Klotho protein in renal and brain aging
Introduction to the Klotho Paradigm
Since its serendipitous discovery in 1997 by Makoto Kuro-o and colleagues, the KLOTHO gene has fundamentally reshaped the theoretical and clinical landscape of molecular gerontology 123. Originally identified as an "aging-suppressor" gene during the creation of transgenic mice, an insertional mutation disrupting its expression resulted in a syndrome remarkably indistinguishable from human premature aging 145. The gene was aptly named after Clotho, the Greek goddess of fate who spins the thread of life 146. Mice homozygous for the Klotho mutation (kl/kl) exhibit a severe, accelerated aging phenotype characterized by a drastically truncated lifespan, widespread arteriosclerosis, ectopic vascular and soft tissue calcification, profound osteoporosis, emphysema, gonadal dysplasia, skin atrophy, ataxia, and severe cognitive deficits, ultimately perishing within two months of birth 478.
Conversely, transgenic overexpression of the KLOTHO gene extends the murine lifespan by 20% to 30%, establishing the encoded protein not merely as a passive biomarker of biological vitality, but as an active, therapeutic driver of mammalian longevity 249. Over the past two and a half decades, extensive biomedical research has elucidated the multifaceted nature of the $\alpha$-Klotho protein. It operates at the critical nexus of mineral metabolism, cellular oxidative stress regulation, and neuroprotection 3410. While foundational studies firmly established its localized role in the kidney-bone endocrine axis - acting as an obligate co-receptor for fibroblast growth factor 23 (FGF23) to manage phosphate homeostasis - more recent literature spanning 2023 to 2026 has increasingly pivoted toward its potent systemic applications, particularly within the central nervous system (CNS) 31112.
This comprehensive report synthesizes the expansive body of literature surrounding Klotho. It critically examines the structural biology of its various transcribed and cleaved isoforms, delineates its foundational mechanisms in renal phosphate regulation, and thoroughly explores its neuroprotective functions. Furthermore, this analysis will explicitly address the highly debated blood-brain barrier (BBB) paradox of peripheral Klotho, scrutinize the severe methodological limitations of commercial enzyme-linked immunosorbent assays (ELISAs) that currently confound clinical translation, and provide an exhaustive review of the emerging therapeutic landscape. This therapeutic review will distinguish highly successful rodent outcomes from preliminary human clinical trials, focusing on advanced plasmid gene therapies, novel small-molecule enhancers such as SBI-797812, and the role of lifestyle interventions acting as endogenous Klotho secretagogues.
Molecular Architecture and Processing of Klotho Isoforms
The physiological influence of the Klotho protein is governed by its complex structural biology and dynamic post-translational processing. In humans, the KL gene is located on chromosome 13q12, encompasses approximately 50 kilobases of DNA, and comprises five structural exons 11314. Through alternative RNA splicing and proteolytic cleavage, the gene gives rise to distinct protein isoforms, each exhibiting specific tissue distributions and highly divergent biological roles.
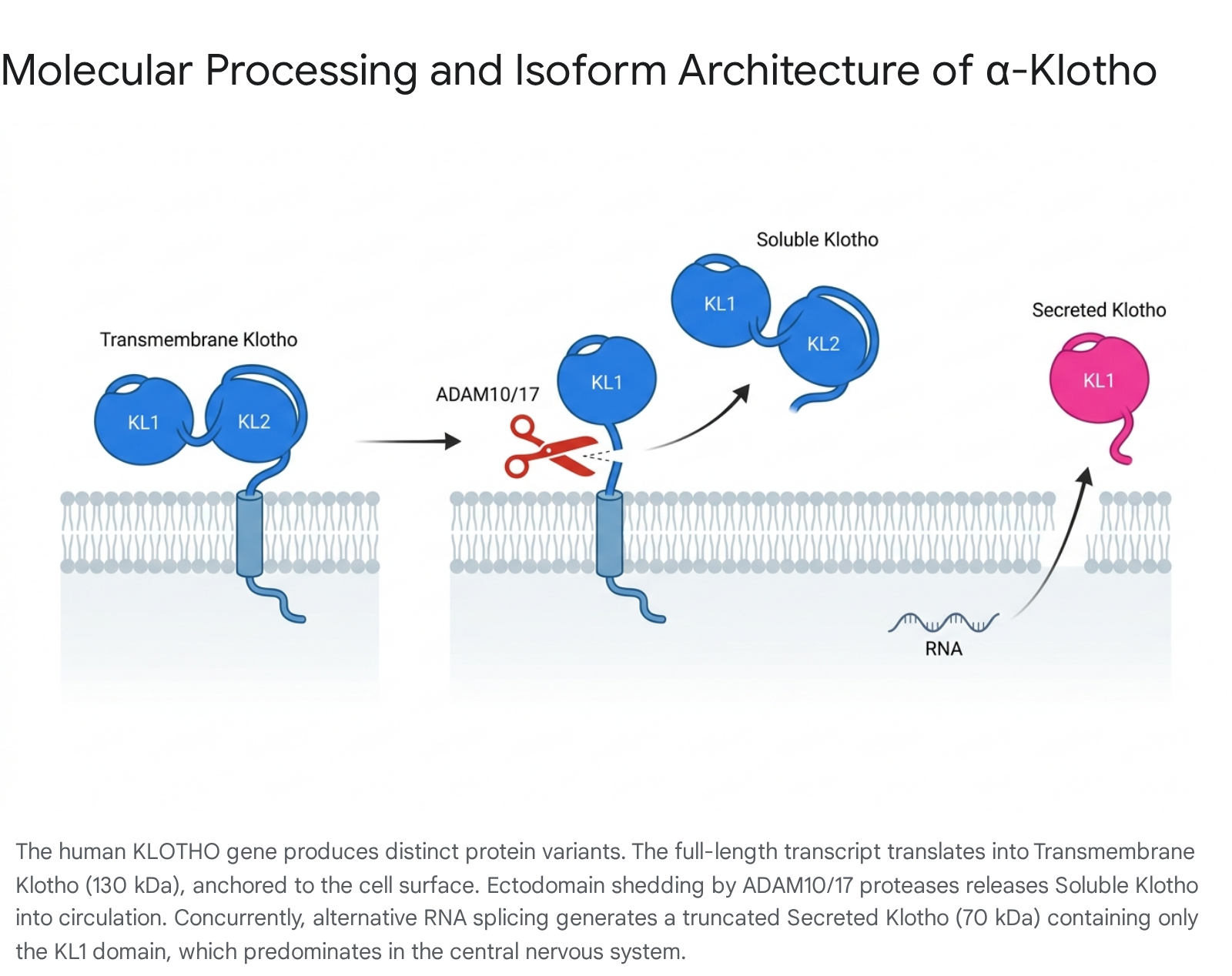
Understanding the structural divergence between these forms is critical for navigating the complex therapeutic literature, as antibodies and assays often fail to distinguish between them.
Transmembrane (Full-Length) Klotho
The primary transcript of the KL gene encodes a single-pass, Type I transmembrane protein consisting of 1,014 amino acids with a molecular weight of approximately 130 kDa 141015. This full-length architecture features a short N-terminal signal sequence, a massive extracellular domain, a transmembrane domain, and a notably brief cytoplasmic tail composed of only 10 amino acids that completely lacks any known intracellular signaling motifs 101516. The extracellular region is composed of two internal tandem repeat sequences, designated as the KL1 and KL2 domains 41114. Each of these domains forms a core structure characterized by an eight-stranded parallel $\alpha$-barrel surrounded by eight $\beta$-helices, sharing 20% to 40% sequence homology with bacterial and plant $\beta$-glucosidases 11114. Transmembrane Klotho is predominantly expressed in the distal convoluted tubules of the kidney, the chief cells of the parathyroid glands, and the epithelial cells of the choroid plexus in the brain 3111718. Its primary recognized function is structural, complexing with canonical fibroblast growth factor receptors (FGFRs) to facilitate high-affinity endocrine signaling 23.
Soluble (Cleaved) Klotho
Transmembrane Klotho is not permanently confined to the cell surface; its extracellular domain is subject to continuous proteolytic shedding, a highly regulated process termed "ectodomain shedding" 235. Membrane-bound metalloproteinases, specifically ADAM10 and ADAM17 (a disintegrin and metalloproteinase), alongside $\beta$-secretases (such as BACE1), cleave the extracellular domain near the plasma membrane through an $\alpha$-cut mechanism 28111920. This enzymatic cleavage releases the entire 130 kDa extracellular fragment - comprising both the KL1 and KL2 domains - into the systemic blood circulation, cerebrospinal fluid (CSF), and urine 2192122. This highly active soluble form circulates throughout the body, functioning as an endocrine, paracrine, and autocrine hormone that exerts pleiotropic effects on distant organs that do not inherently express the KL gene 31123. Acting as a glycan-modifying enzyme and a humoral factor, soluble Klotho influences ion transport, suppresses insulin and insulin-like growth factor-1 (IGF-1) signaling pathways, inhibits the pro-fibrotic Wnt/$\beta$-catenin and TGF-$\beta$ pathways, and activates FoxO-mediated antioxidant responses 31116.
Secreted (Alternatively Spliced) Klotho
Distinct from the proteolytically shed soluble form, a third variant arises directly from alternative mRNA splicing utilizing a donor site within the third exon of the KL gene 41319. This truncated transcript generates a smaller, 65 to 70 kDa secreted protein comprising approximately 549 to 550 amino acids 41419. Crucially, this alternatively spliced form contains only the KL1 domain and a unique 15-amino-acid C-terminal sequence, entirely lacking the KL2 domain, the transmembrane region, and the intracellular tail 41019. Foundational molecular studies have established a critical species divergence: while the full-length transmembrane form overwhelmingly predominates in the mouse, the alternatively spliced secreted form is the major product of the KL gene in humans 413. This secreted isoform is most robustly expressed in the human central nervous system, particularly within the choroid plexus, where it is secreted directly into the cerebrospinal fluid 131922.
Table 1: Structural and Functional Comparison of Klotho Protein Isoforms
| Characteristic | Transmembrane Klotho | Soluble (Shed) Klotho | Secreted (Spliced) Klotho |
|---|---|---|---|
| Origin Mechanism | Full-length mRNA transcription of the KL gene | Proteolytic cleavage (ADAM10/17, BACE1) of the transmembrane form | Alternative mRNA splicing (third exon donor site) of the KL gene |
| Amino Acid Length | 1,014 AA | ~960 AA | 549 - 550 AA |
| Molecular Weight | ~130 kDa | ~130 kDa | ~65 - 70 kDa |
| Domain Architecture | KL1, KL2, Transmembrane, Cytoplasmic tail | KL1 and KL2 domains | KL1 domain + unique 15-AA C-terminal sequence |
| Primary Localization | Renal distal tubules, parathyroid glands, choroid plexus | Systemic blood circulation, urine, cerebrospinal fluid (CSF) | Predominantly Central Nervous System, cerebrospinal fluid (CSF) |
| FGF23 Coreceptor Role | Obligate co-receptor (High affinity) | Retains limited co-receptor capability, functions primarily as a hormone | No FGF23 co-receptor function (lacks the requisite KL2 domain) |
| Primary Physiological Functions | Mineral homeostasis, renal phosphate excretion, Vitamin D suppression | Anti-oxidative, Wnt/TGF-$\beta$ inhibition, systemic anti-aging humoral factor | Neuroprotection, anti-inflammation, cognitive resilience maintenance |
Renal Function and the Phosphate-Aging Axis
To understand the systemic consequences of Klotho decline, one must first examine its foundational biochemical role within the kidney. The kidney is the primary endogenous source of circulating soluble Klotho, and the organ is highly vulnerable to the loss of the protein during the progression of acute kidney injury (AKI) and chronic kidney disease (CKD) 351723.
The FGF23-Klotho Obligate Partnership
The most well-characterized physiological role of transmembrane Klotho is its function as an obligate co-receptor for Fibroblast Growth Factor 23 (FGF23) 351724. FGF23 is a bone-derived phosphaturic hormone, predominantly synthesized and secreted by osteocytes and osteoblasts in response to elevated dietary phosphate intake and rising serum phosphate levels 172025. Canonical FGF receptors, such as FGFR1c, FGFR3c, and FGFR4, inherently possess an exceedingly low binding affinity for FGF23 81126. Transmembrane Klotho physically associates with these FGFRs, creating a highly stable ternary complex (FGF23-FGFR-Klotho) on the surface of renal proximal and distal tubular cells, effectively converting canonical FGFRs into highly specific, high-affinity receptors for FGF23 81125.
Upon binding the FGF23 ligand, this tripartite complex activates critical intracellular downstream signaling cascades, most notably initiating the phosphorylation of FGF receptor substrate 2$\alpha$ (FRS2$\alpha$) and activating the extracellular signal-regulated kinase 1/2 (ERK1/2) pathway 8112526. This signaling cascade achieves two vital, complementary outcomes for whole-body mineral homeostasis: 1. Induction of Phosphaturia: The signaling cascade downregulates the expression and promotes the rapid cellular internalization and degradation of sodium-dependent phosphate cotransporters (NaPi-2a and NaPi-2c) located on the apical membrane of renal proximal tubules. This action drastically curtails renal phosphate reabsorption, promoting its mass excretion into the urine and lowering serum phosphate levels 11172427. 2. Vitamin D Counter-Regulation: Simultaneously, the signaling axis acts on vitamin D metabolism. It suppresses the expression of renal CYP27B1 ($1\alpha$-hydroxylase), the critical enzyme responsible for synthesizing active 1,25-dihydroxyvitamin D3. Concurrently, it upregulates CYP24A1 (24-hydroxylase), the enzyme that degrades the active vitamin D metabolite. This dual action prevents excessive intestinal calcium and phosphate absorption, closing a critical negative feedback loop 571127.
Phosphatopathy: The Link Between Mineral Metabolism and Accelerated Aging
The absolute requirement of the Klotho co-receptor for effective FGF23 signaling elegantly explains a foundational biological mystery: why mice lacking the KLOTHO gene are phenotypically identical to mice lacking the FGF23 gene 51725. Both transgenic knockout models suffer from severe hyperphosphatemia, hypercalcemia, and hypervitaminosis D, leading to massive soft tissue calcification and premature death 3520.
This profound relationship birthed the concept of "phosphatopathy," a paradigm suggesting that systemic phosphate retention acts as a primary, insidious driver of mammalian aging 52224. Excess serum phosphate directly promotes severe vascular calcification by driving the osteogenic transdifferentiation of vascular smooth muscle cells (VSMCs), physically stiffening the arterial tree and accelerating cardiovascular mortality 11222428. In human chronic kidney disease (CKD), the progressive loss of functional renal mass leads to a profound, early decline in endogenous Klotho production 517. To compensate for accumulating phosphate, bone osteocytes secrete massive quantities of FGF23. However, without sufficient renal Klotho to act as a receptor, this FGF23 is rendered impotent at the kidney level, leading to a vicious, maladaptive cycle of severe hyperphosphatemia, extreme FGF23 elevations, secondary hyperparathyroidism, and calciphylaxis 5172427.
Furthermore, extreme elevations of circulating FGF23 in the absence of adequate Klotho can be highly toxic. Massive concentrations of FGF23 can bypass the need for Klotho, binding weakly to FGFR4 in cardiac tissue via a Klotho-independent pathway. This interaction pathologically activates pro-hypertrophic calcineurin/NFAT signaling cascades, directly contributing to left ventricular hypertrophy and heart failure, a leading cause of death in end-stage renal disease 1126. Thus, maintaining endogenous Klotho expression is not just a passive marker of healthy aging, but an active, indispensable shield against the cardiovascular toxicity of phosphate and unmitigated FGF23.
Neuroprotection and the Central Nervous System
While the renal phosphate axis dominates classical Klotho biology, intense research efforts spanning 2023 to 2026 have revolutionized our understanding of Klotho's role in the central nervous system. As global populations age and life expectancies rise, combating cognitive frailty, Alzheimer's disease (AD), and Parkinson's disease (PD) has become a paramount biomedical challenge 629. Within this context, Klotho has emerged as a premier "resilience factor," a protein that does not merely target a specific, downstream pathology like amyloid-beta plaques or tau tangles, but broadly enhances the brain's foundational capacity to withstand multiple forms of neurodegeneration and toxic protein aggregation 123230.
The Choroid Plexus: The Brain's Klotho Epicenter
Within the central nervous system, Klotho is overwhelmingly expressed in the choroid plexus (CP), the highly vascularized, secretory intraventricular structure responsible for producing cerebrospinal fluid (CSF) and maintaining the blood-CSF barrier 18223431. Recent highly granular transcriptomic and proteomic analyses confirm that CP epithelial cells produce vast quantities of Klotho, primarily the alternatively spliced, truncated secreted isoform, releasing it directly into the CSF 181922.
The physiological function of CP-derived Klotho is uniquely tied to neuro-immune modulation and barrier integrity. The CP acts as a primary site of peripheral-central immune surveillance, one of the few CNS structures where T cells and macrophages reside 1831. Under healthy, youthful conditions, high concentrations of CSF Klotho act as a neuro-immunological "gatekeeper," maintaining the structural integrity of the tight junctions comprising the blood-CSF barrier and suppressing aberrant immune activation 3233.
However, CP Klotho production declines precipitously with advancing age and in neurodegenerative conditions, a loss that destabilizes the entire neurovascular unit. A landmark 2025 clinical study utilizing advanced magnetic resonance imaging (MRI) revealed that significantly reduced CSF Klotho levels strongly correlate with pathological CP enlargement and volumetric dysfunction in patients suffering from neurodegenerative diseases 18. Mechanistically, experimental deletion of Klotho specifically from the CP via viral vectors in young murine models triggers a cascade of spontaneous neuroinflammation that simulates advanced aging 3233. The specific absence of Klotho in the CP leads to the profound upregulation of Thioredoxin-Interacting Protein (TXNIP), which subsequently hyperactivates the NLRP3 inflammasome within localized CP macrophages 732. This molecular breakdown of the Klotho-mediated gate allows peripheral innate immune cells and pro-inflammatory cytokines to infiltrate the adjacent hippocampus, exacerbating microglial activation, generating severe oxidative stress, and accelerating cognitive decline - a phenomenon heavily implicated in age-related "inflammaging" 313233.
The Blood-Brain Barrier (BBB) Paradox of Peripheral Klotho
One of the most intensely debated, counterintuitive, and fascinating discoveries in recent neurogerontology is the BBB Paradox of peripheral Klotho. Abundant preclinical evidence from multiple independent laboratories demonstrates that artificially increasing circulating Klotho levels - whether through genetic overexpression, viral gene therapy, or direct peripheral injection of recombinant Klotho protein into the bloodstream - profoundly enhances learning, spatial memory, and cognitive resilience in both healthy animals and transgenic models of Alzheimer's and Parkinson's diseases 691238.
The physiological paradox lies squarely in the pharmacokinetics of the molecule: the 130 kDa soluble Klotho protein is physically far too massive to cross the intact, highly restrictive blood-brain barrier 12. How, then, does a peripheral, intravenous, or subcutaneous infusion of a massive protein orchestrate such profound, rapid, and direct neurological enhancements within the isolated CNS?
Research advanced significantly between 2023 and 2026, largely spearheaded by investigative teams such as Dr. Dena Dubal's laboratory at the University of California, San Francisco (UCSF), to elucidate this indirect mechanism of action 12. The current prevailing scientific paradigm suggests that peripheral soluble Klotho exerts its central neuroprotective effects through an elaborate cascade of indirect systemic signaling pathways, rather than requiring direct physical penetration into the brain parenchyma 12.
The proposed mechanism involves a highly complex, multi-step biological relay: 1. Peripheral Receptor Engagement and Modulation: Soluble Klotho circulating in the peripheral blood likely interacts with unidentified cell-surface receptors on endothelial cells or modulates the activity of peripheral circulating factors. It acts systemically to drastically reduce oxidative stress, inhibit pro-inflammatory systemic TGF-$\beta$ signaling, and alter the secretion of circulating osteokines from bone marrow stroma 11123440. 2. Transduction via Secondary Pathways: It is strongly hypothesized that this peripheral biological interaction generates a secondary physiological signal. This signal is transmitted either through neural pathways, such as the extensive vagus nerve network that links gut and organ immunity directly to the brainstem, or via the enzymatic generation of small, highly lipophilic, BBB-permeable neuroactive metabolites that easily cross the endothelial tight junctions 3034. 3. Synaptic Strengthening via NMDA Receptors: Once this secondary molecular or electrical signal reaches the hippocampus and prefrontal cortex, it induces robust, physical changes in synaptic plasticity. Specifically, it has been demonstrated that peripheral Klotho elevation leads to the rapid strengthening of glutamatergic synapses, primarily mediated through the enhanced activation, phosphorylation, and cell-surface trafficking of N-methyl-D-aspartate (NMDA) receptor pathways within the brain, fundamentally augmenting the hardware required for memory formation and retention 12.
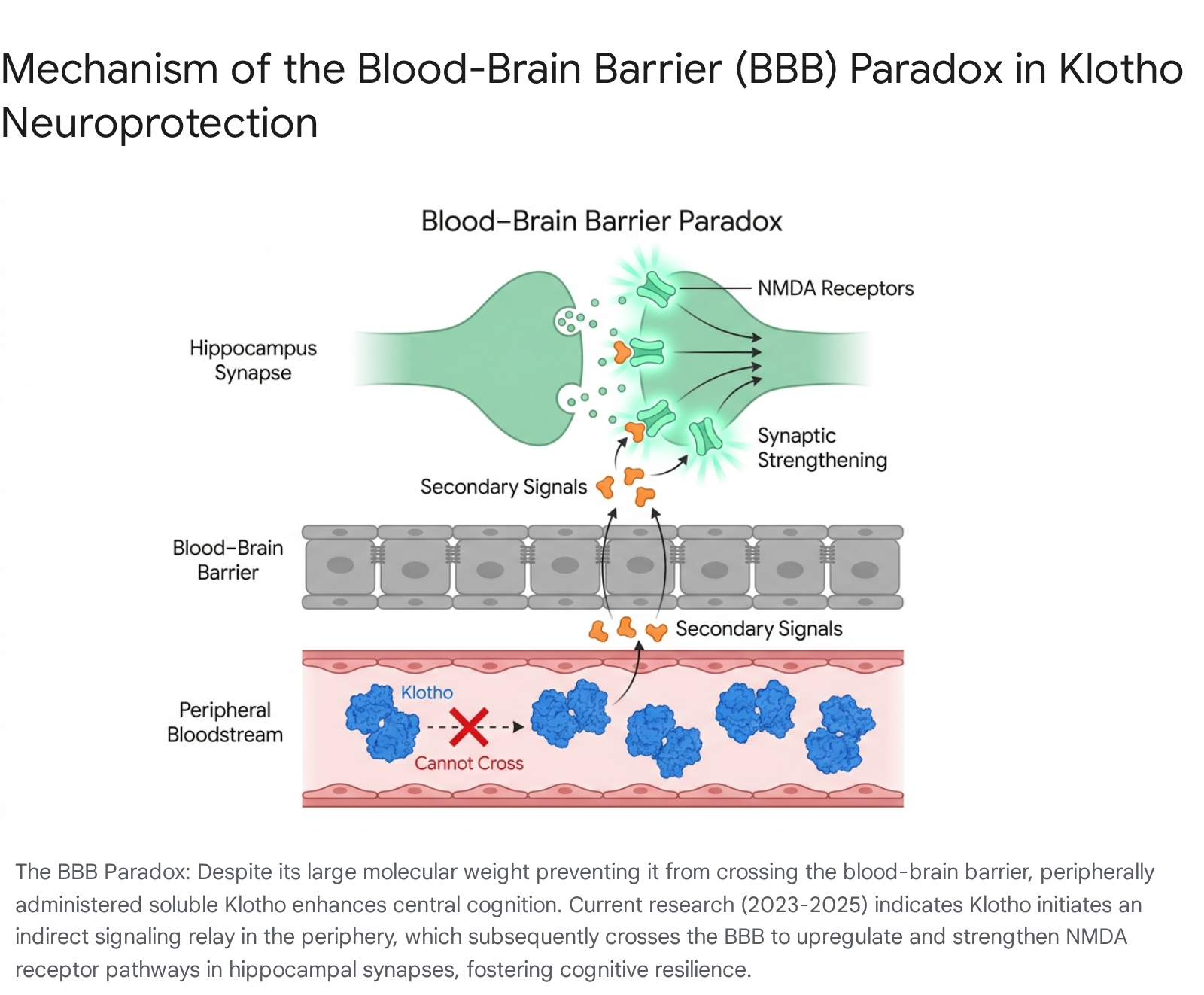
This indirect signaling mechanism represents a massive paradigm shift in neuropharmacology. It indicates that intractable neurodegenerative diseases may be effectively combatted not only by small molecules meticulously designed to cross the notoriously restrictive BBB, but by systemic biological modifiers that leverage the body's existing peripheral-to-central communication axes to fortify brain resilience from the outside in.
Human Clinical Validation: The KL-VS Genetic Variant Controversy
The elegant neuroprotective theories derived from animal models find compelling, albeit highly complex and sometimes contradictory, correlates in human population genetics. Approximately 15% to 20% of the Caucasian population is heterozygous for a specific, well-studied genetic haplotype of the KLOTHO gene known as KL-VS 622. This haplotype includes a cluster of six single nucleotide polymorphisms, two of which result in functional missense mutations (F352V and C370S) in exon 2 22. In vitro studies suggest these amino acid substitutions alter the protein's inherent transmembrane shedding dynamics and half-life, paradoxically resulting in higher systemic levels of circulating Klotho in heterozygous carriers compared to wild-type individuals 622.
Extensive observational studies over the past decade demonstrated that carrying a single copy of the KL-VS variant is associated with enhanced overall longevity, greater baseline cortical volumes (specifically in the right dorsolateral prefrontal cortex), and superior performance on cognitive executive function tests throughout adulthood 635. Furthermore, the KL-VS allele appears to confer a powerful neuroprotective buffer against the onset of Alzheimer's disease. In vulnerable individuals carrying the high-risk Apolipoprotein E4 (ApoE $\epsilon$4) allele, co-inheritance of the KL-VS variant reportedly mitigates the risk of developing clinical AD by approximately 30% to 40% 2242. A detailed 2025 study of patients diagnosed with amnestic mild cognitive impairment (aMCI) confirmed that KL-VS heterozygotes displayed significantly better memory performance and a lower likelihood of classification as full AD dementia, particularly among those who were ApoE $\epsilon$4 carriers 36.
However, the interpretation of this genetic data requires extreme caution and nuance. A massive, paradigm-challenging 2025 population-based study utilizing a vast cohort of over 320,000 individuals challenged the universality of these benefits. When researchers heavily adjusted the data for sex, age, and broad genetic backgrounds across a massive sample size, they found completely no significant protective effects of the Klotho-VS genotype on general cognitive decline, nor did it rescue the cognitive decline mediated by ApoE $\epsilon$4 status in older adults 35. Heterozygotes showed slightly better cognition before age 11, but actually performed worse in adolescence, indicating complex antagonistic pleiotropy 35. This deep controversy suggests that while elevated Klotho likely provides profound baseline cognitive reserve, its interactions with other genetic, environmental, and developmental factors are extraordinarily complex. This unresolved tension emphasizes the critical need for targeted, randomized clinical interventional trials, rather than relying solely on genetic epidemiology to dictate therapeutic direction.
Methodological Hurdles: The Crisis in Human Klotho Measurement
A critical bottleneck severely hindering the successful translation of Klotho research into routine clinical practice is the severe unreliability of current commercial assays used to measure soluble Klotho in human serum. Much of the early clinical literature correlating declining Klotho levels with disease states (such as CKD progression, cardiovascular events, and aging) relied heavily on commercially available enzyme-linked immunosorbent assays (ELISAs), specifically the widely adopted assay manufactured by Immuno-Biological Laboratories (IBL) 374546. However, recent rigorous comparative studies have exposed profound limitations in these tests, calling into question the validity of decades of observational data 3745.
The IBL ELISA vs. Immunoprecipitation-Immunoblot (IP-IB)
The performance of the commercial IBL ELISA has been subjected to intense biomedical scrutiny. While the manufacturer claims high analytical precision, independent clinical researchers evaluating the assay across broad cohorts - including healthy volunteers and patients with varying stages of kidney disease - have reported alarming inconsistencies.
In rigorous head-to-head comparisons between the commercial ELISA and a highly specific, labor-intensive immunoprecipitation-immunoblot (IP-IB) assay utilizing novel synthetic antibodies, the commercial ELISA failed across multiple fundamental analytical metrics 3745: * Poor Correlation with Renal Function: Given that soluble Klotho is primarily produced by the kidneys, its circulating levels should theoretically track closely with renal health. Measurements utilizing the IP-IB assay demonstrated a highly significant, strong correlation with estimated Glomerular Filtration Rate (eGFR) ($R = 0.80$, $P < 0.001$). In stark contrast, the commercial ELISA exhibited minimal to no correlation with eGFR ($R = 0.18$, $P = 0.12$) within the exact same patient cohorts, including those in the landmark SPRINT trial 3745. * Suboptimal Recovery Rates: When human serum was experimentally "spiked" with known quantities of exogenous recombinant Klotho, the IP-IB assay successfully recovered 81% to 115% of the protein across the spectrum of kidney function. The commercial ELISA only managed a 60% to 81% recovery rate, indicating severe capture inefficiency, especially in complex uremic serum from advanced CKD patients 3745. * High Inter-Assay Variability: Independent clinical biochemistry laboratories have reported inter-assay coefficients of variation (CV) for the commercial ELISA soaring as high as 44.9%, vastly exceeding acceptable clinical limits and making longitudinal or cross-institutional comparisons nearly impossible 37. * Susceptibility to Pre-analytical Variables: The performance of the commercial ELISA is highly, and negatively, sensitive to standard sample handling procedures. The addition of protease inhibitor cocktails (often routinely used to preserve blood samples) artificially inflated measured Klotho concentrations and vastly increased intra-subject variability when measured by ELISA, while single freeze-thaw cycles further degraded measurement accuracy 3745. * Lack of Isoform Specificity: Furthermore, a severe lack of standardization implies that different commercial antibodies recognize different Klotho fragments. The assays frequently cannot distinguish between the large 130 kDa cleaved soluble form (KL1-KL2) and the smaller 70 kDa alternatively spliced secreted form (KL1 only), adding biological ambiguity to the analytical failure 819.
Implications for the Field
The deep unreliability of commercial ELISAs offers a highly compelling explanation for the conflicting, noisy results observed in human observational trials over the last decade 37. Many studies have failed to find the expected declines in Klotho during disease progression simply because the assays utilized were functionally incapable of accurately capturing the protein in pathological serum 37. Until high-throughput assays match the fidelity of the IP-IB method, or until mass spectrometry-based absolute quantification becomes standardized and financially viable, circulating Klotho cannot be reliably utilized as a clinical biomarker. Consequently, data from older observational trials utilizing standard ELISAs must be interpreted with extreme caution and skepticism.
Translational Therapeutics: Bridging the Preclinical-Clinical Divide
Driven by the spectacular, life-extending results observed in animal models, the pursuit of therapeutics capable of increasing systemic Klotho has accelerated at a breakneck pace. However, a highly critical analysis of the literature from 2024 to 2026 reveals a stark contrast between established, triumphant rodent outcomes and the nascent, highly experimental reality of human clinical translation 38384839.
The Triumph of Murine Gene Therapy
In well-controlled animal models, virus-mediated gene therapy has yielded unequivocally profound results. A landmark 2025 study, led by Dr. Miguel Chillón and researchers at the Universitat Autònoma de Barcelona, utilized an Adeno-Associated Virus serotype 9 (AAV9) vector to deliver the genetic code for the secreted isoform of Klotho (s-KL) to middle-aged mice (12 months old, roughly equivalent to 44-year-old humans) 9323850.
The outcomes were transformative and unprecedented: the single-dose AAV9 treatment resulted in a 15% to 19.7% absolute extension in the murine lifespan 93850. Beyond mere longevity, the older mice exhibited a drastically expanded "healthspan." By the time they reached 24 months of age, the treated mice demonstrated significant increases in muscle fiber size and physical fitness, enhanced muscular regenerative capacity, reductions in fibrotic tissue, and preserved internal trabecular bone architecture, pointing to a protective effect against osteoporosis 950. Furthermore, the s-KL treatment stimulated adult neurogenesis and boosted immune-modulatory phagocytosis in the aged hippocampus, directly correlating with improved cognitive performance 950.
The Human Reality: Limitations and Preliminary Clinical Trials
Despite these triumphs, translating AAV9 gene therapy directly to humans presents formidable biological, ethical, and regulatory barriers 3838. 1. Delivery Vectors and the BBB: In the successful 2025 murine trials, the researchers only achieved adequate CNS transduction by combining standard intravenous (IV) injections with highly invasive intracerebroventricular (ICV) injections directly through the skull and into the brain 3850. Such invasive intracranial surgical procedures are clinically unjustifiable for prophylactic, anti-aging treatments in healthy human adults. 2. Overexpression Toxicity: Unlike small-molecule drugs that wash out of the system, AAV therapies integrate and are notoriously difficult to "turn off." Long-term, unregulated overexpression of Klotho raises theoretical concerns regarding the severe disruption of mineral homeostasis (potentially driving hypophosphatemia and osteomalacia via unchecked FGF23 signaling). Furthermore, hyper-elevated Klotho has been controversially linked in some observational data to elevated risks for specific psychiatric phenotypes, such as schizophrenia, highlighting the absolute necessity for precise spatiotemporal control of gene expression 1738. 3. Protein Supplementation Failures: Early biomedical attempts to circumvent gene therapy by simply injecting recombinant Klotho protein into humans faced severe limitations. The protein has an exceedingly short circulating half-life, the cost of large-scale manufacturing is prohibitive, and recombinant proteins produced by bacterial or yeast vectors lack the proper human post-translational modifications (such as complex glycosylation), which drastically blunts their in vivo biological activity 3851.
The Emergence of Plasmid Gene Therapy (2025 - 2026)
To circumvent the immense limitations of viral vectors and recombinant proteins, cutting-edge biotechnology firms have recently advanced non-viral plasmid gene therapies into human clinical trials. As of late 2025 and 2026, two highly notable Phase 1 interventional clinical trials (NCT07216781 and NCT07285629), spearheaded by Minicircle Inc., began actively recruiting healthy adult volunteers 394053.
These groundbreaking trials utilize "minicircle" DNA plasmids - engineered, stripped-down circular vectors entirely lacking viral and bacterial backbone sequences - that are administered via a simple subcutaneous injection directly into abdominal adipose tissue 394054. The localized fat tissue acts as a biological bioreactor, taking up the plasmids and continuously secreting endogenous, perfectly folded, and heavily glycosylated human Klotho directly into the systemic circulation. One trial (NCT07216781) focuses on Klotho plasmid therapy alone, while a secondary trial (NCT07285629) evaluates a combinatorial gene therapy delivering both Klotho and Follistatin, attempting to simultaneously promote muscle hypertrophy and lifespan extension 3940.
These trials represent the bleeding edge of interventional gerontological therapeutics. They will track highly rigorous clinical endpoints over a six-month period, including subjective well-being, epigenetic age clocks, dual-energy X-ray absorptiometry (DXA) body composition, brain perfusion measures, and comprehensive metabolic profiles, focusing specifically on high-fidelity renal markers like Cystatin C and serum chloride 3940. However, as Phase 1 pilot studies, it must be stressed that their primary, immediate objective is merely to establish fundamental safety, tolerability, and proper dosing kinetics in humans, not to validate efficacy.
Broadening the Horizon: Small-Molecule Enhancers and Lifestyle Interventions
Recognizing the steep regulatory hurdles, prohibitive costs, and potential irreversibility of gene therapy, the pharmacological field has aggressively shifted toward identifying modalities capable of safely inducing the endogenous expression of the KLOTHO gene. By chemically stimulating the body's own KL promoters, researchers hope to achieve safe, titratable, and physiological increases in systemic Klotho production 3848.
Small-Molecule Klotho Enhancers
A comprehensive 2026 pharmacological review systematically classified emerging small-molecule Klotho inducers based on their specific mechanistic targets 3848. These diverse compounds aim to reverse the epigenetic and transcriptional silencing that typically represses Klotho expression during natural aging and chronic disease. * Epigenetic Modulators: Klotho silencing in aging, chronic kidney disease, and certain carcinomas is frequently driven by profound hypermethylation of its promoter region and extensive histone deacetylation 2948. DNA methyltransferase (DNMT) inhibitors, such as the chemotherapeutic agent 5-Azacytidine, and Histone deacetylase (HDAC) inhibitors, including Valproic acid, Trichostatin A, and the natural isoflavone Genistein, have demonstrated immense efficacy in restoring chromatin accessibility. In both in vitro cervical cancer cell lines and preclinical tissue models, these agents dramatically un-silenced and upregulated endogenous Klotho transcription 3848. * Metabolic Reprogramming and NAMPT Activators (SBI-797812): A highly promising, novel avenue involves metabolic reprogramming through NAD+ restoration. Nicotinamide phosphoribosyltransferase (NAMPT) is the rate-limiting enzyme in the NAD+ salvage pathway. In a landmark 2026 study, researchers deployed SBI-797812, a novel, structurally unique small-molecule "super catalyst" that directly binds and activates NAMPT, aggressively boosting intracellular NAD+ and NMN levels 5556. In murine models of acute kidney ischemia-reperfusion injury, SBI-797812-induced NAD+ restoration rapidly activated the SIRT1/PGC-1$\alpha$ signaling pathway. SIRT1, a crucial longevity-associated deacetylase, subsequently drove a profound upregulation of Klotho expression. This cascade ultimately restored renal urine flow, significantly lowered blood urea nitrogen (BUN) and creatinine, inhibited NF-$\kappa$B-mediated inflammation (lowering TNF-$\alpha$ and IL-6), and halted apoptotic cascades, proving the viability of small-molecule Klotho induction 55. * Repurposed Pharmacologics: Existing, clinically approved drugs have also been repurposed as effective Klotho enhancers. Renin-angiotensin system (RAS) blockers, such as Losartan and Valsartan, mitigate angiotensin II-induced oxidative stress in the kidney, thereby relieving the transcriptional repression of the KL gene and significantly increasing systemic soluble Klotho 3848. Similarly, the mTOR inhibitor Rapamycin and various anti-diabetic medications (Metformin, SGLT2 inhibitors like empagliflozin, and GLP-1 receptor agonists) have been shown to indirectly promote Klotho expression through the broad suppression of pro-aging, pro-inflammatory metabolic pathways 38415842. Antioxidants such as N-acetylcysteine, curcumin, and resveratrol also mitigate the oxidative stress-induced repression of the KL promoter 303848.
Lifestyle Interventions: Exercise as an "Exerkine" Stimulator
While synthetic pharmacological interventions traverse long, rigorous regulatory pathways, extensive clinical data validate that robust lifestyle interventions directly and potently upregulate Klotho safely today. * Exercise Physiology: A massive, comprehensive 2022 systematic review and meta-analysis comprising 12 randomized controlled trials (involving 621 participants, aged 30 to 65 years) definitively established that chronic exercise training significantly elevates circulating soluble Klotho (Hedges' g = 1.3, $P < 0.0001$) 434462. This highly robust, statistically significant effect was observed across varied health conditions and exercise modalities (both aerobic and resistance), though the interventions strictly required a minimum continuous duration of 12 weeks to achieve sustained baseline elevations 4344. Interestingly, acute, highly intense exercise may transiently depress Klotho levels immediately post-workout, followed by a dramatic rebound over subsequent days, highlighting the protein's dynamic role as a stress-responsive "exerkine" secreted by skeletal muscle to mediate whole-body metabolic adaptation, induce antioxidant enzymes (like catalase and superoxide dismutase), and combat age-related insulin-like growth factor-1 (IGF-1) toxicity 624546. * Dietary Modulation: Nutritional strategies also profoundly impact the Klotho axis. Intermittent energy restriction and caloric reduction decrease systemic oxidative stress, allowing baseline Klotho transcription to proceed unhindered 4246. More specifically, strict dietary phosphate restriction directly unburdens the renal FGF23-Klotho axis. By reducing the obligate physiological demand for continuous phosphaturia, phosphate restriction preserves renal functional reserve and highly upregulates tubular Klotho expression, offering a highly actionable, non-pharmacological strategy for delaying vascular aging and CKD progression 112246.
Table 2: Comparative Overview of Klotho-Targeted Therapeutics
| Intervention Category | Specific Modality / Compound | Mechanism of Action | Current Translational Status |
|---|---|---|---|
| Gene Therapy (Viral) | AAV9 vector encoding secreted Klotho (s-KL) | Transduction of hepatocytes/CNS to continuously secrete Klotho protein. | Preclinical (High rodent success; +19.7% lifespan); limited by BBB delivery hurdles and human overexpression toxicity risks. |
| Gene Therapy (Non-Viral) | Minicircle Plasmid DNA | Subcutaneous fat injection acts as a bioreactor for transient, titratable endogenous Klotho production. | Phase 1 Clinical Trials (NCT07216781; NCT07285629; recruiting healthy adults 2025/2026). |
| Epigenetic Modulators | Valproic Acid, 5-Azacytidine, Genistein | Inhibits HDACs and DNMTs to reverse the hypermethylation and un-silence the KLOTHO promoter. | Experimental / Preclinical for aging applications; Drugs are FDA approved for other specific indications (cancer, epilepsy). |
| Metabolic/NAD+ Boosters | SBI-797812 | "Super catalyst" that activates NAMPT, boosting NAD+ and driving SIRT1/PGC-1$\alpha$-mediated Klotho transcription. | Preclinical in vivo validation (2026); High potential for future small-molecule translation. |
| Re-purposed Drugs | RAS Blockers (Losartan, Valsartan), Rapamycin | Suppresses angiotensin/mTOR pathways and reduces inflammatory oxidative stress, lifting KL repression. | Clinically established globally; Klotho enhancement acts as a beneficial, secondary pleiotropic effect. |
| Lifestyle - Exercise | Chronic Aerobic or Resistance Training | Acts as a stress-responsive 'exerkine'; muscle-derived signaling stimulates endogenous systemic production. | Highly validated in human meta-analyses; strictly requires long-term (12+ weeks) protocol compliance. |
| Lifestyle - Diet | Phosphate Restriction, Intermittent Fasting | Reduces demand on renal Klotho-FGF23 axis; minimizes oxidative stress and inflammation. | Clinically accessible immediately; highly effective in CKD management and longevity models. |
Conclusion
The Klotho protein represents one of the most compelling, structurally elegant, and biologically complex targets in modern molecular gerontology. Over the last two decades, its paradigm has expanded dramatically. Originating as a strictly renal-centric co-receptor tasked with mitigating the cardiovascular and calcific toxicities of phosphate metabolism, it is now globally recognized as a premier, systemic resilience factor capable of profoundly defending the central nervous system against age-related cognitive decline, Alzheimer's, and Parkinson's disease.
The elucidation of the Blood-Brain Barrier paradox - demonstrating that massive peripheral Klotho molecules rely on an elaborate, indirect systemic signaling relay to ultimately fortify central NMDA receptor synapses - solves a longstanding pharmacokinetic mystery. This breakthrough fundamentally redefines neuropharmacology, proving that the brain can be armored against degeneration without requiring drugs to physically penetrate its barriers.
However, realizing the ultimate promise of Klotho in human medicine demands the immediate, rigorous resolution of critical translational barriers. The biological ambiguity and severe unreliability of commercial ELISA assays must be rectified through the widespread, mandated adoption of highly rigorous IP-IB techniques or mass spectrometry standardizations; without accurate measurement, clinical observational data remains hopelessly confounded. Furthermore, while AAV-mediated gene therapies have definitively proven the concept of Klotho-induced longevity and tissue regeneration in rodents, the future of human intervention likely resides in the more titratable, safer avenues currently undergoing intense evaluation. The pioneering non-viral Minicircle plasmid trials of 2025 - 2026, alongside the rapid, exciting development of sophisticated small-molecule NAMPT activators like SBI-797812, represent the vanguard of this monumental effort. Until these pharmacological breakthroughs clear the rigors of late-stage clinical trials, rigorous exercise regimens and dietary phosphate restriction remain the only clinically validated, immediately accessible methodologies to harness the profound, life-extending power of the Klotho axis.