Longevity Biotechnology Landscape in 2026
Industry Overview and Market Maturation
The year 2026 marks a structural inflection point for the longevity biotechnology industry, representing a definitive transition from an era characterized by theoretical geroscience and foundational biological research into a phase defined by active clinical translation, regulatory precedent-setting, and sophisticated capital allocation. Historically constrained by the immense biological complexity of the aging process and a lack of validated measurement tools, the sector has matured into a multi-tiered industry encompassing epigenetic reprogramming, artificial intelligence-driven drug discovery, cellular clearance mechanisms, and advanced diagnostic biomarkers.
The aggregate scale of the longevity sector reflects sustained institutional and private interest, with total deployed venture capital in longevity pure-play companies exceeding $13.1 billion 1. The investment thesis driving this capital influx is underpinned by macro-economic projections; utilizing quality-adjusted life years (QALYs) valued at $100,000 each, analysts project that the total addressable market for longevity interventions could reach $1.2 quadrillion over the coming decades 2. Despite the current global biotechnology market capturing only a fraction of a percent of this potential, the landscape in 2026 is defined by stark concentrations of capital and a bifurcation between early-stage preventative interventions and highly capitalized clinical platforms.
While heavily funded entities pursuing fundamental cellular rejuvenation attract multibillion-dollar valuations based on preclinical promise, a parallel tier of computationally driven biopharmaceutical companies is quietly securing significant pharmaceutical partnerships and public market validation 23. Concurrently, global regulatory agencies are beginning to adopt flexible frameworks for intermediate biomarkers, and international hubs in the Middle East and Asia are aggressively deploying state-backed capital to position themselves as centers for longevity medicine and preventative healthcare 451.
Regulatory Frameworks and Surrogate Endpoints
The core regulatory challenge for longevity biotechnology has historically been the U.S. Food and Drug Administration's (FDA) position that aging itself is not a recognizable disease indication, necessitating that therapies target specific, recognized age-related pathologies to gain approval 7. In 2026, while aging remains officially unclassified as a disease, regulatory agencies have demonstrated a critical paradigm shift in their willingness to accept intermediate biomarkers as surrogate endpoints for neurodegenerative and age-related conditions.
The Shift Toward Biomarker Endpoints
The FDA's evolving framework emphasizes the compression of time in clinical trials. Instead of requiring multi-year observational data to confirm that a therapeutic intervention extends lifespan or permanently halts disease progression, regulators are increasingly considering whether changes in specific biological markers reliably reflect the slowing of disease 2. A prominent example occurred in May 2026, when the FDA acknowledged that neurofilament light chain (NfL) could potentially serve as a "reasonably likely surrogate endpoint" to support the accelerated approval of Clene's CNM-Au8 under the Subpart H pathway for amyotrophic lateral sclerosis (ALS) 2.
This regulatory flexibility compresses the timeline from hypothesis to treatment, allowing signals of preserved biological resilience to emerge within months rather than years 2. The shift is supported by mounting pressure from clinicians who advocate moving biomarkers from purely informative roles to formal efficacy measures that can form the basis of new drug approvals 9. Further evidence of this trend is the unanimous 12-to-0 decision by the FDA's Oncologic Drugs Advisory Committee (ODAC) to approve minimal residual disease (MRD) negativity as a biomarker for multiple myeloma 9.
Context Dependence and Regulatory Limitations
Despite these advancements, the regulatory acceptance of longevity and aging biomarkers remains strictly context-dependent. The FDA evaluates the acceptability of surrogate endpoints on a case-by-case basis, heavily weighing the specific disease, patient population, mechanism of action, and existing therapeutic alternatives 3. A biomarker validated in one clinical setting cannot be assumed appropriate for another, and the agency publishes a table of accepted surrogate endpoints updated every six months to reflect current scientific consensus 3.
This conservative posture was underscored by the FDA's issuance of a Complete Response Letter (CRL) to Regenxbio for its gene therapy RGX-121, citing issues with the use of the cerebrospinal fluid biomarker CSF HS D2S6 as a surrogate endpoint to predict clinical benefit 9. The FDA maintains that surrogate endpoints must demonstrate robust correlation with definitive clinical outcomes, and the agency explicitly warns that evolving scientific understanding may render previously accepted endpoints obsolete 3. Consequently, longevity companies in 2026 must navigate a dual pathway: developing therapies intended to address the root mechanisms of aging while designing clinical trials anchored to highly specific, traditionally recognized disease outcomes 7.
Clinical Pipelines and Therapeutic Modalities
The clinical pipeline in 2026 spans a diverse array of therapeutic modalities, moving beyond broad dietary and lifestyle interventions into precise molecular engineering. The most prominent strategies currently undergoing human evaluation include partial epigenetic reprogramming, targeted cellular autophagy enhancement, and computationally derived therapeutic discovery.
Partial Epigenetic Reprogramming
Partial epigenetic reprogramming utilizes specific transcription factors - most commonly the Yamanaka factors (OCT4, SOX2, KLF4, and c-Myc) - to reset the epigenetic signatures of aged or damaged cells to a more youthful state without inducing pluripotency or stripping the cell of its functional identity 114. This process aims to modulate gene activity and correct epigenetic drift rather than permanently splicing or replacing DNA 13.
Life Biosciences Life Biosciences has emerged as a clinical leader in this sub-sector. Following an $80 million Series D funding round, the company transitioned to a clinical-stage biotechnology firm and commenced Phase 1 human clinical trials for its lead candidate, ER-100, after receiving FDA Investigational New Drug (IND) clearance in January 2026 71314. ER-100 utilizes an adeno-associated virus (AAV) gene therapy delivery mechanism to introduce three reprogramming factors (OCT4, SOX2, and KLF4, actively excluding the oncogenic c-Myc to satisfy FDA safety requirements regarding tumorigenesis) 715.
The initial trial focuses on non-arteritic anterior ischemic optic neuropathy (NAION) and open-angle glaucoma (OAG) 135. The human eye serves as an ideal regulatory entry point because it is a highly compartmentalized organ containing post-mitotic neurons, which minimizes the systemic risks commonly associated with cellular reprogramming 76. In the clinical protocol, participants receive a single administration of ER-100 to one eye, which is subsequently activated by systemic doxycycline for eight weeks 7. The trial evaluates safety, tolerability, immune responses, and preliminary visual function endpoints, tracking bodily fluid clearance and utilizing an independent Data Safety Monitoring Board for dose escalation oversight 57. Life Biosciences operates with a clinical runway extending into the second half of 2027 and plans to advance subsequent candidates, such as ER-300, targeting metabolic dysfunction-associated steatohepatitis (MASH) in the liver 713146.
Turn Biotechnologies Turn Biotechnologies approaches epigenetic reprogramming through transient mRNA delivery rather than permanent viral gene therapy. Utilizing its proprietary ERA platform and eTurna lipid particle carrier system, Turn Bio delivers reprogramming factors temporarily, functioning similarly to a traditional dosed pharmaceutical rather than a permanent genetic modification 198. This mRNA modality is theoretically safer, as it mitigates the risk of permanent oncogenic alterations to DNA 19. Throughout 2025 and into 2026, Turn Bio has been preparing its IND submission for TRN-001, a therapy targeting dermatology, skin aging, and scarring, with the aim of becoming the first mRNA-based cell rejuvenation therapy to reach clinical trials 1989. In late 2025, Klotho Neurosciences signed a letter of intent to acquire Turn Bio's ERA platform and eTurna RNA delivery system, reflecting ongoing consolidation in the sector 9.
Foundational Cellular Rejuvenation
Altos Labs With $3 billion in initial funding and a valuation exceeding $6.33 billion, Altos Labs remains the most heavily capitalized entity in the longevity sector 221024. Operating initially in deep stealth under the scientific advisement of Nobel laureate Shinya Yamanaka, the company began signaling a shift toward clinical application in late 2025 by appointing Joan Mannick as Chief Medical Officer 1110. By 2026, reports indicated Altos was preparing to initiate its "Sentinel Cell" program, aimed at identifying and reprogramming specific cells responsible for propagating inflammation and aging signals throughout the body 1122. To navigate the inherent dangers of in vivo reprogramming, Altos has developed specialized artificial intelligence models to predict biological "safe zones" to ensure cells are rejuvenated without becoming cancerous 22. Furthermore, the company is actively exploring ex-vivo reprogramming techniques, wherein isolated donor organs are rejuvenated on perfusion machines prior to human transplantation 19.
NewLimit Founded by a coalition of technology and biology executives, NewLimit focuses on extending human healthspan through epigenetic reprogramming and AI-enabled drug discovery 1112. Following a $130 million Series B raise in 2025 that brought its valuation to $810 million, NewLimit reported significant milestones in early 2026 1112. The company advanced its first candidate medicine - a liver mRNA therapeutic aimed at restoring youthful function in old hepatocytes - into large-scale manufacturing 12. Utilizing in silico reprogramming models, NewLimit has doubled its rate of discovery, identifying over 19 payloads designed to rescue regulatory activity in T-cells and successfully establishing preclinical models for assessing renal endothelial cell age 12.
Autophagy and Cellular Clearance Mechanisms
A separate tier of the industry avoids genetic reprogramming entirely, instead targeting the removal of accumulated cellular waste and the restoration of natural homeostatic recycling systems.
Retro Biosciences Backed heavily by OpenAI CEO Sam Altman, Retro Biosciences pursues multiple therapeutic tracks, with a primary focus on cellular autophagy 192713. In late 2025, Retro dosed the first participant in a Phase 1 randomized, double-blind, placebo-controlled trial in Australia for RTR242 2714. RTR242 is an oral small-molecule therapy designed to restore lysosomal function and acidity, a core component of the cellular waste-handling system 27. By reactivating the cell's internal cleanup machinery, Retro aims to clear toxic protein aggregates upstream of disease presentation, positioning the therapy as a potential intervention for neurodegenerative conditions like Alzheimer's disease before significant cognitive decline begins 2714. Supported by a $180 million seed round and seeking a subsequent $1 billion Series A, Retro is also developing programs in plasma exchange and T-cell rejuvenation 1013.
Cambrian Biopharma Operating under a unique "hub-and-spoke" model, Cambrian Bio functions as a central parent company incubating multiple subsidiary programs, each targeting different hallmarks of aging 1516. As of 2026, its diversified pipeline hedges against the clinical failure of any single modality. Its subsidiary, Tornado Therapeutics, is advancing selective mTORC1 inhibitors (novel rapalogs) into IND-enabling studies to extend human healthspan 1617. Another subsidiary, Amplifier Therapeutics, is evaluating ATX-304, a small-molecule AMPK activator, in Phase 1b/2a trials to enhance mitochondrial respiration and offset the catabolic side effects of GLP-1 weight loss drugs 15. Additionally, Oviva Therapeutics is advancing an agent that suppresses folliculogenesis to preserve ovarian reserve and delay ovarian decline 16. In February 2026, Cambrian was awarded up to $30.8 million in non-dilutive funding by the Advanced Research Projects Agency for Health (ARPA-H) for its PROSPR program to test whether mTORC1 inhibitors can actively preserve intrinsic metabolic and physical capacity in older adults, marking one of the first direct human tests of the fundamental geroscience hypothesis 18.
Mechanistic Target Modulators and Gene Therapy
Rejuvenate Bio A spin-out from George Church's laboratory at Harvard, Rejuvenate Bio is taking a distinct path by combining partial reprogramming with established longevity genes 19. While the company gained prominence for extending the lifespan of murine models by 100% using an OSK approach, its first human clinical trials focus on gene therapy delivery platforms targeting heart failure and kidney disease 19.
| Company | Lead Program / Modality | Biological Target / Mechanism | Clinical Status (2026) | Capitalization / Valuation |
|---|---|---|---|---|
| Life Biosciences | ER-100 (AAV Delivery) | Partial Epigenetic Reprogramming (OSK) | Phase 1 (NAION, Glaucoma) | $80M Series D |
| Retro Biosciences | RTR242 (Oral Small Molecule) | Autophagy / Lysosomal Restoration | Phase 1 (Healthy Volunteers) | $180M Seed / ~$4.5B Est. Valuation |
| Turn Biotechnologies | TRN-001 (mRNA Delivery) | Epigenetic Reprogramming | IND-Enabling (Dermatology) | $10M Convertible Note |
| Altos Labs | Sentinel Cell Program | Cellular Rejuvenation (iPSC approaches) | Preclinical / Target Discovery | $3.0B Initial Funding / $6.33B Valuation |
| NewLimit | Liver mRNA therapeutics | Epigenetic Reprogramming / T-cells | Preclinical / IND-Enabling | $130M Series B / $810M Valuation |
| Cambrian Bio | ATX-304 / mTORC1 Inhibitors | AMPK Activation / mTOR pathway | Phase 1b/2a (ATX-304) | $160M VC / $30.8M ARPA-H Grant |
Artificial Intelligence and Target Discovery
The integration of generative artificial intelligence has drastically reduced preclinical development timelines across the biotechnology sector, shifting the discovery paradigm from manual, high-throughput wet-lab screening to predictive computational modeling.
Generative Computational Platforms
Insilico Medicine Insilico Medicine represents the vanguard of AI-driven longevity therapeutics. The company completed a $293 million initial public offering on the Hong Kong Stock Exchange (HKEX: 3696) in late 2025, achieving the largest biotech IPO in Hong Kong for the year and establishing a market capitalization of approximately $4.1 billion (HKD $20.7 billion) 1241920. Insilico's proprietary end-to-end generative AI platform, Pharma.AI, integrates sub-engines spanning biology, chemistry, and clinical outcome prediction, generating significant software revenue from 13 of the top 20 global pharmaceutical companies 2122.
Insilico's lead internal candidate, rentosertib (formerly ISM001-055), successfully completed a Phase 2a trial for idiopathic pulmonary fibrosis (IPF) in 2025, demonstrating lung function stabilization and a favorable safety profile 192022. Rentosertib acts as a first-in-class inhibitor of TNIK, a target identified entirely by AI that plays a critical role in fibrotic scarring 2022. In 2026, the company is expanding clinical trials to include an inhalable formulation for IPF and new applications in kidney fibrosis 22. The strategic validation of Insilico's platform is evidenced by its expansive roster of out-licensing agreements and co-development partnerships, generating milestone agreements with global leaders including Sanofi, Exelixis, and Eli Lilly, with maximum total deal values potentially exceeding $2.1 billion 119.
Human Data and Physics-Based Machine Learning
Gero Singapore-based Gero utilizes a human data-first AI platform built upon physics-based machine learning to identify targets driving age-related diseases and human aging processes 923. In July 2025, Gero entered a transformative joint research and license agreement with Chugai Pharmaceutical, a Japanese company majority-owned by Roche 2324. Under the agreement, Chugai committed up to $250 million in development and sales milestones, plus royalties, to leverage Gero's AI discoveries 2324. Chugai will apply its proprietary antibody engineering technologies to create novel biologic drug candidates aimed at restoring lost physiological function based entirely on the therapeutic targets identified by Gero's algorithmic analysis of human datasets 2324.
Longitudinal Human Data Cohorts
Calico Life Sciences Alphabet's subsidiary, Calico Life Sciences, continues to transition from foundational biological research into independent clinical execution, armed with deep expertise derived from large human data cohorts like the UK Biobank 2526. Calico maintains over 20 early-stage programs and several assets in active clinical development 2527. Notably, ABBV-CLS-7262, an eIF2B activator developed in collaboration with AbbVie, is currently being evaluated in a Phase 1b trial for Vanishing White Matter (VWM) disease, an incredibly rare autosomal recessive disorder 2829. While the drug previously failed to meet primary endpoints in the HEALEY ALS Platform Trial, the ongoing VWM trial is expected to conclude in late 2026 28. Additionally, Calico is progressing ABBV-CLS-484, a dual PTPN2/N1 inhibitor that enhances anti-tumor activity and represents the first active-site phosphate inhibitor to enter Phase 1 trials as a cancer immunotherapy 2628.
Preclinical Science and Translational Bottlenecks
While clinical pipelines mature and artificial intelligence expedites target discovery, the broader scientific community continues to uncover novel biological mechanisms governing the aging process. However, the translation from murine models to human therapies remains the most formidable bottleneck in the longevity industry.
Breakthroughs in Animal Models and Combinatorial Pharmacology
Recent laboratory successes have highlighted the potential of combinatorial pharmacology and microbiome modulation to achieve systemic lifespan extension. In 2025, researchers demonstrated a 30% extension in mouse lifespan using a cocktail combining the cancer drug trametinib with the established longevity molecule rapamycin 46. The additive effects of these compounds suggest that targeting multiple, independent biological pathways simultaneously is more effective than monotherapy, though researchers caution that the necessary dosing of trametinib in mice far exceeds tolerable human safety limits 46.
In early 2026, a study published in Aging reported that a combination of oxytocin (a hormone tied to tissue repair and social bonding) and an Alk5 inhibitor (a compound that blocks the TGF-beta inflammatory signaling pathway) increased the remaining lifespan of extremely old, frail male mice by 73% and overall median lifespan by 14% 4730. This dual strategy, known as OT+A5i, restored youthful protein patterns in circulating blood and improved muscle strength and memory 4730. Notably, the intervention demonstrated significant sexual dimorphism, showing no substantial lifespan benefit in female mice, highlighting the hormonal and metabolic complexities that complicate broad clinical applications 4730.
Further emphasizing the systemic nature of aging, a March 2026 Nature publication by a Stanford research team detailed how age-related memory loss is governed by a three-step pathway originating in the gastrointestinal microbiome 31. The pathway links specific gut bacteria to GPR84-driven myeloid inflammation, which transmits signals through the vagus nerve directly to the hippocampus 31. Crucially, interrupting this inflammatory cascade successfully restored memory function to youthful levels in aged mice, suggesting that cognitive decline may be mitigated without accessing the brain directly 31.
The Translational Gulf and Systems Biology
Despite these profound in vivo results, researchers caution against premature clinical enthusiasm. The realistic timeline for transitioning findings - such as microbiome-driven cognitive reversal - into human trials is measured in years 31. The translational bottleneck is driven by massive variables in human biological complexity, substantially longer human lifespans, and the lack of a standardized regulatory framework for aging as an actionable clinical endpoint 31.
This translational friction is prompting a major conceptual reset across the academic and industrial spheres. Rather than viewing aging as a series of isolated molecular defects to be fixed via single-target interventions (e.g., purely administering senolytics or isolated mTOR inhibitors), the vanguard of the field is redefining aging as a progressive loss of biological coordination 3233. Emerging models suggest that true longevity interventions will require the simultaneous, coordinated modulation of complex networks - spanning mitochondria, the microbiota, immune signaling, and metabolic regulation - to engineer overall system resilience 33. This paradigm shift was the central focus of the "Targeting Longevity 2026" conference in Berlin, reflecting an industry-wide pivot toward holistic biological stabilization rather than localized cellular rejuvenation 3233.
Venture Capital Allocation and Market Valuations
The financial architecture of the longevity biotechnology sector in 2026 exposes a complex dynamic of highly concentrated capital allocation, distinct outlier effects, and an emerging disconnect between private market valuations and objective clinical validation.
Funding Concentrations and Validation Disconnects
Data analyzing pure-play longevity equity funding reveals a sharp apparent contraction in early 2026. From January through May 2026, the sector recorded only 5 qualifying deals totaling $233.9 million 52. This represents a steep statistical decline from the $2.2 billion raised across 31 deals in the full year of 2025 52. However, analysts note that the 2025 data was heavily distorted by massive outlier rounds - specifically Retro Biosciences' reported $1 billion Series A and Insilico Medicine's $110 million Series E 352.
The industry remains remarkably top-heavy, driven by individual conviction rather than traditional venture capital patterns. In the 12 months leading up to May 2026, the single largest deal - Function Health's $298 million Series B - accounted for 42% of all disclosed capital, and the top four rounds captured nearly 73% of the funding 53. Seed deals represented 25% of the transaction volume but less than 5% of the total capital deployed, indicating that large checks require deep institutional credibility, regulatory pathways, or significant platform depth 53. The median funding round for longevity startups hovers around $24 million, indicating that while mega-rounds capture headlines, standard venture deployment remains highly calibrated 13.
A critical divergence exists between capital momentum and validation momentum. Between April 2024 and April 2026, the cellular reprogramming segment absorbed roughly $1.28 billion across 6 deals 3.
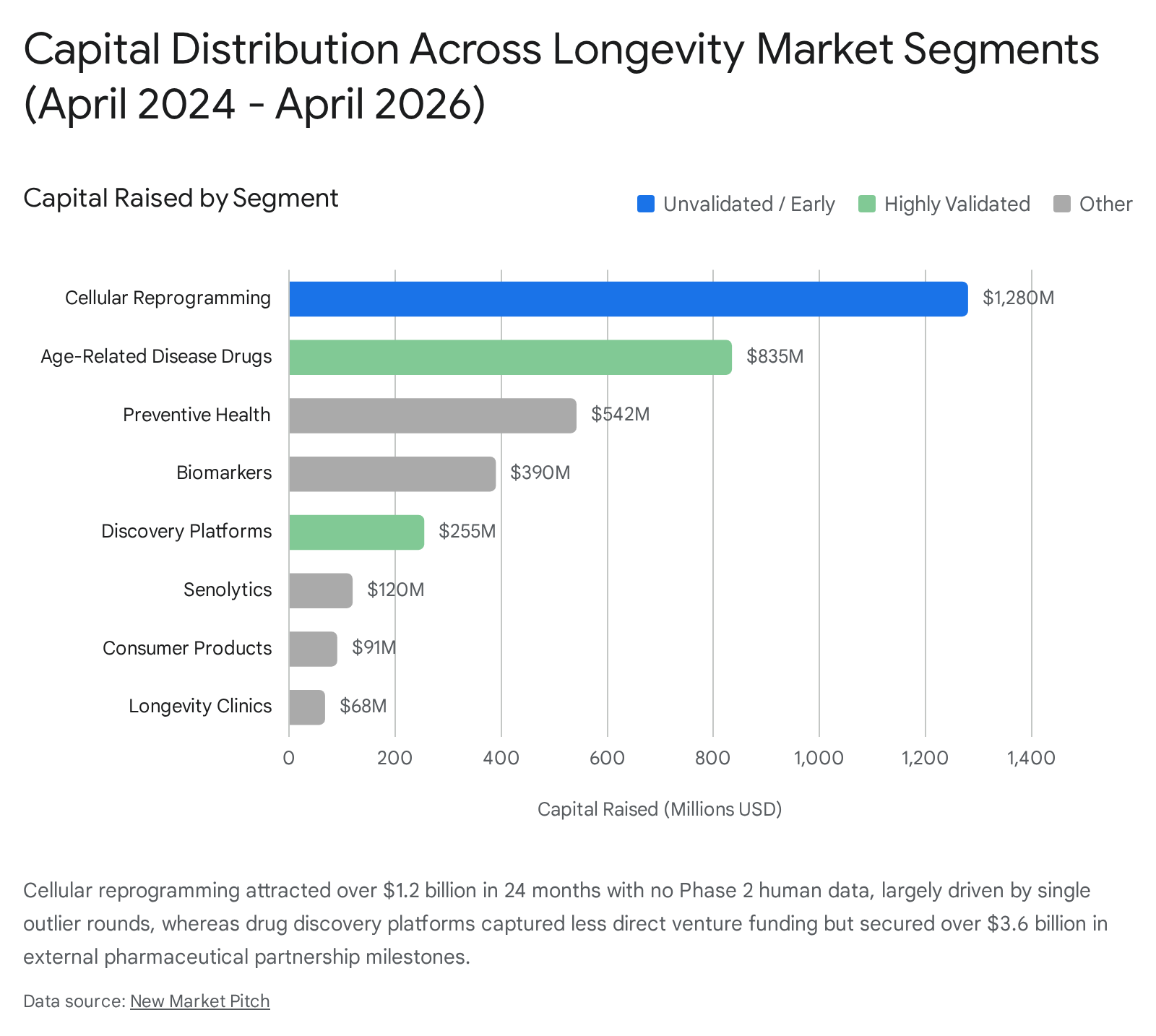
Despite this massive influx of capital, no reprogramming company has yet to produce human Phase 2 efficacy or safety data 3. Conversely, drug discovery platforms secured comparatively modest direct venture funding ($255 million across 5 deals), but demonstrated the strongest validation momentum 3. Companies like Insilico Medicine and Gero have aggregated billions in potential pharmaceutical partnership value, underscoring that legacy pharma views computational targeting as a derisked entry point compared to foundational age-reversal 3. Senolytics, following the high-profile clinical failures of earlier pioneers like Unity Biotechnology, raised only $120 million over 24 months, suffering from intense investor hesitancy despite ongoing clinical trials 3.
Top Longevity Valuations
The valuation landscape further illustrates the premium placed on consumer engagement and audacious foundational science over traditional clinical revenue.
| Rank | Startup | Core Focus / Modality | Estimated 2026 Valuation | Capital Raised |
|---|---|---|---|---|
| 1 | Oura | Smart health-tracking hardware | $11.0 Billion | ~$1.2 Billion |
| 2 | Altos Labs | Cellular reprogramming (Stealth/Preclinical) | $6.33 Billion | $3.0 Billion |
| 3 | Retro Biosciences | Cellular autophagy & Reprogramming | $4.5 - $5.5 Billion | ~$1.2 Billion |
| 4 | Insilico Medicine | AI-driven drug discovery (Public HKEX) | $4.1 Billion (Market Cap) | ~$688 Million |
Note: Valuations represent a mix of public market capitalization, announced private rounds, and active raise estimates 1024.
The valuation disconnect is stark when comparing highly valued preclinical firms to operational entities. For instance, Altos Labs and Retro Biosciences combined represent over $10 billion in theoretical value despite minimal or zero human clinical data 1024. In contrast, unheralded companies like Celljevity - which maintains an estimated $200 million valuation - have quietly treated over 1,000 patients across seven years of safety monitoring with no serious adverse events and active revenue generation 10.
Philanthropic and Sovereign Market Shapers
The influx of capital is not strictly driven by traditional venture capital. The market is increasingly subsidized by sovereign wealth and specialized philanthropic institutions designed to alter the risk calculus of aging research. The Riyadh-based Hevolution Foundation operates with an annual budget of up to $1 billion and has rapidly become a central architect of the industry 3435. In three years, Hevolution has deployed over $400 million to over 250 grantees and executed numerous biotech impact investments 36.
Guided by Chief Investment Officer Dr. William Greene, Hevolution's strategy explicitly seeks to frame aging science as a global economic imperative 35. The foundation funds early-stage breakthroughs that traditional venture capitalists may consider too protracted, such as investments in Aeovian Pharmaceuticals' development of next-generation rapamycin analogs 5335. Hevolution also supports scientific infrastructure globally, launching the Hevolution Foundation Scientific Conferences Fund to supply up to $10,000 per event to facilitate academic exchange across North America, the UK, and the EU 37. In the public sector, the U.S. government's ARPA-H initiative represents a similar de-risking mechanism, injecting non-dilutive capital into programs testing the core tenets of geroscience directly in human populations 18.
Diagnostic Biomarkers and Clinical Implementation
As therapeutic interventions enter clinical trials, the standard of care in the commercial preventative health sector is undergoing a massive shift. The longevity clinic market in 2026 has fully transitioned from "treatment theater" - characterized by generic IV drips and cryotherapy - into a data-dense, diagnostic-first ecosystem 38.
The Evolution of Biological Age Testing
Biological age testing is no longer a vanity metric; it is evolving into a complex clinical assessment tool. Historically, discrepancies between methodologies hampered clinical utility; an individual could simultaneously possess a "young" epigenetic age and "old" telomeres due to the asynchronous aging rates of different biological systems 39. In 2026, the industry is moving away from single-layer clocks toward integrated, multi-omics models.
Recent advancements include composite algorithms like OMICmAge, which integrates epigenetic biomarker proxies, proteomic markers, metabolomic markers, and electronic medical record data to generate a multi-system risk estimate linked accurately to mortality and chronic disease 4061. Proteomic clocks are particularly valuable because circulating blood proteins dynamically reflect tissue stress, immune activation, and organ-specific biology 40.
Epigenetic tests relying on DNA methylation analysis remain the gold standard for accuracy. Early iterations like the Horvath Clock (analyzing 353 CpG sites) and the Hannum Clock (71 CpG sites) have been superseded by advanced platforms 61. The DunedinPACE algorithm and the extensive TruAge COMPLETE test - which analyzes over 900,000 CpG sites utilizing Illumina EPIC chip technology - are widely regarded as the most scientifically validated single measures of the pace of aging 3961.
Clinical Utility and Limitations
Despite technological sophistication, biological age testing faces practical adoption hurdles. Most advanced epigenetic clocks carry a standard error margin of ±3 to 5 years, limiting their utility as definitive diagnostic tools for acute individual treatment decisions 39. Furthermore, the lack of universal insurance coverage and high out-of-pocket costs restrict broad clinical implementation 41.
Consequently, the most effective longevity clinics in 2026 utilize biological age tests as triage and baseline tools rather than absolute mandates for intervention 40. Geriatricians and functional medicine practitioners rely on highly actionable, routinely monitored blood biomarker panels to track the immediate efficacy of lifestyle and pharmacological interventions (e.g., sleep optimization, metabolic control, and off-label geroprotective drugs), reserving deep-omic clocks for annual, high-level trajectory analysis 3942. Concurrently, imaging-derived biological age models - utilizing AI to assess the biological age of specific organs via MRI, CT, or DEXA scans - are gaining traction as they sit in the sweet spot between robust medical utility and actionable patient behavior modification 3840.
Global Expansion and Regional Hubs
The pursuit of longevity is inherently global, yet specific geographical regions are rapidly structuring their economies and healthcare infrastructures to capture the massive projected commercial value of the space.
The Middle East Longevity Ecosystem
The Gulf Cooperation Council (GCC), particularly the United Arab Emirates (UAE), has established itself as the premier global hub for commercial longevity and preventative health. The regional longevity market is currently valued between $9 billion and $13 billion, with the UAE specifically forecasting sector growth to $32 billion by 2026 443. This growth is driven by state-backed mandates shifting healthcare frameworks from reactive disease management to proactive biological optimization, recognizing that the UAE demographic over age 60 is expected to rise from 1% in 2016 to 16% by 2050 443.
Dubai exemplifies this integration by merging clinical rigor with luxury hospitality. The 2026 market introduces the "Uberization" of biohacking, wherein elite longevity protocols, genetic screening, and targeted treatments are delivered on-demand within high-end hospitality environments 465. This "Medical Butler" model - pioneered by entities like XLR8 Wellness and Clubtogether Hospitality - decouples wellness revenue from labor-intensive traditional spa services, creating high-margin integrated recovery ecosystems utilizing automated, non-invasive technologies 6566. Institutionally, the Dubai Healthcare City (DHCC) expanded its footprint at the World Health Expo 2026 by integrating specialized longevity clinics like Proto Clinic and launching "The Formula," the Middle East's first AI-driven tele-compounding pharmacy platform to deliver customized, precision-based longevity medicines at scale 5.
European and Asian Market Integration
Europe continues to foster a robust ecosystem of highly specialized, preclinical and clinical-stage biopharmaceutical companies. The landscape includes firms operating in distinct therapeutic sub-sectors, such as Switzerland's Vandria (developing mitophagy inducers for Alzheimer's, advancing its VNA-318 asset), Germany's Seamless Therapeutics (focusing on programmable recombinases for precision gene editing), and the UK's Genflow Biosciences (utilizing a centenarian variant of the SIRT6 gene) 4445. Additionally, UK-based diagnostic and AI companies like GlycanAge, Mitra Bio, and 360Me provide critical infrastructure for biological age tracking and personalized health insights 44.
In Asia, Hong Kong has positioned itself as the premier financial conduit for the commercialization of longevity and AI-biotech. The successful $293 million public listing of Insilico Medicine on the Hong Kong Stock Exchange signaled the region's capacity to absorb highly complex, AI-driven biopharmaceutical ventures 11922. Concurrently, deep integrations between Asian pharmaceutical leaders (e.g., Japan's Chugai) and regional AI platforms (e.g., Singapore's Gero) illustrate a highly collaborative trans-Pacific approach to uncovering novel targets for age-related decline 23. As China accelerates its biotechnology innovation under its new Five-Year Plan, the convergence of global capital, regional clinical research organizations, and advanced digital health ecosystems ensures that Asia will remain a critical frontier for scaling longevity therapeutics 46.