Lymphatic system decline during aging
The lymphatic system is a ubiquitous and vital network of vessels, nodes, and organs responsible for maintaining interstitial fluid homeostasis, facilitating intestinal lipid absorption, and orchestrating systemic immune surveillance. Unlike the cardiovascular system, which relies on the mechanical force of the heart to circulate blood, the lymphatic vasculature depends on a delicate combination of extrinsic mechanical tissue forces and the intrinsic, spontaneous contractions of lymphatic muscle cells. This decentralized transport network propels fluid, macromolecules, and immune cells against gravity and pressure gradients, ultimately returning them to the venous circulation.
With advancing age, the structural integrity and functional capacity of the lymphatic system undergo profound deterioration. This age-related decline - characterized by endothelial hyperpermeability, muscle cell atrophy, impaired contractility, and lymph node fibrosis - precipitates far-reaching systemic consequences. Contemporary research demonstrates that lymphatic senescence is not merely an isolated physiological shift but a core driver of systemic inflammaging, impaired metabolic regulation, and the pathogenesis of neurodegenerative conditions such as Alzheimer's disease. Understanding the precise mechanisms of lymphatic aging provides crucial insight into the progressive failure of tissue waste clearance and opens novel avenues for therapeutic intervention.
Biological Mechanisms of Lymphatic Senescence
The degradation of the lymphatic system with age occurs at multiple biological levels, spanning gross anatomical architecture, cellular signaling pathways, and extracellular matrix composition. These compounding deficits transform the lymphatic vasculature from a highly efficient drainage and filtration network into a sluggish, highly permeable system that fosters chronic inflammation and tissue dysfunction.
Structural Degradation of Lymphatic Capillaries and Collectors
Lymphatic fluid transport relies on an intricate balance between initial lymphatic capillaries, which absorb interstitial fluid, and collecting lymphatic vessels, which transport the fluid toward regional lymph nodes. Initial lymphatics are lined with endothelial cells connected by discontinuous, "button-like" junctions that permit the entry of fluid, immune cells, and large macromolecules into the vessel lumen 1. Conversely, collecting vessels feature tight "zipper-like" junctions, intraluminal bi-leaflet valves to prevent retrograde flow, and a functional covering of lymphatic muscle cells (LMCs) that provide the intrinsic pumping force necessary for lymph propulsion 12.
Aging induces severe structural remodeling within both types of vessels. In aged organisms, the density and branching of initial lymphatic vessels decrease significantly. Studies of the cutaneous lymphatic network indicate age-related reductions of up to 35% in initial vessel density and 25% in vessel branching 23. Concurrently, the collecting vessels undergo pathological dilation, LMC atrophy, and the destruction of essential elastic elements. This loss of elasticity frequently results in the formation of aneurysm-like "varicose bulges" along the vessel walls 4. Furthermore, the morphological orientation of LMCs shifts from a functional, force-generating circular arrangement to a disorganized longitudinal orientation, severely compromising their ability to generate contractile force 2.
At the molecular level, this loss of contractility is driven by a reduction in the expression of crucial contractile and cytoskeletal proteins, including myosin, troponin, dynein, actin, and gelsolin 5. Furthermore, the physiological generation of nitric oxide (NO) by endothelial nitric oxide synthase (eNOS) - a critical regulator of vessel relaxation and subsequent contraction frequency - is disrupted. In aged collecting vessels, the responsiveness to physiological shear stress and external inflammatory stimuli is blunted, leading to a diminished capacity to adjust pumping rates in response to localized tissue edema 56.
Endothelial Cell Senescence and Barrier Permeability
The endothelial cells lining the lymphatic vasculature are highly susceptible to cellular senescence, an irreversible state of cell cycle arrest accompanied by a senescence-associated secretory phenotype (SASP). Normal lymphatic endothelial cell (LEC) survival, proliferation, and function are primarily driven by Vascular Endothelial Growth Factor Receptor 3 (VEGFR-3) signaling. Aging significantly diminishes VEGFR-3 signaling, leading to increased LEC apoptosis and widespread cellular senescence across the lymphatic network 23.
A primary hallmark of an aged lymphatic endothelium is increased barrier permeability, which allows lymph fluid, unneutralized pathogens, and metabolic waste to leak back into the surrounding interstitial space. This hyperpermeability is caused by the progressive degradation of the endothelial glycocalyx - a protective, sugar-rich glycoprotein layer lining the vessel lumen - and the dissociation of critical junctional protein complexes, such as the VE-cadherin and catenin complex 47. The loss of the glycocalyx directly impairs the vessel's ability to prevent pathogen adhesion and allows potentially harmful macromolecules to escape from the collectors into the peripheral tissues 4. Consequently, aged lymphatic vessels exhibit an increased accumulation of peri-lymphatic leukocytes and elevated nitrosative stress, establishing a localized feed-forward loop of chronic, low-grade inflammation 23.
Lymph Node Architecture and Immunosenescence
Lymph nodes function as critical filtration checkpoints and physical hubs for the adaptive immune system. The structural integrity of a lymph node is maintained by a specialized network of lymph node stromal cells (LNSCs), including fibroblastic reticular cells (FRCs), lymphatic endothelial cells (LECs), and blood endothelial cells (BECs) 89. As age advances, the architecture of the lymph node undergoes progressive deterioration, marked heavily by lipomatosis (the accumulation of adipose tissue) and fibrosis (the excessive deposition of collagen and extracellular matrix proteins) 810.
This structural degradation directly impairs systemic immune function. The network of FRCs, which typically elongates and relaxes to accommodate the influx and rapid proliferation of lymphocytes during an acute immune response, loses its vital plasticity. Aged FRCs exhibit delayed proliferative responses and reduced activation, severely crippling the node's ability to expand and coordinate defenses against new pathogens 8. Furthermore, the expression of key chemokines, such as CCL21, which is necessary for attracting dendritic cells from the periphery into the lymphatic vessels and subsequently into the draining lymph nodes, is significantly reduced in aged lymphatic endothelia 810.
These localized nodal changes are inextricably linked to the broader phenomenon of systemic immunosenescence. The T-cell compartment within both the lymph nodes and the peripheral blood undergoes extensive, lifelong remodeling. There is a dramatic decline in the absolute number and functional output of naive T-cells - driven heavily by the age-related involution of the thymus gland - and a corresponding massive expansion of terminally differentiated, senescent memory T-cells 101112. This compositional shift severely restricts the T-cell receptor (TCR) repertoire, limiting the adaptive immune system's ability to recognize novel antigens and mount effective responses to new vaccines 101112. Simultaneously, the accumulation of senescent B-cells - often referred to as age-associated B cells (ABCs) - actively inhibits normal B-cell lymphopoiesis and disrupts central immune tolerance, increasing the incidence of autoantibodies and autoimmune conditions in the elderly 1013.
Quantitative Physiological Decline in Lymphatic Transport
The qualitative observation of lymphatic senescence is firmly corroborated by strict quantitative metrics derived from both murine models and human clinical studies. These multi-species data illustrate a progressive, age-dependent failure in the system's mechanical ability to transport fluid and clear waste from peripheral and central tissues.
Human Clinical Measurements of Lymphatic Pumping
In humans, the intrinsic pumping pressure of collecting lymphatic vessels demonstrates a clear, measurable inverse relationship with age. In a definitive study involving 399 healthy volunteers aged 20 to 91, researchers utilized minimally invasive indocyanine green (ICG) fluorescence lymphography combined with a custom-made transparent sphygmomanometer cuff to accurately measure leg lymphatic pumping pressure 141515. The cuff was inflated and gradually deflated until the fluorescent dye was visually observed exceeding the upper border, defining the absolute pumping pressure.
The analysis revealed a statistically significant negative correlation between leg lymphatic pumping pressure and advancing age (r = -0.34, p < 0.0001) 1415. Individuals exhibiting poor pumping pressures - defined as falling below 20 mmHg in both legs - reported significantly higher rates of clinical leg edema and diminished overall quality of life compared to subjects with moderate (20-40 mmHg) or good (>40 mmHg) pumping pressures 15. Notably, this age-associated decline in lymphatic pumping pressure was found to be most pronounced in women of postmenopausal age, suggesting a potential hormonal influence on lymphatic muscle cell function 1415.
Murine Models of Cervical Lymphatic Clearance
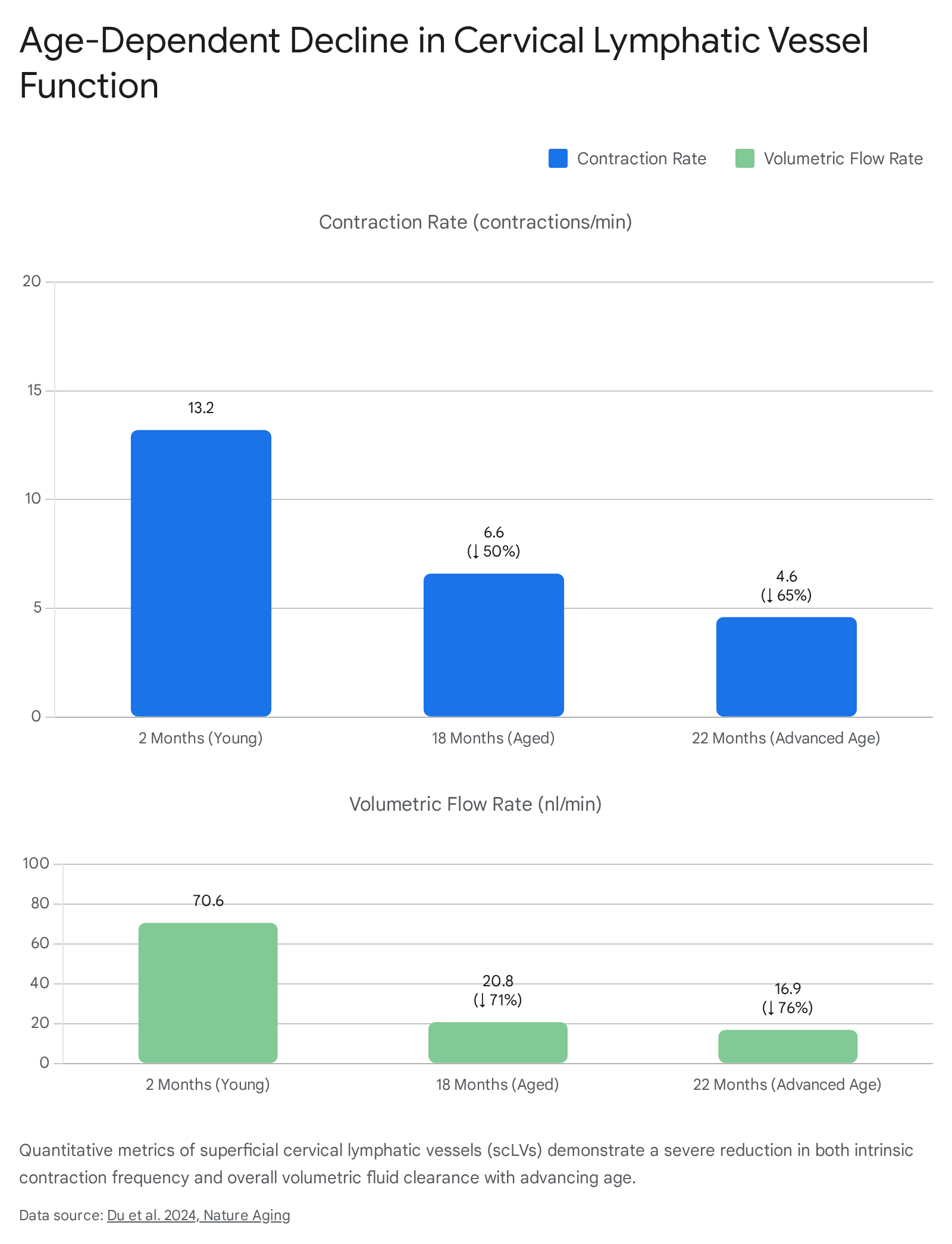
Animal models provide further high-resolution quantitative data regarding the specific trajectory of this mechanical decline. A 2024 study assessing the function of superficial cervical lymphatic vessels (scLVs) in mice successfully mapped the exact progression of contractile failure over a standard mammalian lifespan 16.
In young mice (2 months of age), the intrinsic contraction rate of the superficial cervical lymphatic vessels averaged 13.2 contractions per minute. This baseline contractility maintained an average volumetric flow rate of 70.6 nanoliters per minute, ensuring efficient fluid turnover from the craniocervical compartment 16. By 18 months of age, representing the onset of physiological aging, the contraction rate precipitously declined by 50% to roughly 6.6 contractions per minute, and the volumetric flow rate dropped correspondingly to 20.8 nanoliters per minute 16. In advanced age (22 months), the contraction rate fell by 65% compared to baseline, yielding a severely depressed flow rate of only 16.9 nanoliters per minute and a 63% reduction in downstream flow velocity 16.
Translational Limitations and Diversity Gaps
While murine models have exponentially accelerated the discovery of lymphatic mechanisms, translational gaps complicate the direct application of these findings to human aging. Fundamental differences exist in lymphatic architecture, physiology, and gravitational exposure. For instance, mice are quadrupeds and do not experience the chronic gravitational loads on the lower extremities that bipedal humans do, fundamentally altering the mechanics of lower-limb lymphatic failure and subsequent lymphedema 1718. Furthermore, while human visceral and thoracic collecting lymphatics rely heavily on intrinsic pumping to move lymph fluid upward, aspects of mouse thoracic duct physiology do not accurately mirror this specific contractility 1718.
There is also a pressing need for diverse human cohort studies. The incidence of specific lymphatic pathologies varies globally, with secondary lymphedema driven heavily by the parasitic nematode infection lymphatic filariasis in endemic regions of Asia, Africa, and the Americas, while surgical and radiotherapeutic oncologic interventions primarily drive incidence in developed nations 1921. Emerging multinational longitudinal studies - such as the Health and Aging in Africa: A Longitudinal Study in South Africa (HAALSI) and the Asian Cohort for Alzheimer's Disease (ACAD) - are currently attempting to harmonize data on how biological aging, genetic markers, and environmental stressors intersect to influence cognitive and systemic lymphatic decline across diverse populations 202122.
| Metric Category | Human Presentation | Murine Model Presentation | Key Translational Difference |
|---|---|---|---|
| Lower Extremity Fluid Dynamics | Heavily influenced by bipedal gravity; high incidence of dependent edema with age. | Quadrupedal posture minimizes dependent gravitational pooling. | Mechanical stress on initial lymphatics differs drastically. |
| Thoracic Duct Pumping | High reliance on intrinsic smooth muscle contractility to move fluid vertically. | Thoracic collecting lymphatics do not exhibit the same robust intrinsic pumping. | limits direct modeling of human central lymph transport. |
| Primary Pathological Drivers | Oncologic surgery (developed nations); Lymphatic filariasis (developing nations). | Genetic knockout models (e.g., Chy mice); surgical ablation. | Murine models rarely replicate the chronic parasitic or radiotherapeutic tissue damage seen in humans. |
Systemic Consequences of Lymphatic Dysfunction
The deterioration of the lymphatic system is not an isolated mechanical failure; it creates a cascade of physiological deficits that influence nearly every major organ system. The most profound and highly researched implications of lymphatic senescence are observed in the central nervous system, systemic lipid metabolism, and global immune homeostasis.
Central Nervous System Clearance and Neurodegeneration
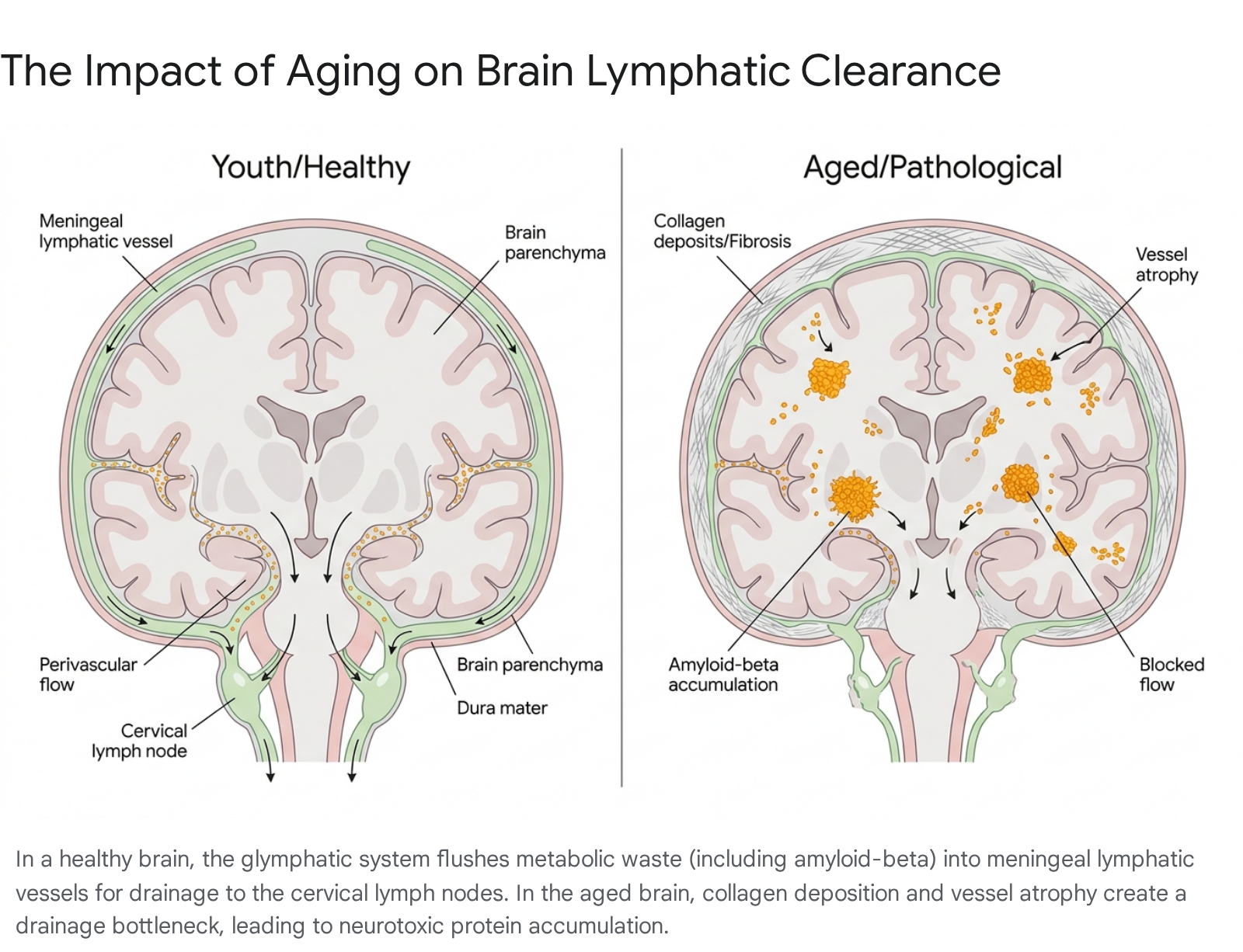
The brain represents a unique immunological environment because it lacks a conventional parenchymal lymphatic vessel network. Instead, it relies on a highly specialized waste clearance mechanism known as the glymphatic system, which works in seamless tandem with the meningeal lymphatic vessels 2523242526. Cerebrospinal fluid (CSF) flows into the brain tissue along perivascular spaces, facilitated by aquaporin-4 (AQP4) water channels located on the endfeet of astrocytes 2527. This fluid actively mixes with the interstitial fluid of the brain, collecting metabolic waste, cellular debris, and neurotoxic proteins, before effluxing back out toward the meninges. The meningeal lymphatic vessels, located within the dura mater, absorb this waste-laden fluid and drain it out of the skull into the deep cervical lymph nodes (dCLNs) for systemic processing and clearance 24282930.
Aging induces catastrophic failures along this entire clearance axis. Meningeal lymphatic vessels undergo age-related narrowing, reduced spatial branching, and severe functional impairment, significantly slowing the paravascular influx of fresh CSF and the efflux of interstitial waste 23262831. Recent pathological data indicates that this decline is severely exacerbated by extracellular matrix remodeling in the aged dura; specifically, the progressive deposition of peri-lymphatic collagen forms a physical fibrotic barrier that constricts the vessels and prevents the efficient uptake of CSF 32.
The primary consequence of this impaired drainage is the accumulation of amyloid-beta (Aβ) and hyperphosphorylated tau proteins within the brain parenchyma - the widely recognized pathological hallmarks of Alzheimer's disease (AD) and other dementias 24313334.
In transgenic mouse models of Alzheimer's disease (such as the 5xFAD model), the artificial ablation of meningeal lymphatics drastically increases amyloid plaque deposition and visibly worsens cognitive, spatial learning, and memory deficits 28313338. Conversely, impaired lymphatic drainage triggers a maladaptive neuroinflammatory response within the brain. The accumulation of metabolic waste provokes microglia to release inflammatory cytokines, particularly Interleukin-6 (IL-6). Elevated IL-6 alters the balance of cortical excitatory and inhibitory synaptic inputs, actively driving neuronal dysfunction and cognitive decline 3835.
Biomarker Enrichment in Cervical Lymph Nodes
Because the deep cervical lymph nodes act as the primary sink for brain-derived waste, they provide a unique window into central nervous system pathology. A landmark 2025 study investigated the presence of neurodegenerative fluid biomarkers directly in human cervical lymph nodes using ultrasound-guided fine-needle aspiration 363738.
The clinical findings confirmed that neurodegenerative biomarkers are highly enriched in the cervical lymph nodes compared to matched peripheral blood plasma. Specifically, phosphorylated tau 181 (pTau181) was found at concentrations averaging 4,865 pg/ml in CLN aspirates compared to 18.3 pg/ml in plasma, representing an extraordinary 266-fold enrichment 3637. Amyloid-beta 40 and 42 were also significantly elevated, exhibiting an approximate 4.5-fold enrichment over plasma levels 36. Crucially, the concentration of pTau181 in the cervical lymph nodes demonstrated a significant inverse correlation with the subject's age (r = -0.66, P = 0.001) 3637. This negative correlation in living humans strongly supports the preclinical hypothesis that the meningeal-cervical lymphatic drainage pathway progressively loses its physical capacity to extract neurotoxic proteins from the brain as humans age. While absolute concentrations for certain markers like glial fibrillary acidic protein (GFAP) and neurofilament light (NfL) were not uniformly elevated across all cohorts, the profound enrichment of tau highlights the lymphatic system's role in neurodegenerative disease tracking 36.
Intestinal Lipid Transport and Metabolic Homeostasis
Beyond immune and neurological functions, the lymphatic system is exclusively responsible for the transport of dietary lipids from the intestine to the systemic circulation. Within the small intestine, highly specialized lymphatic capillaries known as lacteals absorb dietary fats that have been packaged into large chylomicron particles by enterocytes 139. The entry of chylomicrons into lacteals is not a passive diffusion process; it requires the active, dynamic opening of the button-like junctions between lymphatic endothelial cells, driven by ROCK (Rho-associated kinase) signaling and heavily modulated by the VEGF-A/VEGFR-2 and VEGF-C/VEGFR-3 signaling pathways 140.
While baseline lipid digestion and absorption remain relatively well-preserved during healthy aging, the dynamic adaptability of this intestinal transport system declines precipitously 41. Under physiological stress or intercurrent illness, aged animals and humans display significant impairments in the assembly and transport of lipoproteins from the enterocytes into the lymphatic network 41. Alterations in intestinal lymphatic structure, including changes in junctional integrity (pathological zippering) and the decline of molecular regulators like Calcitonin Receptor-Like Receptor (CALCRL), can lead to systemic dyslipidemia and predispose elderly individuals to compounding metabolic conditions such as severe obesity, insulin resistance, and hepatic steatosis 394042.
Adipose Tissue Senescence and Chronic Systemic Inflammation
Lymphatic dysfunction is recognized as a major contributor to systemic "inflammaging" - the chronic, low-grade inflammation characteristic of advanced age that precedes overt disease. In peripheral tissues, notably visceral adipose tissue, the failure of lymphatic vessels to efficiently clear cellular debris, immune complexes, and metabolic byproducts leads to a significant buildup of senescent cells 43.
These localized senescent cells, including aged macrophages and T-lymphocytes, accumulate in adipose tissue and express markedly elevated levels of classical senescence markers like CDKN2A (p16) and CDKN1A (p21) 43. In response to stress, they secrete a potent, toxic cocktail of pro-inflammatory cytokines, chemokines, and matrix metalloproteinases collectively known as the senescence-associated secretory phenotype (SASP) 134344. The sustained SASP environment aggressively upregulates CD38 in surrounding immune cells, leading to the rapid depletion of systemic NAD+ levels. This decline in NAD+ further drives metabolic dysfunction and compromises distant vital organs, ultimately contributing to age-related cardiovascular disease and multiorgan fibrosis 1343.
Clinical Interventions and Pharmacological Targets
The recognition of lymphatic decline as a primary root cause of age-related pathology has catalyzed the rapid development of novel pharmacological and surgical interventions. These treatments are aimed at rejuvenating lymphatic structure, restoring fluid homeostasis, and actively clearing neurotoxic waste.
Pharmacological Restoration of Lymphatic Function
Pharmacological strategies for addressing lymphatic and systemic aging are broadly categorized by their mechanistic targets. These approaches focus either on the active restoration of vessel growth and contractility or the targeted elimination of the toxic cellular environments that paralyze lymphatic function.
- Lymphangiogenic Growth Factors: Vascular Endothelial Growth Factor C (VEGF-C) is the primary signaling protein responsible for the physiological generation and maintenance of lymphatic vessels 3845. In aged mouse models, the delivery of VEGF-C via viral vectors (such as AAV-VEGF-C) directly into the cerebrospinal fluid has successfully stimulated the growth of new meningeal lymphatics, increased existing vessel diameter, and restored the drainage of macromolecules to the cervical lymph nodes 233846. Crucially, this targeted intervention was shown to rapidly reduce IL-6 inflammatory signaling and reverse age-associated cognitive and memory deficits 3835.
- Contractility Enhancers: Researchers have explored pharmacological agents that directly target the smooth muscle tone of lymphatic vessels to restore the mechanical pumping action lost to age. The topical application of prostaglandin F2α analogs to the exposed cervical lymphatic vessels in aged mice successfully restored spontaneous vessel pulsation, diminished retrograde lymph flow, and enhanced overall CSF clearance 162446.
- Senolytics: Senolytic compounds are designed to selectively induce apoptosis in senescent cells, thereby permanently removing the source of the tissue-destructive SASP 474849. The combination of Dasatinib (a targeted tyrosine kinase inhibitor) and Quercetin (a naturally occurring flavonoid) is the most extensively studied senolytic regimen 495051. By clearing senescent cells from the peri-lymphatic tissue and degraded lymph nodes, senolytics actively reduce chronic inflammation, enhance localized tissue repair, and have been shown to improve cardiac function, exercise endurance, and cognitive performance in preclinical models 4950.
- Senomorphics: Unlike senolytics, senomorphics (such as rapamycin and metformin) do not kill the senescent cell. Rather, they modulate cellular behavior by suppressing the active secretion of SASP factors, effectively muting the inflammatory signal that degrades lymphatic and systemic health without altering total cell counts 4750.
| Intervention Class | Primary Mechanism of Action | Target Biological Pathway | Current Level of Evidence |
|---|---|---|---|
| Lymphangiogenics (e.g., VEGF-C, HGF) | Stimulates lymphatic endothelial cell proliferation and vessel generation; expands meningeal drainage. | VEGFR-3 / Non-VEGFR-3 pathways | Preclinical (Mouse Models); highly effective in reversing cognitive decline in AD models. |
| Contractility Agents (e.g., Prostaglandin F2α) | Restores smooth muscle tone and pumping frequency of collecting lymphatic vessels. | ROCK / Smooth Muscle Actin | Preclinical; demonstrates acute restoration of CSF outflow. |
| Senolytics (e.g., Dasatinib + Quercetin) | Selectively induces apoptosis in senescent cells, completely removing the source of SASP inflammation. | Apoptosis Resistance Pathways | Early Clinical / Off-label use; visibly reduces systemic senescent cell burden. |
| Senomorphics (e.g., Rapamycin, Metformin) | Suppresses the secretion of inflammatory SASP factors without eliminating the underlying senescent cell. | mTOR, AMPK | Clinical; broad multi-hallmark anti-aging physiological effects. |
Surgical Interventions in Central Nervous System Drainage
In human medicine, the translation of lymphatic enhancement has briefly taken an aggressive surgical route, particularly for the treatment of severe neurodegenerative diseases. Deep cervical lymphaticovenous anastomosis (dcLVA) is an innovative, albeit highly controversial, microsurgical procedure designed to bypass physical lymphatic bottlenecks 3452. The procedure involves manually conjoining the deep cervical lymphatic vessels that drain the brain directly with adjacent veins in the neck, establishing a low-resistance, high-flow outflow tract for cerebrospinal fluid and metabolic waste 3452.
Before being formally restricted pending further standardized safety trials in 2025, dcLVA was performed on over 2,000 patients across numerous provinces in China. Preliminary, uncontrolled case series reported short-term improvements in cognitive scores (e.g., MMSE improved by an average of two points) and favorable trends in AD biomarkers (Aβ and tau) 3452. However, the broader international medical community maintains strict, calibrated uncertainty regarding the procedure. Critics highlight that the complex anatomy of human lymphatic drainage is still incompletely mapped, translation from rodent lymphatic pathways is uncertain, and the reported cognitive improvements may be heavily subject to placebo effects, perioperative physiological fluctuations, or natural disease variations 3452.
Evidence-Based Lymphatic Medicine Versus Commercial Detoxification
As the scientific understanding of the lymphatic system has firmly entered the public consciousness, a robust, highly profitable commercial wellness industry has emerged. This industry heavily promotes "lymphatic detox" products, unproven supplements, and rapid-weight-loss routines. It is crucial to delineate evidence-based physiological support from marketing trends that fundamentally misrepresent basic lymphatic biology.
Deconstructing Lymphatic Detoxification Claims
The concept of chemically "detoxifying" the body via the lymphatic system is a biological misnomer. The physiological breakdown, metabolism, and neutralization of environmental toxins, heavy metals, and xenobiotics are performed almost exclusively by the phase I and phase II enzymatic pathways of the liver and ultimately excreted by the kidneys 535455. The lymphatic system's actual role in waste clearance is strictly limited to the physical transport of large biological macromolecules, dead cellular debris, excess interstitial fluid, and immune complexes to the lymph nodes for filtration and eventual return to the bloodstream 5354.
Products marketed heavily for "lymphatic detox" - such as transdermal adhesive patches (e.g., NutriSpot neck and foot patches) or aggressive botanical supplements - lack any robust, independent clinical validation 5556. Adhesive patches that turn a dark, sludgy color overnight rely entirely on simple chemical reactions between body sweat and the patch ingredients (often wood vinegar or ginger), not the physical extraction of systemic toxins through the skin barrier 56. Similarly, herbal supplements containing ingredients like cleavers, dandelion, or heavy doses of selenium may offer mild, general diuretic or antioxidant properties. However, there is zero peer-reviewed evidence that they chemically "flush" the lymphatic system, reverse established secondary lymphedema, or induce fat release independent of a strict caloric deficit 53555758.
Clinically Validated Lymphatic Therapies
While the commercial "detox" paradigm is medically unsupported, evidence-based physical interventions can successfully optimize lymphatic fluid dynamics. Because the lymphatic system relies heavily on external mechanical forces to compensate for intrinsic pumping deficits, physical movement and targeted mechanical therapies are paramount 535459.
Manual Lymphatic Drainage (MLD) is a clinically validated, gentle, rhythmic massage technique specifically designed to stimulate the flow of lymph fluid and visibly reduce interstitial fluid pressure 535460. Medical-grade MLD, particularly when combined with rigorous compression therapy, remains the established gold standard for managing secondary lymphedema and post-surgical oncologic swelling 5360. MLD does not chemically "cleanse" organs of toxins. Instead, by physically directing pooled fluid toward functioning nodal clusters, it actively alleviates the mechanical burden of tissue edema and functionally supports the body's natural immunological filtration processes 5354.
Additionally, emerging non-invasive technologies like photobiomodulation (Red and Near-Infrared Light Therapy) are being actively studied for their direct cellular effects on lymphatic contractility. Preliminary evidence indicates that specific, targeted wavelengths (660nm and 850nm) may interact with lymphatic endothelial cells to enhance mitochondrial ATP production and promote localized nitric oxide release. This process potentially increases the intrinsic contraction frequency of lymphatic muscle cells, aiding in the systemic reduction of localized swelling and supporting post-surgical recovery 61.