Molecular Circadian Rhythms in Human Health and Longevity
The temporal organization of biological processes is a fundamental property of life on Earth, driven by the planet's 24-hour rotation and its yearly orbit around the Sun. Empirical evidence of internal biological timekeeping dates back to 1729, when it was first observed that specific plants maintain rhythmic leaf movements even in complete darkness 1. Today, chronobiology reveals that these innate mechanisms - known as circadian (approximately 24-hour) and circannual (seasonal) rhythms - govern virtually every aspect of human physiology, behavior, and metabolism 123. Rather than relying on a single localized timer, the mammalian circadian system consists of a highly integrated, hierarchical network of molecular oscillators located in the brain and in nearly every peripheral cell 456.
In modern society, exposure to artificial light at night (ALAN), shift work, and irregular dietary patterns routinely disrupts this finely tuned temporal alignment. This phenomenon, known as circadian desynchrony, is increasingly recognized as a profound pathophysiological driver of metabolic syndrome, cardiovascular disease, neurodegeneration, psychiatric disorders, and carcinogenesis 167. Understanding the molecular architecture of the circadian clock - and the biochemical pathways it uses to synchronize human physiology - provides critical insights into the maintenance of health, the process of biological aging, and the optimization of medical interventions through chronopharmacology.
Central Pacemakers and Extra-SCN Clocks
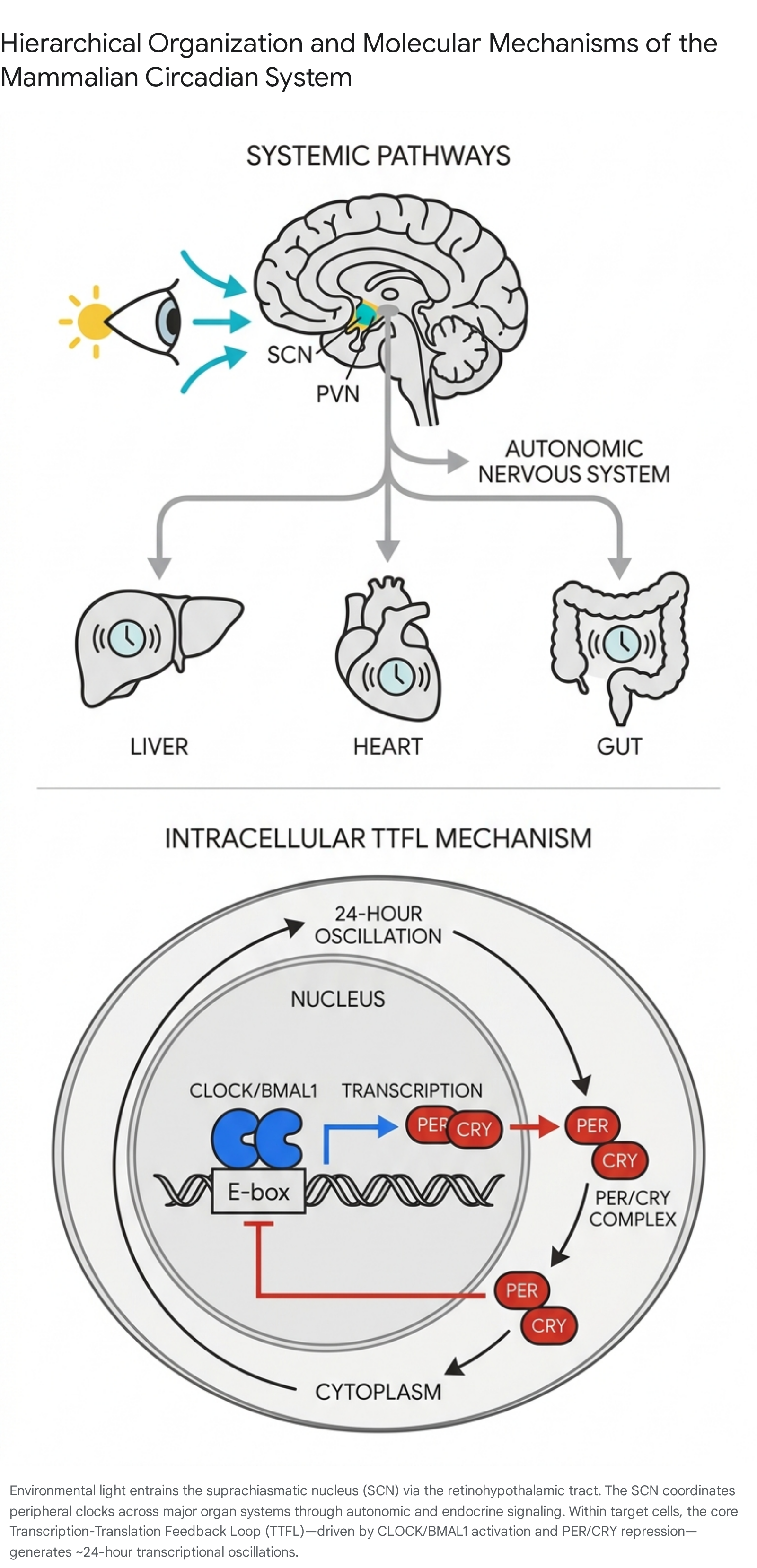
The master pacemaker of the mammalian circadian system is the suprachiasmatic nucleus (SCN), a bilaterally symmetric structure in the anterior hypothalamus comprising approximately 20,000 highly interconnected neurons 45. The SCN operates as the central biological clock, capable of generating self-sustaining, diurnal oscillations in electrical activity and gene expression that persist in the absence of external time cues 57. However, to maintain biological utility, the SCN must constantly synchronize, or entrain, its internal rhythms to the external environment.
Photic Entrainment and Retinal Signaling
Light serves as the primary environmental synchronizer, or zeitgeber 89. Photic information bypasses standard visual processing and is captured directly by intrinsically photosensitive retinal ganglion cells (ipRGCs) that express the photopigment melanopsin 8. These specialized cells project directly to the SCN via the retinohypothalamic tract 810.
Upon receiving photic input, excitatory neurotransmitters - primarily glutamate and pituitary adenylate cyclase-activating polypeptide (PACAP) - are released into the SCN 10. This signaling triggers calcium influx through N-methyl-D-aspartate (NMDA) receptors, activating intracellular cyclic AMP (cAMP) and calcium-dependent kinase pathways 710. Ultimately, this cascade leads to the cAMP response element-binding protein (CREB)-mediated transcription of specific clock genes, rapidly phase-shifting the master pacemaker to align with the geophysical dawn or dusk 710.
Neurochemical Architecture of the SCN
The SCN achieves robust internal synchronization through a highly organized neurochemical topography. The ventral core of the SCN, which receives direct retinal innervation, is populated predominantly by neurons expressing vasoactive intestinal polypeptide (VIP) and gastrin-releasing peptide (GRP) 7. VIP neurons act as critical coupling agents; mutations in VIP or its receptor (VPAC2) result in dampened and desynchronized cellular oscillations across the SCN, manifesting behaviorally as weakened rhythmicity 710.
In contrast, the dorsal shell of the SCN primarily contains neurons that express arginine vasopressin (AVP) 7. SCN-AVP neurons represent the principal molecular output of the master clock, projecting rhythmic signals to downstream target regions such as the paraventricular nucleus (PVN), the subparaventricular zone (SPVZ), and the dorsomedial hypothalamus (DMH) to coordinate behavioral and hormonal rhythms 711.
Extra-SCN Central Oscillators
While the SCN is the undisputed master pacemaker, 24-hour oscillatory activity is not exclusive to it. Extensive networks of secondary, extra-SCN clocks exist throughout the brain. Regions such as the arcuate nucleus, the ventromedial hypothalamus (VMH), the lateral septum, and the paraventricular thalamic nuclei exhibit distinct circadian transcriptomic patterns 511. These localized oscillators play specialized roles in temporal homeostasis; for example, clocks in the DMH and VMH integrate external thermal cycles and nutritional signals, while oscillators in the hippocampus independently gate time-of-day variations in synaptic plasticity and memory formation 512.
The Molecular Clockwork: Transcription-Translation Feedback Loops
The generation of circadian rhythms within individual cells - both in the SCN and in peripheral tissues - is driven by an intricate, highly conserved molecular engine. This engine consists of interlocked transcription-translation feedback loops (TTFLs) that mandate a roughly 24-hour periodicity in gene expression 51314.
The Primary Negative Feedback Loop
The core architecture of the TTFL relies on the stoichiometric balance between transcriptional activators and their respective repressors. The positive arm is formed by two basic helix-loop-helix-PAS (bHLH-PAS) domain transcription factors: BMAL1 (Brain and Muscle ARNT-Like 1, also known as ARNTL) and CLOCK (Circadian Locomotor Output Cycles Kaput) 515. In certain neural tissues, neuronal PAS domain protein 2 (NPAS2) functions as a homolog capable of replacing CLOCK 1316.
During the subjective morning, the CLOCK-BMAL1 heterodimer binds to E-box enhancer elements (CACGTG consensus sequences) within the promoter regions of target genes 1315. This initiates the transcription of thousands of clock-controlled genes (CCGs), estimating to represent 10 - 15% of the human genome, thereby imparting temporal control over tissue-specific physiology 517. Concurrently, CLOCK-BMAL1 drives the transcription of its own repressors: the Period (PER1, PER2, PER3) and Cryptochrome (CRY1, CRY2) gene families 131518.
As the day progresses, PER and CRY mRNAs are exported to the cytoplasm and translated into proteins. To ensure the cycle requires a full 24 hours to complete, the accumulation of these repressor proteins is subject to strict post-translational gating 18. Kinases, notably Casein Kinase 1 delta and epsilon (CK1δ/ε), continually phosphorylate PER proteins, targeting them for rapid ubiquitination and proteasomal degradation 1418. It is only by late afternoon and early evening that PER and CRY synthesis outpaces their degradation, allowing them to reach critical concentrations. They subsequently heterodimerize into large macromolecular repressor complexes and translocate back into the nucleus 1819.
Upon nuclear entry, the PER-CRY complex interacts directly with CLOCK-BMAL1, displacing coactivators and halting further E-box-mediated transcription 1418. As the repressive phase persists through the biological night, the transcription of Per and Cry ceases. Eventually, the existing PER and CRY proteins are degraded by specific E3 ubiquitin ligases (such as FBXL3 for CRY), lifting the transcriptional repression and permitting a new cycle to begin the following morning 1420.
Secondary Stabilizing Loops
To confer robustness and precision against environmental noise, auxiliary feedback loops operate in parallel to the primary TTFL. The most prominent involves the nuclear hormone receptors REV-ERB (α/β) and ROR (α/β/γ) 1518. CLOCK-BMAL1 promotes the rhythmic transcription of both families. Upon translation, the REV-ERB proteins act as transcriptional repressors, while ROR proteins act as activators. Both compete for binding at ROR/REV-ERB response elements (ROREs) present in the promoter region of the Bmal1 gene 1718. This dynamic competition ensures that Bmal1 transcription is tightly regulated, maintaining high-amplitude cyclic expression that peaks in exact antiphase to the PER and CRY repressors 1821.
Systemic Synchronization of Peripheral Clocks
While molecular clocks are ubiquitous across human tissues, maintaining temporal cohesion among trillions of cells requires orchestrated systemic communication. Peripheral clocks in the liver, heart, lungs, and gut are highly autonomous, yet they depend on the SCN to remain synchronized with external geophysical time 82223.
Endocrine and Autonomic Pathways
The SCN broadcasts temporal data primarily via neuroendocrine cascades and the autonomic nervous system. A crucial output pathway involves the hypothalamic-pituitary-adrenal (HPA) axis 824. SCN neurons synthesizing AVP and VIP coordinate the rhythmic release of corticotropin-releasing hormone (CRH) from the paraventricular nucleus (PVN) 71024. This triggers the release of adrenocorticotropic hormone (ACTH) and establishes a pronounced circadian rhythm of glucocorticoid secretion (cortisol in humans), which typically peaks sharply just prior to awakening 824.
Because glucocorticoid receptors (GRs) are expressed on virtually all peripheral cell types - but are notably absent in SCN core neurons - circulating cortisol serves as a unidirectional systemic messenger. Upon binding to peripheral GRs, glucocorticoid complexes interact with response elements located upstream of the Per1 promoter, directly inducing its transcription and resetting the phase of local molecular clocks 10. This ensures that peripheral organs prepare metabolic and energetic substrates in anticipation of the active phase 810.
Physical and Non-Photic Zeitgebers
In addition to humoral signals, the SCN dictates daily rhythms in core body temperature, characterized by a steady rise toward the late active phase and a 1 - 2°C nadir during the rest phase 10. Similar to glucocorticoid signaling, the SCN itself is largely impervious to physiological temperature fluctuations, preventing self-disruption. However, peripheral clocks in tissues such as the lungs, pituitary gland, and liver are exquisitely temperature-sensitive; the recurring thermal cycle acts as an essential physical zeitgeber that sustains peripheral clock gene activity in vivo 1025.
Furthermore, precise inter-organ communication utilizes complex kinome networks. Recent data demonstrate that peripheral entrainment involves receptor tyrosine kinases (RTKs) independently of standard neuroendocrine axes. In glandular tissues, specific RTK ligands (such as anaplastic lymphoma kinase and Pvr pathways) function additively to modulate peripheral timing, underscoring the biochemical diversity required for systemic coordination 26.
Chronobiology of the Human Transcriptome
Advances in functional genomics, particularly analyses aggregating massive donor databases like the Genotype-Tissue Expression (GTEx) project, have illuminated the sheer scale of circadian regulation across the human body 2728. Algorithms designed to infer tissue-specific local phases from bulk RNA-sequencing data reveal that previous methodologies vastly underestimated circadian transcriptional volume.
A comprehensive multi-tissue study mapping the 24-hour rhythmic transcriptome across 45 human tissues identified a median of 5,747 rhythmic transcripts per tissue 28. In the brain alone, over 5,500 genes exhibit nycthemeral (day-night) rhythms, enriching critical pathways such as the synaptic vesicle cycle, neurotransmitter signaling, and major neurodegenerative disease cascades 28.
Seasonal Variations and Demographic Dimorphism
Beyond 24-hour cycles, human biology exhibits profound circannual rhythmicity. Research indicates that the human circadian system is functionally partitioned, acting as two interconnected clocks - one tracking dawn and the other tracking dusk - to continuously adapt to seasonal variations in daylight length 29. Transcriptomic mapping identifies distinct gene sets exhibiting seasonal expression patterns; tissues within the thoracic cavity (e.g., the lung and heart) show extreme sensitivity to daily photic cycles, whereas the brain and testes demonstrate higher sensitivity to seasonal, circannual shifts 30.
At the demographic level, the fundamental architecture of the molecular clock is conserved across sex and age groups. Nonetheless, overall gene expression rhythms are highly sexually dimorphic, with females generally exhibiting more sustained and robust rhythmic programs 27. Conversely, rhythmic gene expression steadily dampens across almost all peripheral tissues with advancing age, marking systemic temporal decay as a fundamental feature of human aging 27.
Circadian Control of Metabolism and Energy Homeostasis
Metabolism and circadian rhythms maintain an intimate, bidirectional relationship. The molecular clock ensures that biochemical processes are temporally segregated; catabolic and anabolic pathways are scheduled to optimize resource utilization and prevent futile enzymatic cycling 132231.
Nutrient Sensing and Metabolic Feedback
In metabolic tissues such as the liver, skeletal muscle, and adipose tissue, the clock regulates a vast array of rate-limiting enzymes. Up to 40% of the hepatic transcriptome is subject to circadian control, governing glucose homeostasis, lipogenesis, cholesterol synthesis, and xenobiotic detoxification 2232. The integration of the TTFL with nutrient-sensing pathways provides an adaptive mechanism through which energetic states directly alter clock velocity and amplitude.
A primary metabolic feedback loop involves the nicotinamide adenine dinucleotide (NAD+) salvage pathway. The CLOCK-BMAL1 heterodimer regulates the transcription of Nampt, the gene encoding the rate-limiting enzyme in NAD+ biosynthesis 1314. The resulting circadian oscillation of intracellular NAD+ levels governs the activity of SIRT1, an NAD+-dependent deacetylase. In turn, SIRT1 directly deacetylates PER2, promoting its degradation, and deacetylates BMAL1, thereby lifting transcriptional repression 1433. Similarly, nutrient deprivation increases the AMP/ATP ratio, activating AMP-activated protein kinase (AMPK). AMPK phosphorylates CRY1, accelerating its interaction with the FBXL3 ubiquitin ligase and hastening its destruction 1433. Consequently, feeding and fasting states fundamentally tune the timing machinery at the molecular level.
Internal Desynchrony and Time-Restricted Eating
Because peripheral clocks are highly responsive to metabolic metabolites (e.g., glucose, lipids, bile acids), food intake acts as an independent zeitgeber. While light remains the ultimate entraining cue for the SCN, the timing of meals dictates the phase of peripheral organs 222532. In scenarios such as shift work, chronic jet lag, or late-night eating, food is consumed out of phase with the light-dark cycle. This decoupling forces the liver and gut clocks to shift out of alignment with the central SCN pacemaker, a state of internal desynchrony strongly associated with the pathogenesis of obesity, insulin resistance, and hepatic steatosis 223334. The disruptive effects extend to the microbiome, where jet-lag-induced host desynchrony promotes dysbiosis and glucose intolerance 34.
Correcting this misalignment offers profound therapeutic utility. Time-Restricted Eating (TRE) - limiting all caloric intake to a consistent 6- to 10-hour window during the active phase - acts as a powerful behavioral chronotherapy 1533. By reinforcing a predictable feeding-fasting cycle without necessarily altering total caloric volume, TRE optimally engages AMPK and SIRT1 pathways, restores the high-amplitude oscillations of peripheral clock genes, and mitigates diet-induced metabolic syndrome in both human and animal cohorts 1833.
Cardiovascular Physiology and Pathology
The cardiovascular system is subject to stringent temporal regulation designed to match the rhythmic nature of human activity. Critical hemodynamic parameters, including blood pressure, heart rate, vascular endothelial tone, coagulation cascade components, and circulating catecholamines, exhibit robust circadian periodicity 432.
The heart harbors an autonomous intrinsic clock, driven by the standard BMAL1, CLOCK, PER, and CRY loops, which controls an estimated 6 - 10% of the cardiac transcriptome 1632. This molecular regulation primarily dictates energy substrate flexibility. During the active daytime phase, the clock gears cardiac metabolism toward highly efficient fatty acid oxidation to meet increased workload demands. During the resting nocturnal phase, the system shifts toward glucose utilization and glycolysis 32.
Dysregulation of these rhythms entails severe clinical consequences. The sharp morning surge in sympathetic tone and blood pressure - a physiological anticipation of awakening - correlates epidemiologically with the peak daily incidence of acute myocardial infarctions, sudden cardiac death, and strokes 3236. In murine models, cardiomyocyte-specific deletion of Bmal1 or Clock severely impairs myocardial contractility, precipitates mitochondrial dysfunction, and magnifies the tissue damage inflicted by ischemic events 432. Chronic environmental misalignment, such as shift work or persistent sleep fragmentation, significantly elevates the longitudinal risk for hypertension and accelerated atherosclerosis by dismantling these protective temporal barriers 432.
Oncology and the Disrupted Clock
A functional circadian system serves as a systemic tumor suppressor, tightly coordinating the cell cycle, apoptosis, and cellular stress responses 313536. Disruptions in circadian timing - whether initiated by environmental light at night, lifestyle factors, or internal genetic alterations - compound the risk of carcinogenesis and modulate the trajectory of tumor progression 731.
DNA Repair and The Tumor Microenvironment
One of the most critical circadian outputs in maintaining genomic stability is the regulation of DNA repair pathways. The expression of key nucleotide excision repair proteins, such as XPA, oscillates predictably, aligning peak repair capacity with historical patterns of environmental mutagenic exposure (e.g., daytime ultraviolet radiation) 31.
In active malignancies, bidirectional interactions between the circadian clock and the tumor microenvironment (TME) fuel disease progression. For instance, the stiffened, fibrotic extracellular matrix characteristic of advanced breast cancer physically dampens the amplitude of the epithelial clock via mechanotransduction pathways 31. This loss of local rhythmicity fosters unchecked inflammation, cancer stem cell expansion, and metastasis 31. The circadian gating of immune surveillance dictates that specific immune cell trafficking and immunosurveillance pathways are only highly active at certain times of day, a rhythm that tumors often exploit to evade detection 31.
Genomic and Epigenetic Clock Alterations in Cancer
Extensive analyses of data from The Cancer Genome Atlas (TCGA) reveal a widespread downregulation of core clock gene expression across nearly all human cancer types 35. Unlike classical oncogenes, core clock genes (PER1-3, CRY1-2, CLOCK, BMAL1) rarely harbor structural mutations or copy number alterations. Instead, their suppression is frequently driven by aberrant DNA hypermethylation at their promoter regions 35.
The clinical implications of clock gene silencing are profound. In hepatocellular carcinoma (HCC), decreased expression of PER1, PER3, and CRY2 is strongly associated with abbreviated overall survival rates 37. In cholangiocarcinoma (bile duct cancer), the targeted experimental disruption of BMAL1 accelerates epithelial-to-mesenchymal transition (EMT), enhances invasive cell migration, and critically induces resistance to chemotherapeutic agents like gemcitabine 16. Consequently, restoring functional circadian rhythms or targeting the molecular clock directly represents an emerging paradigm in adjuvant oncology 1631.
Neurobiology of Mood and Psychiatric Disorders
The neural circuits responsible for emotion, cognition, and stress response are deeply intertwined with the circadian system. Mood disorders - including major depressive disorder (MDD), bipolar disorder (BD), and seasonal affective disorder (SAD) - are universally characterized by severe disruptions in sleep architecture and circadian alignment 3839.
Pathophysiological Links to Depression and Bipolar Disorder
Clinical markers of circadian dysfunction are highly prevalent in depressed populations, manifesting as flattened diurnal cortisol slopes, an advanced or delayed onset of melatonin secretion, and shortened REM sleep latency 4041. This behavioral desynchrony is mirrored at the molecular level. Post-mortem transcriptomic analyses of cortico-limbic brain regions in patients with MDD demonstrate a pervasive dampening of normal 24-hour gene expression cycles, indicating a localized failure of the molecular clock within mood-regulating circuits 3842.
Furthermore, assays of peripheral blood leukocytes in individuals with a history of depression frequently reveal anomalous over-expression of CLOCK, PER1, and BMAL1 mRNA compared to healthy controls 41. The mechanistic bridge connecting the clock to mood regulation primarily involves monoamine signaling. The synthesis, release, and receptor sensitivity of both serotonin and dopamine - neurotransmitters essential for emotional stability - are under strict circadian governance 384344. Genetic variations that disrupt this timing machinery invariably cascade into dysregulated monoamine dynamics, precipitating depressive or manic episodes 44.
Sleep and Circadian Rhythm Sleep Disorders (CRSDs)
The molecular underpinnings of sleep and wakefulness rely heavily on the clock. Mutations in core TTFL components can lead to profound alterations in sleep timing, collectively categorized as Circadian Rhythm Sleep Disorders (CRSDs) 45. Conditions such as Advanced Sleep Phase Syndrome (ASPS) and Delayed Sleep Phase Syndrome (DSPS) highlight how subtle molecular changes - such as altered phosphorylation kinetics of the PER protein - shift the entire physiological framework relative to the solar day 4546.
Neurodegeneration and Cognitive Decline
The deterioration of circadian rhythmicity is not solely a feature of psychiatric distress; it is a primary, precipitating event in the pathogenesis of neurodegenerative diseases, including Alzheimer's disease (AD), Parkinson's disease (PD), and Huntington's disease (HD) 93247.
In neurodegenerative cohorts, disrupted rest-activity rhythms and flattened melatonin secretion profiles frequently precede the onset of overt cognitive or motor decline by years 947. The SCN itself undergoes severe anatomical and functional degradation with advancing age and disease pathology, leading to diminished expression of BMAL1, PER2, and CRY1, reduced neural synchronization, and a weakened response to photic zeitgebers 47.
This loss of central timing exacerbates cellular damage. The BMAL1-CLOCK complex is directly responsible for transcribing genes critical for redox homeostasis, such as the NAD(P)H quinone dehydrogenase (Nqo1) and aldehyde dehydrogenase 2 (Aldh2) 1245. Diminished BMAL1 activity results in an unchecked accumulation of reactive oxygen species (ROS), driving oxidative stress and accelerating neuronal apoptosis 124548. Concurrently, the failure of consolidated sleep patterns hinders the glymphatic clearance of neurotoxic proteins like amyloid-beta and misfolded tau, creating a vicious cycle of circadian decay and neuroinflammatory damage 4347.
Aging, Longevity, and the Epigenetic Clock
Aging is universally accompanied by a progressive decay in the amplitude, precision, and synchrony of the circadian network 214950. As individuals age, temporal consolidation weakens, leading to fragmented sleep, blunted hormonal peaks, and metabolic inflexibility 2150.
The Circadian Foundation of Healthspan
Experimental data across model organisms suggest that a functional clock is an absolute requisite for extreme longevity. Centenarians are distinguished not merely by genetics, but by their ability to preserve robust circadian rhythms, including high-quality sleep architecture and rhythmic lipid profiles, well into their advanced years 51.
The profound life-extending benefits of interventions such as caloric restriction are intrinsically tied to circadian pathways. Studies reveal that the metabolic adaptations induced by caloric restriction - such as altered SIRT1, mTOR, and AMPK signaling - fail to extend lifespan in the absence of a functional molecular clock, positioning the TTFL as the ultimate executor of geroprotective protocols 2154. Furthermore, aging fundamentally alters the systemic milieu that synchronizes peripheral cells. Research demonstrates that culturing young fibroblasts in human serum derived from older individuals causes critical cellular pathways, notably oxidative phosphorylation cascades, to lose their characteristic 24-hour rhythmicity 52. This age-induced circadian reprogramming highlights the systemic nature of temporal decay 52.
Biological Age and Epigenetic Clocks
The necessity to precisely measure the rate of human aging has led to the development of sophisticated "epigenetic clocks." Utilizing algorithms that track dynamic DNA methylation patterns across specific cytosine-guanine (CpG) dinucleotides, these molecular models provide a highly accurate estimation of an individual's biological - rather than chronological - age 535754.
Advanced iterations, such as DNAm PhenoAge and the newly developed Intrinsic Capacity (IC) Clock, correlate specific methylation signatures with real-world functional outcomes. The IC Clock transcends mere mortality prediction by integrating methylation data with the six pillars of intrinsic capacity defined by the World Health Organization: mobility, cognition, mental health, vision, hearing, and nutrition/vitality 5556. Deviations indicating a biological age older than chronological age strongly predict the onset of multimorbidity, cognitive decline, and physical frailty, positioning these clocks as critical endpoints for assessing the efficacy of future anti-aging or circadian-restoring interventions 545556. Astoundingly, epigenetic methylation analysis can even reliably predict the maximum inherent lifespan across mammalian species, independent of the individual specimen's current age 57.
Chronopharmacology and Precision Medicine
The realization that human biochemistry fluctuates systematically over a 24-hour period necessitates a fundamental paradigm shift in clinical therapeutics. Chronopharmacology - the practice of timing drug administration to align with endogenous biological rhythms - seeks to maximize therapeutic efficacy while minimizing adverse toxicity 575859.
Modulating Pharmacokinetics and Pharmacodynamics
The pharmacokinetic profile of most pharmaceuticals is governed by circadian rhythms. Hepatic detoxification enzymes, renal blood flow, and gastrointestinal motility exhibit marked diurnal variation, ensuring that drug absorption, distribution, metabolism, and excretion rates differ drastically between morning and evening administration 5759. Simultaneously, drug pharmacodynamics are impacted by the rhythmic expression of cellular receptors and target enzymes 5960.
Clinical applications of chronopharmacology are rapidly advancing: * Oncology: Chemotherapy regimens are increasingly scheduled to coincide with periods when healthy cells are least replicative, and thus least vulnerable, while targeting specific diurnal vulnerabilities in the tumor cycle. This strategy significantly improves patient tolerability and drug effectiveness 365759. * Cardiology: Groundbreaking prospective trials, such as the MAPEC and HYGIA studies, demonstrated that shifting the administration of routine antihypertensive medications to bedtime improves nocturnal blood pressure control and dramatically reduces the incidence of subsequent major cardiovascular events, compared to standard morning dosing 5960. * Rheumatology and Immunology: Autoimmune pathologies like rheumatoid arthritis exhibit distinct temporal symptom profiles, typically peaking in the morning due to the nocturnal rise in pro-inflammatory cytokines. Administering non-steroidal anti-inflammatory drugs (NSAIDs) or immunosuppressants such as methotrexate with specific timing ensures peak plasma concentrations align with peak symptom severity 36. Similar timing strategies are routinely employed for nocturnal asthma exacerbations 36.
The Future of Chronotherapeutics
The future of precision medicine relies on the seamless integration of chronobiological principles. Beyond drug timing, a new class of pharmacological agents designed to directly target molecular clock proteins - such as REV-ERB agonists or CRY modulators - is under investigation to chemically "reset" misaligned clocks in metabolic syndrome or mood disorders 145758.
Translating this science into routine clinical practice requires accurate, non-invasive assessment of a patient's internal circadian phase. Advancements in digital health, artificial intelligence, and wearable actigraphy devices now permit the continuous monitoring of rest-activity rhythms 576061. By quantifying multidimensional actigraphy metrics - such as the Circadian Function Index (CFI), interdaily stability (IS), intradaily variability (IV), and relative amplitude (RA) - clinicians can generate individualized movement behavior profiles 61. This data enables the prescription of highly personalized interventions, utilizing appropriately timed exercise, light exposure, and feeding as therapeutic modalities for circadian re-entrainment 61.
Genetic Polymorphisms in the Human Circadian System
While environmental factors heavily influence circadian alignment, an individual's inherent chronotype and susceptibility to circadian-related diseases are strongly mediated by genetics. Single nucleotide polymorphisms (SNPs) within core clock genes can alter the stoichiometric balance, structural stability, and binding kinetics of clock proteins, profoundly impacting the TTFL 2062.
The following table summarizes prominent clock gene polymorphisms and their established clinical consequences in human populations.
| Gene | Polymorphism (SNP) | Clinical Phenotypes and Systemic Dysregulation |
|---|---|---|
| CLOCK | rs1801260 (3111T>C) | Enhances CLOCK expression. Associated with extreme evening preference, delayed sleep-wake cycles, and heightened risk for major depressive disorder (MDD) and bipolar disorder (BD). In metabolic contexts, linked to increased small dense LDL, higher daily energy intake, and elevated type 2 diabetes risk. 46626364 |
| CLOCK | rs4580704 | Intronic variant associated with altered risk profiles for cardiovascular disease and diabetes, though functional directionality varies by population cohort. 6364 |
| BMAL1 | rs7950226 | Genotype distributions demonstrate a significant association with inflammatory bowel diseases; the G allele shows higher frequency in patients with ulcerative colitis. 65 |
| BMAL1 | rs11022780 | Intronic variant exhibiting significant association with early-onset preeclampsia (eoPE), likely mediated via disrupted vascular endothelial function and placental oxidative stress. 66 |
| CRY1 | rs2287161 | Significantly linked to delayed sleep phase disorder (DSPD), prolonged nocturnal activity, major depressive disorder (MDD), anxiety, and impaired glucose tolerance. 63656768 |
| CRY2 | rs10838524 | Strongly associated with dysthymia, poor seasonal affective outcomes, and heightened vulnerability to MDD. 4663 |
| PER2 | rs2304672 (C>G) | The minor allele correlates with abdominal obesity, aberrant dietary behaviors (e.g., excessive snacking, skipping breakfast), and metabolic dysregulation. 64 |
| PER3 | rs10746473 | Associated with a lower risk of insulin resistance and lower fasting blood glucose, with effects heavily modified by lifestyle interactions such as alcohol consumption. 68 |
| CK1δ | rs104894561 (T44A) | Autosomal dominant mutation reducing kinase activity. Leads to rapid PER degradation, causing early onset Familial Advanced Sleep Phase (FASP) syndrome. 46 |
The presence of these genetic variations highlights the critical necessity of patient stratification in chronomedicine. Individuals carrying specific risk alleles may inherently require stricter adherence to environmental zeitgebers (e.g., rigid sleep schedules, rigorous bright light therapy) to prevent the cascade of metabolic and psychiatric comorbidities associated with their genetic predisposition 466269.
Conclusion
Chronobiology demonstrates that time is not a passive backdrop to human physiology, but an active, structural component encoded deeply within our molecular biology. The circadian clocks oscillating within nearly every cell are not isolated timers; they form an incredibly complex, systemic network coordinated by the master pacemaker in the suprachiasmatic nucleus and seamlessly entrained by environmental cues.
When this temporal system operates in alignment, it optimizes metabolic efficiency, bolsters immune surveillance, orchestrates essential DNA repair, and maintains neurobehavioral and cognitive stability. Conversely, circadian misalignment - whether induced by modern lifestyle pressures, inappropriate light exposure, asynchronous feeding, or age-related biological decay - serves as an underlying catalyst for a broad spectrum of chronic diseases, ranging from metabolic syndrome to neurodegeneration and cancer.
As the understanding of transcription-translation feedback loops, multi-omics epigenetic clocks, and precise inter-organ communication continues to mature, it offers unprecedented opportunities for clinical intervention. By respecting and actively managing our biological rhythms through targeted chronopharmacology, time-restricted eating, and rigorous light modulation, medical science can fundamentally shift from reactive disease management to proactive resilience building, paving a viable pathway toward extended human longevity and optimal systemic health.