Network neuroscience of cognition and disease
For over a century, the prevailing paradigm in clinical neuroscience and cognitive psychology leaned heavily upon localizationism - the theoretical premise that specific, circumscribed regions of the cerebral cortex are exclusively responsible for distinct cognitive functions. Rooted in early lesion studies, such as the localized mapping of Broca's area, and reinforced by half a century of single-electrode neurophysiological recordings, this framework dominated the biological understanding of the mind 123. However, the advent of advanced non-invasive neuroimaging methodologies and high-throughput computational modeling has precipitated a profound paradigm shift toward network neuroscience. This modern discipline views the human brain as an intricate, highly dynamic connectome - a vast topological map of neuronal elements and interconnecting pathways that continuously integrate and segregate information 134.
Contemporary measurements generated from high-throughput neuronal activity tracking suggest that cognitive variables are reflected broadly across the brain, including within regions traditionally believed to be strictly engaged in controlling movement or sensory processing 25. Network neuroscience has revealed that complex behaviors, fluid intelligence, and psychiatric pathologies do not reside in isolated brain tissue. Instead, they emerge from the synchronized activation and deactivation of distributed neural networks 256. By abstracting the physical brain into mathematical graphs composed of nodes (representing anatomical regions or individual neurons) and edges (representing structural tracts or functional correlations), researchers can quantitatively decode the global and local architecture of human cognition 7810. This analytical abstraction has unlocked unprecedented capabilities in predicting real-world cognitive performance and has provided a neurobiological substrate for understanding neurodegenerative and psychiatric disorders, which have historically eluded precise, biomarker-driven clinical diagnosis 9101112.
Foundations of Network Neuroscience
To decode the organizational principles of the brain, network neuroscience relies on a dual-framework classification of connectivity: structural and functional. While these two modalities are intrinsically linked within the connectome, they represent fundamentally different biological realities, imaging modalities, and timescale dynamics.
Structural and Functional Architecture
Structural connectivity defines the anatomical architecture of the brain, consisting of physical white matter tracts - bundles of myelinated axons - that link spatially distinct cortical and subcortical gray matter regions 131417. This physical wiring diagram is typically mapped in vivo using diffusion-weighted imaging (DWI) or diffusion tensor imaging (DTI), combined with deterministic or probabilistic tractography algorithms 113. Variables such as fractional anisotropy, mean diffusivity, and the sheer volume of fibers passing between regions are utilized to estimate the integrity and strength of these anatomical connections 17. Structural networks are relatively static over short periods, changing primarily through long-term developmental maturation, experience-dependent neuroplasticity, or pathological neurodegeneration 1315. They impose the foundational physical constraints - a spatial and physiological manifold - upon which all dynamic neural communication must unfold 314.
Functional connectivity, conversely, is a statistical construct rather than a physical one. It measures the temporal dependency, coherence, or cross-correlation of neurophysiological signals between anatomically separated regions 131716. Most commonly derived from low-frequency blood-oxygen-level-dependent (BOLD) signal oscillations recorded during resting-state or task-based functional magnetic resonance imaging (fMRI), functional connectivity identifies networks of brain regions that activate and deactivate synchronously 141516. Unlike static structural connections, functional networks are highly dynamic and state-dependent. They can reconfigure within milliseconds to seconds in response to evolving cognitive demands, novel sensory inputs, or spontaneous internal thought 315.
Interestingly, while strong structural connections reliably predict strong functional connectivity, the reverse is not inherently true. Robust functional connectivity frequently occurs between regions that entirely lack direct structural links, mediated instead by complex, polysynaptic pathways or common subcortical drivers projecting to multiple cortical regions simultaneously 13. This divergence suggests that the brain utilizes a relatively fixed anatomical white matter infrastructure to host a near-infinite repertoire of transient functional states 315. Furthermore, developmental neuroimaging studies reveal that the alignment between structural and functional networks is not congenital but matures over the human lifespan. For instance, along anterior-posterior white matter tracts such as the cingulum bundle, structural and functional connectivity are highly positively correlated in adults. However, in young children, no significant correlation exists between the physical integrity of these tracts and their functional synchrony, indicating that experience-dependent plasticity and myelination progressively strengthen these structure-function alignments as the brain matures 13.
Graph Theory and Brain Topology
Network neuroscience relies heavily on graph theory, a branch of discrete mathematics that models complex interacting systems to quantify their topological architecture. In modern neuroimaging analysis, parcellation atlases (such as the AAL3 atlas or the Glasser parcellation) divide the continuous brain volume into discrete nodes, while the extracted edges represent the weighted strength of structural tracts or functional correlations bridging them 8161718. The application of graph theory algorithms has uncovered several universal, scale-invariant organizational principles that define the healthy human connectome.
The most defining characteristic of the brain's topology is its "small-world" architecture. Topologically, networks can be conceptualized along a spectrum. At one extreme lies a regular lattice network, characterized by nodes connecting only to their immediate physical neighbors, resulting in extreme local clustering but highly inefficient, long global path lengths. At the other extreme lies a random network, where edges are distributed uniformly by chance, resulting in short global path lengths but an absence of structured local clusters. The brain avoids the inefficiency of the lattice and the chaos of the random network by adopting a small-world topology 719202122. A small-world network perfectly balances two opposing topological demands: high local clustering (segregation) and short characteristic path lengths (integration) 71920. High clustering allows localized communities of neurons to perform highly specialized processing, such as visual feature extraction in the occipital lobe or fine motor planning in the precentral gyrus. Simultaneously, short path lengths - facilitated by long-range white matter "shortcut" connections - ensure that these segregated communities can rapidly share information across the entire brain with minimal synaptic relays 2026. This configuration is mathematically and metabolically optimal; it maximizes the capacity for complex information processing while minimizing the physical wiring cost and energy expenditure required to sustain long-range anatomical tracts 212223.
Beyond basic small-worldness, deep connectivity analysis utilizing percolation theory demonstrates that brain networks exhibit a hierarchical, fractal modularity 2123. Nodes are organized into distinct modules, or communities, that are densely interconnected internally but only sparsely connected to nodes residing in other modules 2425. This modularity provides biological resilience, restricting the potential spread of localized neurological damage or aberrant electrical noise 25. Percolation models indicate that the brain consists of large-world, self-similar fractal modules composed of exceptionally strong functional links. These dense modules are subsequently bridged by weaker ties to create the overarching small-world framework, a topological solution remarkably similar to the "strength of weak ties" theory observed in complex human social networks 21.
These diverse modules are interconnected via central "hubs" - neural nodes characterized by exceptionally high degree (a massive number of individual connections) and high betweenness centrality (a metric quantifying the frequency with which a specific node falls on the shortest path between other nodes) 61925. Central hubs are often densely interconnected with one another, forming a "rich-club" core. This rich-club serves as the brain's primary communication backbone, integrating outputs from disparate sensory and cognitive modules to generate cohesive, unified perception and executive action 2226.
The Core Neurocognitive Networks
While the human brain contains dozens of specialized sub-networks, contemporary cognitive neuroscience heavily focuses on three large-scale intrinsic connectivity networks that govern high-order cognition, emotion, and directed attention. This architecture, frequently referred to as the Triple Network Model, comprises the Default Mode Network (DMN), the Central Executive Network (CEN, occasionally denoted as the Executive Control Network or ECN), and the Salience Network (SN) 272829.
The Default Mode Network
Discovered initially as a distributed set of brain regions that consistently deactivated during cognitively demanding, externally focused tasks, the DMN is now recognized as the fundamental neurological substrate for internally directed, self-referential thought 3031. The core anatomical nodes of the DMN include the medial prefrontal cortex (mPFC), the posterior cingulate cortex (PCC), the precuneus, the inferior parietal lobule (IPL), and regions of the medial temporal lobe, specifically the parahippocampal gyrus and hippocampus 28303233.
Functionally, the DMN dominates brain activity during states of wakeful rest, spontaneous mind-wandering, day-dreaming, theory of mind computations (understanding the mental states of others), and the retrieval of autobiographical episodic memories 313234. Advanced component analysis further divides the DMN into specialized functional subsystems. An anterior sub-network, centered on the ventromedial and dorsomedial prefrontal cortex, primarily handles self-referential processing, affective decision-making, and emotional regulation 293132. Conversely, a posterior sub-network, centered on the PCC and its connections to the hippocampus, governs spatial navigation, environmental context, and long-term memory consolidation 2932. The DMN operates in a strictly anti-correlated, or out-of-phase, manner with task-positive networks; for a healthy mind to successfully execute a complex external task or focus outward attention, the DMN must be actively suppressed 3035.
The Central Executive and Salience Networks
The Central Executive Network represents the primary opposing functional force to the DMN. Comprising the dorsolateral prefrontal cortex (DLPFC) and the posterior parietal cortex (PPC), the CEN is a robustly task-positive network recruited exclusively during externally directed, cognitively demanding behavior 27282930. It governs the active maintenance and manipulation of information in working memory, complex problem solving, sustained attention, conflict monitoring, and the execution of goal-directed behavioral sequences 283233.
Modulating the continuous competitive dynamic between the internally focused DMN and the externally focused CEN is the Salience Network. The SN is anatomically anchored by the dorsal anterior cingulate cortex (dACC) and the frontoinsular cortices (FIC), specifically the right anterior insula 2735. Subcortical components, including the amygdala, hypothalamus, ventral striatum, and the substantia nigra/ventral tegmental area, also play vital integrative roles within this network 35. The primary cognitive function of the SN is to act as the brain's central switchboard or executive moderator. It continuously monitors the external environment and internal homeostatic physiological state for highly salient, relevant stimuli - whether they represent immediate biological threats, potent rewards, or novel contextual information 3235. Upon detecting a salient stimulus, the anterior insula generates a rapid, transient signal that functionally disengages the introspective DMN and dynamically recruits the CEN, shifting the brain's massive computational resources toward addressing the immediate environmental demand 3536.
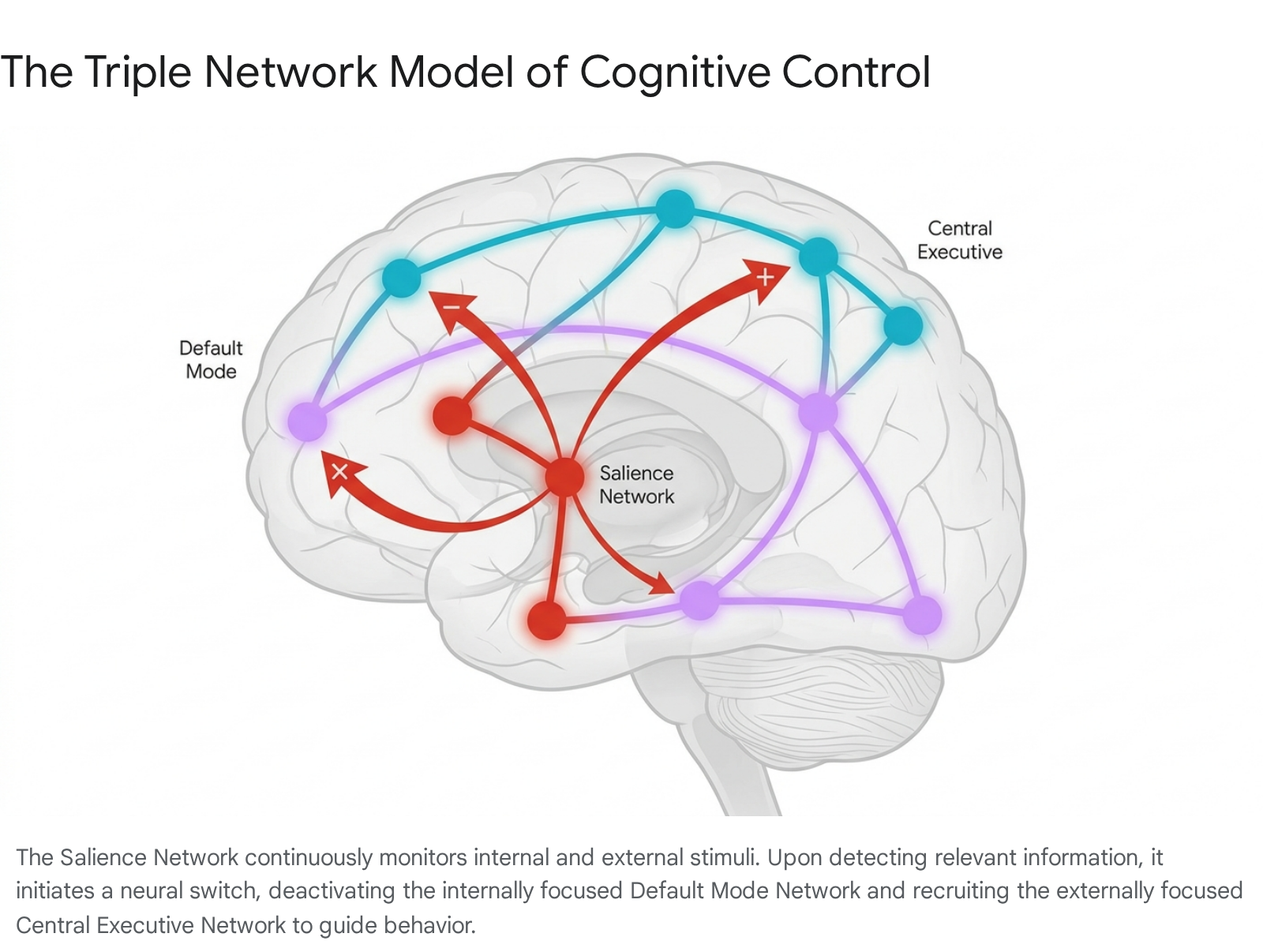
The Lesion Network Mapping Debate and Distributed Processing
The identification of these highly distributed networks has forced a reevaluation of traditional anatomical localization. Early cognitive neuroscience often fell into a "localization fallacy," attributing complex neurobehavioral syndromes solely to focal anatomical damage 23. However, emerging neuroimaging techniques like Lesion Network Mapping (LNM) demonstrate that neurological and psychiatric symptoms resulting from focal brain lesions - such as prosopagnosia, amnesia, or severe apathy - rarely localize to a single, shared anatomical coordinate across different patients 3738. Instead, diverse lesions causing identical symptoms invariably intersect with different nodes of the exact same functional network 37.
This methodology has sparked intense academic debate. Critics recently argued that LNM produces statistically biased results, positing that it highlights the same highly connected generic brain networks regardless of the specific lesion or condition analyzed 38. Furthermore, some models relying strictly on LNM fail to accurately predict longitudinal behavioral deficits following stroke 3738. However, detailed reanalyses utilizing massive healthy reference connectomes counter these criticisms, demonstrating that when appropriately corrected for regional baseline connectivity, LNM findings remain highly disease-specific 38. This distributed framework confirms that cognitive variables are represented broadly across the brain. Therefore, while specialized processing certainly occurs within specific cortical columns, the emergent properties of complex cognition rely fundamentally on dynamic, brain-wide integration rather than isolated anatomical centers 25.
Predicting Cognitive Traits Through Connectivity
One of the most profound achievements of network neuroscience is the empirical demonstration that individual differences in resting and active brain connectivity reliably predict specific cognitive traits and complex behavioral outcomes. Connectome-based predictive modeling leverages high-dimensional machine learning algorithms to decode complex fMRI data manifolds and map them to standardized human behavioral metrics 93940.
Functional Connectome Fingerprinting
Just as human fingerprints uniquely identify individuals, intrinsic functional connectivity profiles act as a highly specific "neural fingerprint." Research spanning the last decade confirms that resting-state connectomes are stable enough to accurately identify specific individuals out of massive demographic cohorts 4041. More importantly, these unique connectivity patterns serve as biological templates for predicting general intelligence, sustained attention, creative thinking, and executive decision-making capabilities 940.
Studies evaluating vast longitudinal cohorts, such as the Adolescent Brain Cognitive Development (ABCD) study (encompassing over 6,700 participants), demonstrate that the global interplay of the cingulo-parietal, retrosplenial-temporal, and dorsal attention networks is highly predictive of general adolescent cognition 3942. Furthermore, the specific brain networks responsible for predicting distinct sub-types of intelligence exhibit measurable neuroanatomical differences. For example, crystallized intelligence (the application of accumulated knowledge and vocabulary) is robustly predicted by latent functional connectivity factors active across multiple non-specific task states. Conversely, fluid intelligence (the capacity for abstract reasoning and solving novel problems independent of past knowledge) relies heavily on highly dynamic, state-specific connectivity within distinct frontoparietal architectures, demanding rapid structural reconfiguration 4043.
Ecological Validity and Real-World Assessment
Historically, much of this neuro-predictive modeling was confined to rigidly controlled laboratory assessments measuring simple reaction times or working memory spans. However, recent advancements have proven the true ecological validity of the functional connectome in real-world scenarios. By deploying rigorous algorithmic protocols - including Linear Ridge Regression (LRR), Kernel Ridge Regression (KRR), Brain Basis Set (BBS) algorithms, and Meta-Matching techniques - researchers can now significantly predict complex, real-world psychometric outcomes directly from resting-state fMRI data 910.
For instance, studies evaluating university applicants successfully predicted their global performance on standardized Psychometric Entrance Tests, alongside sub-domain scores in quantitative reasoning, verbal reasoning, and foreign language proficiency 10. Node-level and edge-level contribution analyses in these models reveal that different real-world abilities are governed by unique connectivity sub-graphs. Quantitative reasoning relies on specific frontoparietal edges entirely distinct from the temporal lobe connectivity patterns that govern language skills 910. In a demonstration of sophisticated analytics, scientists implemented transfer learning approaches within these connectomic datasets. By first calculating predicted domain-specific cognitive scores (e.g., verbal reasoning) and subsequently using those intermediate biological predictions as input features, machine learning models achieved significantly higher accuracy in predicting the subject's overarching global cognitive score than when relying exclusively on the raw, whole-brain functional matrix 10. This confirms that the brain's functional organization intrinsically partitions specific cognitive skills into independent network modules before integrating them to produce generalized intelligence.
Pathological Disruptions in the Connectome
In the context of aging, neurodegeneration, and severe psychiatric illness, the finely tuned architecture of the functional connectome breaks down in highly predictable, disease-specific sequences. Network neuroscience provides the capability to map these deteriorations long before profound structural atrophy is visible on standard clinical neuroimaging 4344.
Cognitive Aging and Dementia
During nonpathological aging, overall brain connectivity decreases, particularly within the DMN 2745. However, specific networks offer protective resilience. Research indicates that heightened intra-network coherence specifically within the Cingulo-Opercular Network (CON) strongly correlates with the preservation of fluid cognitive abilities - such as episodic memory, sustained attention, and executive function - in healthy older adults, explaining significantly more variance in cognitive test scores than chronological age or educational background 43.
Alzheimer's disease (AD) provides the clearest paradigm of pathological network disruption. Advanced neuroimaging confirms that AD molecular pathology, specifically the accumulation of amyloid-beta plaques and hyperphosphorylated tau proteins, preferentially targets the highly connected "rich-club" hubs of the brain 2744. This targeted metabolic assault leads to a progressive dismantling of the Default Mode Network. In patients transitioning from normal cognitive aging into amnestic Mild Cognitive Impairment (aMCI), resting-state fMRI reveals a distinct, tri-phasic pattern of triple-network alteration. Connectivity within the core nodes of the DMN decreases significantly, correlating directly with early clinical declines in episodic memory retrieval and self-referential processing 333646.
Concurrently, connectivity within the Central Executive Network frequently increases in aMCI. This initial hyperconnectivity within the CEN is largely interpreted as a maladaptive compensatory mechanism - the brain attempting to recruit additional frontal cognitive resources to offset the early degradation of the posterior DMN 2936. The Salience Network exhibits the most complex temporal dynamics during this disease progression. In the early stages of MCI, SN connectivity often appears heightened; the SN seemingly acts as an overworked functional bridge attempting to maintain the delicate signaling balance between a deteriorating DMN and an overcompensating CEN 3646. However, as the pathology advances inexorably into severe clinical Alzheimer's disease, the structural integrity and functional organization of the SN ultimately collapse. This terminal network failure mirrors the profound clinical loss of environmental awareness, emotional regulation, and cognitive flexibility seen in late-stage dementia 272946.
| Network Configuration | Healthy Brain State | Amnestic Mild Cognitive Impairment (aMCI) | Advanced Alzheimer's Disease (AD) |
|---|---|---|---|
| Default Mode Network | Highly synchronized; anti-correlated with CEN during active tasks. | Early hypoconnectivity, especially in posterior nodes (PCC, hippocampus). | Severe hypoconnectivity and structural atrophy; failure to deactivate. |
| Central Executive Network | Activated efficiently during working memory and goal-directed actions. | Compensatory hyperconnectivity; increased reliance on frontal nodes. | Hypoconnectivity; widespread loss of functional integrity and integration. |
| Salience Network | Intact switching mechanism; precise detection of external/internal stimuli. | Aberrant hyperconnectivity; attempting to balance deteriorating DMN/CEN dynamics. | Network collapse; correlating with profound apathy and loss of environmental awareness. |
Table 3: Sequential disruption of the Triple Network Model across the continuum of Alzheimer's disease progression 27293646.
Primary Psychiatric Disorders
The application of connectomic biomarkers to primary psychiatric disorders - such as schizophrenia (SCZ), bipolar disorder (BD), and major depressive disorder (MDD) - reveals that these conditions, long classified purely by behavioral observation, are fundamentally disorders of network integration 47484950. In schizophrenia, researchers consistently observe widespread disruptions in striatal resting-state functional connectivity. Indices measuring functional striatal abnormalities are currently under intense investigation not just for initial diagnostic staging, but as predictive biomarkers to prospectively determine whether a first-episode patient will respond favorably to standard dopaminergic antipsychotic pharmacotherapy 48.
Single-cell transcriptomics combined with fMRI connectivity analysis confirms that the polygenic genetic risk architecture for schizophrenia uniquely and disproportionately impacts the functional dynamics of the central and lateral amygdala, the hippocampal body, and specific subregions of the prefrontal cortex 51. This multidisciplinary alignment beautifully bridges the gap between massive genome-wide association studies (GWAS) and macroscopic, observable brain networks 5152.
Recent molecular discoveries further underscore the immense value of integrating cerebrospinal fluid signatures with network connectivity. For example, a newly identified brain protein, Cacna2d1, has been found circulating at substantially reduced levels in the cerebrospinal fluid of human schizophrenia patients. This exact protein deficiency directly results in overexcited, highly disorganized functional brain circuits 5354. When a synthetic replication of this protein (designated SEAD1) was injected into genetic mouse models of schizophrenia, it successfully corrected the aberrant circuit-level activity and reversed the associated behavioral deficits, representing a massive breakthrough in targeting the intractable cognitive symptoms - rather than merely the positive hallucinatory symptoms - of the disease 5354.
In major depressive disorder, resting-state imaging consistently identifies pathological hyperconnectivity within the anterior nodes of the DMN, correlating with the persistent, uncontrollable rumination typical of the condition 4955. Furthermore, elevated systemic inflammatory markers (such as C-reactive protein) can be utilized to biologically segment the MDD patient population; patients presenting with this specific inflammatory-connectomic biotype demonstrate a significantly superior clinical response to pharmacological agents that modify dopaminergic tone, rather than traditional Selective Serotonin Reuptake Inhibitors (SSRIs) 555662.
Clinical Translation and Diagnostic Readiness
Despite standard medical specialties routinely relying on definitive laboratory assays, cellular biopsies, and quantitative imaging biomarkers, psychiatry and much of behavioral neurology remain overwhelmingly confined to syndromal, symptom-based diagnostic frameworks, relying heavily on the subjective clinical criteria outlined in the DSM-5 and ICD-11 44505256. Network neuroscience presents an unparalleled opportunity to bridge this historical gap by offering quantitative, objectively measurable connectomic biomarkers that map directly onto disease pathogenesis rather than mere symptom presentation 1252.
The Limitations of Connectomic Biomarkers
Despite decades of intense global research and massive financial investment, a candid assessment of the field reveals that functional connectomic biomarkers are not yet fully prepared for widespread, unguided clinical application in routine psychiatric practice 485657. The obstacles preventing immediate clinical integration are complex and multifaceted. First, many promising candidate biomarkers identified in neuroimaging literature show only modest statistical effect sizes and suffer from severely limited generalizability across diverse real-world patient cohorts. Machine learning models trained on highly controlled, homogenous laboratory datasets frequently fail when deployed in chaotic clinical environments, as they struggle to account for the massive confounding effects of normal physiological aging, diverse medical comorbidities, sex differences, and the chronic use of psychotropic medications 485257.
Furthermore, while advanced neuroimaging techniques like LNM and predictive connectomics excel at identifying group-level statistical differences between cohorts (e.g., "healthy controls" vs. "MDD patients"), regulatory bodies such as the US Food and Drug Administration (via its Biomarker Qualification Program) require extraordinarily rigorous analytical and clinical validation to prove that a biomarker possesses actionable diagnostic sensitivity and specificity at the individual patient level 126257.
Breakthroughs in Molecular and Blood Biomarkers
Currently, only neurological biomarkers related directly to definitive molecular pathology have successfully cleared this stringent regulatory threshold. In the realm of Alzheimer's disease, the integration of amyloid and tau PET tracers, alongside the recent FDA clearance of highly precise plasma assays (blood tests measuring p-tau181, p-tau217, and neurofilament light chain [NfL]), represents a monumental leap forward 47625865.
However, even these validated molecular markers face complex implementation challenges. For instance, the diagnostic accuracy of AD blood biomarkers can be skewed by routine medical comorbidities; the common heart failure medication sacubitril-valsartan has been shown to artificially inflate plasma Aβ42 concentrations and alter the Aβ42/40 ratio by approximately 30%, though fortunately, it does not alter p-tau or NfL readings 65. To successfully integrate functional connectivity biomarkers into standard psychiatric care, the field must replicate the rigorous validation pathways established by the Alzheimer's research community. This will require massive, multi-center longitudinal validation cohorts to transition from identifying interesting statistical correlations to generating robust, clinically actionable diagnostic tools 125556.
Global Brain Initiatives and Artificial Intelligence
The astronomical complexity of decoding the human connectome - mapping roughly 86 billion neurons and trillions of synaptic connections - has pushed the boundaries of traditional scientific methodology. This scale has prompted the launch of massive, state-sponsored international scientific consortia and catalyzed the deep integration of next-generation artificial intelligence algorithms 5960.
Global Mapping Consortia
The European Union's Human Brain Project (HBP), a massive Future and Emerging Technologies Flagship that concluded its decade-long mandate in 2023, laid the vital technical and digital foundation for computational neuroscience. Uniting over 500 researchers, clinicians, and engineers, the HBP generated massive multiscale digital brain atlases, deployed specialized neuromorphic computing hardware architectures, and successfully developed "digital twin" methodologies. These personalized virtual brain simulations now allow clinicians to model patient-specific neural dynamics to guide targeted medical interventions in conditions like drug-resistant epilepsy and Parkinson's disease 5961626364. The extensive data and computational infrastructure established by the HBP currently lives on through EBRAINS, an open research platform continuing to push collaborative network neuroscience 6364.
In parallel, the China Brain Project (CBP), initiated in 2016 and extending through 2030, operates under a highly strategic "one body, two wings" structural framework. The central "body" focuses on unraveling the basic neural mechanisms underlying human cognition, while the "two wings" seek to develop advanced clinical diagnostics for major brain diseases and drive the rapid development of brain-inspired artificial intelligence 596566. Utilizing China's extensive non-human primate research resources, CBP scientists have made rapid progress in creating highly accurate macaque and marmoset models to map higher-order primate cognition - such as the neural basis of consciousness, language processing, and empathy - phenomena that cannot be accurately or functionally modeled in rodent brains 6667.
The CBP has also launched groundbreaking, multi-system clinical initiatives. The Brain-Gut Health Initiative (BIGHI) is currently executing the most comprehensive mapping of the microbiota-gut-brain axis in psychiatric patients. Utilizing advanced multi-omics data, the project has successfully linked pro-inflammatory gut microbiomes and deficits in short-chain fatty acid-producing bacteria with widespread brain network alterations, differentiating schizophrenia and bipolar disorder patients from healthy controls while identifying specific neural microstates tied to suicidal ideation 68. Concurrently, the CBP is aggressively accelerating neurotechnology; by early 2025, Chinese research institutes commenced successful human trials for high-throughput, minimally invasive brain-computer interfaces (BCIs). Devices such as the Beinao No. 1 wireless chip have successfully enabled tetraplegic patients to flawlessly control external digital environments and robotic prosthetics via thought alone, with massive expanded clinical trials slated for late 2025 6977.
Algorithmic Advances in Neural Decoding
Handling the extreme dimensionality and temporal resolution of modern connectomic data has necessitated a completely symbiotic merger between neuroscience and artificial intelligence. Traditional deep learning architectures - such as Convolutional Neural Networks (CNNs) and Recurrent Neural Networks (RNNs) - have achieved remarkably high accuracy in interpreting EEG signals for brain-machine interfaces and categorizing basic cognitive states 7071. However, their massive computational energy requirements and inherent "black-box" nature pose severe challenges for generating biologically meaningful interpretability 7072.
To overcome these barriers, computational neuroscientists are deploying novel topological data analysis tools, such as persistent graph homology. This advanced mathematical framework allows researchers to analyze the functional connectome across its entire spectrum of spatial scales without relying on arbitrary, researcher-defined connectivity thresholds. By extracting high-order topological invariants (such as independent functional cycles and discrete connected components), persistent graph homology preserves the underlying biological mechanisms of the neural network while remaining computationally scalable for massive datasets 72.
Simultaneously, the deployment of advanced Transformer architectures - originally designed to process massive sequences in natural language processing - is revolutionizing the speed and accuracy of real-time neural decoding. Breakthrough models like NuCLR (a spatiotemporal transformer utilizing contrastive learning to map within-neuron dynamics) and POSSM (a highly efficient hybrid state-space model) are trained on massive, aggregated multi-animal recording datasets 73. These cutting-edge AI models capture both complex temporal neural dynamics and vast across-network interactions. This dual capability allows researchers to accurately predict complex physical behavior directly from neural activity (decoding) and conversely predict neural firing patterns based on observed behavior (encoding). Crucially, these Transformer models exhibit zero-shot generalization to entirely unseen biological data and operate at the extreme microsecond speeds necessary for seamless, real-time closed-loop neuroprosthetic applications 73. Furthermore, the emerging field of Organoid Intelligence (OI) seeks to bypass silicon entirely, utilizing 3D cultures of human brain cells as bio-hybrid computing systems capable of executing complex AI algorithms with vastly superior energy efficiency and learning plasticity compared to traditional hardware 74.
Conclusion
The evolution of network neuroscience has fundamentally rewritten the operational rulebook of the human brain. The mind is no longer viewed as a fragmented collection of isolated anatomical processors, but rather as an exquisitely elegant, small-world topology of rapidly shifting dynamic networks. The constant push-and-pull between the internally reflective Default Mode Network and the externally directed Central Executive Network, moderated vigilantly by the Salience Network, forms the very foundation of human consciousness, self-awareness, and cognitive flexibility.
When this intricate architecture falters - whether through the slow, targeted neurodegenerative deterioration of critical hubs in Alzheimer's disease, or the hyperconnected, neurodevelopmental disarray observed in severe psychiatric disorders - the resulting clinical deficits reflect a systemic communication failure across the entire connectome rather than a localized anatomical lesion. While the immediate clinical translation of functional connectomic biomarkers remains temporarily hindered by strict regulatory requirements and the necessity for massive longitudinal validation, the trajectory is clear. The rapid integration of multi-modal deep learning, the unparalleled scope of global brain mapping initiatives, and the deployment of advanced, closed-loop brain-computer interfaces signal an imminent and profound breakthrough. Ultimately, deciphering the human connectome promises not merely to refine our diagnostic manuals, but to unlock targeted, circuit-level, precision interventions that address the true biological and topological origins of cognitive and psychiatric disease.