Insulin and IGF-1 signaling in longevity and mammalian aging
Introduction to the Somatotropic Axis and the Biology of Aging
For decades, the biological processes underlying physiological aging were predominantly viewed through the lens of thermodynamic inevitability - an accumulation of random cellular wear and tear, oxidative damage, and entropic decay that progressively eroded organismal function over time. However, the emergence of modern geroscience has radically transformed this paradigm, revealing that aging is heavily dictated by highly conserved, genetically modifiable nutrient-sensing pathways. Chief among these is the somatotropic axis, comprising growth hormone (GH), insulin-like growth factor-1 (IGF-1), and the mechanistic target of rapamycin (mTOR) signaling network. This intricate biochemical cascade orchestrates the fundamental biological trade-off between somatic growth, reproductive maturation, and long-term cellular maintenance 123.
The groundbreaking discovery that genetic downregulation of the insulin/IGF-1 signaling (IIS) pathway could profoundly extend lifespan was first documented over two decades ago in the nematode Caenorhabditis elegans, where mutations in the daf-2 receptor gene resulted in a doubling of the organism's maximum lifespan 45. This revelation catalyzed a global research initiative, subsequently demonstrating that diminished IIS and mTOR activity could delay age-related decline across yeast, fruit flies, and mammalian models, including rodents 267. Yet, the translation of these foundational findings into human clinical applications has proven exceptionally complex, fraught with physiological paradoxes and confounding species-specific biology.
In humans, the relationship between circulating IGF-1 levels, healthspan, and mortality does not follow a linear trajectory where biological suppression is universally beneficial. Instead, the pathway is governed by the evolutionary principle of antagonistic pleiotropy, resulting in a paradoxical U-shaped mortality curve where both hyper-activation and severe deficiency of IGF-1 confer significant, albeit distinct, health risks 889. Consequently, the contemporary geroscience translational pipeline is shifting away from blunt, systemic suppression of the somatotropic axis and moving toward highly selective, context-dependent pharmacological modulation. This exhaustive report examines the evolutionary underpinnings of the IGF-1/mTOR axis, dissects the human genetic evidence surrounding FOXO3a longevity variants across diverse global cohorts, evaluates the translational limitations between invertebrate and mammalian models, and provides a rigorous analysis of the late-stage clinical therapeutics currently defining the field of longevity medicine in the 2024 - 2026 period.
The Evolutionary Paradigm: Antagonistic Pleiotropy
To comprehend why a signaling pathway essential for early-life development and physiological maturation becomes a primary driver of senescence and chronic disease in late life, one must apply the evolutionary concept of antagonistic pleiotropy. Originally formalized by evolutionary biologist George C. Williams in 1957, antagonistic pleiotropy posits that genes or biological pathways that confer a strong fitness advantage early in life - such as rapid organismal growth, early sexual maturation, and high reproductive output - will be positively selected for by evolutionary pressures, even if those exact same genes exert detrimental, pro-aging effects during post-reproductive phases 91012. Because natural selection exerts minimal pressure on organisms post-reproduction - a period colloquially referred to as the "selection shadow" - traits that accelerate later-life decay are not actively purged from the gene pool provided they successfully guarantee early-life propagation.
The Dual Role of the IGF-1/mTOR Axis in Cellular Fate
The IGF-1/mTOR axis is the quintessential biological manifestation of antagonistic pleiotropy. During embryonic, neonatal, and juvenile developmental windows, high systemic levels of GH and its downstream effector IGF-1 are absolutely critical for tissue accretion, bone elongation, skeletal muscle hypertrophy, and central nervous system development 1112. The primary intracellular effector of this axis, mTOR Complex 1 (mTORC1), acts as the cellular master nutrient sensor. When nutrients (such as amino acids and glucose) and growth factors (including IGF-1 and insulin) are abundant, mTORC1 is hyper-activated. It drives cellular anabolism by promoting ribosomal biogenesis and protein translation via the phosphorylation of S6 Kinase 1 (S6K1) and eukaryotic translation-initiation factor 4E-binding proteins (4E-BPs) 613. Simultaneously, active mTORC1 shuts down vital catabolic processes, notably macroautophagy, via the phosphorylation and subsequent inhibition of Unc-51-like autophagy-activating kinase (ULK1) 1314.
In youth, this hyper-anabolic state ensures that an organism rapidly achieves reproductive maturity and possesses the physical robustness necessary for survival. However, in late life, the continuous, unabated signaling of the IGF-1/mTOR axis becomes profoundly maladaptive. Once somatic growth has ceased, chronic mTORC1 activation drives cellular hyperfunction. This state leads to the rapid depletion of adult stem cell pools, the toxic accumulation of misfolded protein aggregates and damaged organelles (due to chronically suppressed autophagic clearance), and the increased secretion of pro-inflammatory cytokines associated with the senescence-associated secretory phenotype (SASP) 91317. Furthermore, chronic IGF-1 signaling activates the PI3K/Akt kinase cascade, which phosphorylates FOXO transcription factors. This phosphorylation event prevents the nuclear translocation of FOXO, thereby inhibiting the robust expression of vital stress-resistance genes, antioxidant enzymes, and DNA-repair mechanisms 715. Thus, the very pathway that builds the organism actively prevents its long-term maintenance.
Ecological Evidence from Wild Mammalian Populations
The evolutionary trade-offs mediated by systemic IGF-1 are not merely theoretical constructs confined to laboratory environments; they are clearly observable in wild mammalian populations, acting as a biological metronome that dictates the "fast-slow" life-history continuum 1620. Comprehensive comparative studies spanning 41 diverse mammalian species reveal that naturally higher plasma IGF-1 concentrations are intricately linked to "faster" life histories. These species exhibit rapid postnatal development, earlier age at first female reproductive maturity, larger litter sizes, but a correspondingly abbreviated maximum lifespan 16.
In wild Soay sheep populations, for example, researchers have documented profound population-level phenotypic plasticity tied directly to systemic IGF-1. Lambs exhibiting elevated summer IGF-1 levels achieved significantly greater body mass, post-natal growth rates, and skeletal size. Consequently, these high-IGF-1 individuals were significantly more likely to survive the harsh conditions of their first winter and reproduce the following spring 20. However, this accelerated early-life vitality comes at a definitive physiological cost. The energetic allocation toward rapid growth, mediated by IGF-1, inherently diverts resources away from cellular repair mechanisms. Across mammalian species, the physiological commitment to elevated IGF-1 and hyper-anabolism invariably correlates with accelerated physiological decline post-maturity and a higher propensity for age-related morbidities 1620. Furthermore, resource limitation effects on juvenile IGF-1 can act via maternal traits, where lambs born to young, still-growing mothers, or older, resource-depleted mothers, exhibit reduced IGF-1 levels, showcasing the pathway's exquisite sensitivity to environmental and generational energetic availability 20.
Physiological Trade-Offs of Systemic IGF-1 Across the Lifespan
The dynamic shifts driven by systemic IGF-1 across different stages of mammalian life illustrate the precarious balance between growth and longevity. The table below summarizes these physiological trade-offs:
| Life Stage | Systemic IGF-1 Level | Evolutionary/Biological Purpose | Physiological Consequences & Phenotypic Expression | Long-Term Trade-offs |
|---|---|---|---|---|
| Fetal / Neonatal | Highly Elevated | Maximize tissue accretion, organogenesis, and survival probability. | Rapid cellular proliferation, bone elongation, and neurological development. Over-activation leads to macrosomia. | High nutrient demand; establishes baseline metabolic setpoints that may predispose to later-life metabolic syndrome if nutrients become scarce. |
| Juvenile / Pubertal | Peak Elevation | Accelerate time to reproductive maturity and peak physical robustness. | Muscle hypertrophy, sexual maturation, peak bone mineral density acquisition. Drives the "fast" life history strategy (early reproduction). | Secures immediate genetic propagation but initiates the accumulation of sub-lethal cellular damage due to the suppression of autophagic clearance. |
| Adult (Post-Reproductive) | Moderately High (Pathological) | Vestigial evolutionary drive for anabolism in a post-growth state. | Cellular hyperfunction, stem cell exhaustion, suppression of FOXO-mediated stress resistance, increased cellular senescence. | Accelerated biological aging, increased incidence of neoplasia, reduced metabolic flexibility, and reduced maximum lifespan. |
| Adult (Post-Reproductive) | Attenuated / Low (Optimal) | Shift energy allocation from growth toward somatic maintenance. | Upregulation of autophagy, enhanced DNA repair via FOXO nuclear translocation, reduced oxidative stress, delayed onset of age-related decline. | Prolonged healthspan and delayed mortality, provided levels do not drop below the critical physiological threshold required for tissue homeostasis. |
| Advanced Age (Elderly) | Severely Deficient | Exhaustion of the somatotropic axis and hepatic synthetic capacity. | Sarcopenia, osteopenia, cognitive decline, frailty, immunosenescence, and impaired recovery from physiological stressors. | Increased susceptibility to cardiovascular disease, mobility disability, overt hepatic encephalopathy, and elevated all-cause mortality. |
The Invertebrate Foundation and the Translational Gap
The structural and functional conservation of the insulin/IGF-1 signaling (IIS) cascade across immense evolutionary distances is a testament to its fundamental biological importance. The seminal discoveries in geroscience were forged using the invertebrate models Caenorhabditis elegans (nematode) and Drosophila melanogaster (fruit fly), which share striking homologous alignments with human metabolic and longevity pathways.
Mapping the Homologous Pathway Components
In both invertebrates and mammals, the IIS pathway operates through a sequential cascade of phosphorylation events acting as molecular toggles. Ligand binding activates a transmembrane tyrosine kinase receptor, which recruits intracellular adaptor proteins (such as IRS). These adaptors recruit lipid kinases, predominantly Phosphoinositide 3-kinase (PI3K), to convert the membrane lipid PIP2 to PIP3 71722. This action, which is directly antagonized by the PTEN lipid phosphatase, recruits downstream serine/threonine kinases (PDK1 and Akt/PKB) to the cell membrane for activation. Activated Akt then directly phosphorylates FOXO transcription factors. This critical phosphorylation event forces FOXO to bind with 14-3-3 anchor proteins, which physically sequesters the transcription factors in the cytoplasm, entirely preventing them from translocating to the nucleus to activate longevity and stress-resistance gene networks 172218.
The highly conserved homologous components of this signaling cascade across diverse species are detailed below:
| Functional Role in IIS Cascade | C. elegans (Nematode) | Drosophila (Fruit Fly) | Mammals (Mice / Humans) |
|---|---|---|---|
| Circulating Ligands | INS-1 through INS-39 (39 distinct insulin-like peptides) | DILP 1-8 (Drosophila insulin-like peptides) | Insulin, IGF-1, IGF-2 |
| Transmembrane Receptor | DAF-2 (Sole insulin/IGF-1 receptor ortholog) | dInR | Insulin Receptor (IR), IGF-1 Receptor (IGF-1R) |
| Receptor Substrate (Adaptor) | IST-1 | CHICO | IRS-1, IRS-2, IRS-3, IRS-4 |
| Lipid Kinase (Activator) | AGE-1 (PI3K ortholog) | PI3K (Dp110/p60) | PI3K (Class IA, p110/p85) |
| Lipid Phosphatase (Inhibitor) | DAF-18 | dPTEN | PTEN |
| Intermediate Kinase | PDK-1 | dPDK1 | PDK1 |
| Primary Effector Kinase | AKT-1, AKT-2, SGK-1 | dAkt1 (PKB) | Akt (PKB), SGK-1 |
| Master Transcription Factor | DAF-16 | dFOXO | FOXO1, FOXO3a, FOXO4, FOXO6 |
| Cytoplasmic Anchor | FTT-2, PAR-5 (14-3-3 orthologs) | 14-3-3 epsilon, 14-3-3 zeta (Leonardo) | 14-3-3 protein family |
The Translational Gap: Complexities of the Mammalian System
While the genetic homology is elegant and mechanistically informative, severe translational limitations exist when extrapolating longevity data directly from simple invertebrates to complex mammals. The primary confounding variable in C. elegans aging research is the dauer diapause state. In the wild, when nematode larvae face environmental stressors such as starvation or overcrowding, diminished IIS forces the animal into the dauer stage - a dormant, highly stress-resistant, and morphologically distinct larval form that can survive for months in suspended animation without undergoing biological aging 45.
The classical daf-2 mutations that famously double adult worm lifespan inherently trigger aspects of this dauer-associated genetic program even when the worm bypasses the larval diapause and enters adulthood. For decades, it remained unclear whether the extended lifespan of daf-2 mutants was a true retardation of aging or merely the pathological misexpression of an alternate developmental trajectory 45. Recent methodological advances, utilizing specific temperature-sensitive alleles across varying thermal ranges (15-25°C), have successfully decoupled dauer-dependent from dauer-independent longevity pathways, revealing that true aging delay is possible outside of diapause 524. Furthermore, cutting-edge techniques employing the auxin-inducible degradation (AID) system have allowed researchers to conditionally deplete DAF-2 protein levels exclusively in adult or elderly worms, proving that late-life suppression of IIS can still yield survival benefits independent of early-life development 525. However, the vestigial diapause mechanisms present in worms possess no direct physiological equivalent in mammalian biology, complicating the translation of effect sizes.
Furthermore, the mammalian neuroendocrine system introduces a layer of evolutionary bifurcation entirely absent in invertebrates. C. elegans possesses a staggering 39 insulin-like peptides but only a single receptor (DAF-2) to process all metabolic and longevity signals 1819. In stark contrast, mammals have evolved highly specialized distinct receptors (the Insulin Receptor and the IGF-1 Receptor) and separate ligands (Insulin, IGF-1, IGF-2) to delineate functions. While GH and IGF-1 levels are intimately linked in mammals via the hepatic somatotropic axis, their systemic actions are often divergent and even contradictory. For instance, while IGF-1 promotes systemic insulin sensitivity and drives peripheral fat deposition, GH induces profound insulin resistance and is highly lipolytic 1.
Genetically engineered dwarf mice lacking GH receptors exhibit a staggering 70% extension in maximum lifespan, making them among the longest-lived laboratory mammals 12. However, mice deficient strictly in IGF-1 experience only marginal lifespan extensions, and frequently suffer from high early-life mortality and severe growth retardation due to the indispensable role of IGF-1 in early somatic development and metabolic buffering 20.
Sex-specific dimorphism further profoundly complicates translation. Interventions targeting the IGF-1/mTOR axis in adult mammals consistently yield highly skewed, sex-specific success rates that are rarely observed in hermaphroditic nematode models. For example, the conditional, ubiquitous degradation of DAF-2 in male C. elegans utilizing the AID system resulted in a staggering 440% increase in median lifespan, whereas degradation localized solely to the male germline actually decreased lifespan - an effect completely opposite to that observed in hermaphrodites 2528. In mammalian models, reductions in the GH/IGF-1 axis, or the administration of late-stage preclinical monoclonal antibodies targeting the IGF-1R, preferentially and robustly extend median lifespan and healthspan in female mice, with male mice showing blunted or entirely non-existent longevity benefits despite achieving similar systemic metabolic parameters 2212223. These stark physiological disparities underscore why interventions that radically extend invertebrate life routinely fail to map directly to human clinical success.
Human Exceptional Longevity and the FOXO3a Architecture
If mammalian laboratory models present significant translational hurdles, the rigorous study of human exceptional longevity offers direct, undeniable evidence of the IIS pathway's role in human aging. As established, when IIS activity is attenuated, the FOXO family of transcription factors escapes 14-3-3 protein sequestration, translocates into the nucleus, and activates a vast network of target genes responsible for DNA repair, antioxidant defense, and stem cell maintenance 715. In humans, genetic variation within the FOXO3a gene is one of only two universally replicated genetic determinants of extreme longevity (the other being the APOE locus) 1024.
Specifically, the single nucleotide polymorphism (SNP) rs2802292 (a G > T transversion) within the intronic regulatory region of FOXO3a has emerged as the focal point of genetic gerontology. The presence of the minor "G" allele is consistently overrepresented in centenarian and supercentenarian populations worldwide, acting as a powerful biological shield against age-related morbidity 252627. Exhaustive genetic sequencing and rigorous meta-analyses across geographically, culturally, and genetically diverse cohorts have solidified the pleiotropic protective nature of this specific variant.
Geographically Diverse Centenarian Cohorts
The Hawaiian-Japanese Cohort: The initial breakthrough linking FOXO3a to human longevity occurred within the Honolulu Heart Program (HHP), an exhaustive 40-year prospective study tracking thousands of American men of Japanese ancestry. Researchers discovered that men carrying the homozygous "GG" genotype of rs2802292 were 2.75 times more likely to survive into their late 90s compared to those with the major "TT" genotype 2837. Crucially, the survival advantage was not merely an extension of decrepitude; G-allele carriers demonstrated a highly significant 10% reduction in all-cause mortality. This overarching survival benefit was driven primarily by a massive 26% reduction in coronary heart disease (CHD) mortality, alongside remarkably preserved insulin sensitivity and lower systemic cancer prevalence 2728. Interestingly, phenotypic analysis of this cohort revealed an inverse correlation between the longevity allele and physical height - subjects with the protective genotype were, on average, shorter than their peers. This finding perfectly echoes the antagonistic pleiotropy theory, where suppressed early-life growth signaling via attenuated IGF-1 yields late-life longevity 29.
The Okinawan Cohort: In the legendary longevity hotspot of Okinawa, Japan, detailed analysis of the rs2802292 variant revealed profound cellular mechanisms of preservation. Okinawan G-allele carriers aged 55 and older exhibited significantly higher telomerase activity in peripheral blood mononuclear cells (PBMCs), directly translating to the preservation of telomere length - a fundamental hallmark of delayed cellular senescence 30. Furthermore, FOXO3a appears to actively modulate the age-related inflammatory state known as "inflammaging." Older female carriers demonstrated a distinct age-dependent decline in the pro-inflammatory cytokine IL-6, while male carriers exhibited progressive increases in the anti-inflammatory cytokine IL-10, showcasing sex-specific mechanisms of disease resistance 30.
The Ashkenazi Jewish Cohort: Validating the variant's influence across entirely different genetic backgrounds, the Longevity Genes Project analyzed over 2,000 cases and 6,000 controls of Ashkenazi Jewish descent. The results robustly replicated the association of the FOXO3a rs2802292 G-allele with extended lifespan, confirming that the genetic advantage persists independently of specific cultural diets or lifestyles 3141. Whole-exome sequencing of Ashkenazi centenarians further revealed an intense enrichment of rare, protein-coding variants clustered specifically within the insulin/IGF-1 signaling pathway. These rare variants collectively shield individuals from common age-related conditions, providing a genetic buffer that allows them to bypass the cardiovascular and neurodegenerative diseases that typically limit human lifespan to eight decades 41.
The Sardinian Blue Zone Cohort: The mountainous Ogliastra region of Sardinia, Italy, was the first demographic "Blue Zone" formally identified, characterized by an unprecedented concentration of centenarians and an anomalous 2:1 female-to-male ratio (compared to the standard 5:1 observed in the broader Western world) 3233. Validation studies, including the comprehensive Southern Italian Centenarian Study, confirmed that the rs2802292 variant is significantly associated with extreme longevity in Sardinian and mainland Italian males 102534. While geographic isolation, low environmental pollution, a traditional Mediterranean diet, and strong communal support undeniably contribute to the Sardinian phenotype, the FOXO3a variant provides the underlying genetic architecture that enables these environmental factors to manifest as exceptional longevity 1033.
Global Replications and Gene-Environment Interactions: The robust pleiotropic benefits of the FOXO3a G-allele have been subsequently confirmed in German, French, and Danish cohorts, where it demonstrates consistent associations with exceptional aging 2435. Even in highly admixed populations, such as in southeastern Brazil, the FOXO3a GT genotype in the oldest-old is intimately linked with healthier lipid profiles, specifically lower systemic triglyceride levels, proving that the variant's metabolic benefits transcend strict ethnic boundaries 36.
However, emerging research underscores that these genetic benefits are intimately tied to environmental interactions, particularly diet. In the European NUTRIHEP cohort, individuals carrying the protective FOXO3a alleles who consumed high amounts of added sugars and processed meats still developed metabolic dysfunction-associated steatotic liver disease (MASLD), demonstrating that while the gene confers resilience, it can be overridden by sufficiently hostile environmental exposures 26. This gene-environment interaction highlights why lifestyle interventions remain paramount even for those with favorable genetic profiles.
The Misconception of Continual Suppression and the U-Shaped Mortality Curve
Given the overwhelming evolutionary, laboratory, and genetic evidence pointing to the inherent dangers of hyper-active IIS, a pervasive and dangerous misconception has permeated the anti-aging community: the reductionist belief that "lower IGF-1 is always better." This perspective is biologically flawed when applied to adult humans. The reality is that systemic IGF-1 operates on a precarious U-shaped mortality curve.
While laboratory interventions in model organisms frequently seek to ablate or drastically suppress the somatotropic axis to maximize overall lifespan, human physiological systems require a definitive baseline threshold of IGF-1 to maintain tissue integrity, cognitive function, and metabolic homeostasis. Pushing human IGF-1 levels too low induces catastrophic physiological decay.
Evidence from Large-Scale Human Cohorts
Recent, highly powered epidemiological analyses have definitively mapped this U-shaped relationship. A landmark 18-year prospective study utilizing the EPIC-Heidelberg cohort (over 7,400 individuals tracked with repeated biomarker assessments) and corroborating data from the UK Biobank (encompassing nearly 450,000 participants) demonstrated that participants with both the lowest and the highest levels of circulating IGF-1 experienced significantly elevated hazards of mortality from cardiovascular disease (CVD), cancer, and all causes 889.
A comprehensive meta-analysis encompassing over 30,000 participants pinpointed the optimal "trough" of the mortality curve: mid-range IGF-1 levels between 120 and 160 ng/mL were associated with the lowest absolute risk of death 89.
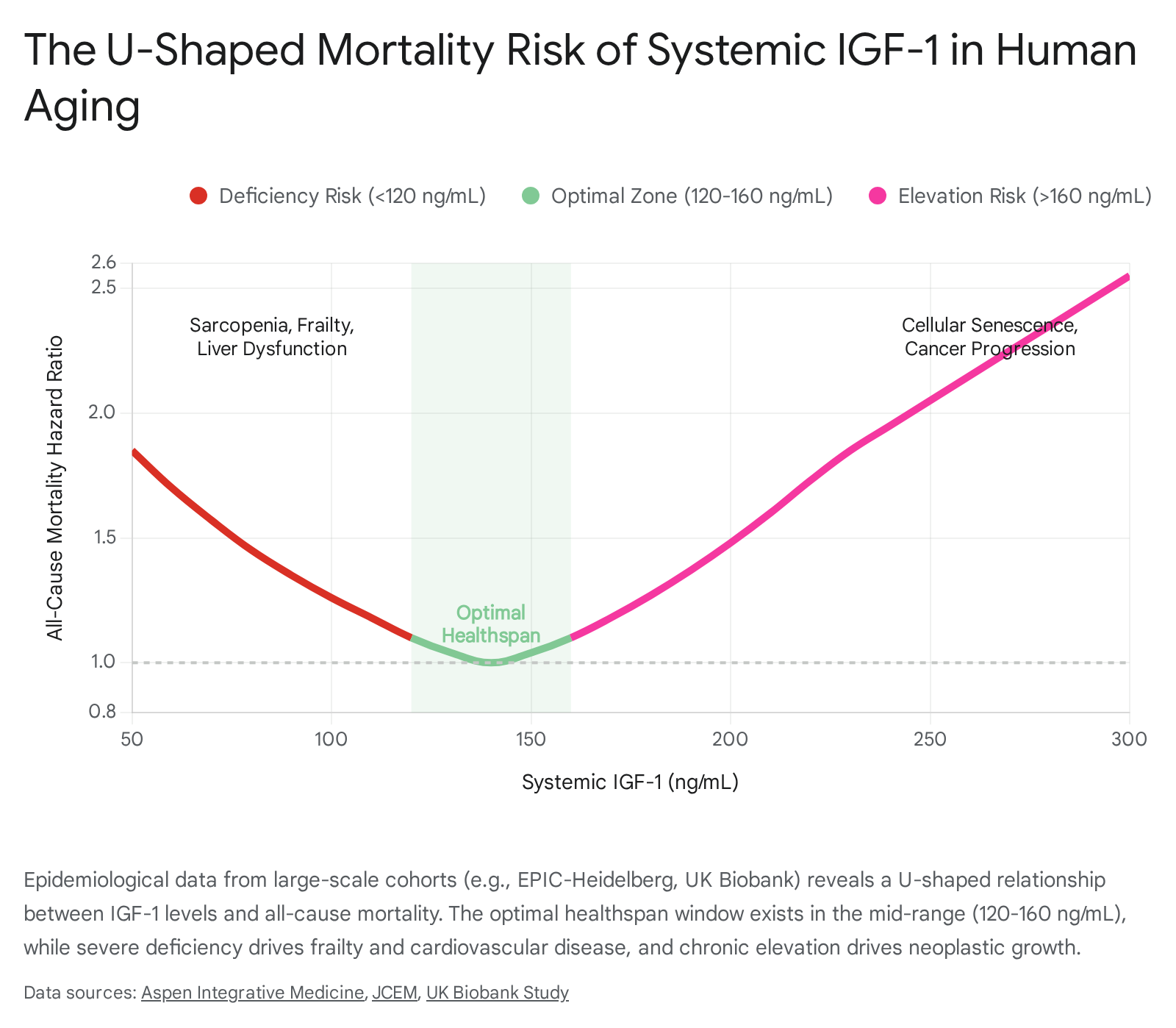
The Risks of the Upper Tail (High IGF-1): Chronic elevation of IGF-1 in adulthood acts as a potent, unabated mitogen. Elevated levels are directly correlated with accelerated cellular turnover, suppression of apoptosis, and a markedly increased incidence of neoplastic transformations, particularly in breast, prostate, and colorectal tissues 8847. Furthermore, high IGF-1 coupled with high-protein diets in middle-aged cohorts strongly predicts cancer-related mortality. For individuals aged 50 - 65, a high-protein diet was linked to a 75% higher overall mortality rate, reflecting the pathway's antagonistic pleiotropy where an environment rich in anabolic precursors actively overrides cellular maintenance 47.
The Risks of the Lower Tail (Severe IGF-1 Deficiency): Conversely, dropping to the lower extreme of the curve introduces catastrophic physiological failures, particularly in the elderly. Severe IGF-1 deficiency critically impairs the body's regenerative capacity. Older adults with steeply declining IGF-1 trajectories face drastically heightened risks of all-cause mortality, completely independent of gender 37. The clinical manifestations of this deficiency are severe and multi-systemic:
- Sarcopenia and Frailty: IGF-1 is mandatory for skeletal muscle protein synthesis, the prevention of proteolysis, and the activation of muscle satellite cells for repair. Severe deficits directly precipitate sarcopenia - the progressive, debilitating loss of muscle mass and physical function that cascades into falls, fractures, and devastating loss of independence 4950.
- Cardiovascular and Hepatic Failure: The liver is the primary endogenous producer of systemic endocrine IGF-1. Plunging IGF-1 levels are heavily implicated in the rapid progression of metabolic dysfunction-associated steatotic liver disease (MASLD) and decompensated cirrhosis 81226. Furthermore, cardiovascular mortality spikes in the lowest IGF-1 quintile due to failing endothelial repair mechanisms, deteriorating cardiomyocyte health, and the inability of the vascular system to withstand oxidative stress 8.
- Neurodegeneration: Within the central nervous system, both local and systemic IGF-1 provide vital neurotrophic support. Deficiencies accelerate the onset of cognitive decline and significantly exacerbate the pathology of Alzheimer's disease, as IGF-1 is required for the efficient clearance of amyloid-beta plaques, the maintenance of synaptic plasticity, and the promotion of neurogenesis 911.
Translational Geroscience: Pharmacological Modulators of the IGF-1/mTOR Axis (2024-2026)
Armed with the nuanced clinical understanding of the U-shaped IGF-1 mortality curve, the contemporary geroscience pipeline has pivoted dramatically. Rather than utilizing blunt genetic knockouts or severe, lifelong caloric restriction - which carry unacceptable adherence and physiological risks in humans - researchers are advancing sophisticated pharmacological modulators designed to finely tune the IGF-1/mTOR axis. The overarching objective is to compress morbidity and extend healthspan by gracefully shifting the cellular machinery from growth to maintenance, without ever plunging the patient into the devastating consequences of severe IGF-1 deficiency.
Targeting the Apex: mTORC1 vs. mTORC2 Selectivity
Rapamycin, an FDA-approved macrolide initially deployed as a potent immunosuppressant for organ transplant patients, remains the most robust pharmacological life-extending compound identified in biogerontology to date. It acts by directly binding to the intracellular protein FKBP12, forming a complex that allosterically inhibits the mTOR kinase 38. Extensive, rigorously controlled data from the National Institute on Aging's Interventions Testing Program (ITP) demonstrates that rapamycin significantly extends lifespan in diverse, genetically heterogeneous mouse strains 217.
However, the direct translation of chronic rapamycin therapy to human anti-aging protocols has been severely throttled by significant side effects, most notably "starvation diabetes" (characterized by hyperglycemia and severe peripheral insulin resistance) and hyperlipidemia 239. Recent mechanistic breakthroughs have illuminated that these detrimental metabolic effects are not caused by the inhibition of mTORC1 (the specific protein complex responsible for accelerated aging, protein synthesis, and suppressed autophagy). Rather, they are driven by the off-target disruption of a second complex, mTORC2. While mTORC1 is acutely and exquisitely sensitive to rapamycin, mTORC2 is structurally distinct - the presence of the RICTOR protein masks the rapamycin-binding domain 13. Therefore, acute rapamycin does not inhibit mTORC2. However, chronic or high-dose exposure over prolonged periods physically sequesters the entire cellular pool of free mTOR kinase, effectively starving the cell of the raw materials needed to assemble new mTORC2 complexes 1339. Because mTORC2 is the primary downstream effector of insulin signaling in the liver and skeletal muscle, its subsequent disruption induces severe systemic insulin resistance 1439.
Consequently, the 2024 - 2026 clinical landscape is heavily focused on achieving selective mTORC1 inhibition. Advanced dosing regimens - such as intermittent, pulsed, or exceedingly low-dose administration - are demonstrating clinical viability by effectively suppressing mTORC1 signaling while leaving mTORC2 physically and functionally intact 1339. Notably, a landmark 2025 pilot clinical trial published in the journal GeroScience demonstrated that just 8 weeks of low-dose rapamycin (1 mg daily) in older men yielded statistically significant improvements in diastolic heart function, enhanced transmitral blood flow, and broad endothelial health, all without incurring severe metabolic penalties 17. These rapid cardiovascular improvements suggest that mTORC1 inhibition targets root-cause functional decline. Furthermore, Phase 2 clinical trials evaluating the rapalog everolimus (e.g., NCT05835999) are currently underway, assessing the drug's ability to safely modulate aging biomarkers and reverse functional decline in older adults specifically enriched for metabolic risk 653.
Modulating the Receptor: Late-Stage Preclinical IGF-1R Inhibitors
Moving upstream from the mTOR complexes, scientists are actively investigating the direct pharmacological inhibition of the IGF-1 Receptor (IGF-1R) itself. This approach seeks to chemically mimic the profound longevity benefits observed in daf-2 mutant worms and heterozygous Igf1r+/- laboratory mice 423.
Recent late-stage preclinical evaluations of orally bioavailable, small-molecule IGF-1R inhibitors, such as picropodophyllin (PPP) and NVP-ADW742, have shown profound healthspan benefits in wild-type mice when administration begins in mid-life (13 months of age). These compounds successfully protected subjects against short-term memory decline, abolished the development of grey hair, rescued declining glucose tolerance, and reduced physical frailty parameters, such as declining grip strength 4055. Notably, while the overall maximum lifespan extension was statistically modest, the survival curve for the NVP-ADW742 cohort was significantly "squarer." A squarer survival curve indicates a massive compression of morbidity, effectively extending the disease-free healthspan by an average of 93 days in mice before a rapid end-of-life decline 4041. However, these specific small molecules exhibit significant toxicity profiles - such as severe gastrointestinal bleeding with PPP, and potential cardiotoxicity and brain bioaccumulation with NVP-ADW742 - that currently preclude their direct translation into human trials 1240.
A safer, highly promising alternative lies in the development of targeted monoclonal antibodies. Robust preclinical trials utilizing the IGF-1R monoclonal antibody L2-Cmu (developed by Amgen) administered to 18-month-old mice yielded a 9% increase in median lifespan, alongside highly significant reductions in systemic inflammation and overall neoplastic tumor burden 2122. Crucially, aligned with the known sex-specific dimorphism of the mammalian IIS pathway, these healthspan and lifespan benefits were realized almost exclusively in female mice. This dimorphism highlights the critical need for highly personalized, sex-aware precision medicine approaches as geroscience therapeutics advance toward the clinic 2122.
Combating the Lower Tail: Pharmacological Rescues for Sarcopenia
Because severe suppression of the IGF-1 axis induces catastrophic muscle wasting, the most critical translational challenge in applied geroscience is successfully preserving muscle mass while simultaneously suppressing systemic, pro-aging cellular pathways. To safely navigate this biological paradox, biotechnology firms are advancing targeted therapeutics specifically engineered to reverse sarcopenia and frailty without globally stimulating the IGF-1/mTOR axis.
BIO101 (Sarconeos): Developed by Biophytis, BIO101 is a highly innovative small molecule (20-hydroxyecdysone) that completely bypasses the traditional IGF-1/mTOR growth axis. Instead, it acts as a specific, non-peptide agonist of the Mas receptor. By activating this alternative receptor pathway, BIO101 triggers an independent anabolic cascade that promotes targeted muscle protein synthesis without elevating systemic cancer risks 4957. Following robust Phase 2b (SARA-INT) trial results in 2023 and 2024 - which demonstrated clinically meaningful improvements in gait speed via the 400-meter walk test (400MWT) and an excellent safety profile with no severe product-related adverse events - BIO101 is now formally poised to enter pivotal Phase 3 trials. These upcoming trials will target severe sarcopenic populations globally in 2025 and 2026, making it the most advanced sarcopenia therapeutic currently in development 49574243.
LPCN 1148: Lipocine's LPCN 1148 operates on a different, highly targeted mechanism. It is a novel oral prodrug of bioidentical testosterone functioning as a direct androgen receptor agonist. In December 2024, the US FDA granted LPCN 1148 Fast Track Designation specifically for the treatment of severe sarcopenia in patients suffering from decompensated cirrhosis 606162. Phase 2 clinical trials demonstrated that just 24 weeks of oral treatment induced a highly significant increase in the L3-skeletal muscle index (achieving a mean group difference of 4.4 cm2/m2 compared to placebo) 4445. Furthermore, the therapy significantly reduced the incidence of overt hepatic encephalopathy, providing a vital, life-saving intervention for patients whose endogenous liver-derived IGF-1 production has completely failed 44.
MYMD-1 (Isomyosamine): Recognizing that the SASP and chronic, low-grade systemic inflammation are the primary drivers of age-related muscle catabolism, TNF Pharmaceuticals is advancing MYMD-1. This compound is a first-in-class, orally administered, highly selective TNF-alpha inhibitor designed to regulate the immuno-metabolic system without causing broad, dangerous immunosuppression 5065. Following statistically significant Phase 2a results presented in December 2024 - which demonstrated robust, targeted decreases in key pro-inflammatory biomarkers (including TNF-α, IL-6, and sTNFR1) in elderly patients suffering from sarcopenia and frailty - the company is launching a fully funded Phase 2b study in early 2025 506566. Furthermore, moving aggressively into emerging markets, TNFA is expanding MYMD-1 trials in 2025 to actively combat the rapidly growing phenomenon of GLP-1-induced sarcopenia. This strategic move positions the drug as a critical adjunct therapy necessary to preserve lean muscle mass in patients undergoing rapid, pharmacologically induced weight loss via incretin mimetics .
Indirect Modulators: GLP-1 Agonists and SGLT2 Inhibitors as Gerotherapeutics
Finally, the boundary delineating traditional metabolic disease treatment from cutting-edge geroscience is blurring at an unprecedented rate. GLP-1 receptor agonists (such as semaglutide and tirzepatide) and Sodium-Glucose Cotransporter-2 (SGLT2) inhibitors, originally engineered strictly for the management of type 2 diabetes and obesity, are now demonstrating profound, multi-systemic pleiotropic geroprotective effects 464748. These classes of drugs indirectly, yet powerfully, modulate the IIS/mTOR axis by biochemically mimicking the physiological state of caloric restriction. They enhance metabolic flexibility, drastically reduce low-grade systemic inflammation (specifically downregulating the IL-6/NF-κB axis), and resensitize systemic insulin signaling without plunging circulating IGF-1 into dangerous, sarcopenia-inducing deficiency zones 1746.
Remarkably, a breakthrough 2025 human clinical study published in Cell Reports Medicine demonstrated that a 6-month intervention with an SGLT2 inhibitor induced a robust and statistically significant increase in actual telomere length in human subjects 17. This represents a fundamental, biological reversal of a primary, universally recognized hallmark of aging - an achievement previously thought impossible with currently available, FDA-approved pharmaceuticals 17. Consequently, leading gerontologists and medical panels are now formally proposing the widespread repurposing of these existing metabolic modulators as safe, first-in-class, broad-spectrum gerotherapeutics designed for prophylactic administration in aging populations 1747.
Conclusion
The pursuit of human longevity is no longer a speculative or fringe scientific endeavor; it is a meticulously mapped, biologically grounded pharmacological frontier. By framing the IGF-1/mTOR axis through the evolutionary lens of antagonistic pleiotropy, geroscience has successfully reconciled the pathway's indispensable role in youth with its destructive, senescence-driving hyper-anabolism in late life. The translational journey - from the simple daf-2 mutant nematode, surviving indefinitely in a suspended dauer state, to highly complex mammalian systems - has laid bare the staggering intricacies of human biology. This rigorous scientific progression confirms that the blunt, unrefined downregulation of growth pathways is not a viable clinical strategy, due entirely to the perilous U-shaped mortality curve of IGF-1, where severe deficiency is just as lethal as chronic hyper-activation.
The human genetic blueprint, specifically the geographically ubiquitous FOXO3a protective variants found in centenarians from Hawaii to Sardinia to Brazil, provides absolute proof that nuanced, downstream modulation of this metabolic pathway can yield exceptional human longevity, free from the crushing morbidities of heart disease and metabolic failure. As we navigate the rapidly evolving 2024 - 2026 clinical landscape, the era of true precision geroscience has firmly arrived. Through the highly selective inhibition of mTORC1, the advent of specialized IGF-1R monoclonal antibodies, and the deployment of targeted, non-IGF-dependent anabolics to rescue sarcopenic muscle, the medical community is moving closer to its ultimate, overarching objective: not merely extending the maximum human lifespan, but profoundly and equitably expanding the boundaries of the disease-free human healthspan.