Impact of Early Adversity on Brain Development
The architecture of the developing human brain is fundamentally shaped by continuous interactions between genetic predispositions and environmental inputs. Historically, the prevailing clinical consensus posited that adverse childhood experiences - ranging from severe emotional neglect to physical trauma - resulted in irreversible neural damage. Contemporary developmental neuroscience, however, has fundamentally revised this paradigm. Evidence increasingly demonstrates that the developing brain does not merely sustain damage from early adversity; rather, it actively adapts its structural and functional organization to maximize survival within a hostile or unpredictable environment 1234. While these neurobiological adaptations are highly advantageous for immediate short-term survival, they establish complex developmental cascades that significantly increase vulnerability to cognitive deficits, emotional dysregulation, and severe psychiatric disorders later in life 567.
The study of early life stress encompasses multiple neurobiological domains, including macroscopic changes in regional brain volume, microstructural alterations in white matter integrity, shifts in the functional connectivity of large-scale neural networks, and epigenetic modifications at the cellular level. This report provides an exhaustive analysis of the mechanisms through which early adversity alters the developing brain. It examines the foundational mechanisms of neuroplasticity, the distinct neural signatures of different adversity typologies, the critical role of sensitive periods, the framework of biological sensitivity to context, and the robust emerging evidence demonstrating that targeted behavioral and environmental interventions can successfully reverse early neurodevelopmental alterations.
Foundational Mechanisms of Neuroplasticity
To contextualize how adversity alters neural architecture, it is necessary to examine the underlying mechanisms of neuroplasticity. Neuroplasticity refers to the inherent capacity of neural circuits to change, reorganize, and adapt in response to internal biological signals or external environmental stimuli 8910. This process is highly metabolically demanding and varies significantly across the lifespan and across different regions of the brain.
Cellular Maturation and Synaptic Pruning
Brain development begins with a massive overproduction of neurons and synapses during the perinatal period and the first years of life. This initial overproduction is subsequently followed by synaptic pruning, a highly selective process wherein frequently activated neural connections are strengthened and stabilized, while unused connections are eliminated 41112. This competitive elimination fine-tunes neural networks, making the brain more efficient and adapted to the specific demands of its surrounding environment.
At the cellular and molecular level, the onset and closure of plasticity windows are regulated by the excitatory/inhibitory (E/I) balance within local cortical circuits. Typical synaptogenesis relies on a delicate equilibrium between excitatory glutamatergic signaling and inhibitory gamma-aminobutyric acid (GABA) signaling 1213. The maturation of specific inhibitory interneurons, notably those expressing parvalbumin, triggers a sequence of molecular events that stabilize local networks 1314.
A critical mechanism in putting the "brakes" on plasticity is the formation of perineuronal nets (PNNs). PNNs are specialized extracellular matrix structures that condense around the cell bodies and proximal dendrites of parvalbumin-positive interneurons 1214. By physically restricting the formation of new synaptic connections and buffering local ion concentrations, PNNs effectively close the window of heightened neuroplasticity, solidifying the network's architecture 12.
Experience-Expectant versus Experience-Dependent Processes
The brain's structural development is driven by two distinct but overlapping mechanisms: experience-expectant and experience-dependent plasticity. Experience-expectant development dictates that the brain requires certain universal environmental signals - such as visual input, language exposure, and basic caregiver nurturance - at specific developmental stages to mature normally 412. If these expected inputs are absent due to severe deprivation or sensory deficits, the corresponding brain regions undergo profound reorganization. For instance, in individuals with early blindness, the occipital cortex, typically dedicated to visual processing, undergoes cross-modal plasticity to process tactile and auditory information 15.
Conversely, experience-dependent plasticity refers to neural changes driven by idiosyncratic, individual-specific experiences that occur throughout the lifespan. This form of plasticity is responsible for learning specific skills or forming discrete memories 12. Early adverse rearing conditions fundamentally represent violations in expected caregiving input, thereby disrupting experience-expectant development and forcing the brain to adapt its fundamental architecture to an atypical, high-stress baseline 47.
The Dimensional Model of Early Adversity
Historically, researchers and clinicians utilized cumulative risk models, measuring adversity by simply tallying the number of adverse childhood experiences (ACEs) an individual endured. This approach treated highly distinct experiences - such as physical abuse, extreme poverty, and emotional neglect - as additive but fundamentally equivalent stressors 1617. Recent neuroimaging meta-analyses have largely abandoned cumulative risk in favor of the Dimensional Model of Adversity. This framework disaggregates adverse experiences into distinct typologies, demonstrating that different forms of adversity exert highly specific, differential effects on neural architecture 171819.
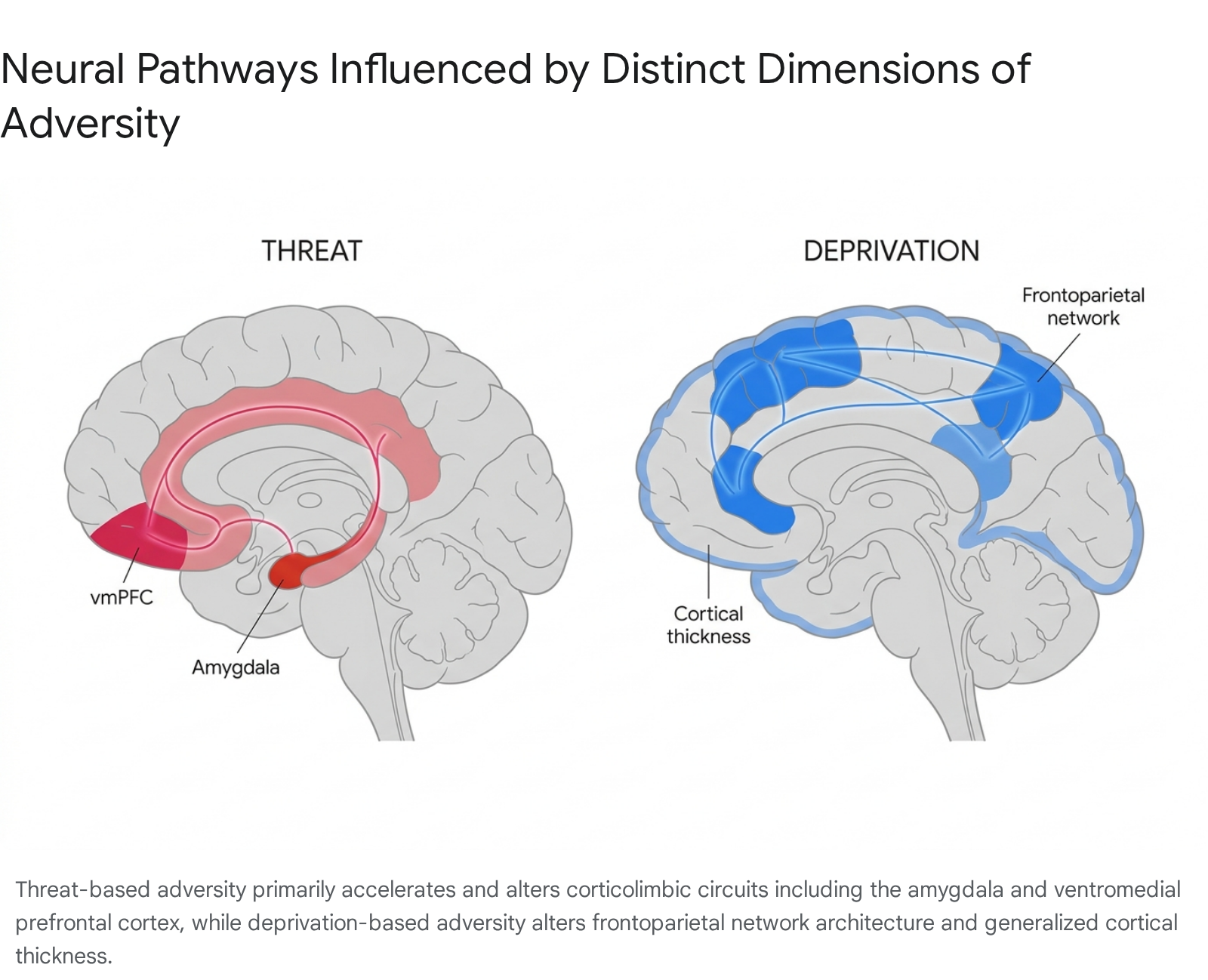
Threat and Corticolimbic Circuitry
The dimension of threat is defined as experiences involving actual or threatened harm to the physical integrity of the child, encompassing physical abuse, sexual abuse, and chronic exposure to domestic or community violence 1718. Threat represents a direct challenge to survival, prompting an immediate and profound neurobiological adaptation within the brain's emotional appraisal and stress-response systems.
Research consistently demonstrates that exposure to threat is associated with specific structural and functional alterations in corticolimbic circuitry. Structurally, threat exposure correlates with widespread reductions in cortical surface area across the prefrontal cortex, as well as distinct reductions in the thickness of the ventromedial prefrontal cortex (vmPFC) 16171920. The vmPFC is crucial for the top-down regulation of emotional responses and the extinction of fear. Reductions in its structural integrity significantly impair an individual's capacity to modulate emotional arousal 1619.
Functionally, threat exposure is robustly associated with heightened reactivity in the amygdala, the brain's primary threat-detection hub. Meta-analyses of fMRI studies reveal that individuals exposed to severe early threat demonstrate significantly higher amygdala activation in response to negative or fearful stimuli compared to controls 161820. This hyper-reactivity operates in tandem with lower prefrontal cortical reactivity, resulting in an impaired regulatory circuit that heavily predisposes individuals to anxiety, post-traumatic stress disorder (PTSD), and other internalizing disorders 51821.
Deprivation and Frontoparietal Networks
Deprivation is characterized by the absence of expected cognitive, social, and emotional environmental inputs. It is most starkly observed in cases of institutionalized rearing, profound emotional neglect, and extreme material poverty 16171819. Unlike threat, which actively stimulates the nervous system with noxious input, deprivation represents an environmental void that disrupts experience-expectant synaptogenesis.
Deprivation primarily manifests structurally within the frontoparietal networks and sensory association areas. While threat is associated with reduced cortical surface area, deprivation is frequently associated with anomalous increases in cortical thickness, particularly within the occipital cortex, insula, and cingulate 171920. Furthermore, studies of children raised in profoundly depriving institutional environments demonstrate reduced overall gray matter volume and altered development of the corpus callosum 192223.
Functionally, the neural signatures of deprivation are distinct from threat. While threat reliably induces heightened amygdala reactivity across various task domains, deprivation does not consistently produce this hyper-reactivity unless specifically tested with fearful stimuli lacking appropriate perceptual controls 1618. Instead, deprivation typically impairs higher-order cognitive processing, working memory, and language acquisition, reflecting the stymied development of associative cortical networks 2425.
Unpredictability as an Emerging Dimension
Building upon the dichotomy of threat and deprivation, contemporary research has identified unpredictability as a crucial third dimension of early life adversity. Unpredictability involves frequent, stochastic changes in the caregiving environment, physical location, or parental emotional availability 192326. Exposure to chronic unpredictability alters the fundamental economic constraints that govern the formation of the structural connectome 26.
Computational modeling of neural network formation reveals that unpredictable postnatal stress increases the stochasticity of structural brain development 26. The brain trades the wiring cost of long-distance neural connections for localized topological homophily, resulting in a network that is highly robust against sudden external perturbations but less efficient at integrated, complex cognitive processing 26. Clinical evaluations further indicate that unpredictability, specifically measured through metrics like "maternal mood entropy," sets the stage for distinct neurodevelopmental trajectories linked to stress sensitivity and psychiatric symptomology independent of direct abuse or neglect 28.
| Adversity Dimension | Phenomenological Definition | Key Structural Signatures | Key Functional Signatures |
|---|---|---|---|
| Threat | Presence of harm or threat of harm (e.g., physical/sexual abuse, violence). | Reduced vmPFC volume/thickness; Widespread cortical surface area reduction. | Heightened amygdala reactivity to negative stimuli; Reduced prefrontal regulatory activity. |
| Deprivation | Absence of expected input (e.g., profound neglect, institutionalization). | Increased cortical thickness (occipital, insula, cingulate); Reduced overall gray matter. | Altered frontoparietal activation; Distinct deficits in working memory and executive function. |
| Unpredictability | Frequent, stochastic changes in environment or caregiver mood. | Increased randomness in structural connectome formation. | Heightened stress sensitivity; Altered economic constraints on neural network wiring. |
Macroscopic Structural and Functional Alterations
The specific neurobiological consequences of early adversity heavily dictate the long-term cognitive and emotional capabilities of the individual. Analyzing alterations at the macroscopic level - encompassing subcortical volumes, white matter integrity, and large-scale functional networks - provides critical insight into the mechanisms of developmental psychopathology.
Subcortical Volume Variations
The hippocampus, a seahorse-shaped structure critical for episodic memory consolidation and contextualizing emotional responses, is exceptionally vulnerable to early adversity. This vulnerability arises because the hippocampus contains a remarkably high density of glucocorticoid receptors, rendering it acutely sensitive to the neurotoxic effects of chronically elevated cortisol resulting from toxic stress 2127. Sustained exposure to glucocorticoids severely reduces neurogenesis and dendritic arborization within the developing hippocampus 27. Volumetric MRI studies consistently demonstrate reduced bilateral hippocampal volume in adolescents and adults who experienced early adversity 1132127. This structural atrophy is directly implicated in difficulties with declarative memory retrieval and the contextual modulation of fear responses 1321.
The amygdala's structural response to adversity is more complex, exhibiting patterns of both hypertrophy and atrophy depending on the timing of the assessment and the nature of the trauma. In normative development, amygdala volume increases gradually. However, extreme early stress forces an accelerated adaptation. Some preclinical and human studies demonstrate that early adversity results in basolateral amygdala hypertrophy (enlargement) as the brain upregulates its threat-detection capacity to survive a hostile environment 12128. Conversely, large-scale meta-analyses and data from patients with trauma-related psychopathology, such as severe PTSD, often reveal significant reductions in bilateral amygdala volume 212829. This paradox may be explained by the timeline of toxic stress: initial structural hypertrophy to manage immediate threats may eventually succumb to excitotoxicity and volumetric loss following decades of chronic hyperactivation.
Prefrontal Cortex and Executive Function
The prefrontal cortex (PFC), specifically the ventromedial prefrontal cortex (vmPFC) and the anterior cingulate cortex (ACC), is responsible for higher-order executive functions, impulse control, and the critical top-down inhibition of the amygdala. The structural integrity of these regions is compromised following childhood trauma, often presenting as decreased gray matter volume and reduced cortical thickness 16192130.
Because the PFC heavily modulates the affect regulation center, damage to the ACC directly results in severe difficulties with emotion regulation, leading to heightened emotional outbursts and maladaptive coping mechanisms 321. Structurally, functional imaging of adversity-exposed adolescents demonstrates lower prefrontal cortical reactivity during emotional tasks, proving an impaired capacity to apply logical, executive control over an overactive limbic system 31821.
White Matter Integrity and Structural Connectivity
Beyond isolated brain regions, the physical communication infrastructure of the brain - the white matter tracts - is profoundly impacted by early trauma. Toxic stress initiates a severe biological cascade beginning with the immune system. When a child's environment is chronically unstable or abusive, the immune system remains permanently activated, overproducing inflammatory molecules 233. Over time, these elevated inflammatory markers compromise the blood-brain barrier, allowing cytokines to cross into the brain and induce chronic neuroinflammation 2.
This inflammation actively disrupts the myelination process and degrades existing white matter integrity. Diffusion tensor imaging (DTI) reveals that individuals with histories of early life adversity exhibit reduced fractional anisotropy - a measure of white matter structural coherence - indicating a less efficient internal communication system 230. Reductions in the quality and quantity of white matter communication tracts directly correlate with measurable deficits in cognitive performance, specifically in receptive language processing, vocabulary development, and mental arithmetic 2431.
Large-Scale Functional Network Dynamics
At the level of global brain dynamics, adversity visibly disrupts resting-state functional connectivity (rs-FC) within large-scale neural networks. The brain's operations are largely governed by the interplay of the "Triple Brain Network," comprising the Default Mode Network (DMN), the Salience Network (SN), and the Central Executive Network (CEN).
The SN, anchored by the insula and dorsal ACC, integrates sensory and emotional information to detect relevant environmental stimuli. The CEN manages cognitive control and working memory, while the DMN typically activates during introspective, internally directed thought 3236. Meta-analyses of rs-FC data indicate that individuals exposed to adverse childhood experiences present a consistent pattern of hypoconnectivity (weakened neural coordination) within the SN, and critically, between the SN and the CEN 3236.
This uncoupling between salience detection and executive control creates significant cognitive vulnerabilities. When the CEN cannot effectively communicate with the SN, the individual struggles to assert top-down control over threat-related signals, leading to the clinical manifestations of anxiety, hypervigilance, and difficulty suppressing repetitive negative thoughts 36. Furthermore, hyper-flexibility or "supra-optimal" reconfiguration within these networks during executive tasks - a lack of stable network segregation - is identified as a maladaptive consequence of childhood adversity that persists into mid-life 33.
The specific neural outcomes of adversity also inform the differential etiologies of distinct psychiatric disorders. Structural MRI analyses demonstrate that adverse childhood experiences correlate with significant cortical thinning in emotion-regulating regions among patients with Borderline Personality Disorder (BPD), whereas patients with Major Depressive Disorder (MDD) exhibit general cortical thinning that is not specifically modulated by the severity of early adversity, suggesting different mechanistic pathways for trauma embedding 34.
Sensitive Periods in Neurodevelopment
The precise neurobiological consequences of early adversity are heavily dependent on the exact developmental timing of the exposure. The brain does not develop uniformly; rather, different regions and circuits mature according to specific biological timetables 10. "Sensitive periods" are distinct time windows during which a specific brain circuit exhibits heightened neuroplasticity, rendering it highly malleable to environmental influences 1112153536.
During a sensitive period, environmental experiences have an exceptionally strong impact on neural organization. If the environment provides traumatic, adverse, or deprived inputs during these windows, the resultant structural and functional alterations become deeply embedded 1115. The literature delineates two primary epochs of heightened sensitivity to stress: early childhood and adolescence.
Infancy and Early Childhood Vulnerabilities
The first five years of life encompass a massive wave of synaptic overproduction and subsequent pruning 4. The hippocampus, which undergoes rapid and complex structural changes during this time, appears governed by a distinct sensitive period. Longitudinal neuroimaging assessments demonstrate that the severity of stressful experiences occurring strictly between birth and age five is significantly associated with reduced bilateral hippocampal volume later in adolescence. Notably, equivalent levels of stress experienced after age six yield no such structural association, explicitly defining the temporal bounds of this vulnerability 2827.
Animal models provide the mechanistic underpinning for this hippocampal sensitive period. In mice, the emergence of episodic-like memory precision occurs alongside the formation of perineuronal nets (PNNs) within the dorsal CA1 subfield of the hippocampus 14. Subjecting the developing organism to early life adversity significantly decelerates the maturation of these CA1 PNNs, delaying the onset of precise memory formation and cementing enduring anxiety-like behavioral phenotypes 14. This establishes that the timing of episodic memory development is not rigidly hard-wired but is flexibly set by the quality of the environment during this early sensitive window.
Adolescence as a Secondary Window of Plasticity
Adolescence represents a second major sensitive period, characterized by profound synaptic pruning, extensive white matter myelination, and the critical refinement of prefrontal-subcortical circuitry 6123536. While basic sensory-motor regions lose their high plasticity early in childhood, the associative regions of the brain - those supporting high-level cognitive, social, and emotional functions - remain structurally malleable well into the mid-twenties 1012.
Because the prefrontal cortex does not achieve full myelination until early adulthood, the neural circuits connecting the PFC to the amygdala remain highly sensitive to environmental input throughout adolescence 41213. This developmental timetable explains a crucial clinical phenomenon: the delayed emergence of stress-related psychopathology. Children exposed to severe early trauma may not exhibit full-blown depressive or anxiety disorders immediately. Instead, the pathology often surfaces during early adolescence. This delay occurs because the dysfunction embedded by early abuse is "unmasked" only when the prefrontal-amygdala regulatory circuits actively attempt to integrate and functionalize during the adolescent maturation phase 1213.
Stress Acceleration and Premature Maturation
When severe stress occurs during a sensitive period, the developing brain frequently engages in a dramatic adaptive response known as the Stress Acceleration Model 1116. Chronic adversity signals to the biological system that the environment is dangerous and survival is uncertain, prompting a truncation of normative childhood and an accelerated maturation of the organism's stress-response systems 11.
At the neural level, stress acceleration manifests as the premature structural and functional maturation of amygdala-mPFC connectivity. In healthy development, children exhibit positive functional connectivity between the amygdala and the mPFC, which slowly transitions to a mature, inhibitory (inverse) connection during adolescence 37. Institutionalized children and those exposed to extreme early life stress display this adult-like inverse connectivity prematurely 1937.
This premature closure of the neural sensitive period is a biological trade-off. It prioritizes immediate survival, rapid threat detection, and early independence at the severe cost of long-term neuroplasticity, memory retention, and emotional regulation 1137. At the cellular level, this stress acceleration correlates with biological aging, evidenced by the accelerated erosion of telomeres - the protective chromosomal caps necessary for healthy DNA replication - leading to long-term health risks and premature mortality 111.
Biological Sensitivity and Epigenetic Embedding
A central question in developmental neuroscience is why a subset of children exposed to severe early adversity develops profound neurobiological deficits, while others exhibit remarkable resilience. The divergence in outcomes is explained by the theory of Biological Sensitivity to Context (BSC) and the highly dynamic mechanisms of epigenetics 3839444046.
Differential Susceptibility Framework
Traditional diathesis-stress hypotheses posited that high biological stress reactivity was an inherent vulnerability, strictly leading to negative health outcomes in the face of adversity 39. The BSC theory fundamentally revises this view through the lens of evolutionary biology and Differential Susceptibility. BSC theory proposes that natural selection favored developmental mechanisms capable of adjusting an individual's neurobiological responsivity to match the precise socioecological conditions encountered during early life 38444046.
Empirical data reveals a non-linear, U-shaped relationship between the quality of the childhood environment and the magnitude of physiological stress reactivity 4046.
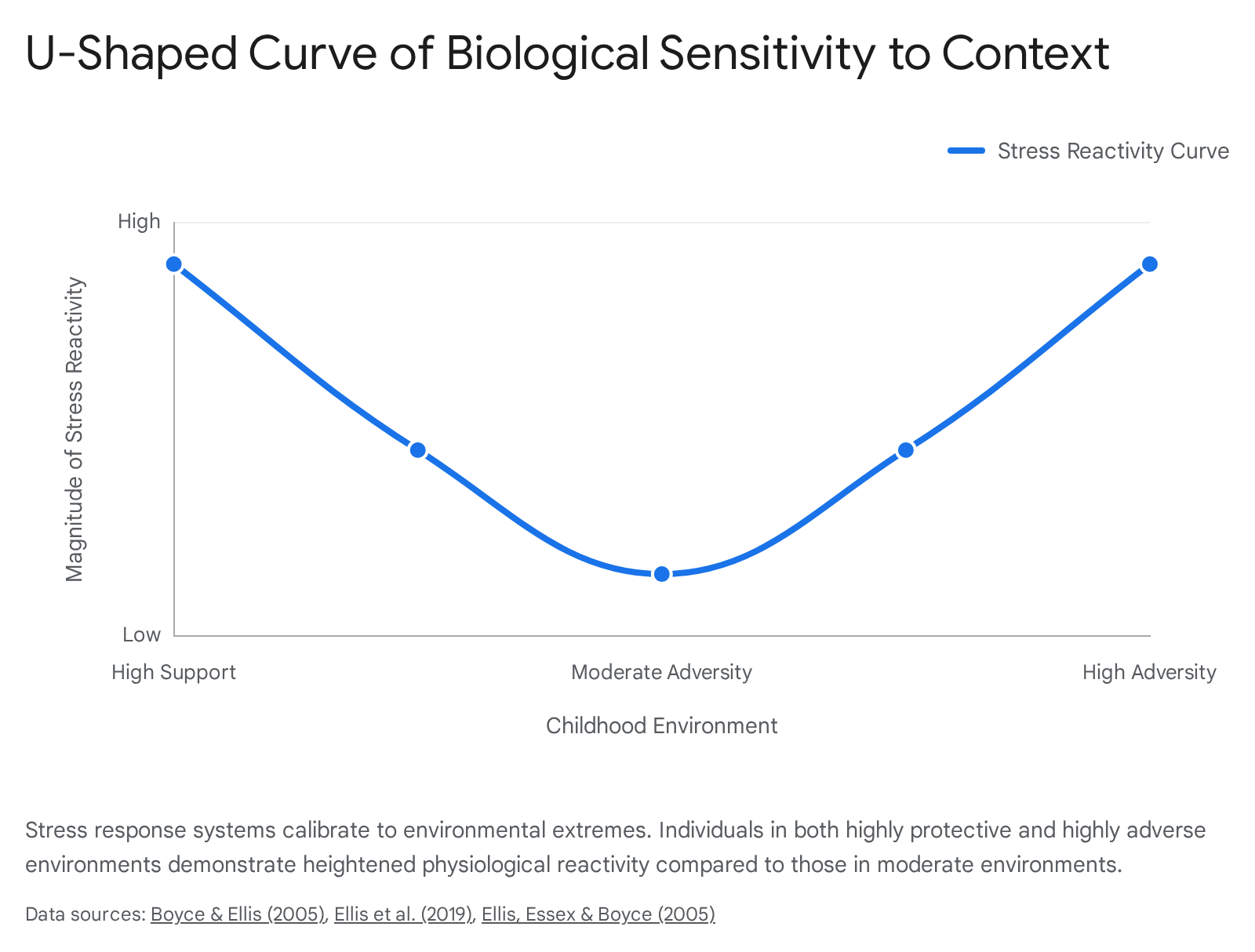
Children reared in moderately stressful or average environments develop low physiological reactivity, resulting in a generally buffered phenotype. However, children reared in environments at either extreme - highly adverse/threatening or highly supportive/enriched - develop heightened physiological reactivity 4440.
For the child in an adverse environment, heightened reactivity manifests as a "vigilant" pattern, maximizing the detection of threats necessary for survival. For the child in a highly supportive environment, heightened reactivity manifests as a "sensitive" pattern, maximizing their susceptibility to the abundant social cues and educational opportunities available 40. Therefore, high biological sensitivity is a bidirectional trait: it is severely maladaptive in toxic contexts, leading to psychopathology, but highly advantageous in nurturing contexts, leading to exceptional outcomes 3839.
Genetic Polymorphisms and Gene-Environment Interactions
The calibration of this biological sensitivity is heavily mediated by gene-by-environment (GxE) interactions. Inherited genetic variations do not encode trauma itself; rather, they shape traits such as baseline sensitivity to threat and emotional regulation capacity 47.
Genetic association studies indicate that the long-lasting alterations to the hypothalamic-pituitary-adrenal (HPA) axis are mediated in part by specific polymorphisms within the CRHR1 and FKBP5 genes 41. When individuals possessing these specific biological risk alleles are exposed to childhood abuse, the combination of elevated stress-dependent cortisol and genetic vulnerability permanently alters the development of amygdala-dependent emotional circuits. This interactive process creates an adult neural network that is inherently primed for stress responsiveness and unable to appropriately differentiate safe versus threatening stimuli 41. Notably, heritability accounts for roughly 30% to 50% of the variance contributing to stress resilience and the risk for subsequent mood disorders, emphasizing the equal weight of environmental exposure 4142.
Epigenetic Modifications and DNA Methylation
The specific mechanism by which environmental trauma embeds itself biologically is through epigenetics. Epigenetics involves chemical modifications to the DNA structure - such as methylation - that alter the expression of genes (turning them "on" or "off") without fundamentally changing the underlying genetic sequence 434452.
Adverse experiences induce widespread epigenetic changes at genome sites responsible for modulating the physiological stress response. For example, severe early neglect can lead to chemical modifications that silence the genes required to turn off the HPA axis stress response, leaving the individual physically incapable of absorbing stress in a healthy manner 44. The timing of these epigenetic modifications is critical. Research measuring DNA methylation (DNAm) across multiple time points found that children exposed to adversity specifically between the ages of 3 and 5 exhibited the most dramatic differences in DNAm levels by age 15, confirming the preschool period as a highly sensitive biological window for epigenetic embedding 43.
However, the field of epigenetics provides a robust framework for optimism. Epigenetic markers function dynamically, responding continuously to changing environments throughout the lifespan 474352. The biological imprint of early stress is not an immutable scar; subsequent experiences of family stability, social support, and targeted therapeutic interventions can successfully remodel these epigenetic patterns, reversing the neurobiological damage 474452.
Neurobiological Reversibility and Intervention Models
A pervasive and damaging misconception regarding early trauma is the assumption that it induces permanent, irreversible brain damage 124554. Modern neuroscientific consensus, driven by large-scale longitudinal intervention studies, definitively proves that the same mechanisms of neuroplasticity that allow adversity to alter the brain can be successfully harnessed to repair it 92246. Because the brain retains significant adaptive plasticity - particularly during the secondary sensitive window of adolescence - targeted environmental shifts and clinical interventions can effectively reverse structural and functional deficits.
Environmental Shifts and the Bucharest Early Intervention Project
The most compelling causal evidence regarding the reversibility of profound early deprivation is derived from the Bucharest Early Intervention Project (BEIP). Initiated in 2001, the BEIP is a landmark randomized controlled trial involving 136 infants raised in severely depriving Romanian institutions under the Ceaușescu regime 47484950. Researchers randomized the children either to remain in institutional care (care-as-usual) or to transition into newly established, high-quality family foster care 49.
Across nearly two decades of follow-up spanning from 30 months to 18 years of age, the BEIP demonstrated massive developmental recovery resulting from the environmental intervention 49. By age 18, individuals randomized to the foster care intervention achieved IQ scores an average of 9 points higher than those assigned to institutional care, driven by specific recoveries in verbal comprehension and processing speed 2550. Furthermore, children in foster care exhibited significantly lower rates of reactive attachment disorder, internalizing symptoms, and ADHD compared to the care-as-usual cohort 74749.
The behavioral recovery was matched by profound neurobiological normalization. Utilizing resting-state electroencephalogram (EEG) assessments at age 16, researchers found that children remaining in institutions continued to exhibit an immature pattern of brain electrical activity, characterized by high theta power and low alpha power 51. Conversely, children removed from institutions and placed into foster care normalized their EEG activity, matching the power frequency patterns of community controls who had never been institutionalized 51.
Crucially, the BEIP identified clear parameters for success based on sensitive periods and placement stability. Children placed into foster care before the age of 24 months achieved the greatest degree of cognitive and neurobiological recovery 5052. Additionally, the protective neural and behavioral effects persisted into adolescence only for those children who maintained stable, undisrupted foster placements; children experiencing placement disruptions showed neurological regressions resembling the institutionalized cohort 74951.
Targeted Behavioral Therapeutics: ABC and PCIT
Beyond broad environmental shifts, highly specific, time-limited therapeutic interventions demonstrate the capacity to rewire brain networks altered by adversity.
Attachment and Biobehavioral Catch-up (ABC): ABC is a 10-session home-visiting intervention targeted at parents and foster caregivers of infants (birth to 48 months) who have experienced maltreatment or disruptions in care. Based on attachment theory and stress neurobiology, ABC trains caregivers to consistently follow the child's lead, provide nurturing physical contact during distress, and avoid harsh or frightening behaviors 53545556. The biological results are striking: ABC significantly normalizes the child's diurnal cortisol production rhythm, with effects sustained years post-intervention 545657. Furthermore, ABC significantly reduces disorganized attachment, achieving secure attachment rates of 52% among treated children compared to only 33% in control groups 5455. Notably, the intervention physically alters the parent's brain; Event-Related Potential (ERP) imaging demonstrates that caregivers receiving ABC develop larger neural responses (N170 and LPP components) to their children's emotional facial expressions, fundamentally enhancing biobehavioral co-regulation 5357.
Parent-Child Interaction Therapy (PCIT): PCIT is a behavioral parent training program uniquely successful in interrupting coercive parent-child interactions and drastically reducing child maltreatment recidivism 5859. Modern neuroimaging confirms that PCIT directly improves the neurological markers of self-regulation in children who have suffered early trauma. Electroencephalogram (EEG) analyses demonstrate that adversity-exposed children (ages 3 - 8) who complete PCIT exhibit significant reductions in the theta/beta ratio, serving as a direct neural marker of enhanced attention regulation 58. In caregivers, PCIT participation produces measurable gains in inhibitory control on cognitive tasks and significantly reduces self-reported parental depression and anxiety, proving that modifying the caregiving environment actively buffers and repairs both generations' neural architecture 5960.
Physical Activity and Mindfulness Interventions
Adults carrying the biological imprint of childhood trauma can also utilize targeted lifestyle and psychological interventions to reverse structural anomalies. Sustained, lifelong physical activity (e.g., swimming, cycling) has been empirically shown to alter neuroplasticity and rework the connective wiring between the amygdala, hippocampus, and anterior cingulate cortex, effectively reversing the weak stress-circuit connectivity laid down by early adversity 54.
Similarly, mindfulness-based interventions and exposure-focused cognitive behavioral therapy (EF-CBT) exhibit targeted neurological repair. EF-CBT applied to clinically anxious youth selectively normalizes the functional connectivity density between the Central Executive Network (CEN) and the Salience Network (SN), physically strengthening the brain's top-down control over threat-related signals 3661. At the cellular level, meditation-based practices demonstrate the power to restore telomere length and actively reverse the basolateral amygdala hypertrophy induced by early life stress, providing concrete evidence that trauma-related brain changes are reversible through systematic mental training 12228.
| Intervention Model | Target Population | Key Neurobiological & Physiological Outcomes | Reversibility Mechanism |
|---|---|---|---|
| Bucharest Early Intervention Project (BEIP) | Institutionalized infants | Reversal of IQ deficits; Normalization of EEG (alpha/theta power); Improved white matter integrity. | Removal from deprivation into enriched family environments before age 2 years. |
| Attachment & Biobehavioral Catch-up (ABC) | Maltreated/Foster infants | Normalized diurnal cortisol rhythms; Altered parent ERP responses; Amygdala-PFC circuitry regulation. | Enhancing caregiver sensitivity, responsiveness, and biobehavioral co-regulation. |
| Parent-Child Interaction Therapy (PCIT) | Maltreated children (Ages 3-8) | Reduction in EEG theta/beta ratio (improved attention regulation); Improved parent inhibitory control. | Interrupting coercive parent-child interactions and strengthening positive reinforcement. |
| Exposure-Focused CBT & Mindfulness | Trauma-exposed youth and adults | Normalized CEN-SN network connectivity; Reversal of amygdala hypertrophy; Telomere restoration. | Systematic mental training, neuroplasticity engagement, and top-down cognitive restructuring. |
Cross-Cultural Variances in Neuroimaging Research
While the neurobiological mechanisms linking early adversity to changes in brain architecture are overwhelmingly robust, the field of developmental neuroscience is currently constrained by severe geographical and cultural representation disparities.
Representation Disparities in Global Databases
The vast majority of foundational structural and functional MRI data concerning childhood adversity originates exclusively from Western, Educated, Industrialized, Rich, and Democratic (WEIRD) societies 716263. Systemic analyses of major global neuroimaging databases, such as the ENIGMA Consortium and the Brain Charts Consortium, reveal extreme disparities: between 90% and 95.4% of all structural MRI scans utilized in psychiatric and neurological research originate from High-Income Countries (HICs), overwhelmingly concentrated in North America and Europe 6263.
Low-income countries contribute functionally zero structural MRI data to these major international consortiums, leaving the populations of Africa, Latin America, Southeast Asia, and the Eastern Mediterranean critically underrepresented 7162. This data exclusion is highly problematic because the baseline exposure to systemic adversity varies radically across global populations. Cohort studies analyzing Adverse Childhood Experiences (ACEs) in non-Western contexts - such as the FAMELO project assessing populations in Mexico, Mozambique, and Nepal - reveal significantly higher average rates of ACE exposure (e.g., an average of 2.7 ACEs per adolescent in Mozambique) driven by compounded socioeconomic vulnerabilities, severe food insecurity, and political instability 6465.
Universal Mechanisms versus Culturally Specific Adaptations
When neuroimaging research is successfully conducted in low- and middle-income countries, the core biological principles of adversity embedding appear universally conserved, though cultural contexts significantly shift the developmental parameters. For example, a study utilizing portable functional Near-Infrared Spectroscopy (fNIRS) to measure visual working memory in infants in rural India successfully replicated the exact prefrontal cortex activation patterns observed in Midwest US cohorts 66. This confirms that structural stressors like severe poverty and low maternal education universally disrupt canonical working memory networks 66. Similarly, large-scale Voxel-Based Morphometry (VBM) studies assessing brain development in Chinese children (ages 7 to 23) demonstrate identical age-related trajectories in gray and white matter to Western cohorts, and Mendelian randomization utilizing the UK Biobank confirms causal links between specific adversity traits and cortical thickness globally 636768.
However, severe methodological errors arise when Western clinical definitions of "adverse" parenting are applied blindly to non-Western data. Cross-cultural observational studies comparing mother-infant dyads in the UK and India demonstrate that Indian mothers utilize substantially more instructional, directing, and controlling comments during play compared to UK mothers 69. In a Western clinical paradigm, this high level of instruction might be mischaracterized as "intrusive" or indicative of a lack of maternal sensitivity. In the Indian cultural context, however, these practices are deeply rooted in cultural values prioritizing interdependence, obedience, and parental guidance, and they do not inherently trigger the neurobiological stress cascades associated with true emotional deprivation or threat 69. Future developmental neuroscience research must integrate diverse socio-ecological variables and localized definitions of normative caregiving to accurately distinguish universal neurobiological trauma responses from adaptive, culturally specific developmental pathways 716269.
Conclusions
The consensus of modern developmental neuroscience is that the developing brain does not passively suffer irreversible damage from early adversity; rather, it engages in an active, highly plastic process of structural and functional adaptation designed to maximize survival in a hostile environment. By disaggregating adverse experiences into distinct dimensions of threat, deprivation, and unpredictability, researchers have mapped precise neurobiological signatures. Threat-based environments accelerate and alter corticolimbic circuitry - specifically the amygdala, hippocampus, and vmPFC - to prioritize rapid threat detection. Conversely, deprivation-based environments lead to anomalous cortical thickening and deficits in frontoparietal networks due to the severe absence of expected environmental, cognitive, and social input.
The severity and permanence of these neural alterations are dictated by the developmental timing of the exposure. The brain exhibits profound, specialized vulnerability during the sensitive periods of infancy and early adolescence, during which time toxic stress can force a premature maturation of regulatory networks and embed trauma at the epigenetic level. However, the framework of Biological Sensitivity to Context proves that this heightened plasticity is fundamentally bidirectional. The exact same neuroplasticity that renders a highly sensitive child susceptible to severe psychiatric disorders in a toxic environment enables that child to achieve exceptional developmental outcomes when placed in an enriching, supportive environment.
As definitively proven by large-scale longitudinal efforts such as the Bucharest Early Intervention Project, and supported by targeted clinical models like the ABC and PCIT interventions, the neural consequences of childhood adversity are rarely an irreversible destiny. Provided that interventions are initiated thoughtfully - particularly before the closure of early sensitive windows or capitalized upon during the secondary plasticity window of adolescence - it is entirely possible to restore structural connectivity, normalize physiological stress responses, and successfully repair the architectural foundation of the developing brain.