Human efficacy of NR and NMN for raising NAD+ levels
Introduction
Nicotinamide adenine dinucleotide (NAD+) is an indispensable dinucleotide coenzyme present in all living cells, mediating over 500 distinct enzymatic reactions 12. Functioning primarily as a critical electron carrier in cellular energy production pathways - including glycolysis, the tricarboxylic acid (TCA) cycle, and mitochondrial oxidative phosphorylation - NAD+ also serves as an obligate consuming substrate for a wide array of signaling enzymes. These include the sirtuin (SIRT) family of protein deacetylases, poly(ADP-ribose) polymerases (PARPs) involved in DNA damage repair, and the cyclic ADP-ribose synthase CD38, which regulates calcium signaling and immune cell function 114.
Extensive physiological profiling has consistently demonstrated that intracellular and systemic tissue NAD+ concentrations undergo a progressive, age-associated decline 156. This pervasive depletion is mediated by a dual mechanism: a downregulation of endogenous biosynthetic enzyme capacity, coupled with an aggressive upregulation of NAD+-consuming enzymes in response to cumulative genomic instability, chronic inflammatory states, and elevated oxidative stress 72. Diminished NAD+ bioavailability is heavily implicated in the biochemical pathogenesis of numerous age-related systemic dysfunctions, including metabolic syndrome, neurodegenerative pathology, skeletal muscle sarcopenia, and compromised immune resilience 523.
To counteract this cellular deficit, the exogenous administration of NAD+ precursors has emerged as a primary therapeutic intervention in the fields of aging and metabolic health. Because the intact NAD+ molecule is too large and carries a high negative charge that prevents efficient direct transport across the cellular plasma membrane, supplementation strategies rely on smaller metabolic intermediate molecules 110. Cells can internalize these precursors and enzymatically convert them into NAD+ through localized biosynthetic pathways. Currently, the two most prominent and heavily researched precursors are nicotinamide riboside (NR) and nicotinamide mononucleotide (NMN) 1114.
While preclinical murine models have demonstrated profound, systemic age-reversing and metabolic benefits following the administration of both compounds, a complex scientific debate has evolved regarding their comparative efficacy, divergent pharmacokinetic behaviors, and the precise fidelity of their functional translation in human clinical trials 111314.
Biochemical Pathways and Cellular Uptake Mechanisms
The intracellular metabolism of NAD+ precursors is governed by distinct biochemical routes, primarily the salvage pathway and the Preiss-Handler pathway 115. A persistent mechanistic dispute between advocates of NMN and NR centers on their respective requirements for extracellular modification prior to cellular internalization.
Structural Requirements for Cellular Entry
In mammalian cells, NAD+ biosynthesis occurs via three primary pathways: the de novo synthesis pathway utilizing the amino acid tryptophan; the Preiss-Handler pathway, which converts nicotinic acid (NA); and the dominant salvage pathway, which recycles nicotinamide (NAM) and utilizes the precursors NR and NMN 15.
Within the salvage pathway, NR functions as a direct substrate for the nicotinamide riboside kinase enzymes (NRK1 and NRK2). These kinases phosphorylate NR to generate NMN within the cytoplasm. Subsequently, NMN is adenylated by a family of nicotinamide mononucleotide adenylyltransferases (NMNAT1-3) to yield the final functional NAD+ molecule 11718.
Conversely, the cellular uptake mechanics of exogenous NMN are significantly more complex. Under physiological conditions, the NMN molecule possesses an intact phosphate group, which generally prevents spontaneous diffusion or unassisted transport across the lipid bilayer of the plasma membrane 113. For years, the established biochemical consensus maintained that extracellular NMN must undergo an obligatory dephosphorylation event, catalyzed by the cell-surface ectonucleotidase enzyme CD73, converting it into NR before it can enter the cell via equilibrative nucleoside transporters (ENTs) 1319. Once internalized as NR, it requires re-phosphorylation by NRKs, ultimately following the same terminal route to become NAD+. This mandatory extracellular conversion step theoretically introduces thermodynamic and metabolic inefficiency, providing a classical biochemical argument for NR's structural superiority as a supplement 113.
While researchers have identified a specific mammalian NMN transporter, Slc12a8, which is expressed in the murine gastrointestinal tract, the broader physiological relevance, transport efficiency, and systemic expression levels of Slc12a8 in human tissues remain a subject of active debate 1420. Consequently, from a purely molecular standpoint, NMN and NR are not fundamentally distinct parallel molecules; rather, they are adjacent, sequential intermediates integrated into the exact same biosynthetic cascade 20.
The Role of the Gut Microbiome in Precursor Metabolism
Recent analytical advances have fundamentally altered the pharmacokinetic model of oral NAD+ precursor supplementation in humans. A pivotal 2025 study by Christen et al., published in Nature Metabolism, demonstrated that following standard oral ingestion, neither NR nor NMN is absorbed into the systemic circulation entirely intact 15206. Instead, both precursors are subjected to heavy preliminary metabolism by the host gut microbiota.
Utilizing both ex vivo human microbiota fermentation models and in vivo human trials, researchers revealed that specific gut bacteria - most notably the species Enterocloster aldensis - rapidly metabolize both NR and NMN 156. The microbes deamidate these precursors into nicotinamide (NAM), and subsequently convert the NAM into nicotinic acid (NA) 156. This microbially derived NA is then slowly released and absorbed into the systemic circulation, where it is robustly processed via the Preiss-Handler pathway to boost whole-blood NAD+ pools 156.
This critical discovery functionally equalizes the two compounds at the level of gastrointestinal processing. It indicates that their primary mechanism for elevating human whole-blood NAD+ relies on a shared, microbially mediated conversion sequence rather than direct, intact tissue absorption across the intestinal epithelium 206. Furthermore, the Christen study demonstrated that the microbial metabolism of these specific precursors (NR and NMN, but not NAM alone) significantly increased the intestinal concentrations of short-chain fatty acids (SCFAs) 15. SCFA generation was sustained for up to 40 hours post-exposure, providing independent anti-inflammatory, anti-diabetic, and mucosal health benefits that operate collaterally to direct NAD+ synthesis 15.
Comparative Efficacy in Elevating Human Systemic NAD+
The central clinical dispute within the "NMN vs. NR" debate is the determination of whether one precursor inherently elevates systemic NAD+ concentrations more effectively than the other. Until recently, indirect comparisons drawn across highly heterogeneous, single-arm trials yielded no definitive answer. The pharmacokinetic landscape was substantially clarified by two landmark head-to-head human clinical trials published in 2025 and 2026, though their conflicting conclusions have perpetuated ongoing scientific division.
The 2025 Parallel-Group Trial (Christen et al.)
The first direct, comprehensive human comparison was a randomized, open-label, placebo-controlled, parallel-group study conducted by Christen and colleagues (2025) involving 65 healthy adult participants 1206. The subjects were randomized to receive 1,000 mg/day of NR, 1,000 mg/day of NMN, 500 mg/day of NAM, or a matching placebo over a 14-day intervention period 115.
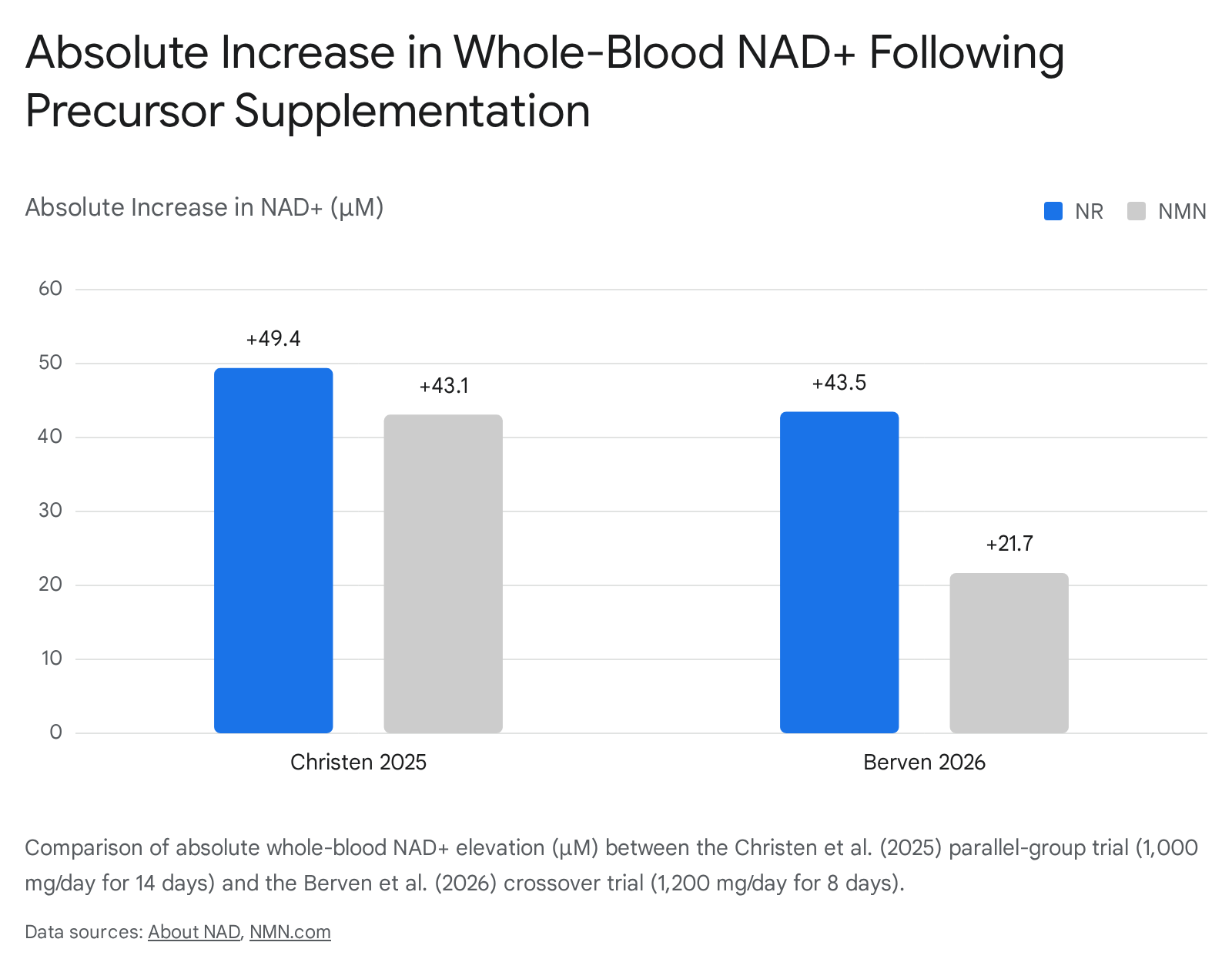
The resulting data demonstrated that both NR and NMN sustainably and significantly elevated circulating NAD+ by approximately two-fold compared to baseline measurements and the placebo cohort 156. Specifically, absolute whole-blood NAD+ increased by 49.4 μM in the NR cohort and 43.1 μM in the NMN cohort 115. While NR exhibited a nominal ~15% statistical edge in absolute concentration yield, the authors formally concluded that the two compounds are highly comparable in their ability to chronically increase baseline whole-blood NAD+ over a two-week span 16. Nicotinamide (NAM), by contrast, provided only an acute, transient spike in NAD+ that did not translate to sustained, long-term steady-state elevation, precluding its viability as a chronic restorative intervention 15206.
The 2026 Crossover Trial (Berven et al.)
In early 2026, an independent research team at Haukeland University Hospital in Bergen, Norway, published a phase I pharmacokinetic trial in the journal iScience that presented a starkly different kinetic narrative 113. Berven et al. administered 1,200 mg/day of both NR and NMN to six healthy middle-aged adults (three men, three women) over an eight-day period, utilizing a randomized crossover design separated by a controlled washout phase 15. The crossover architecture meant that each participant acted as their own absolute control, effectively eliminating inter-individual genetic and metabolic variance as a confounding factor 113.
The pharmacokinetic profiling revealed a pronounced, unambiguous superiority for NR. Whole-blood NAD+ rose from an average baseline of approximately 27 μM to 70.5 μM (representing a 161% increase from baseline) under NR supplementation 1513. Conversely, under NMN supplementation, whole-blood NAD+ rose from an average baseline of 32 μM to 53.7 μM (an average increase of 67% to 69%) 1513. When controlling for precise gram dose and mathematically correcting for molecular weight discrepancies, NR produced a 2.3-fold greater NAD+ elevation compared to NMN 113.
The kinetic divergence observed was so profound that the researchers made an unprecedented decision mid-study: they permanently discontinued the NMN arm and executed all subsequent experimental phases of their research program exclusively utilizing NR 13. Furthermore, comprehensive tracking of the broader NAD metabolome revealed elevated baseline levels of nicotinic acid adenine dinucleotide (NAAD) in participants taking both NR and NMN 17. NAAD acts as a reliable metabolic fingerprint, indicating that multiple parallel routes - including the microbially driven Preiss-Handler pathway - were saturated to build the systemic NAD+ pool 17.
Resolving Pharmacodynamic Discrepancies
The apparent contradiction between the Christen and Berven trials can be reconciled through careful examination of their respective methodological contexts and limitations. The Berven trial's crossover design provides inherently superior statistical power to control for individual genetic, enzymatic, and metabolic variance, lending exceptionally high internal validity to its finding that NR possesses a distinct early kinetic advantage 1317. However, the study was constrained by a highly restrictive sample size (n=6) and a brief 8-day intervention duration 117. Crucially, the authors explicitly noted in their findings that NMN's absolute pharmacokinetic peak in human subjects typically occurs on day nine - a full 24 hours after the Berven supplementation window had already closed 1.
Conversely, the Christen trial, utilizing a much larger cohort of 65 participants over a 14-day window, permitted NMN sufficient time to reach and demonstrate a stable steady-state concentration 117. Furthermore, human absorption pharmacokinetics for both nucleoside compounds are highly sensitive to gastrointestinal variables, specifically fed versus fasted administration states, which may not have been perfectly aligned across the two disparate study protocols 13. Consequently, the consensus emerging among independent analysts is that while NR may exhibit superior short-term absorption kinetics and direct whole-molecule bioavailability, both precursors are highly comparable in their ultimate capacity to maintain elevated systemic NAD+ pools over sustained, chronic periods 2022.
Structural Limitations of the Current Evidence Base
Beyond the isolated head-to-head trials, the broader literature attempting to compare NMN and NR suffers from severe, systematic methodological asymmetries that actively preclude reliable network meta-analysis. A rigorous 2026 systematic transitivity assessment sought to map the evidence gap across 15 randomized controlled trials (5 evaluating NMN, 10 evaluating NR, comprising a total of 740 participants) 214.
The analysis identified three critical, quantifiable barriers that prevent a valid indirect cross-precursor comparison:
- Molar Dose Asymmetry: Across the established literature, NR is consistently administered at doses 1.9 to 9.2 times higher than NMN on a strict molar basis 1423. NMN trials have predominantly utilized highly conservative doses of 250 to 300 mg/day (equating to 0.75 to 0.90 mmol/day). In stark contrast, NR trials routinely evaluate doses between 500 and 2,000 mg/day (equating to 1.72 to 6.88 mmol/day) 1423. This substantial dose discrepancy creates an inherent bias in evaluating aggregate efficacy.
- Population Bias: The vast majority of NMN trials (such as Yoshino 2021, Huang 2022, Igarashi 2022, Katayoshi 2023, and Morifuji 2024) have been conducted exclusively within East Asian cohorts (primarily in Japan and India) 1423. Conversely, NR trials have heavily skewed toward Western populations across Europe and North America. This geographic disparity introduces unquantified, systematic confounding variables related to ethnicity, baseline dietary patterns, and genetic metabolic predispositions 1423.
- Biomarker Incompatibility: The analytical assays utilized to measure the primary pharmacodynamic endpoint - NAD+ concentration - are historically non-standardized. Different trials report NAD+ values utilizing entirely incompatible matrices and scaling units. For example, some NMN trials (e.g., Huang 2022) reported blood cellular NAD+/NADH ratios mapped in pmol/mL, whereas contemporary NR trials reported whole-blood NAD+ mapped in μM 1423.
These severe methodological asymmetries render indirect statistical comparisons of precursor efficacy structurally unreliable until future multi-center clinical trials enforce rigorous equimolar dosing parameters, enroll diverse populations, and utilize harmonized assay matrices 214.
Characteristics of Key Pharmacodynamic Trials
| Precursor Evaluated | Key Study / Author | Dose Evaluated | Trial Duration | Cohort Characteristics | Primary Pharmacodynamic Outcome |
|---|---|---|---|---|---|
| NR vs NMN vs NAM | Christen et al. (2025) | 1,000 mg/day (NR & NMN) | 14 days | 65 healthy adults (Parallel) | NR and NMN comparably doubled blood NAD+; NAM produced transient spike only 156. |
| NR vs NMN | Berven et al. (2026) | 1,200 mg/day | 8 days | 6 healthy adults (Crossover) | NR produced 2.3-fold greater blood NAD+ elevation over NMN at matched weight 113. |
| NMN | Yoshino et al. (2021) | 250 mg/day | 10 weeks | 25 prediabetic, postmenopausal women | Increased immune cell NAD+ by 43%; no increase in skeletal muscle NAD+ pool 22. |
| NR | Conze et al. (2019) | 100, 300, 1,000 mg/day | 8 weeks | Healthy overweight adults | Dose-dependent whole blood NAD+ increase (22%, 51%, 142% respectively) 22. |
Translation to Clinical and Functional Outcomes
The intense debate regarding which precise precursor produces a steeper or faster pharmacokinetic NAD+ spike is largely secondary to a much more critical physiological question: does raising blood NAD+ actually yield patient-relevant functional and clinical benefits? The prevailing evidence regarding functional outcomes presents a stark, sobering contrast between spectacular preclinical murine data and highly modest, often null, human clinical translation.
Glycemic Control and Metabolic Health
In preclinical laboratory settings, studies have consistently demonstrated that the administration of NAD+ precursors can rapidly restore glucose homeostasis, improve peripheral insulin sensitivity, and reverse diet-induced obesity in rodents. These benefits are mechanistically driven by the robust activation of the SIRT1/AMPK/PGC-1α signaling pathway, which enhances mitochondrial biogenesis and normalizes oxidative stress 278. However, when these identical interventions are transitioned to human trials, the results are overwhelmingly muted 7.
Systematic reviews and comprehensive meta-analyses of human trials consistently reveal a "negligible effect" on standard cardiometabolic parameters 14179. A highly comprehensive 2024 meta-analysis conducted by Zhang et al. evaluated 12 human RCTs involving a total of 513 participants taking NMN 27. The analysis concluded that while NMN supplementation successfully and effectively elevated blood NAD+ concentrations, it failed to produce any statistically significant improvements in fasting blood glucose, total cholesterol, LDL-C, HDL-C, HbA1c, HOMA-IR, body weight, or general BMI in adults 27. The single positive metabolic signal observed was a minor decrease in triglyceride (TG) levels in overweight participants 27. Similarly, an independent 2024 meta-analysis by Zheng et al. encompassing 9 RCTs and 412 participants corroborated these non-significant effects across the entire lipid profile, glycemic indices, and systolic/diastolic blood pressure metrics 14.
Clinical results for NR mirror these disappointing metabolic findings. Robustly designed trials, such as the 2018 study by Dollerup et al., administered 2,000 mg/day of NR to obese, insulin-resistant men for a duration of 12 weeks. Despite confirmed, safe NAD+ elevation, the intervention produced absolutely no measurable improvements in whole-body insulin sensitivity, endogenous glucose production, lipid oxidation, resting energy expenditure, or body composition 1011.
The sole notable exception within the human metabolic literature is a highly specific 2021 trial by Yoshino et al. The researchers evaluated 250 mg/day of NMN over 10 weeks in a narrowly defined cohort of postmenopausal, prediabetic, overweight women 1130. In this specific population, the intervention yielded an approximate 25% improvement in skeletal muscle insulin sensitivity - a magnitude of biological benefit comparable to a 10% total body weight loss or the initiation of certain diabetes medications 1130. However, this clinical benefit was entirely tissue-specific; hepatic and adipose tissue insulin sensitivity remained completely unaffected by the NMN 30. Furthermore, this specific outcome has proven exceedingly difficult to replicate broadly in mixed-gender or healthy volunteer cohorts 1430.
Skeletal Muscle Function and Physical Performance
Results regarding macroscopic muscle strength, exercise capacity, and physical performance are mildly more favorable than metabolic parameters, but remain structurally inconsistent. An NMN trial in healthy older men (Igarashi et al., 2022) administering 250 mg/day for 12 weeks reported no measurable change in total skeletal muscle mass, but did note statistically significant, albeit marginal, improvements in left-hand grip strength (p=0.019) and gait walking speed (p=0.033) 2212. Another trial involving middle-aged adults receiving 300 mg/day of NMN for 60 days reported improvements in six-minute walk distances 32.
However, larger systematic meta-analyses assessing NR and NMN collectively have concluded that there is no significant aggregate effect on muscle mass or absolute physical strength across general older adult populations 2732.
At the microscopic and cellular level, the biological machinery is indeed being stimulated. An NR trial administering 1,000 mg/day for six weeks demonstrated an increase in sleeping metabolic rate and altered the muscle metabolome - specifically elevating levels of acetylcarnitine, a compound linked to efficient muscle energy utilization 30. A 2023 trial involving BMI-discordant identical twins given escalating doses of NR (up to 1,000 mg/day) confirmed notable improvements in muscle myoblast differentiation and mitochondrial biogenesis, highlighting that NR actively reshapes cellular energy dynamics, even if those microscopic changes fail to translate into macroscopic, clinically obvious strength gains 2010.
Neurological Bioavailability and Disease Intervention
The selective permeability of the blood-brain barrier poses a significant hurdle for metabolic and systemic therapeutics. The 2026 pharmacokinetic study by Berven et al. sought to determine if systemic NAD+ elevation translates to the central nervous system by assessing brain NAD+ pools utilizing magnetic resonance imaging (MRI) 15. The study found that 8 days of standard NR or NMN supplementation failed to alter neurological NAD+ levels 5. However, when researchers extended the NR supplementation protocol to four full weeks, it successfully raised brain NAD+ levels in both healthy adults and patients suffering from Parkinson's disease 15.
Subsequent phase I/II clinical trials administering exceedingly high doses of NR (escalating up to 3,000 mg/day) to Parkinson's patients proved to be both safe and well-tolerated. These high-dose interventions generated mild but observable clinical symptom improvements and decreased neuroinflammation, firmly establishing the targeting of the autophagy-NAD+ axis as a viable, promising frontier for neurodegenerative disease research 1109.
Similarly, MetroBiotech's pharmaceutical-grade polymorph of NMN (designated MIB-626) is currently undergoing rigorous Phase II clinical trials targeting Alzheimer's disease. The primary objective of these ongoing trials is to precisely quantify NMN's capacity to cross the blood-brain and cerebrospinal fluid barriers to mitigate pathological amyloid-beta concentrations and delay cognitive neurodegeneration 3334.
Summary of Human Clinical Translation Outcomes
| Physiological Domain | NMN Clinical Translation Data | NR Clinical Translation Data | Overall Scientific Consensus |
|---|---|---|---|
| Pharmacodynamics | Reliably doubles blood NAD+ at doses of ~1g/day 12022. | Reliably doubles blood NAD+ at ~1g/day; potential early kinetic advantage 12022. | Both precursors are highly effective at elevating systemic NAD+ 2022. |
| Metabolic Health | Null effect on HbA1c, fasting glucose, BMI, and lipids in general populations 27. Isolated benefit observed in prediabetic female muscle insulin sensitivity 1130. | Null effect on whole-body insulin sensitivity, lipolysis, or resting energy expenditure in obese and insulin-resistant cohorts 11. | Negligible impact on broad cardiometabolic health markers in human populations 1417. |
| Physical Performance | Modest improvements in gait speed and grip strength in select elderly cohorts 2212. No significant muscle mass gain 32. | Increases muscle mitochondrial markers and biogenesis 2010; no significant changes in macroscopic strength 32. | Weak physical functional translation despite confirmed tissue-level bioenergetic signaling 32. |
| Neurological Access | Currently undergoing phase II trials (MIB-626) to assess efficacy in Alzheimer's and Friedreich's ataxia 3334. | Proven to elevate brain NAD+ at 4 weeks via MRI; safe in Parkinson's patients at doses up to 3g/day 59. | Highly promising frontier; NR has established human proof-of-concept for CNS penetration 5. |
The Senescence Paradox and Oncological Considerations
While the commercial and public narrative surrounding NAD+ precursors centers almost exclusively on "anti-aging" and metabolic rejuvenation, a critical, highly nuanced debate is rapidly evolving within the cellular biology community regarding the potential long-term risks of chronic NAD+ augmentation. This scientific debate focuses heavily on the dynamics of cellular senescence and the associated senescence-associated secretory phenotype (SASP).
The Dual Role of NAD+ Depletion in Aging
As cells accumulate genetic and oxidative damage over a lifespan, they eventually enter senescence - a protective state of irreversible cell cycle arrest that acts as a primary biological defense mechanism against rampant tumorigenesis and cancer development 3513. However, these senescent cells do not simply remain dormant or quietly die. They exhibit remarkably high metabolic activity and actively secrete a pathogenic, highly toxic cocktail of pro-inflammatory cytokines (including IL-6, IL-8, IL-1β, and TNF-α), chemokines, and tissue-remodeling proteases collectively known as SASP 1337.
SASP induces chronic, low-grade systemic tissue inflammation (often termed "inflammaging"), drives paracrine senescence by infecting neighboring healthy tissues with the senescence phenotype, and actively creates an immunosuppressive tumor microenvironment that can paradoxically fuel cancer progression and treatment resistance later in life 133738.
Recent research demonstrates that the well-documented age-related decline in systemic NAD+ may not be exclusively pathological; it may partially serve as an evolutionary, tumor-suppressive physiological adaptation. Overactivation of PARP1 in response to severe DNA damage purposefully and rapidly drains cytosolic NAD+ pools. This mechanism essentially starves damaged cells of the energetic resources required to complete malignant transformation and suppresses excessive cellular proliferation 739.
The Risk of Fueling Senescent Macrophages
Because senescent cells - particularly M2-polarized senescent macrophages - are highly glycolytic, they rely heavily on robust supplies of NAD+ for survival and to meet the massive bioenergetic demands required for continuous SASP production 737. A fundamental, unresolved physiological question currently dividing longevity researchers is whether supplementing with potent precursors like NR or NMN inadvertently provides the raw metabolic fuel required by these highly damaged cells to sustain and amplify their pro-inflammatory SASP output 713.
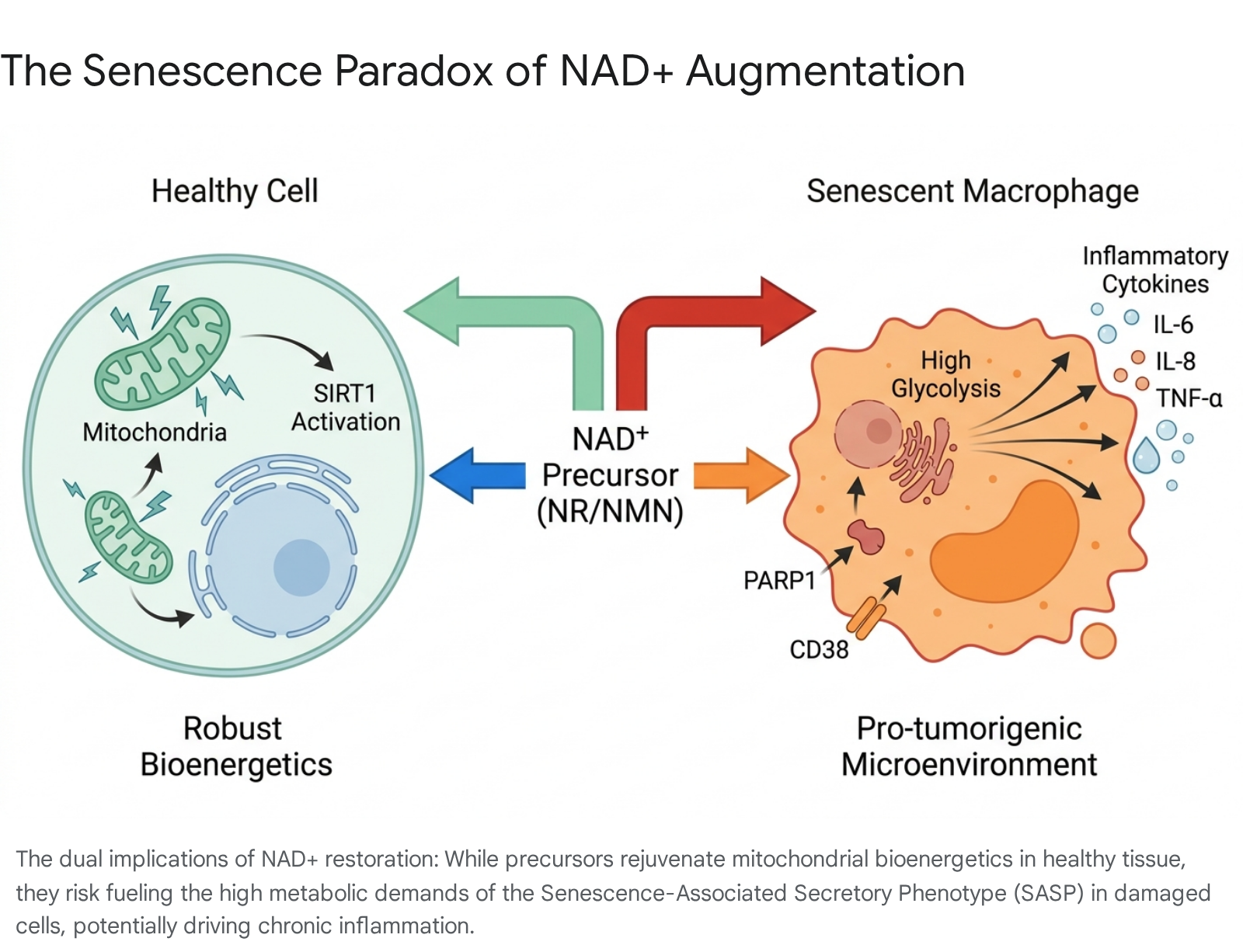
If NAD+ precursors successfully restore mitochondrial function and SIRT1 activation in healthy cells, but simultaneously rescue the metabolic capacity of senescent cells to secrete inflammatory cytokines, the net systemic effect could be highly deleterious over decades of unmonitored use 353839.
Consequently, an emerging consensus in the anti-aging field suggests that generalized, long-term NAD+ precursor supplementation should ideally be administered sequentially, or in direct combination with, senolytics (pharmacological agents that selectively induce targeted apoptosis in senescent cells) 1338. By clearing the biological debris and clearing the high CD38-expressing senescent macrophage populations first, practitioners can safely restore systemic energy networks without inadvertently fueling pre-malignant pathology 133814.
Regulatory Landscape and the Drug Preclusion Clause
The intense scientific debate regarding NMN's efficacy and structural biology has been severely compounded by massive regulatory volatility and legal maneuvering within the United States. While NR has historically maintained a secure, globally recognized status as a generally recognized as safe (GRAS) dietary supplement (most notably commercialized and patented as Tru Niagen) 13, NMN spent several years in strict regulatory limbo.
In late 2022, the U.S. Food and Drug Administration (FDA) formally intervened and excluded NMN from the official definition of a dietary supplement under the Dietary Supplement Health and Education Act (DSHEA) of 1994 414243. This sweeping regulatory action was predicated entirely on the "drug preclusion" clause (specifically 21 U.S.C. § 321(ff)(3)(B)), which explicitly prohibits a substance from being legally marketed as a supplement if it has been formally authorized for investigation as a new drug (IND) and was not previously marketed as a food or supplement prior to that authorization 414344.
The IND in question belonged to Metro International Biotech - a heavily funded pharmaceutical company co-founded by prominent Harvard aging researcher Dr. David Sinclair 3442. MetroBiotech is currently developing MIB-626, a proprietary, pharmaceutical-grade microcrystalline polymorph of NMN, as a highly regulated therapeutic drug targeting specific conditions such as Alzheimer's disease, Friedreich's ataxia, and diabetic kidney disease 333442.
This FDA decision resulted in immediate, widespread commercial market disruption, culminating in major global e-commerce platforms like Amazon systematically removing all NMN dietary products from their storefronts 4315. In response, the Natural Products Association (NPA) and the Alliance for Natural Health (ANH) launched a massive counter-offensive, filing an extensive citizen petition and eventually launching a federal lawsuit against the FDA in the D.C. District Court. Their legal argument rested on the fact that NMN had demonstrably been marketed as a supplement in the U.S. as early as 2017 - well prior to the formal authorization of MetroBiotech's IND 424315.
In a landmark legal and administrative reversal on September 29, 2025, the FDA officially rescinded its prohibition on NMN 4143. In official response letters penned by Dr. Donald Prater, the agency revised its strict interpretation of the "race-to-market" clause. The FDA concluded that its prior stance - that the historical marketing of the supplement must have been technically and completely lawful at the time to establish priority - was an incorrect reading of the congressional statute 414415. Because NMN was demonstrably sold in the U.S. market before the drug investigations formally commenced, it is legally no longer excluded from the definition of a dietary supplement 4115. This pivotal decision has fully restored NMN's commercial legality in the United States, allowing widespread consumer distribution to proceed in parallel with MetroBiotech's highly targeted pharmaceutical clinical trials 4143.
Conclusion
The clinical and academic debate between Nicotinamide Riboside (NR) and Nicotinamide Mononucleotide (NMN) is defined by a vast surplus of preclinical optimism offset by a frustrating deficit of definitive human translation.
From a purely biochemical and pharmacokinetic standpoint, both distinct precursors successfully fulfill their primary pharmacodynamic objective: they safely, reliably, and dose-dependently elevate systemic NAD+ levels in human subjects 202227. While recent, highly robust crossover data strongly suggests that NR may possess a notable kinetic advantage in peak whole-blood absorption efficiency 113, emerging data regarding microbiome-mediated metabolism equalizes their long-term physiological efficacy, rendering them highly comparable in establishing and maintaining steady-state NAD+ elevation 15206.
However, the foundational assumption that elevating blood NAD+ directly and automatically equates to systemic human rejuvenation is simply not supported by the current clinical evidence base. Despite their massive commercial popularity, neither NR nor NMN has demonstrated an ability to reliably reverse major cardiometabolic risk factors - such as severe insulin resistance, clinical obesity, or advanced dyslipidemia - in broad, general human populations 172711. Occasional, isolated signals of biological efficacy in localized muscle function and neuroprotection warrant continued Phase II and III exploration, particularly at aggressive, high doses for targeted disease states like Parkinson's and Alzheimer's 11034.
Ultimately, until the scientific field comprehensively resolves the profound paradox of potentially fueling the senescence-associated secretory phenotype 1337, and successfully executes adequately powered, equimolarly dosed, and biomarker-harmonized multi-center clinical trials 214, the choice between NR and NMN remains a matter of kinetic preference and theoretical delivery mechanics rather than proven, differential clinical superiority.