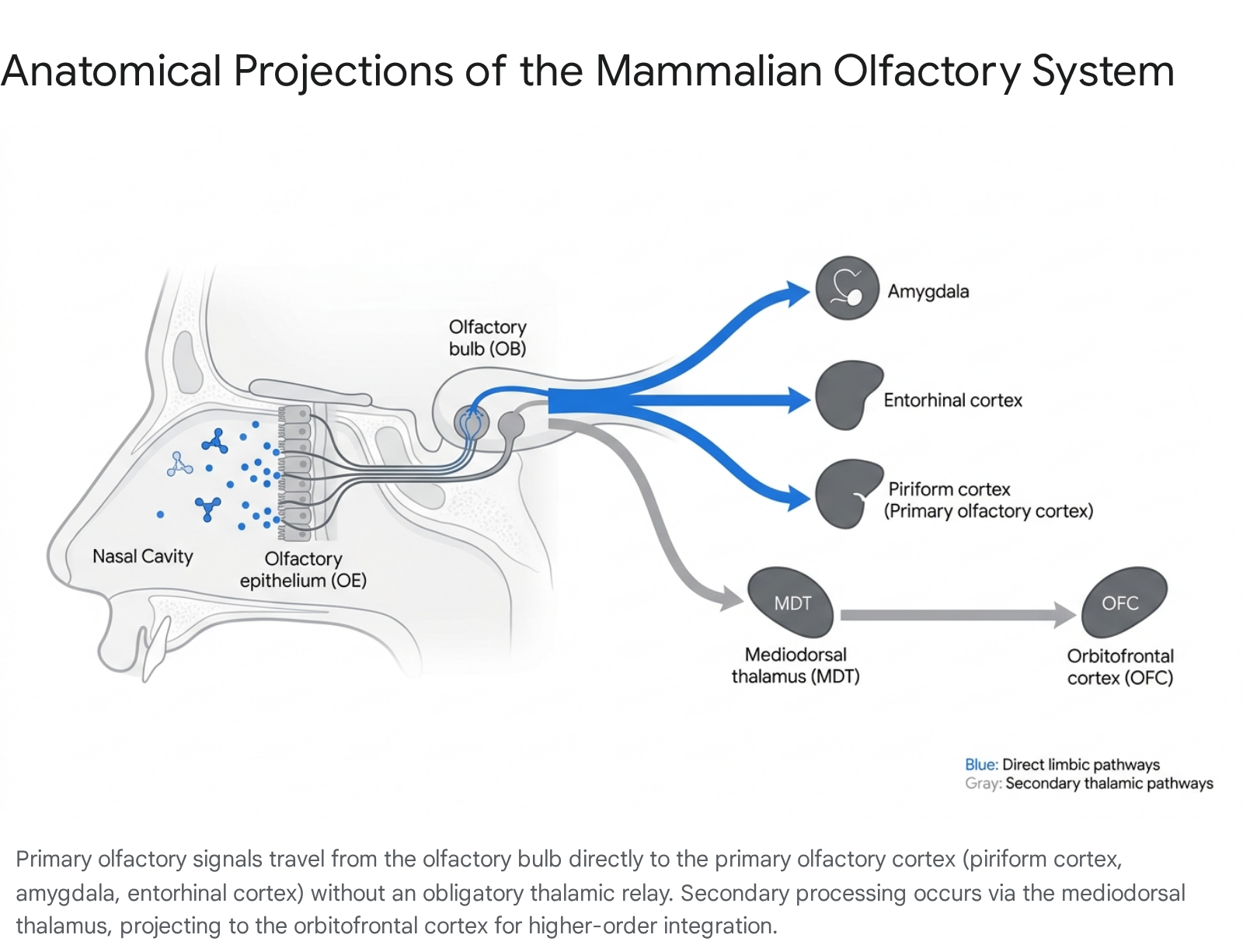
The MDT receives dense, asymmetric input from the primary olfactory cortex, amygdala, and insula, and maintains reciprocal "matrix" projections with the orbitofrontal cortex (OFC) and prefrontal cortex (PFC) 212324.
Clinical stroke data and targeted lesion studies demonstrate that the MDT is not strictly necessary for basic conscious odor detection or the instantiation of basic odor representations 2025. Complete anosmia does not result from isolated MDT lesions 20. Instead, the MDT serves as a critical connector hub for higher-order olfactory cognition, directing sensory attention and facilitating decision-making by providing feedforward information regarding stimulus type and value 2225.
Specifically, the MDT is vital for complex olfactory behaviors including odor discrimination, olfactory identification, and the processing of olfactory hedonics 202122. Patients with MDT damage report a markedly decreased ability to perceive the hedonic value of experienced odors and odor-taste mixtures 22. In behavioral models, MDT inactivation severely disrupts cognitive flexibility. While rodents with MDT lesions can learn initial odor-reward associations and perform simple reversals, they are profoundly impaired when required to execute extradimensional (ED) shifts - tasks requiring rapid, within-trial updates to choice response strategies based on changing olfactory rules 25. Furthermore, electrical stimulation of the human MDT predominantly elicits visceral and olfactory conscious experiences, underscoring its role in integrating visceral signals with higher-order cortical evaluation 24.
| Processing Tier | Primary Anatomical Nodes | Functional Contributions | Thalamic Involvement |
|---|---|---|---|
| Primary (Direct) | Piriform Cortex, Amygdala, Entorhinal Cortex | Rapid affective evaluation; threat detection; initial episodic memory encoding; basic valence coding. | Bypassed. Direct monosynaptic input from Olfactory Bulb. |
| Secondary (Indirect) | Mediodorsal Thalamus, Orbitofrontal Cortex, Prefrontal Cortex | Conscious odor identification; sensory attention modulation; cognitive flexibility (ED shifts); complex hedonic valuation. | Central relay hub. Integrates primary cortex signals and provides feedback. |
The Proust Phenomenon and Odor-Evoked Autobiographical Memory
The specific phenomenon wherein a scent evokes a highly vivid, sudden, and involuntary autobiographical memory is widely referred to in cognitive literature as the "Proust phenomenon," honoring Marcel Proust's literary depiction of a madeleine biscuit eliciting a flood of childhood recollections 262728. Behavioral, physiological, and neuroimaging studies confirm that odor-evoked autobiographical memories (OEAMs) operate via distinct neurocognitive mechanisms compared to memories cued by visual or auditory stimuli.
Behavioral and Temporal Characteristics
Empirical data acquired through double-cuing methodologies (where participants are presented with an odor's verbal label followed by the actual odor) reveal that OEAMs possess unique qualitative traits. Memories recalled in the physical presence of an odor are consistently rated as significantly more emotional, more arousing, more self-relevant, and eliciting a stronger sensation of "mental time travel" than memories recalled via visual or verbal variants of the same cue 1826282930.
Furthermore, the temporal distribution of OEAMs is heavily skewed toward early developmental stages. Standard autobiographical memory distributions (the "reminiscence bump" for word or picture cues) typically peak for events occurring between ages 11 and 25 1631. In contrast, the distribution for odor-cued memories peaks much earlier, for events occurring between the ages of 6 and 10 1631. This early-life encoding preference is hypothesized to stem from the precocious developmental maturation of the olfactory system and its unmediated connectivity with the developing limbic system during childhood 31.
The privileged mnemonic power of odors remains robust even in clinical populations. In individuals with Major Depressive Disorder (MDD) - a condition characterized by overgeneralized memory recall and deficits in autobiographical specificity - odor cues successfully elicit a significantly higher percentage of specific, episodic memories compared to verbal cues, completely bypassing the verbal memory deficits typical of the disorder 3233.
Neural Suppression and Hemodynamic Responses
Functional magnetic resonance imaging (fMRI) has delineated the complex, multi-stage neural networks engaged during OEAMs. The retrieval process requires an intricate balance of localized neural activation and targeted suppression.
When participants engage in episodic odor recognition memory (ORM) tasks, the successful recognition of a familiar odor is correlated with widespread neuronal suppression in regions including the anterior insula, posterior piriform cortex, amygdala, and inferior parietal lobule 3435. This immediate sensory suppression is thought to act as a filtering mechanism, reducing sensory noise to allow for the isolation of the memory trace 34.
Following this initial recognition phase, a secondary network encompassing the hippocampus and the posterior cingulate gyrus engages in a post-recognition process 3435. Dynamic functional connectivity (dFC) analyses utilizing finite impulse response (FIR) models reveal that OEAMs trigger a sequence of four distinct hippocampal networks: the posterior default mode network, the anterior default mode network, a salience network integrating the olfactory cortex, and finally a dorsal attention network 35. The engagement of the left fusiform gyrus and the left posterior orbitofrontal cortex directly correlates with the subjective strength of the emotion and the vividness of the "time travel" sensation induced by the OEAM 2936.
Machine learning predictive modeling (utilizing SHapley Additive exPlanations) further isolates the drivers of OEAMs. These models demonstrate that the emotional strength of an odor - particularly highly unpleasant odors - is the primary predictor of successful baseline odor recognition 17. However, the actual associative retrieval of the episodic context is driven by a non-linear relationship with familiarity: completely unfamiliar or highly familiar odors drastically increase the likelihood of remembering the associated context, whereas moderately familiar odors fail to trigger episodic retrieval 17.
Involuntary Memory Retrieval and Emotion Regulation
Involuntary autobiographical memories (IAMs), such as those triggered by the Proust phenomenon, share retrieval mechanisms with phenomena like déjà vu. Both are considered spontaneous cognitions resulting from the spreading of activation within an internal associative network, moving from a cue representation directly to related concepts in the autobiographical memory system without executive search initiation 3738.
Because odor-cued IAMs bypass executive control, they present unique challenges and opportunities for emotion regulation. The Process Model of Emotion Regulation suggests that unwanted, intrusive emotional memories (a hallmark of posttraumatic stress disorder, or PTSD) can be modulated by altering cue distinctiveness 3739. Interestingly, while individuals with PTSD report more frequent and intense involuntary memories than controls, fMRI analyses reveal that the underlying neural network activated during involuntary retrieval remains structurally similar between the groups 40. The primary difference lies in temporal dynamics: individuals with PTSD exhibit significantly delayed neural responsivity in the parahippocampal gyrus and precuneus during involuntary memory retrieval, alongside heightened ventromedial prefrontal cortex (vmPFC) activity for negative stimuli, indicating an altered temporal processing of spontaneous threat cues 40.
Real-Time Physiological Modulations
The profound emotional impact of olfactory stimuli translates rapidly into measurable peripheral physiological changes. Odors directly engage the hypothalamic-pituitary-adrenal (HPA) axis and the autonomic nervous system (ANS) 4243.
Exposure to pleasant odors significantly increases heart rate variability (specifically HRV MAD), an effect not observed during exposure to equally pleasant auditory stimuli (music) 43. Real-time salivary biomarker profiling demonstrates that specific fragrance profiles elicit immediate, distinct molecular signatures. Highly pleasant and relaxing odors are associated with an increased S2/S1 salivary ratio of oxytocin alongside decreased cortisol levels 41. Conversely, distinct biomarker combinations (e.g., elevated alpha-amylase) emerge in response to stress-related odors 41. These peripheral molecular signatures confirm that olfactory stimuli manipulate autonomic states faster and more deeply than modalities relying on higher-order cortical translation.
Olfactory Dysfunction as a Neurodegenerative Biomarker
Because the olfactory system relies on continuous peripheral neurogenesis and direct, unmediated access to limbic structures, it is extraordinarily vulnerable to early neurodegenerative pathology. Olfactory dysfunction (OD) - ranging from hyposmia (reduced sensitivity) to anosmia (complete loss) - is recognized as one of the earliest prodromal symptoms of neurodegeneration, frequently appearing years or decades prior to the onset of cardinal motor or cognitive symptoms 424344.
Pathology in the Alzheimer's Disease Continuum
In Alzheimer's disease (AD), OD is present in up to 90% of patients 45. The impairment in AD is primarily observed in higher-order olfactory tasks, specifically odor identification and odor discrimination, rather than basic threshold detection 1543.
Within the ATN (Amyloid, Tau, Neurodegeneration) diagnostic framework, declines in olfactory identification precede detectable cognitive impairments, emerging during the stages of subjective cognitive decline (SCD) and mild cognitive impairment (MCI) 15. At a structural level, OD in the AD continuum is closely linked to volumetric atrophy in the medial temporal lobe (MTL), specifically the entorhinal cortex, hippocampus, and amygdala 154950. This structural decline parallels the chronological accumulation of tau-neurofibrillary tangles and amyloid-beta (Aβ) aggregates, which initially deposit in the MTL before propagating outward into the primary olfactory cortices 4550. Furthermore, cognitively unimpaired older adults carrying the APOEε4 allele (a major genetic risk factor for AD) exhibit significantly lower olfactory identification abilities than non-carriers, positioning behavioral olfactory testing as a highly sensitive, non-invasive screening tool for early tau pathology 15.
Pathology in the Parkinson's Disease Spectrum
In Parkinson's disease (PD), olfactory dysfunction is similarly prevalent (affecting 50% to 90% of patients) but exhibits a markedly different pathological and behavioral trajectory 46. According to the Braak hypothesis, misfolded α-synuclein protein aggregates initially deposit in the olfactory bulb and the enteric nervous system, establishing induction sites before spreading retrogradely through the brainstem into the cerebral cortex 4647.
Behaviorally, PD patients demonstrate severe deficits in basic odor threshold and detection tasks, distinguishing idiopathic PD from vascular parkinsonism or essential tremor 43. Longitudinal assessments, such as those utilizing the BioFINDER-1 cohort and the Brief Smell Identification Test (B-SIT), reveal that hyposmia in PD correlates strongly with elevated cerebrospinal fluid (CSF) levels of Glial Fibrillary Acidic Protein (GFAP), an indicator of glial activation and neuroinflammation 46. Critically, PD patients exhibiting both olfactory dysfunction and amyloid-positivity (defined by a low CSF Aβ42/Aβ40 ratio) experience a drastically accelerated rate of cognitive decline and possess a nearly 4.5-fold increased hazard ratio for progressing to full dementia compared to normosmic peers 46.
Transdiagnostic Models and Glymphatic Clearance
To disentangle the overlapping presentations of hyposmia, recent whole-brain voxel-based morphometry studies separating AD and PD cohorts have proposed a transdiagnostic "double hit" model 4950. This model establishes two distinct mechanisms underlying neurodegenerative hyposmia: 1. A process linking medial temporal lobe (MTL) degeneration strictly to cognitive/memory-based olfactory disturbances (characteristic of AD). 2. A separate process linking direct olfactory bulb atrophy to movement disorder phenotypes (characteristic of PD) 4950.
Emerging research also implicates the failure of the brain's glymphatic clearance system in these early olfactory pathologies. The glymphatic system facilitates the movement of brain fluids and the removal of metabolic waste primarily during sleep 45. The nasal pathway has recently been identified as a major outflow exit for cerebrospinal fluid 45. It is hypothesized that age-related glymphatic dysfunction impedes the clearance of misfolded proteins (Aβ, tau, α-synuclein), leading to their localized accumulation in the olfactory bulb and entorhinal cortex. This localized bottleneck triggers early neuroinflammation in the olfactory mucosa, driving preclinical olfactory decline long before widespread cortical neurodegeneration occurs 45.
| Disease Spectrum | Pathological Protein | Primary Olfactory Region Atrophied | Specific Olfactory Functional Deficit | Secondary Biomarker Correlations |
|---|---|---|---|---|
| Alzheimer's Disease (AD) | Amyloid-β, Tau tangles | Medial Temporal Lobe (Entorhinal Cortex, Hippocampus, Amygdala) | Severe impairment in odor identification and episodic discrimination. | APOEε4 allele presence; accelerated subjective cognitive decline (SCD). |
| Parkinson's Disease (PD) | α-synuclein | Olfactory Bulb | Severe impairment in basic odor threshold and detection. | Elevated CSF GFAP; rapid progression to dementia if amyloid-positive. |
Cross-Cultural Variances in Olfactory Perception
Historically, Western science has characterized the human olfactory system as underdeveloped or inferior to that of other mammals (microsmatic). This assumption was largely based on the observation that English speakers, and Westerners generally, face profound difficulty when attempting to explicitly name or categorize odors 4849. However, contemporary cross-cultural linguistics and perceptual research have overturned this framework, revealing that while the physiological mechanics of smell are universal, the cognitive categorization and communication of olfaction are heavily modulated by cultural context.
Lexical Categorization and Abstract Odor Language
In industrialized Western cultures, the olfactory lexicon is highly impoverished. English lacks a dedicated lexical field for smell; speakers typically rely on concrete, source-based descriptors (e.g., "it smells like lemon" or "it smells like smoke") 4950. When asked to identify familiar scents, Western participants demonstrate very low inter-rater agreement and prolonged response times (averaging 17 seconds), frequently attempting to reconstruct a visual source or contextual situation to map the aroma 4950.
Conversely, ethnographic studies of Aslian-speaking hunter-gatherer populations in the Malay Peninsula and Southeast Asia - specifically the Jahai and Maniq communities - demonstrate the existence of highly elaborated, dedicated abstract odor lexicons 484951. The Jahai language contains approximately 12 monolexemic, stative odor verbs, while the Maniq language contains 15 4852. These terms abstract away from specific sources, defining odor qualities directly. For example, the Jahai term ltpit describes a specific olfactory quality shared by bearcats, certain flowers, durian, and soap, in the exact same manner an English speaker uses the abstract visual term "red" to describe both a fire engine and an apple 49.
Under experimental conditions, Jahai speakers depart radically from Western baselines. When presented with monomolecular odorants, Jahai participants name the odors utilizing their abstract vocabulary with exceptionally high inter-rater agreement and rapid response times (averaging 2.7 seconds) 4950. This fluency demonstrates that the human brain's capability for olfactory abstraction is not neurologically constrained by a weak connection between the limbic system and language processing centers, but is instead culturally conditioned by environments that demand high olfactory vigilance 4849.
Dimensionality and Universality in Odor Valence
While the linguistic codification of odors varies drastically, the underlying semantic dimensions used to organize these lexicons - and the raw affective valuation of the odors themselves - show remarkable cross-cultural consistency.
In languages with dedicated olfactory vocabularies, perceptual spaces are organized primarily along a dimension of valence (pleasantness), with a secondary dimension often related to dangerousness, toxicity, or edibility 5152. Despite the vast differences in how the Jahai, Maniq, and Dutch conceptualize smells linguistically, facial expression analyses reveal that all groups exhibit identical initial, involuntary emotional reactions to specific odorants 4950.
Large-scale global studies involving 235 individuals from 9 diverse non-Western cultures and industrialized urbanites further confirm the biological universality of odor valence. When ranking the hedonic value of monomolecular odorants, researchers observed substantial global consistency 53. Frequentist and Bayesian statistical models demonstrate that the molecular identity of an odorant explains 41% of the variance in individual pleasantness rankings, whereas cultural background accounts for a mere 6% 53.
This data reconciles the intersection of biology and culture in the olfactory system: human olfactory perception is fundamentally constrained by universal, structure-based principles at the receptor level that dictate an immediate affective response, while the higher-order cognitive abstraction and linguistic communication of that response are sculpted entirely by cultural frameworks 5053.