Human senolytic clinical trial results 2025-2026
The translation of senolytic therapies - pharmacological and biological agents designed to selectively eliminate senescent cells - from murine models to human clinical applications has reached a critical inflection point during the 2024 - 2026 period. Initially driven by broad aspirations of systemic rejuvenation and generic life extension, the clinical realities of human physiology, off-target toxicities, and strict regulatory endpoints have forced a necessary paradigm shift. Drug developers have transitioned from attempting to treat biological aging as a monolith toward targeting specific, measurable, age-related pathologies such as neurodegeneration, pulmonary fibrosis, dermatological inflammation, and hematological malignancies 123.
As of the first quarter of 2026, no senolytic agent has received regulatory approval from the United States Food and Drug Administration (FDA) or the European Medicines Agency (EMA) specifically for a primary senescence-targeting indication 11. However, the clinical pipeline has expanded dramatically. Over 45 active clinical trials are currently investigating senolytic compounds, a substantial increase from fewer than 15 active trials in 2021 1. This robust expansion is accompanied by a concurrent increase in Phase II trial terminations, reflecting a natural attrition rate exceeding 30% as early safety signals encounter the rigorous demands of functional efficacy endpoints 15.
This report exhaustively analyzes the 2025 - 2026 clinical trial landscape, assessing what has been empirically proven in humans regarding safety, biomarker modulation, and functional efficacy. It examines first-generation repurposed compounds, specialized targeted agents, emerging delivery platforms, and the critical evolution of artificial intelligence-driven biomarker diagnostics that are redefining trial methodologies.
Cellular Senescence and the Therapeutic Premise
To contextualize the clinical trial results, it is necessary to establish the underlying biological mechanisms targeted by senolytics. Cellular senescence is a state of stable, irreversible cell-cycle arrest triggered by various endogenous and exogenous stressors, including telomere attrition, oxidative stress, and oncogene activation 23. While senescence serves as an essential tumor suppressor mechanism in young, healthy organisms by preventing the proliferation of damaged cells (oncogene-induced senescence), the progressive accumulation of these cells in aged tissue becomes highly pathological 34.
Senescent cells remain metabolically active and secrete a highly complex, context-dependent mixture of pro-inflammatory cytokines, chemokines, growth factors, and matrix metalloproteinases. This secretory profile is collectively termed the senescence-associated secretory phenotype (SASP) 31510. The SASP creates a chronically inflamed local tissue microenvironment, contributing to localized tissue dysfunction and propagating secondary senescence in neighboring healthy cells via paracrine signaling 26.
Senescent cells resist their own toxic internal environments by upregulating senescent cell anti-apoptotic pathways (SCAPs), primarily through the B-cell lymphoma 2 (BCL-2) family of proteins and the PI3K/AKT survival networks 137. The central premise of senolytic pharmacology is to transiently and selectively disrupt these SCAPs. This disruption forces the senescent cell into apoptosis, removing the source of the SASP and allowing the tissue to undergo healthy regeneration 789. The therapeutic strategy relies on a "hit-and-run" approach, utilizing intermittent dosing rather than continuous suppression, thereby reducing long-term systemic toxicities while clearing the pathological cellular burden 31516.
First-Generation Kinase Inhibitors and Flavonoids
The combination of dasatinib, an FDA-approved multi-targeted tyrosine kinase inhibitor originally indicated for chronic myeloid leukemia, and quercetin, a naturally occurring flavonoid, forms the benchmark senolytic regimen (D+Q). Dasatinib primarily targets senescent preadipocytes and fibroblasts by inhibiting the SRC kinase pathway, while quercetin targets senescent endothelial cells via modulation of PI3K/AKT and other pathways 117. Together, they provide broad-spectrum senolytic activity.
Neurodegeneration and Cognitive Assessments
In the central nervous system, cellular senescence is deeply implicated in the progression of Alzheimer's disease (AD) and related tauopathies. Senescent microglia and astrocytes accumulate in the aging brain, exacerbating the aggregation of tau proteins and amyloid-beta (Aβ) plaques, ultimately driving sustained neuroinflammation and neuronal apoptosis 61011. Two highly scrutinized, parallel Phase 1/2 pilot trials - SToMP-AD and STAMINA - recently provided foundational human evidence regarding the feasibility of D+Q in neurodegenerative contexts 1610.
The SToMP-AD (Senolytic Therapy to Modulate the Progression of Alzheimer's Disease) vanguard trial evaluated 12 weeks of intermittent D+Q therapy in five older adults (mean age 72 ± 4 years) with early-stage, symptomatic AD 102012. The trial established critical pharmacokinetic proof of central nervous system penetrance. Following the administration of the regimen, dasatinib was detected in the cerebrospinal fluid (CSF) of four out of five participants at concentrations ranging from 0.281 to 0.536 ng/mL 101213. Quercetin, however, was not detected in the CSF, suggesting either rapid local metabolism or an inability to cross the blood-brain barrier effectively in humans 1213.
While SToMP-AD was an open-label safety study unpowered to detect functional cognitive improvements, it provided vital biomarker insights. Participants exhibited significant reductions in systemic plasma inflammatory markers associated with the SASP 102012. Conversely, CSF levels of interleukin-6 (IL-6) and glial fibrillary acidic protein (GFAP) increased post-treatment 1012. Researchers hypothesize that this transient spike in neuroinflammatory markers does not represent disease progression, but rather the active clearance and lysis of senescent astrocytes, alongside a trend toward higher levels of Aβ42 indicating possible mobilization of plaque materials 12.
The complementary STAMINA (Senolytics To Alleviate Mobility Issues and Neurological Impairments in Aging) trial provided preliminary functional signals. This single-arm pilot study enrolled 12 older adults (mean age 77 ± 8 years) with mild cognitive impairment (MCI) and slow gait speed 101424. Participants received six cycles of dasatinib (100 mg) and quercetin (1250 mg) for two consecutive days every two weeks 142415.
The trial revealed nuanced cognitive benefits. While overall cognitive and mobility endpoints trended positively without reaching statistical significance across the entire cohort, participants with the lowest baseline cognitive function (Montreal Cognitive Assessment [MoCA] scores of 18 - 25) exhibited a statistically significant improvement of 2.0 points (95% CI: 0.1, 4.0) 1424. Furthermore, reductions in circulating tumor necrosis factor-alpha (TNF-α) - a primary SASP component - significantly correlated with improvements in MoCA scores (r = -0.65, p = 0.02) 102415. These results confirm the biological activity of D+Q in the human brain and have directly facilitated the launch of fully powered, randomized controlled Phase 2 trials for AD progression 161020.
Systemic Applications: Pulmonary Fibrosis and Physical Frailty
Beyond the central nervous system, D+Q has been extensively evaluated for systemic, age-related fibrotic and musculoskeletal conditions. The rationale for targeting idiopathic pulmonary fibrosis (IPF) stems from preclinical evidence identifying senescent type-II pneumocytes as central drivers of the fibrotic cascade in the lungs 1.
However, translating this biology to human clinical outcomes has proven difficult. A randomized, placebo-controlled Phase 2 trial conducted at the Mayo Clinic evaluating intermittent D+Q in IPF patients yielded modest functional results 131. Published data demonstrated an improvement in the 6-minute walk test of 21.4 meters for the treatment group compared to placebo 1. While biologically active and generally well-tolerated, this functional improvement approaches but does not decisively cross the threshold of transformative clinical significance, highlighting the challenges of reversing established architectural tissue damage in human patients 12.
In the context of physical frailty, systemic senescent cell burden correlates directly with declining muscle endurance and bone density. The NIH-funded AFFIRM-ABLE trial is currently investigating the impact of D+Q on physical function in frail older adults across multiple U.S. clinical centers 1. While definitive Phase 2 results are projected for late 2026, interim analyses from related bone density cohorts indicate that senolytics yield the strongest responses in patients with the highest baseline burden of senescence-associated biomarkers, emphasizing the necessity of precision patient stratification moving forward 12.
Flavonoid Monotherapy and Chemotherapy-Induced Senescence
Fisetin, a naturally occurring polyphenol, functions as a "soft" senolytic. It modulates multiple intracellular pathways, including PI3K/AKT/mTOR, NF-κB, and BCL-xL, demonstrating a high therapeutic index with minimal off-target toxicity 317. In murine models, fisetin frequently outperforms D+Q in clearing senescent cells from specific tissues, including the hippocampus, liver, and kidney 17. Because of its safety profile, human trials have progressed using high daily doses (up to 20 mg/kg/day) 17.
Despite exceptional safety data, human functional efficacy for fisetin remains unproven as of early 2026. A recent trial evaluating fisetin supplementation (200 - 800 mg/day) in men with Gulf War Illness showed no significant differences from placebo regarding symptom severity, fatigue, or pain 26.
Clinical focus has consequently shifted to populations undergoing acute, massive senescent cell induction, such as patients receiving cytotoxic chemotherapy. The ongoing TROFFi (The Role of Fisetin in Frailty) Phase 2 study is investigating whether high-dose fisetin can mitigate the severe physical function decline observed in postmenopausal breast cancer survivors following neo/adjuvant chemotherapy 16. Chemotherapy induces widespread cellular damage, leading to a rapid accumulation of senescent cells that drive long-term fatigue and physical deterioration. If the TROFFi trial produces positive data in late 2026, it will provide the first definitive proof of fisetin's functional efficacy in humans 16.
B-Cell Lymphoma 2 (BCL-2) Family Inhibitors
The BCL-2 family of anti-apoptotic proteins is arguably the most validated therapeutic target in senolytic pharmacology. However, drugs targeting these pathways have generated the most polarizing clinical outcomes due to the inherent tension between high senolytic potency and severe, on-target hematological toxicities 335.
Local Administration in Ophthalmic Indications
To circumvent the systemic toxicities of BCL-2/BCL-xL inhibition, Unity Biotechnology developed UBX1325 (foselutoclax), a highly potent small molecule BCL-xL inhibitor designed exclusively for localized administration. By utilizing intravitreal injections, Unity aimed to clear senescent cells in the retinal vasculature while ensuring negligible systemic exposure 171819.
UBX1325 was evaluated in the Phase 2b ASPIRE trial, a double-masked, randomized, active-controlled study targeting patients with previously treated, active diabetic macular edema (DME) who had experienced suboptimal benefits from standard anti-VEGF therapies 182021. Fifty-two participants were randomized to receive either 10 μg of UBX1325 or 2 mg of the standard-of-care aflibercept (Eylea) every eight weeks for six months 182021.
In March 2025, the release of the topline ASPIRE data sent shockwaves through the longevity biotechnology sector: UBX1325 failed to meet its primary clinical endpoint. The endpoint measured statistical non-inferiority to aflibercept based on the average Best Corrected Visual Acuity (BCVA) letter change from baseline at weeks 20 and 24 182021. The trial required a 90% confidence interval to declare non-inferiority; UBX1325 achieved an 88% confidence interval, falling just short due to slight underperformance specifically at the 20-week measurement 182021.
Despite this narrow statistical failure, the biological and clinical data confirmed that the senolytic mechanism was highly active. UBX1325 demonstrated non-inferiority to aflibercept at nine out of the ten measured time points through 36 weeks. At 24 weeks, UBX1325 patients gained an average of +5.2 ETDRS letters (compared to +4.8 for aflibercept), and by 36 weeks, the gain increased to +5.5 letters (maintaining a +0.2-letter advantage over aflibercept) 182021.
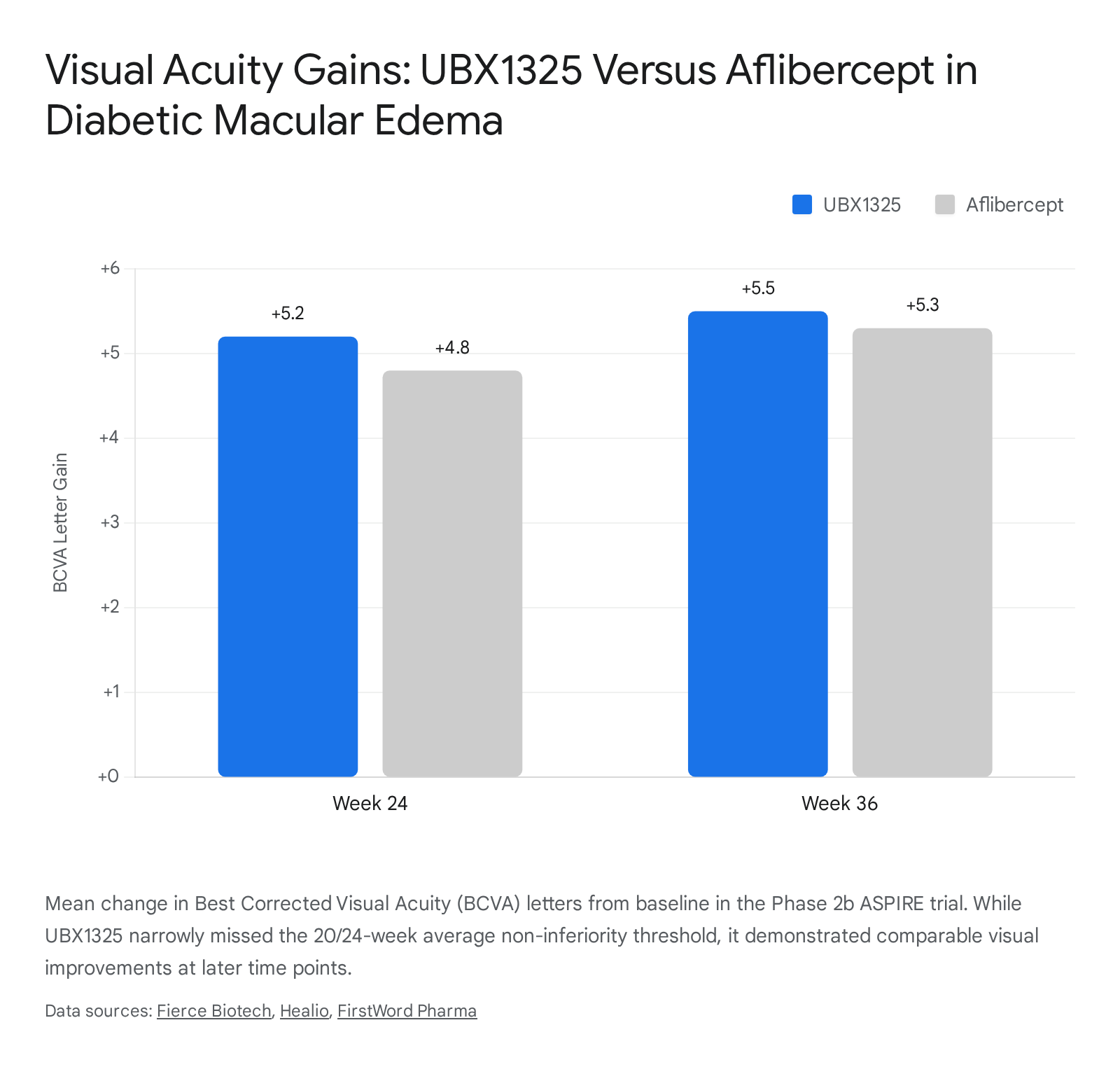
Furthermore, the drug showed excellent safety with no cases of intraocular inflammation, retinal artery occlusion, or endophthalmitis, and it generally outperformed the control in patients with moderate disease severity 18.
However, in the biopharmaceutical industry, primary endpoint failures carry catastrophic financial consequences. Following the data release, Unity's stock price collapsed by nearly 30%, severely restricting the company's ability to raise further capital in a risk-averse market 18203334. Consequently, in September 2025, Unity Biotechnology's board and stockholders approved a plan for complete corporate dissolution and liquidation 222324. The company terminated its laboratory leases in South San Francisco (effective March 31, 2026) and delisted its stock 222438. The collapse of Unity serves as a foundational lesson in senolytic drug development: proving biological activity and securing modest clinical gains are insufficient if the novel asset cannot definitively displace entrenched, highly effective standards of care 23339.
Systemic Administration in Hematological Malignancies
The systemic application of BCL-2 family inhibitors has found its greatest success in oncology, operating under the same biological principles as senolysis. Navitoclax (ABT-263), an oral dual inhibitor of BCL-2 and BCL-xL, forces apoptosis in cells reliant on these anti-apoptotic proteins. In myeloproliferative neoplasms (MPNs) such as myelofibrosis, malignant hematopoietic stem cells exhibit this exact anti-apoptotic dependency 44025.
Clinical data generated throughout 2025 and 2026 validate the profound efficacy of this pathway. The Phase 3 TRANSFORM-1 and TRANSFORM-2 trials evaluated navitoclax in combination with the JAK inhibitor ruxolitinib in patients with advanced myelofibrosis 252627. The TRANSFORM-1 trial met its primary endpoint, demonstrating that the combination therapy achieved a ≥35% reduction in spleen volume (SVR35) at week 24 in 63.2% of JAK-inhibitor-naïve patients, compared to only 31.5% for patients receiving ruxolitinib plus a placebo (p<0.0001) 25.
Crucially, the navitoclax combination demonstrated definitive disease-modifying effects, reducing bone marrow fibrosis by at least one grade in 30% to 33% of patients 2627. Patients achieving this reduction in fibrosis exhibited significantly improved overall survival rates 26.
However, this systemic efficacy is intrinsically linked to dose-limiting toxicity. Human platelets rely entirely on BCL-xL for their survival. Systemic administration of navitoclax inevitably induces rapid, dose-dependent platelet apoptosis. In clinical trials, up to 31% of patients experienced grade 3 or 4 thrombocytopenia 52744. While manageable through strict dose titrations and clinical monitoring in severe oncological indications, this toxicity profile renders first-generation BCL-xL inhibitors inherently unsafe for broad, systemic application in non-terminal, age-related chronic diseases 15.
To overcome this bottleneck, next-generation engineering is required. AbbVie is currently advancing ABV-2580, an engineered BCL-2/BCL-xL dual inhibitor with specifically designed platelet-sparing properties. Having completed IND-enabling studies targeting cardiovascular aging, Phase 1 clinical trials for this asset are scheduled to initiate in the third quarter of 2026 1.
Glutathione Peroxidase 4 (GPX4) Modulation and Ferroptosis
The most significant clinical breakthrough in the 2025 - 2026 senolytic pipeline circumvented the apoptotic pathways entirely, focusing instead on a different mechanism of regulated cell death: ferroptosis.
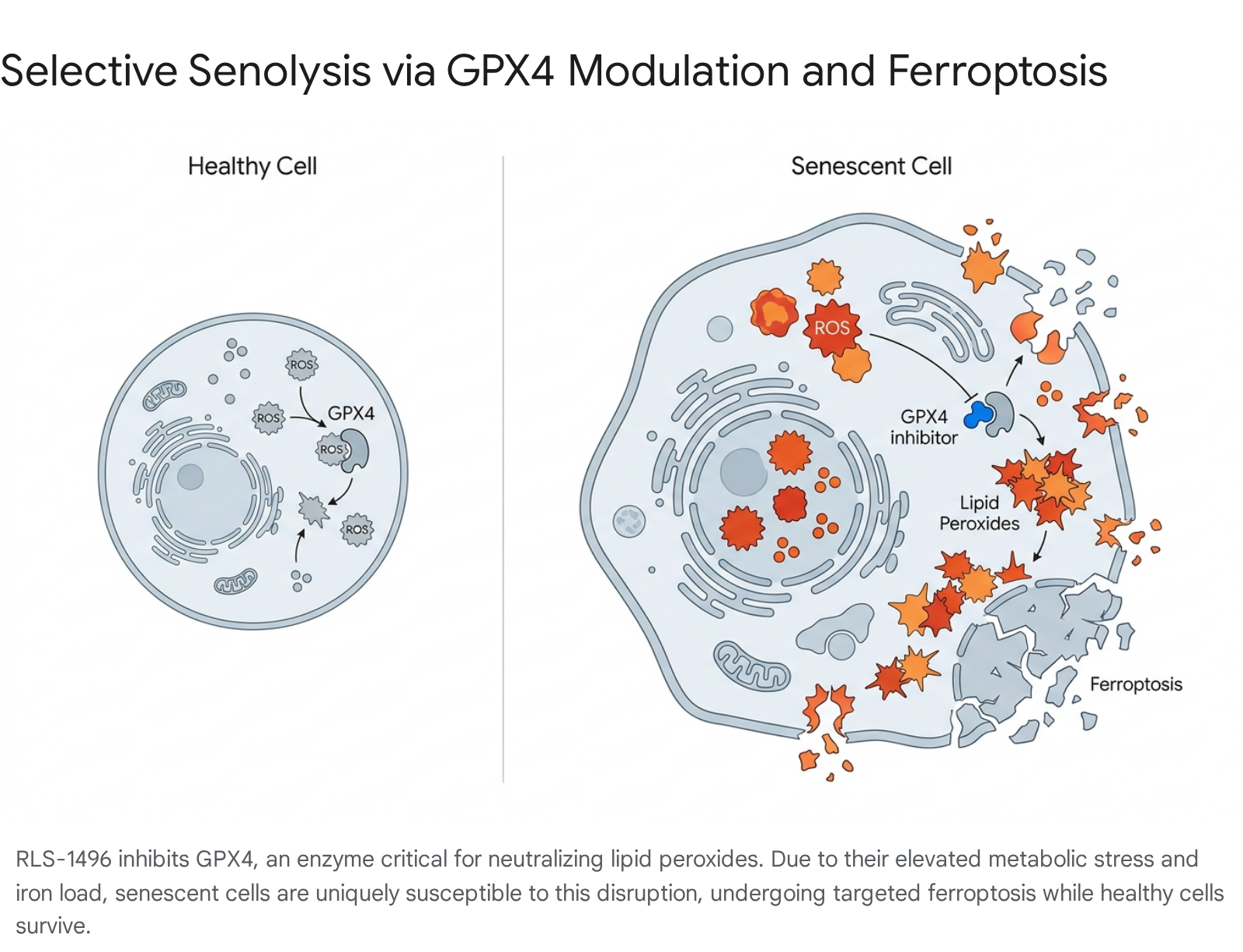
Senescent cells are characterized by high metabolic stress, producing elevated levels of reactive oxygen species (ROS) and accumulating altered intracellular iron 628. This state makes them highly susceptible to lipid peroxidation. In healthy cells, the enzyme glutathione peroxidase 4 (GPX4) acts as the primary defense mechanism, neutralizing lipid peroxides to maintain cellular membrane integrity 282930.
Rubedo Life Sciences developed RLS-1496, a first-in-class small molecule that selectively inhibits GPX4. By removing this protective enzyme, RLS-1496 selectively triggers catastrophic lipid peroxidation and membrane rupture (ferroptosis) in highly stressed senescent cells, while healthy cells - which lack the underlying toxic iron and ROS burden - easily survive the transient GPX4 inhibition 282930.
In March 2026, Rubedo reported positive preliminary data from a European Phase 1 trial evaluating the topical administration of RLS-1496 in patients with plaque psoriasis, atopic dermatitis, and photoaged skin 2831. This trial marked a historic proof-of-concept for the senolytic field. Phase 1 dermatology trials rarely produce definitive clinical efficacy signals due to short durations and small cohorts; however, RLS-1496 succeeded in demonstrating both target engagement and robust clinical outcomes within a four-week period 4950.
The trial evaluated three dose cohorts (0.1%, 0.5%, and 1.0%), establishing a clear dose-response relationship with the 1.0% formulation chosen for further advancement 49. Histological and cellular analyses confirmed a significant reduction in target senescent cells, accompanied by a steep decline in inflammatory SASP cytokines, notably IL-19 and S100A7 3149.
Clinically, psoriasis patients exhibited an average 20% reduction in epidermal thickness following one month of treatment 283149. In the atopic dermatitis cohort, 25% of treated subjects reported a clinically meaningful reduction in pruritus (itching) - defined as a ≥4-point improvement on the numeric rating scale - an outcome absent in the placebo vehicle group 4950. In photoaged skin, researchers noted increased collagen expression and reduced inflammatory signaling, confirming the fundamental rejuvenation of the tissue matrix 3150.
Following these unprecedented results, the FDA cleared an Investigational New Drug (IND) application for a Phase 1b/2a trial in the United States targeting actinic keratosis 293052. Actinic keratosis presents as precancerous, scaly skin patches resulting directly from cumulative sun damage and subsequent cellular senescence 3052. This U.S. trial will gather data on approximately 70 human subjects, bridging the gap between specific inflammatory dermatological diseases and the broader goal of reversing fundamental skin aging 2850. Furthermore, Rubedo plans to initiate human trials for a systemic oral formulation of RLS-1496 by 2027, targeting idiopathic pulmonary fibrosis and metabolic syndromes 392853.
Alternative Targeted Senolytics and Gene Therapies
As the limitations of first-generation kinase and BCL-2 inhibitors become apparent, the late 2025 and 2026 pipelines are increasingly dominated by highly specific, novel mechanisms of action designed to maximize safety in elderly populations.
Precision Protein Inhibition
Cleara Biotech, a Dutch biopharmaceutical firm, focuses on pathways independent of the BCL-2 family. Based on research discovering that senescent cells rely on the FOXO4-p53 interaction to resist apoptosis, Cleara developed FOXO4-DRI, a modified peptide that disrupts this specific interaction. While FOXO4-DRI has demonstrated profound preclinical success in reversing frailty in progeroid mice, it currently remains entirely preclinical 31954.
Consequently, Cleara's clinical strategy has shifted to another target: the p21 CDKN1A pathway. The company is advancing CB-5945, a selective p21 inhibitor. By targeting p21 rather than BCL-xL, CB-5945 is specifically designed to eliminate senescent cells while minimizing the risk of platelet and bone marrow toxicities. Backed by a €62 million Series B financing round in late 2025, Cleara expects Phase 1 clinical trial data for CB-5945 in idiopathic pulmonary fibrosis patients by the end of 2026 115.
Targeted Delivery and Gene Therapy Platforms
A major challenge in senolytic administration is achieving systemic efficacy without exposing healthy tissues to high drug concentrations. Oisín Biotechnologies addresses this challenge by adapting the lipid nanoparticle (LNP) technology that successfully enabled mRNA vaccines.
Oisín utilizes a proprietary proteo-lipid vehicle (FAST-PLV) to deliver specific DNA or RNA payloads 5556. Instead of delivering a cytotoxic small molecule, the LNP delivers a genetic construct containing a suicide gene governed by the p16INK4a promoter 315. The construct is taken up widely by both healthy and senescent cells. However, the suicide gene is only transcribed and activated in cells actively expressing high levels of p16INK4a - the master biomarker of cellular senescence 315. Healthy cells simply degrade the inert payload.
Supported by a $12 million grant from the National Institute on Aging and collaborations with the Salk Institute, Oisín completed IND-enabling studies in 2025. The platform has progressed into Phase 1 clinical trials targeting cardiovascular aging in 2026, marking the first human test of a systemic, gene-targeted senolytic delivery system 1355.
Immunological Clearance Modalities
Harnessing the endogenous immune system to identify and clear senescent cells is emerging as a potent alternative to pharmacological induction of apoptosis. Senescent cells express distinct surface proteins, making them viable targets for immunotherapies.
One primary target is the urokinase-type plasminogen activator receptor (uPAR), which is highly upregulated in senescent populations 932. Preclinical developments have engineered chimeric antigen receptor (CAR) T cells to specifically target uPAR-positive cells 932. In murine models, senolytic CAR-T therapy successfully cleared senescent cells, reversed liver fibrosis, and ameliorated metabolic dysfunction in subjects on a high-fat diet 932. The theoretical advantage of CAR-T senolysis lies in the longevity and "memory" of T cells, potentially allowing a single administration to provide long-term surveillance and clearance of senescent cells as they form 9.
Vaccine-based approaches are also advancing. Researchers have targeted Glycoprotein Nonmetastatic Melanoma Protein B (GPNMB) and Senescence-associated adhesion molecule 1 (SAAM-1) 323334. Prophylactic vaccination against these surface proteins in aged mice led to targeted immune clearance, successfully alleviating cardiovascular and metabolic pathologies 3334. Despite these preclinical triumphs, immunological senolysis has not yet progressed to human clinical trials as of 2026. A significant theoretical risk remains: the artificial depletion of senescent cells via aggressive immune stimulation may inadvertently alter or suppress other necessary vaccine-induced immune responses in vulnerable elderly populations 32.
Summary of the 2026 Clinical Landscape
To synthesize the diverse approaches advancing through human trials, the following table details the most critical clinical-stage assets shaping the senolytic market in 2026.
| Therapeutic Agent | Mechanism of Action | Sponsoring Entity | Target Indication | Clinical Stage (2026) | Key Proven Clinical Outcome |
|---|---|---|---|---|---|
| Dasatinib + Quercetin | Multi-Kinase / PI3K / BCL-2 Inhibition | Mayo Clinic / NIA | Alzheimer's, Frailty, IPF | Phase 2 | Confirmed CNS penetrance; SASP biomarker reduction; preliminary cognitive stabilization signals 101224. |
| UBX1325 (Foselutoclax) | Localized BCL-xL Inhibition | Unity Biotechnology | Diabetic Macular Edema | Phase 2b (Failed) | Missed statistical non-inferiority primary endpoint, but proved visual acuity gains vs. active control 1821. |
| Navitoclax (ABT-263) | Systemic BCL-2/BCL-xL Inhibition | AbbVie / Various | Myelofibrosis | Phase 3 | Significant spleen volume reduction; bone marrow fibrosis modification; dose-limiting thrombocytopenia 402526. |
| RLS-1496 | GPX4 Modulator (Ferroptosis Induction) | Rubedo Life Sciences | Psoriasis, Atopic Dermatitis | Phase 1b/2a | 20% epidermal thickness reduction; clearance of tissue senescence; robust safety profile 3049. |
| Fisetin | Flavonoid (Multi-pathway modulation) | Wake Forest / NIA | Chemotherapy-induced Frailty | Phase 2 | High systemic safety and tolerability; pending definitive functional outcomes (TROFFi trial) 1716. |
| CB-5945 | p21 CDKN1A Pathway Inhibition | Cleara Biotech | Idiopathic Pulmonary Fibrosis | Phase 1/2 | Avoids BCL-2 related platelet toxicity; €62M backing for clinical initiation 115. |
| Oisín LNP Platform | p16INK4a-Targeted Suicide Gene Delivery | Oisín Biotechnologies | Cardiovascular Aging | Phase 1 | Highly specific in vivo targeted delivery; completed IND-enabling studies 31556. |
The Biomarker Paradigm Shift
A historic bottleneck contributing to the high Phase II attrition rate in senolytic trials has been the lack of standardized, non-invasive biomarkers. Until recently, quantifying a patient's senescent cell burden required invasive tissue biopsies to measure senescence-associated beta-galactosidase (SA-β-gal) activity or localized p16INK4a expression 211519.
In April 2026, a landmark study published in Nature Aging revolutionized senolytic trial methodology by introducing the "SASP Score" 1035. Researchers integrated large-scale population proteomics from the UK Biobank Pharma Proteomics Project (UKB-PPP) with a semi-supervised deep learning framework known as the Guided AutoEncoder with Transformer (GAET) model 1035. This model successfully correlates circulating blood-based SASP proteins with systemic cellular senescence burden.
Crucially, the deep learning-derived SASP Score has proven to be a highly accurate, independent predictor of both mortality risk and incident chronic medical conditions, such as dementia, COPD, and myocardial infarction 1035. In independent trial cohorts, interventions (such as multimodal exercise) were shown to significantly alter the SASP Score trajectory over 18 months, validating its use as a dynamic tracking tool for therapeutic efficacy 1035.
Simultaneously, specialized multi-omic metrics are being developed for localized diseases. The Retinopathy Senescence Progression Score (RSPS), derived from analyzing 307 genes, shows strong positive correlations with the core SASP Score and accurately tracks the pathological progression of diabetic retinopathy without requiring retinal biopsies 36.
To standardize these disparate advancements, the Senotherapeutics Biomarker Consortium was formally established in March 2026 62. Comprising leading researchers from institutions such as the NIH SenNet Consortium, Genentech, and Rubedo Life Sciences, the group aims to establish universal, regulatory-grade proxy metrics for cellular senescence 623738. This shift - moving from observing highly variable downstream physical outcomes to measuring precise, upstream biological target engagement via blood-based proteomics - is widely viewed as the essential step required to navigate future Phase 3 pivotal trials successfully 2510.
Conclusion
The clinical trial data compiled between 2025 and 2026 provides unequivocal proof that senolytic therapies are biologically active in humans. It has been empirically established that pharmacological agents - ranging from multi-kinase inhibitors (Dasatinib) and natural flavonoids (Quercetin, Fisetin) to synthetic BCL-2 inhibitors (Navitoclax) and GPX4 modulators (RLS-1496) - can safely engage cellular targets, selectively clear senescent cells from human tissue, and durably suppress systemic inflammatory SASP cascades 102549.
However, the period has also proven that translating biological senescent cell clearance into statistically robust, functional clinical endpoints is a formidable challenge. The dissolution of Unity Biotechnology serves as a harsh reality check: even when a senolytic (UBX1325) successfully clears damaged cells and generates measurable clinical improvements (visual acuity gains), it will face severe commercial and regulatory headwinds if it cannot statistically outperform entrenched, highly effective standards of care 332438.
The most definitive clinical successes of early 2026 belong to localized, targeted interventions rather than broad, systemic "anti-aging" panaceas. The robust efficacy of Rubedo Life Sciences' RLS-1496 in dermatology - achieving histological thickness reductions and significant symptom relief within weeks - demonstrates that exploiting novel cell-death pathways (ferroptosis) in accessible tissues is currently the most viable translational strategy 22949. As the field navigates systemic toxicities by advancing platelet-sparing molecules, lipid nanoparticle gene therapies, and AI-driven proteomic biomarkers like the SASP Score, senolytics are rapidly maturing from experimental longevity science into a highly regulated domain of precision molecular medicine 2310.