Mechanisms and limits of neural plasticity
Neural plasticity, also referred to as brain plasticity or neuroplasticity, defines the intrinsic capacity of the central nervous system to undergo structural and functional reorganization in response to intrinsic or extrinsic stimuli. Historically, the prevailing dogma in neuroscience held that the adult mammalian brain was a static, structurally fixed organ following the closure of early developmental periods. This view postulated that the generation of new neurons and the significant rewiring of synaptic circuits were impossible in adulthood. Contemporary neurobiological research has entirely dismantled this paradigm, demonstrating that the human brain retains a lifelong, albeit highly regulated, capacity for adaptation.
This adaptive capacity forms the biological foundation for continuous learning, memory consolidation, and behavioral adaptation. Furthermore, it provides the fundamental mechanism for functional recovery following severe neurological insults, such as ischemic stroke or traumatic brain injury (TBI) 12. However, neural plasticity is not an unbounded phenomenon. It is governed by strict molecular constraints, temporal critical periods, and anatomical limitations that dictate both the extent of possible rewiring and the magnitude of functional recovery. Understanding the precise cellular mechanisms, spatial limits, and therapeutic modulators of neural plasticity remains a central objective in both fundamental neuroscience and clinical neurorehabilitation.
Cellular and Molecular Mechanisms of Plasticity
The reorganization of neural networks relies on cascading biological processes that range from temporary molecular alterations at individual synapses to long-term structural remodeling of the neuron itself. These adaptations are broadly categorized into functional plasticity and structural plasticity.
Functional Synaptic Plasticity
Functional plasticity involves the dynamic modification of synaptic efficacy without necessarily altering the physical gross architecture of the neural network. This process dictates how effectively a presynaptic neuron can stimulate or inhibit a postsynaptic neuron. The primary mechanisms governing functional plasticity are long-term potentiation (LTP) and long-term depression (LTD), which respectively strengthen or weaken signal transmission based on synchronous firing patterns - a biological realization of Hebbian theory ("neurons that fire together, wire together") 34.
During LTP, high-frequency stimulation of an excitatory synapse initiates a cascade of intracellular events. The release of the neurotransmitter glutamate binds to post-synaptic receptors, causing an influx of calcium ions. This calcium surge triggers the activation of various kinases, most notably calcium/calmodulin-dependent protein kinase II (CaMKII) 5. The immediate functional result is the rapid recruitment and membrane insertion of additional AMPA and NMDA glutamate receptors into the postsynaptic density 4. This heightened receptor density increases the postsynaptic neuron's sensitivity to subsequent neurotransmitter release, thereby amplifying the synaptic connection without requiring immediate physical growth. Conversely, prolonged low-frequency stimulation induces LTD, resulting in the internalization of these receptors and the weakening of the synaptic link 5.
Structural Remodeling of Dendritic Spines
Structural plasticity refers to the physical alteration of neuronal architecture, providing a stable anatomical foundation for long-term memory storage and motor skill acquisition. In the mammalian cortex, the vast majority of excitatory synapses reside on dendritic spines - microscopic, specialized protrusions extending from the dendritic shaft 4. First observed in 1888 by Santiago Ramón y Cajal, these structures were long thought to be static in adulthood. However, the advent of in vivo two-photon microscopy has unequivocally proven that dendritic spines are highly dynamic; they continuously change shape, form de novo, and undergo selective elimination (pruning) throughout the lifespan 46.
The physical enlargement or shrinkage of a dendritic spine is primarily driven by the rapid reorganization of the actin cytoskeleton. Following the induction of LTP, intracellular signaling cascades regulate actin treadmilling. Globular actin (G-actin) rapidly polymerizes into filamentous actin (F-actin), causing the spine head to physically expand and accumulate structural proteins. This expansion can occur within minutes and persist for weeks 57. Conversely, inactive spines lose stabilizing proteins, shrink, and are eventually retracted into the dendritic shaft 5.
Experience-dependent spine remodeling has been directly observed in animal models. For example, when mice are trained on complex motor tasks, researchers observe the rapid formation of new dendritic spines in the motor cortex. The survival rate of these learning-induced spines correlates directly with the degree of behavioral improvement 6. However, the net rate of structural turnover varies significantly by age. In juvenile mice, widespread spine elimination (13 - 20%) and formation (5 - 8%) occur over brief two-week intervals, reflecting massive developmental pruning. In adults, the rates of genesis and elimination drop significantly and reach a homeostatic balance, ensuring the stability of previously encoded memories while permitting limited ongoing plasticity 56.
Non-Neuronal Contributions to Network Plasticity
While research traditionally focuses on neuronal synapses, network plasticity is heavily modulated by non-neuronal glial cells, including astrocytes, microglia, and oligodendrocytes. Astrocytes enwrap synapses and regulate the extracellular concentration of neurotransmitters, effectively controlling the duration and spread of synaptic signals 8. Furthermore, microglia actively participate in structural plasticity by physically engulfing and pruning weakened synapses during developmental refinement and subsequent learning paradigms 8. Oligodendrocytes, which provide the myelin sheath to axons, also exhibit experience-dependent plasticity; changes in complex environments or social behaviors have been shown to alter myelination patterns in the prefrontal cortex, thereby tuning the conduction velocity of specific neural circuits 8.
Cortical Remapping and Topographic Reorganization
Beyond the microscopic remodeling of individual dendritic spines, neural plasticity manifests as the large-scale spatial reorganization of entire cortical territories. The primary sensory and motor cortices contain highly organized topographic maps representing the body surface (the somatosensory homunculus).
Experience-Dependent Map Alterations
Early evidence demonstrating that adult cortical maps remain plastic was established by Michael Merzenich and colleagues in the 1980s through experiments on the primary somatosensory cortex of adult non-human primates 911. Prior to these studies, it was assumed that cortical representations were permanently fixed in early development. Merzenich's team demonstrated that when specific digits of an adult monkey were amputated, or when the median nerve was transected, the cortical territory corresponding to the deafferented tissue did not remain electrically silent 1011.
Instead, over a period of several weeks, the representations of the adjacent, intact digits systematically expanded to occupy the deprived cortical zones 1011. This remapping proved that sensory representations are dynamically maintained by continuous peripheral input. If that input is removed or altered - such as by fusing two fingers together (syndactyly) to create synchronized sensory input - the cortical map reorganizes to reflect the new pattern of experience 9.
Spatial Limits of Cortical Remapping
While cortical remapping demonstrates substantial adaptability, it is bounded by strict anatomical limits. In cases of digit amputation, the translocation of functional sensory representations across the cortex rarely exceeds a physical distance of 600 to 700 microns 1011. This spatial restriction suggests that functional reorganization in the adult brain does not typically involve the long-distance growth of entirely new axonal tracts. Instead, remapping relies heavily on the "unmasking" and strengthening of pre-existing, latent horizontal connections within the local cortical network 1012. When primary input is removed, local GABAergic inhibition decreases, allowing previously sub-threshold excitatory inputs from adjacent cortical regions to drive the deafferented neurons 1011.
Cross-Modal Plasticity
In instances of severe sensory deprivation present from birth or early childhood - such as congenital blindness or deafness - plasticity can transcend individual sensory maps in a phenomenon known as cross-modal plasticity. When the primary visual cortex is deprived of optical input, it does not atrophy completely; instead, it is often recruited to process incoming tactile and auditory information, enhancing spatial hearing and Braille reading capabilities 813. While cross-modal plasticity is most profound when deprivation occurs during early developmental critical periods, limited forms of cross-modal synaptic scaling have also been documented following peripheral nerve injury in adulthood 813.
Developmental Plasticity Versus Adult Plasticity
The magnitude, speed, and underlying mechanisms of neural plasticity differ drastically depending on the organism's developmental stage. While the adult brain retains the capacity to rewire, it is fundamentally constrained compared to the pervasive malleability observed during embryonic, neonatal, and adolescent development.
Critical and Sensitive Periods
During early life, the nervous system passes through specific temporal windows known as critical periods and sensitive periods. A critical period is a strict, biologically predetermined timeframe during which specific environmental stimuli must be present for a neural circuit to develop properly 141516. If the necessary input is absent, the resulting structural deficits are often permanent. A sensitive period refers to a broader, more flexible developmental phase when the brain is optimally responsive to learning (e.g., native language acquisition), though some adaptation remains possible outside this window with significantly greater effort 14161718.
The classic paradigm for critical period plasticity was established by Hubel and Wiesel in the visual system. Suturing one eye of a kitten shut during early postnatal development causes the visual cortex to permanently rewire in favor of the open eye, resulting in cortical blindness in the deprived eye (amblyopia). If the same deprivation occurs in an adult cat, the visual cortex remains largely unchanged, and normal vision returns once the eye is opened 1920.
Initiation and Closure of Plasticity Windows
The timing of critical periods is not dictated simply by chronological age, but by the maturation of local inhibitory interneuron networks. Specifically, the accumulation of the inhibitory neurotransmitter gamma-aminobutyric acid (GABA) shifts the excitatory-inhibitory (E/I) balance of the cortical circuit, acting as the biological trigger that opens the critical period window 14171921.
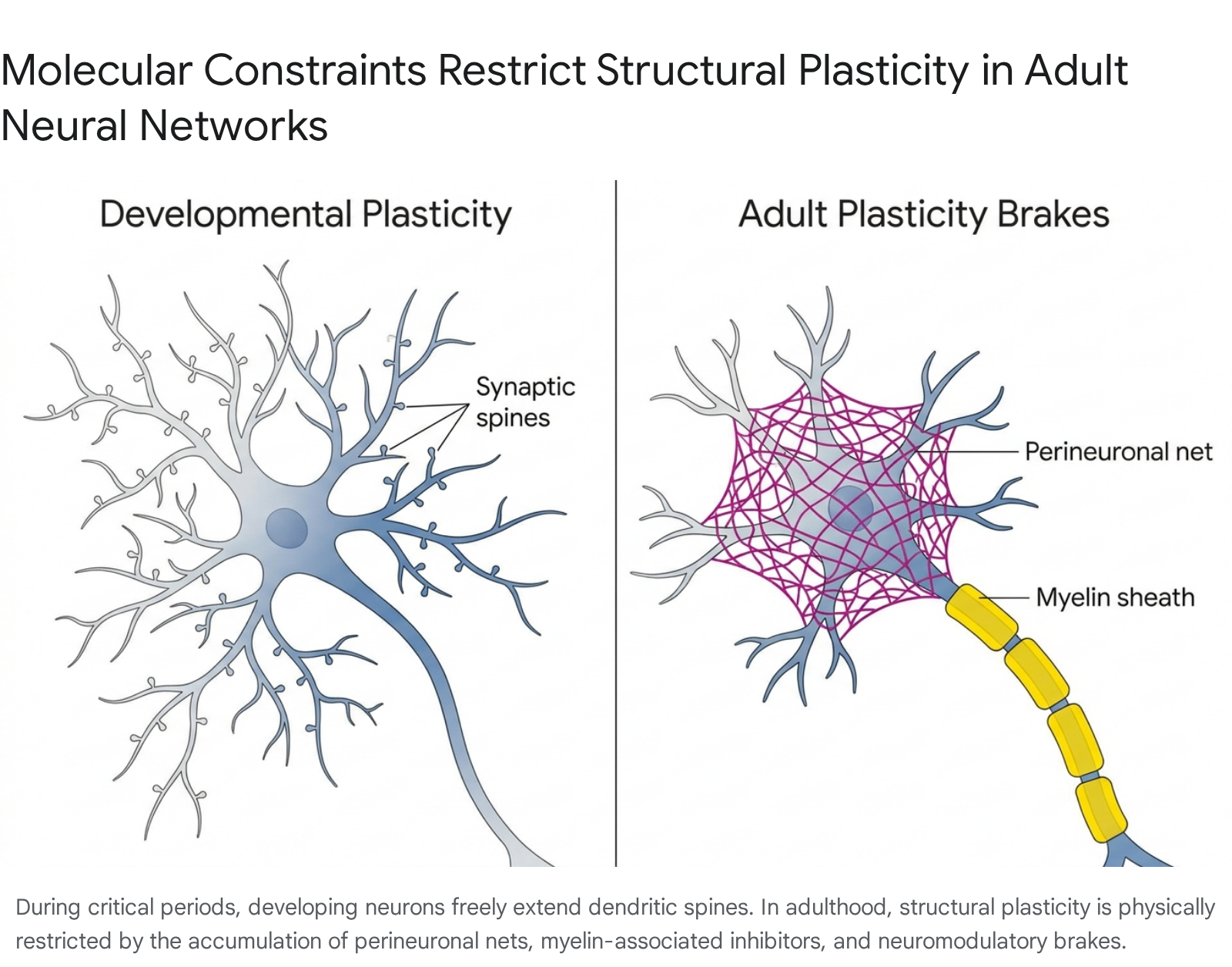
As development progresses, evolutionary mechanisms actively suppress broad plasticity to ensure the stability of established memories and prevent runaway network excitation. The closure of the critical period is enforced by several distinct molecular and structural "brakes" 821.
- Perineuronal Nets (PNNs): As the brain matures, specialized extracellular matrix structures composed of chondroitin sulfate proteoglycans condense around the cell bodies and proximal dendrites of parvalbumin-positive inhibitory neurons. PNNs act as a physical mesh that restricts synaptic remodeling and prevents the outgrowth of new dendritic spines 8171921.
- Myelin-Associated Inhibitors: The maturation of oligodendrocytes coats axons with myelin to increase action potential velocity. However, mature myelin contains potent inhibitory proteins, such as Nogo-66, which actively repel axonal sprouting and limit structural plasticity 821.
- Neuromodulatory Brakes: In the adult brain, the protein Lynx1 binds to nicotinic acetylcholine receptors, dampening the cortex's responsiveness to cholinergic signals. Because acetylcholine is vital for arousal-based learning, Lynx1 functionally limits experience-dependent plasticity. Experimental removal of Lynx1 has been shown to restore critical-period-like visual plasticity in adult animal models 21.
- Epigenetic Silencing: The transition to adulthood alters the chromatin state of neurons. Factors such as Gadd45b, an enzyme involved in DNA demethylation, are down-regulated in adulthood. This epigenetic shift silences the transcription of specific plasticity-related genes that were highly active during juvenile development 22.
The following table summarizes the primary distinctions between developmental critical periods and the constrained plasticity observed in mature neural networks.
| Biological Paradigm | Developmental Critical Periods | Adult Neural Plasticity |
|---|---|---|
| Primary Driver | Experience-expectant (brain requires specific stimuli to form standard architecture) | Experience-dependent (driven by intentional learning, memory encoding, or injury) |
| Magnitude of Remodeling | Massive structural rewiring, pervasive synaptogenesis, and high rates of spine elimination | Localized synaptic unmasking, subtle dendritic spine resizing, constrained spatial remapping |
| Biochemical Initiation | Regulated by the maturation of GABAergic inhibitory circuits and shifting E/I balance | Requires salient behavioral effort, high attention, and neuromodulatory arousal (e.g., dopamine, acetylcholine) |
| Molecular Constraints | Highly permissive environment; low myelin inhibition and absence of PNNs | Actively suppressed by Perineuronal Nets, Nogo-66, Lynx1, and epigenetic methylation |
| Response to Deprivation | Leads to severe, often irreversible functional deficits (e.g., permanent amblyopia) | Causes reversible functional shifts; maps can frequently be restored through targeted rehabilitation |
Data derived from comparative neurobiological analyses of critical periods and adult map reorganization 81416192123.
The Debate on Human Adult Neurogenesis
Beyond the remodeling of existing synapses, a long-standing controversy in neuroscience centers on whether the adult human brain can generate entirely new neurons. In non-human mammals, adult neurogenesis is well-documented in the subventricular zone (supplying the olfactory bulb) and the subgranular zone of the hippocampal dentate gyrus 2425. However, translating these findings to humans has been historically fraught with methodological challenges, leading to highly contradictory publications.
In 2018, the debate fractured sharply. A high-profile study by Sorrells et al. concluded that neurogenesis in the human hippocampus drops to negligible, undetectable levels during early childhood 24. Almost simultaneously, a study by Boldrini et al. reported that robust neurogenesis persists throughout the entire human lifespan 24. The discrepancy stemmed largely from the limitations of postmortem tissue preservation and the reliance on single, potentially unreliable molecular markers (such as doublecortin) to identify immature neurons via immunohistochemistry 2426.
Recent Evidence from Single-Nucleus Transcriptomics
In July 2025, the debate advanced significantly with the publication of a landmark study in Science by researchers at the Karolinska Institute, led by Marta Paterlini and Jonas Frisén. To overcome previous methodological limitations, the team utilized single-nucleus RNA sequencing combined with advanced spatial mapping techniques (RNAscope and Xenium) to analyze over 400,000 individual cell nuclei extracted from 24 human postmortem brains 2728293031. The donor brains ranged in age from neonates to 78 years 272832.
Rather than relying on single protein markers, the researchers trained a machine learning algorithm on the complete transcriptomic signature of neural progenitor cells found in infants, then searched for that identical, complex genetic profile in the adult samples 2630. The analysis identified 354 cells in various stages of neuronal development within the adult dentate gyrus, confirming the presence of dividing precursor cells situated directly adjacent to fully formed mature neurons 272829.
While the Karolinska study provides compelling evidence that adult neurogenesis does not entirely cease, the biological scale and functional necessity of this process in older adults remain a subject of active research. Independent neuroscientists emphasize that the absolute number of these progenitor cells is relatively low, and interindividual variation is high (e.g., several adult brains in the study showed negligible progenitor counts) 263031. Nonetheless, the confirmation that the human hippocampus - a region central to learning, episodic memory, and emotional regulation - retains a reservoir of neurogenic stem cells opens major therapeutic avenues for targeting cognitive decline and neurodegenerative diseases 283233.
Clinical Harnessing of Plasticity: Neurorehabilitation
When the adult central nervous system sustains damage from an ischemic stroke or a traumatic brain injury (TBI), the brain undergoes a complex, biphasic plasticity response. Immediately following the acute injury, there is a pronounced decrease in GABAergic inhibition and widespread synapse loss. This is followed days to weeks later by a phase of heightened neurite expansion, synaptic unmasking, and collateral sprouting within the peri-infarct regions 123.
However, spontaneous recovery is rarely complete. Maladaptive plasticity - where the brain rewires in counterproductive ways - can lead to chronic pain, allodynia, post-stroke spasticity, or the onset of late post-traumatic epilepsy 1134. The goal of modern neurorehabilitation is to leverage the post-injury plasticity window to drive adaptive functional reorganization.
Physical and Behavioral Interventions
Clinical strategies rely heavily on task-oriented, high-repetition physical interventions to force the brain to re-engage damaged networks. One of the most rigorously studied protocols is Constraint-Induced Movement Therapy (CIMT). Designed for patients with unilateral upper limb paresis following stroke, CIMT involves physically restraining the unaffected arm, forcing the patient to utilize the impaired limb 23536. By combating "learned non-use," CIMT stimulates activity-dependent synaptic plasticity and drives the expansion of motor representations in the surviving cortex 35.
The efficacy of rehabilitation protocols is continuously evaluated through systematic reviews, prominently by the Cochrane Collaboration, which utilizes strict hierarchies of clinical evidence. Moderate-quality evidence supports the use of CIMT; a meta-analysis of 28 studies involving 858 participants found a significant standardized mean difference (SMD) of 0.34 in favor of CIMT for improving arm motor function 3637. Similarly, moderate-quality evidence supports Mirror Therapy - where a patient moves their intact limb in front of a mirror, creating a visual illusion that the paretic limb is moving. This technique engages action-observation neural networks, yielding an SMD of 0.47 for motor function improvement across 36 studies 3738.
Conversely, evidence for generic cognitive rehabilitation intended to restore memory or executive function after stroke or TBI remains mixed. While some short-term subjective benefits in memory reporting are noted (moderate-quality evidence, SMD 0.36), there is currently low-to-very-low certainty of evidence that computerized cognitive rehabilitation yields long-term, objective improvements in functional independence or quality of life 394041.
Non-Invasive Neuromodulation
To lower the threshold for synaptic plasticity and accelerate rehabilitation, clinicians increasingly employ non-invasive brain stimulation techniques, primarily Transcranial Direct Current Stimulation (tDCS) and Repetitive Transcranial Magnetic Stimulation (rTMS) 42434445.
tDCS involves passing a weak, continuous electrical current (typically 1 to 2 milliamperes) through the scalp. Anodal stimulation generally depolarizes resting neuronal membrane potentials, facilitating LTP and increasing cortical excitability, while cathodal stimulation hyperpolarizes membranes, promoting LTD 444546. These interventions are highly dependent on the brain's homeostatic plasticity mechanisms. For instance, applying an inhibitory 1 Hz rTMS pulse immediately following inhibitory cathodal tDCS does not compound the inhibition; rather, homeostatic feedback loops reverse the effect, leading to a paradoxical increase in cortical excitability to prevent the network from becoming completely suppressed 45.
Recent advancements pair neurostimulation with advanced imaging techniques. Functional Magnetic Resonance Imaging (fMRI) allows clinicians to precisely map disrupted neural circuits - such as the dorsolateral prefrontal cortex in severe depression or the peri-infarct zone in stroke - and deliver targeted magnetic pulses to strengthen specific synaptic pathways 4750. Combined approaches, such as pairing tDCS with CIMT, have demonstrated synergistic functional improvements by electrically priming the motor cortex for the activity-dependent reorganization demanded by physical therapy 35.
Pharmacological Augmentation: Psychoplastogens
A rapidly expanding frontier in neuroscience is the use of pharmacological agents specifically designed to reopen plasticity windows in the adult brain. Termed "psychoplastogens," these compounds are being investigated for their ability to rapidly alter synaptic density and treat conditions characterized by rigid, maladaptive neural circuitry, including treatment-resistant depression, post-traumatic stress disorder (PTSD), and substance use disorders 4849.
Ketamine and Glutamatergic Modulation
Ketamine, an NMDA receptor antagonist, provides the most well-documented example of pharmacological plasticity. At sub-anesthetic doses, ketamine triggers a paradoxical surge in presynaptic glutamate release, which subsequently over-activates postsynaptic AMPA receptors. This activity stimulates the mammalian target of rapamycin (mTOR) signaling pathway and induces the rapid release of brain-derived neurotrophic factor (BDNF) 484950. The cellular result is a dramatic increase in the growth of new dendritic spines and the strengthening of existing synapses in the prefrontal cortex within 24 to 72 hours of administration - correlating directly with its rapid-acting antidepressant effects 5049.
Serotonergic Psychedelics and Neuropeptides
Classic psychedelics, including psilocybin and LSD, are also potent psychoplastogens. Operating primarily as agonists at the 5-HT2A serotonin receptor, these compounds trigger intracellular cascades that promote significant structural remodeling, increasing dendritic arborization and spine density 4849. Human neuroimaging studies demonstrate that these agents temporarily dissolve established resting-state networks (decreasing network modularity) while increasing global brain connectivity, suggesting a state of enhanced cognitive flexibility and neuroplastic potential 484950. Similarly, MDMA modulates monoamine pathways to enhance BDNF expression, facilitating the fear-extinction learning required in PTSD therapy 49.
Research is also expanding into endogenous neuropeptides. Chronic administration of oxytocin in aged murine models has been shown to upregulate plasticity markers such as glutamate receptor 1 (GluR1) and specific NMDA receptor subunits, while simultaneously increasing markers of hippocampal neurogenesis. These findings position neuropeptide signaling as a potential therapeutic avenue for combating age-related cognitive decline by restoring baseline synaptic adaptability 4850.
Constraints and Efficacy in Cognitive Training
While clinical neurorehabilitation effectively targets focal motor deficits, the pursuit of commercial cognitive enhancement for healthy adults has generated considerable scientific and regulatory scrutiny. A multi-million-dollar industry of digital "brain-training" applications (e.g., Lumosity, Elevate, Peak) markets products based on the premise that repeated practice of simplified cognitive games can leverage neuroplasticity to improve general intelligence, prevent dementia, and enhance real-world occupational performance 51525354.
The Near-Transfer Versus Far-Transfer Debate
The scientific evaluation of these applications hinges on the distinction between near-transfer and far-transfer. Near-transfer occurs when practicing a specific task yields performance improvements on structurally similar tasks (e.g., training on a digit-span memory game improves scores on other laboratory working memory tests). Far-transfer occurs when training on an abstract task produces broad, generalized improvements in untrained, real-world domains, such as fluid intelligence, reading comprehension, or daily executive functioning 5556575859.
Extensive peer-reviewed meta-analyses have consistently demonstrated that while brain-training applications reliably produce near-transfer effects - users inevitably get better at the specific games they play - there is no compelling, high-quality scientific evidence that they produce far-transfer 5456575960. A massive cross-sectional analysis of over 60,000 individuals revealed that individuals who engaged in commercial brain training exhibited no global cognitive advantages in reasoning or verbal abilities compared to individuals who engaged in other active cognitive pursuits, such as regular video gaming or board games 57.
A comprehensive review of 132 journal articles cited by brain-training proponents, published in Psychological Science in the Public Interest, identified severe methodological flaws in the industry's evidence base. Many cited studies utilized inadequate control groups, relied on extremely small sample sizes, or failed to account for robust placebo effects 6061. The absence of empirical backing for far-transfer claims has resulted in regulatory intervention; in 2016, the United States Federal Trade Commission fined Lumos Labs (the creators of Lumosity) $2 million for deceptive advertising regarding their product's ability to delay cognitive decline and improve real-world academic performance 6162. While digital cognitive exercises may offer some modest engagement benefits for specific populations, they do not fundamentally upgrade the brain's generalized "software" in a manner analogous to physical muscle training 54606163.
Global Disparities and Translation in Clinical Practice
Translating complex neurobiological insights regarding plasticity into effective clinical outcomes requires robust, scalable healthcare infrastructure. Currently, access to plasticity-driven interventions is highly unequal, with the vast majority of advanced clinical trials, robotic exoskeletons, and precision neuromodulation technologies concentrated in high-income nations 67646566.
The Burden in Low- and Middle-Income Countries
In regions such as Latin America, the Middle East, and Africa, the burden of neurological disability is accelerating due to rising stroke incidence and unmitigated cardiovascular risk factors. In Latin America alone, the Years Lived with Disability (YLDs) due to stroke rose by over 50% between 2000 and 2019 6768. Despite this immense need, multinational surveys reveal severe discrepancies in rehabilitation care. Post-stroke rehabilitation services are fundamentally restricted by a lack of public health coverage, severe shortages of specialized human resources (such as board-certified physiatrists and speech therapists), and geographic isolation 42656769.
Furthermore, the external validity of Western rehabilitation trials is frequently compromised when applied globally. For instance, a major stroke rehabilitation protocol trial in Brazil screened 571 patients but could only enroll 5.6% due to the extremely high regional rate of recurrent stroke - a factor intrinsically linked to lower socioeconomic status that is often an exclusion criterion in high-income country trials 64.
Protocol Harmonization and Capacity Building
To address these disparities, international consortia are attempting to harmonize clinical trial protocols and build sustainable research capacity in low-resource settings. Initiatives such as the Good Trials Prism - supported by the Wellcome Trust and involving networks like the Africa Health Research Institute - aim to implement rigorous, locally relevant randomized controlled trials across South Asia, Latin America, and South Africa 70. Similarly, academic partnerships, such as the bidirectional training model between the Barrow Neurological Institute and the University of the Witwatersrand in Johannesburg, are training specialized neurologists to establish self-sustaining neurocritical care and rehabilitation centers in underserved regions 71.
The implementation of unified, regional clinical guidelines is critical for maximizing experience-dependent plasticity. In nations with mature healthcare infrastructure, such as Japan, acute stroke rehabilitation is highly structured. Patients are mobilized within 24 to 48 hours to prevent immobility complications and are subsequently transferred to dedicated convalescent rehabilitation wards for intensive, high-dose physical therapy 42727374. Replicating this systematic approach in developing regions requires combining localized clinical guidelines with accessible digital health innovations. Open-source, affordable technologies - such as the 4-channel IoT electrostimulators developed in Brazil for functional electrical stimulation, or widespread telerehabilitation platforms - offer vital pathways for delivering consistent, high-frequency motor training to populations cut off from urban medical centers 676667.
Ultimately, the optimization of neural plasticity transcends microscopic synaptic remodeling; it necessitates the integration of advanced molecular pharmacology, rigorous evidence synthesis utilizing emerging tools like artificial intelligence, and the equitable global distribution of evidence-based rehabilitation protocols 757677.