Human clinical evidence for senolytic drugs
Biological Mechanisms of Cellular Senescence
Cellular senescence is a fundamental biological response characterized by an irreversible arrest of the cell cycle. This terminal state is triggered by a variety of intrinsic and extrinsic stressors, including telomere attrition, radiation-induced DNA damage, oxidative stress, metabolic injury, and oncogene activation 12. While transient cellular senescence plays a highly regulated and beneficial role in embryonic development, tissue repair, wound healing, and tumor suppression, the persistent accumulation of senescent cells over time drives severe tissue dysfunction. This accumulation is now recognized as a primary causal factor in the pathophysiology of organismal aging and a host of chronic, age-related diseases 234.
A defining and pathogenic feature of persistent senescent cells is the Senescence-Associated Secretory Phenotype (SASP). The SASP constitutes a complex, pro-inflammatory secretome comprising various cytokines (such as interleukin-6 and tumor necrosis factor-alpha), chemokines, growth factors, and matrix metalloproteinases 2567. Through the continuous secretion of SASP factors, senescent cells exert potent paracrine effects, inducing a state of chronic, low-grade systemic inflammation frequently termed "inflammaging." Furthermore, these secretions can trigger secondary senescence in neighboring healthy cells, amplifying the biological damage 28. Over decades, this cascading effect contributes to the systemic degradation of tissue homeostasis, intimately linking cellular senescence to conditions such as cardiovascular disease, neurodegeneration, chronic kidney disease, metabolic disorders, and osteoarthritis 699.
To survive in this metabolically hyperactive but non-proliferative state, senescent cells must upregulate specific survival mechanisms to evade programmed cell death. These networks, collectively termed Senescent Cell Anti-Apoptotic Pathways (SCAPs), include the BCL-2 family of proteins, the PI3K/AKT pathway, the p53/p21/p16 signaling axes, and hypoxia-inducible factor (HIF)-1α 81011. By persistently engaging these pathways, senescent cells render themselves highly resistant to the pro-apoptotic signals and immune clearance mechanisms that would normally eliminate damaged tissue 512.
Classification of Senotherapeutics
The rapidly advancing field of geroscience has developed a broad class of pharmacological interventions, known as senotherapeutics, designed to mitigate the deleterious systemic effects of cellular senescence. Based on their distinct mechanisms of action at the cellular level, these interventions are categorized into two primary modalities: senomorphics and senolytics 258.
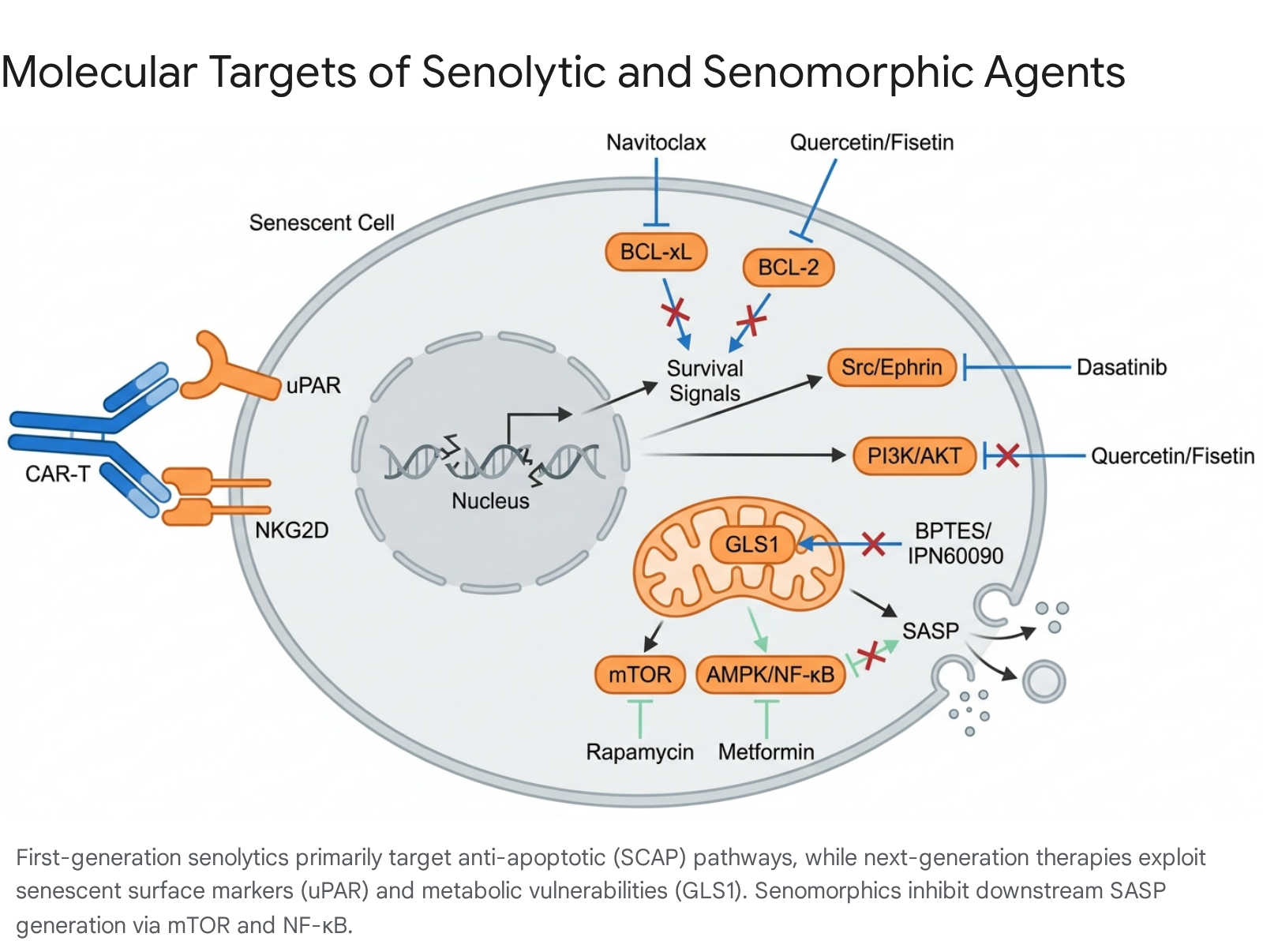
Senomorphics and Inflammatory Modulation
Senomorphics are compounds that modulate the behavior of senescent cells without inducing cell death. Their primary biological objective is to suppress the generation and release of the SASP, thereby reducing localized tissue inflammation and restoring a microenvironment conducive to cellular regeneration 568. By leaving the senescent cells intact, senomorphics preserve the potentially beneficial, tumor-suppressive growth-arrest functions of senescence 6. These agents function by targeting the downstream intracellular signaling pathways responsible for SASP transcription and translation, most notably the mTOR, NF-κB, MAPK, and JAK/STAT pathways 168.
The most extensively studied senomorphics include rapamycin and metformin. Rapamycin, an inhibitor of the mechanistic target of rapamycin (mTOR) complex 1, attenuates the SASP by blocking the translation of IL-1α, a critical upstream regulator of the inflammatory cascade. In murine models, rapamycin has demonstrated considerable efficacy in extending lifespan and decreasing markers of photoaging 68. Metformin, a widely prescribed oral anti-diabetic agent, functions as a senomorphic by activating AMP-activated protein kinase (AMPK). This activation indirectly suppresses NF-κB activity, lowers pro-inflammatory cytokine secretion, and reduces mitochondrial oxidative stress 568.
Despite these benefits, the clinical application of senomorphics faces structural limitations. Because they do not eradicate the underlying dysfunctional cells, senomorphics require continuous, daily administration to maintain SASP suppression 5. Furthermore, achieving geroprotective effects in vivo often requires dosage concentrations that exceed standard clinical parameters. For instance, the concentrations of metformin required to elicit robust senomorphic benefits in animal models (100 mg/kg to 200 mg/kg) are difficult to safely achieve in human clinical trials due to dose-dependent toxicities, such as lactic acidosis and neuropathy associated with vitamin B12 deficiency 4.
Senolytics and Targeted Apoptosis
In stark contrast to senomorphics, senolytic agents are designed to selectively eliminate senescent cells from the body 28. They accomplish this by transiently disabling the specific SCAPs that senescent cells rely upon for survival 12. Because different populations of senescent cells depend on different pro-survival pathways depending on their tissue of origin, senolytics often exhibit narrow cell-type specificity 1013.
The primary clinical advantage of senolytics is their suitability for intermittent, "hit-and-run" dosing regimens. Because senescent cells divide rarely and take weeks or months to reaccumulate to pathological levels, senolytics can be administered in short pulses rather than as daily therapeutics. This pulsed dosing strategy significantly minimizes the risk of off-target toxicity and systemic side effects 381314. Furthermore, by physically eliminating the cells, senolytics prevent the possibility of "senescent bypass," a phenomenon where senescent cells mutate to escape their arrested state, which frequently leads to malignancy 514.
| Pharmacological Feature | Senolytics | Senomorphics |
|---|---|---|
| Primary Mechanism of Action | Induce targeted apoptosis in senescent cells 68. | Suppress the SASP without causing cell death 56. |
| Primary Biological Targets | SCAPs (BCL-2, BCL-xL, PI3K/AKT, Src kinases, p53) 101115. | SASP pathways (mTOR, NF-κB, IL-1α, JAK/STAT) 168. |
| Required Dosing Regimen | Intermittent "hit-and-run" or pulsed dosing 81314. | Continuous, systemic administration required 5. |
| Representative Agents | Dasatinib, Quercetin, Fisetin, Navitoclax 81215. | Rapamycin, Metformin, Apigenin, Luteolin 68. |
| Primary Clinical Advantages | Eliminates the source of damage; prevents "senescent bypass"; low dosing burden 514. | Preserves the beneficial growth-arrest and tumor-suppressive functions of senescence 6. |
First-Generation Senolytic Agents
The first generation of senolytic drugs largely consists of repurposed oncology chemotherapeutics and natural, plant-derived flavonoid polyphenols. Preclinical evidence strongly supports their ability to clear senescent cells, mitigate frailty, reduce chronic inflammation, and extend healthspan across diverse animal models 213.
Dasatinib and Quercetin
Dasatinib and Quercetin were among the initial compounds identified to possess potent senolytic activity. In clinical and preclinical settings, they are almost exclusively administered as a combined cocktail (referred to as D+Q) to achieve a broad-spectrum senolytic effect. This combinatorial approach is necessary because the survival of different senescent cell types is governed by different, distinct SCAPs 1013.
Dasatinib is a potent, FDA-approved tyrosine kinase inhibitor originally developed for the treatment of chronic myeloid leukemia 1316. In the context of senolysis, dasatinib selectively targets Src family kinases and ephrin (EFN) dependence receptors 210. While highly effective at clearing specific senescent populations, chronic administration of dasatinib carries severe toxicity risks, including a high cumulative incidence of pleural effusion and severe hematologic toxicities such as grade 3-4 neutropenia and disproportionate bleeding 13. Dasatinib is also a substrate of the CYP3A4 enzyme, requiring strict contraindication with common metabolic inhibitors 13. The brief, intermittent dosing protocols utilized in senolytic trials are specifically designed to circumvent these long-term toxicities 713.
Quercetin is a naturally occurring redox-active polyphenolic flavonoid. It acts by targeting the BCL-2 family of anti-apoptotic proteins, the PI3K/AKT pathway, and hypoxia-inducible factor (HIF)-1α 1016. Quercetin possesses strong prooxidant activities that are uniquely amplified by transition metals such as copper and iron. Because senescent cells naturally sequester and accumulate high intracellular levels of these transition metals, they become particularly vulnerable to quercetin-induced oxidative damage, triggering apoptosis 13.
Fisetin
Fisetin is a naturally occurring flavonoid structurally related to quercetin. However, in comprehensive screening panels of flavonoid polyphenols, fisetin demonstrated significantly greater senolytic potency and broader biological activity than quercetin 31315. Highly hydrophobic, fisetin easily penetrates cell membranes to accumulate intracellularly, where it exerts both antioxidant and prooxidant effects depending on the cellular microenvironment 12.
Fisetin selectively induces apoptosis in specific senescent populations - such as human umbilical vein endothelial cells (HUVECs) and adipose tissue cells - by targeting BCL-2 and PI3K/AKT pathways, but it shows little to no senolytic activity in human lung fibroblasts or primary preadipocytes 31215. Unlike repurposed synthetic oncology drugs, fisetin boasts a highly favorable safety profile with low systemic toxicity, though its plasma terminal half-life in murine models is notably brief (just over three hours) 121315. In pivotal preclinical studies, the acute or intermittent administration of fisetin to wild-type mice late in life successfully restored tissue homeostasis, reduced age-related pathology, and extended both median and maximum lifespan, establishing it as a primary candidate for human translation 317.
Navitoclax and BCL-2 Inhibition
Navitoclax (ABT-263) is a highly targeted synthetic inhibitor of the BCL-2 protein family, specifically neutralizing BCL-2, BCL-xL, and BCL-w 21215. While navitoclax exhibits remarkably strong clearance of senescent cells across various in vitro and in vivo models, its clinical translation as a systemic senolytic has been severely hampered by dangerous on-target hematological toxicities. Specifically, the inhibition of BCL-xL invariably induces severe, dose-dependent thrombocytopenia due to the reliance of healthy platelets on BCL-xL for survival 21215.
To bypass these limitations, researchers have developed specialized prodrug variants. For example, Nav-Gal is a modified version of navitoclax designed to remain inert until it encounters cells with elevated levels of senescence-associated β-galactosidase (SA-β-gal). Upon cleavage by this enzyme, the active navitoclax is released exclusively within the senescent cell. In preclinical lung cancer models, Nav-Gal achieved 40% higher clearance of senescent cells while simultaneously reducing platelet-related side effects by 70% 15.
Clinical Trial Evidence for First-Generation Senolytics
The transition of senolytics from robust preclinical animal models to human clinical trials represents a critical and challenging phase in geroscience. Initial human studies have rigorously focused on establishing safety profiles, central nervous system penetrance, and preliminary efficacy. To satisfy regulatory frameworks, these trials have largely targeted specific, severe conditions characterized by localized senescence rather than systemic physiological aging.
Idiopathic Pulmonary Fibrosis and Diabetic Kidney Disease
The earliest proof-of-concept human trials for the D+Q combination focused on severe, senescence-associated terminal pathologies. In a landmark study involving patients with idiopathic pulmonary fibrosis (IPF) - a fatal fibrotic lung disease fundamentally driven by cellular senescence - a brief regimen of just nine oral doses of D+Q administered over three weeks yielded marked, clinically significant improvements in physical performance. Trial participants demonstrated measurable gains in their 6-minute walk distance, walking speed, chair rise ability, and overall short physical performance battery scores 13161719.
Subsequent open-label Phase 1 pilot studies in patients with diabetic kidney disease (DKD) provided the critical first direct evidence of senolytic target engagement in human tissue. Following an acute 3-day oral course of dasatinib (100 mg) and quercetin (1,000 mg), researchers obtained subcutaneous adipose tissue biopsies 11 days post-treatment. The biopsies revealed profound reductions in the senescent cell burden, objectively quantified by marked decreases in p16INK4a- and p21CIP1-expressing cells, significantly reduced senescence-associated β-galactosidase (SA-β-gal) activity, and a decline in adipocyte progenitors with limited replicative potential 101319.
Alzheimer Disease and Cognitive Impairment
The definitive role of cellular senescence in driving neuroinflammation and neurodegeneration has prompted multiple clinical trials investigating senolytics for Alzheimer's disease (AD) and mild cognitive impairment (MCI). The Phase 1/2 SToMP-AD (Senolytic Therapy to Modulate Progression of Alzheimer's Disease) trial was explicitly designed to evaluate the safety, feasibility, and central nervous system (CNS) penetrance of D+Q in older adults with early-stage AD who are tau PET positive 1819.
The trial protocol involved administering 100 mg of dasatinib and 1,000 mg of quercetin for two consecutive days every 15 days, spanning a 12-week intervention period 141920. Crucially, cerebrospinal fluid (CSF) analysis demonstrated that dasatinib successfully penetrated the blood-brain barrier within 60 to 90 minutes of oral dosing, whereas quercetin was undetectable in the CNS 19. While the regimen was deemed highly safe and tolerable, yielding no serious adverse events, the 12-week therapy produced no significant clinical changes in cognition, Aβ markers, tau pathology, or circulating senescence biomarkers 1920. This lack of cognitive improvement aligns with contradictory preclinical mouse data, where D+Q improved memory in certain tau models (rTg4510) but failed to reduce senescent markers or improve cognitive behaviors in others (P301S mice) 19.
However, other small pilot studies examining intermittent D+Q in older adults with MCI and slow gait have reported more nuanced signals. While major cognitive overhauls were absent, researchers observed a non-significant overall increase in Montreal Cognitive Assessment (MoCA) scores (mean 1.0 point increase), with a statistically significant 2.0 point increase in the subgroup with the lowest baseline MoCA scores 71415. Importantly, these cognitive improvements inversely correlated with a decrease in circulating tumor necrosis factor-alpha (TNF-α) levels, heavily suggesting that cognitive benefits may be mediated indirectly through the modulation of the systemic SASP 71415.
Adipose Tissue and Metabolic Function
Researchers are also evaluating the capacity of senolytics to rescue systemic metabolic dysfunction driven by adipose tissue senescence. In patients suffering from the lethal combination of heart failure with reduced ejection fraction (HFrEF) and type 2 diabetes mellitus (T2DM), subcutaneous adipose tissue becomes highly dysfunctional 21. Senescent microvascular endothelial cells within this fat secrete high levels of IL-6, which impairs glucose uptake in neighboring healthy adipocytes. Ongoing trials are investigating whether digoxin - a traditional heart failure medication newly identified to possess senolytic properties against endothelial cells - can selectively clear these cells and restore insulin sensitivity and glucose homeostasis in human subjects 21.
In parallel preclinical models highly relevant to human translation, researchers tested intermittent D+Q administration combined with a 10% caloric restriction in middle-aged nonhuman primates over six months. The combined intervention yielded dramatic improvements across multiple systemic health parameters. The primates exhibited significant reductions in adipose tissue senescence markers, circulating PAI-1, and MMP-9, alongside robust improvements in blood urea nitrogen and intestinal barrier function. Furthermore, the therapy induced profound anti-inflammatory shifts in immune cell profiles, reducing white blood cell counts and significantly lowering glycosylated hemoglobin A1c (HbA1c) 22.
Epigenetic Aging Acceleration
Recent longitudinal human data have introduced unforeseen biological complexities regarding how senolytic administration impacts molecular markers of biological aging. A highly scrutinized Phase I pilot study involving 19 participants evaluated the impact of 6 months of intermittent D+Q therapy on DNA methylation (DNAm) clocks. Paradoxically, rather than demonstrating biological rejuvenation, the study found significant increases in epigenetic age acceleration as measured by first-generation and mitotic epigenetic clocks. This acceleration was observed alongside a notable decrease in telomere length at the 3-month and 6-month intervals 161723.
Building upon these confounding findings, researchers initiated a subsequent arm of the study that added the flavonoid Fisetin to the regimen (forming a DQF cocktail). Remarkably, the addition of Fisetin mitigated the adverse epigenetic effects; the DQF group exhibited only non-significant increases in epigenetic age acceleration. Furthermore, extensive immune profiling unveiled notable differences in immune cell proportions between the D+Q and DQF treatment groups. This suggests that the divergent patterns in epigenetic clock evolution may be deeply tied to how specific senolytic combinations transiently alter the immune architecture during the clearance of senescent cells 16172324.
| Clinical Trial / Target Condition | Pharmacological Intervention | Key Findings and Clinical Outcomes | Trial Status |
|---|---|---|---|
| Diabetic Kidney Disease (DKD) 1919 | D+Q (3-day oral course) | Provided the first direct human evidence of target engagement. Significantly reduced p16/p21 and SA-β-gal expression in adipose tissue. | Completed |
| Idiopathic Pulmonary Fibrosis 1619 | D+Q (9 doses over 3 weeks) | Meaningfully improved 6-minute walk distance, walking speed, and chair rise ability in terminal patients. | Completed |
| SToMP-AD (Alzheimer's Disease) 181920 | D+Q (Pulsed over 12 weeks) | Dasatinib successfully penetrated the CSF; quercetin did not. Proven safe, but yielded no significant cognitive or biomarker improvements. | Published / Ongoing |
| Mild Cognitive Impairment & Slow Gait 714 | D+Q (Intermittent pulsed) | Deemed highly safe. A correlated drop in systemic TNF-α levels was linked to slight MoCA score improvements in severe subgroups. | Completed |
| Epigenetic Aging & Methylation Clocks 16172324 | D+Q versus DQF (6 months) | D+Q paradoxically accelerated first-generation DNAm clocks. The addition of Fisetin (DQF) mitigated this acceleration and altered immune subsets. | Published |
Advanced and Next-Generation Senolytics
The recognized limitations of first-generation senolytics - such as off-target toxicities, variable tissue penetrance, poor bioavailability, and incomplete clearance of diverse senescent phenotypes - have rapidly accelerated the development of next-generation biological modalities designed for absolute precision.
Chimeric Antigen Receptor T-Cell Therapies
Initially revolutionized for the treatment of refractory hematological malignancies, Chimeric Antigen Receptor (CAR) T-cell therapy is now being aggressively repurposed to identify and eliminate senescent cells by targeting highly specific senescence-associated surface antigens .
The primary biological target currently under investigation is the urokinase plasminogen activator receptor (uPAR). Under healthy physiological conditions, uPAR expression is minimal; however, it is highly upregulated on the surface of senescent cells in naturally aged tissues, fibrotic models, and the microenvironment of certain solid tumors 2825. In groundbreaking studies involving naturally aged (20-month-old) murine models and non-human primates, infusions of anti-uPAR CAR T cells successfully depleted senescent hematopoietic stem cells and eliminated highly fibrotic tissue. Physiologically, this translated to significantly improved exercise capacity, enhanced epithelial integrity in intestinal regeneration, and the reversal of metabolic dysfunctions, such as impaired glucose tolerance induced by high-fat diets 262728. Notably, these robust results were achieved without inducing cytokine release syndrome (CRS) or other severe CAR T-related toxicities . Furthermore, researchers have developed an anti-NKG2D CAR T-cell variant specifically designed to target senescent cells induced by replicative and oncogenic stress .
The profound therapeutic advantage of senolytic CAR T cells is their persistence. An infusion of engineered T cells acts as a "living drug" capable of long-term immune surveillance, permanently overcoming the need for the repeated, sustained dosing regimens required by small-molecule senolytics 2627. However, the translation of CAR T therapies to human aging faces severe biological hurdles. Foremost is immunosenescence - the natural, age-related functional decline of the endogenous immune system. Harvesting, engineering, and expanding viable, highly active autologous T cells from elderly, frail patients presents a massive logistical and biological challenge that currently limits widespread clinical applicability .
Glutaminase 1 Inhibitors
Glutaminase 1 (GLS1) inhibitors represent a highly novel, purely metabolic approach to senolysis. Senescent cells inherently suffer from structural lysosomal membrane damage, which results in the leakage of hydrogen ions and subsequent severe intracellular acidosis. To survive this toxic internal environment, senescent cells become heavily reliant on enhanced glutaminolysis, utilizing the GLS1 enzyme to convert glutamine into glutamate and ammonia. The resulting ammonia acts to neutralize the acidic environment, ensuring cellular survival 293035.
A landmark 2021 study in Science demonstrated that pharmacologically inhibiting GLS1 (using compounds such as BPTES or IPN60090) selectively triggered senolysis. In aged murine models, this intervention ameliorated age-associated organ dysfunction, resolved atherosclerosis, and improved metabolic parameters 2930. Because GLS1 inhibitors like CB-839 are already progressing through phase I clinical trials for the treatment of solid tumors with minimal significant side effects, they were rapidly highlighted as highly promising, readily translatable candidates for systemic human senolysis 2930.
However, this metabolic approach is currently facing a severe reproducibility crisis. A comprehensive, cross-laboratory study published in 2026 stringently re-tested the senolytic efficacy of GLS1 inhibitors alongside anti-PD-1 antibodies. Contrary to the initial reports, the independent researchers found that GLS1 inhibition completely failed to significantly reduce the burden of p16INK4a-positive senescent cells in aged tissues, nor did it yield any measurable improvements in aging-related health parameters 31. The failure to reproduce these foundational findings has injected necessary calibrated uncertainty into the field, questioning the generalizability of targeting metabolic vulnerabilities across highly heterogenous senescent cell populations 31.
Peptide and Antibody Interventions
Emerging biotechnology ventures are focusing on disrupting specific, pathological protein-protein interactions that are entirely unique to senescent states. * FOXO4-p53 Interacting Peptides: Companies such as Cleara Biotech are developing fourth-generation D-amino acid peptides designed to target cellular "scarring." In senescent cells, the transcription factor FOXO4 sequesters phosphorylated p53 (specifically at Serine 46), trapping it in the nucleus and preventing it from inducing natural apoptosis. Peptides like CL04183 are designed to physically disrupt this specific FOXO4-p53 interaction. By freeing p53, the peptide selectively forces the scarred, senescent cells into immediate apoptosis. Having demonstrated efficacy in models of neurodegeneration, liver fibrosis, and triple-negative breast cancer, CL04183 is progressing through late-stage preclinical GLP-Tox studies, with Phase 1a/1b human trials slated to commence in 2026 3839. * Galectin-9 Antibodies: LYT-200 is a fully human IgG4 monoclonal antibody specifically targeting Galectin-9, an immunosuppressive glycan-binding protein heavily implicated in cellular survival and immune interference. Gal-9 is significantly upregulated in highly senescent microenvironments and various malignancies. Currently advancing through Phase 1/2 clinical trials (NCT04666688) for the treatment of solid tumors and relapsed/refractory acute myeloid leukemia (AML), LYT-200 has demonstrated a favorable clinical safety profile and is being closely monitored by researchers for its potential expansion into broader senotherapeutic applications 323342.
The Mouse-to-Human Translational Gap
The largest impediment in the advancement of modern geroscience is the profound translational gap between laboratory murine models and human patients 634. While senolytics routinely yield remarkable, easily quantifiable healthspan extensions in mice, translating these outcomes to humans has proven exceedingly difficult due to fundamental structural, biological, and physiological divergences between the species.
Telomere Dynamics and Replicative Senescence
A primary biological divergence between humans and mice lies in fundamental telomere biology. In humans, chromosomal telomeres are relatively short (ranging from 5 to 15 kilobases). More importantly, the expression of telomerase reverse transcriptase (hTERT) - the enzyme responsible for rebuilding telomeres - is heavily repressed in the vast majority of human somatic tissues. Consequently, human cells experience progressive, unavoidable telomere shortening with each mitotic division. This establishes telomere attrition as a strict biological clock enforcing the Hayflick limit, ultimately driving replicative senescence as a mechanism to prevent tumor progression 453536.
Conversely, standard laboratory mice possess extremely long telomeres (spanning 50 to 100 kilobases) and constitutively express mouse telomerase (mTERT) across most adult somatic tissues 4535. Because laboratory mice do not naturally exhibit telomere-mediated replicative aging, their cells stop dividing in vitro due to completely different stress responses long before any appreciable telomere erosion occurs. Therefore, standard mice are fundamentally flawed models for investigating human cellular senescence 4535. To bridge this immense gap, bioengineers have recently created "HuT mice" (mice with humanized telomeres). By genetically modifying murine embryonic stem cells to feature human regulatory sequences, these mice exhibit shorter telomeres and restricted telomerase expression, offering a vastly improved, physiologically relevant model for studying organismal aging 48.
Physiological Complexity and Heterogeneity
Human aging is remarkably heterogeneous compared to the highly controlled, inbred environments of laboratory rodents 634. Senescent cells in humans exhibit extreme phenotypic variability depending on their tissue of origin and the initiating stressor. Thus, a senolytic compound that successfully clears senescent fibroblasts in a mouse model may completely fail to clear senescent endothelial cells or neurons in a human 634.
Furthermore, pharmacokinetics differ wildly. Many first-generation, plant-derived senolytics (such as quercetin and fisetin) suffer from very poor oral bioavailability, rapid hepatic metabolism, and severely limited systemic retention in the human body. This complicates all clinical efforts to achieve the sustained intracellular concentrations required for effective senolysis, particularly when attempting to cross complex physiological boundaries like the blood-brain barrier 6.
Clinical Trial Design Constraints
Translating senolytics into standard clinical practice is heavily constrained by modern trial design logistics. Because "aging" is not classified as a curable disease by global regulatory agencies (like the FDA or EMA), clinical trials cannot be designed to test "anti-aging" therapeutics directly. Instead, trials must target specific, severe, age-associated pathologies - such as idiopathic pulmonary fibrosis, osteoarthritis, or Alzheimer's disease 38.
This regulatory environment necessitates identifying specific clinical cohorts that possess a sufficiently high localized burden of senescent cells to detect a statistically significant clinical benefit. Senolytics fundamentally require a target to destroy; therefore, they will not produce measurable outcomes in younger individuals or those with a low baseline senescent burden, complicating the pursuit of purely preventative, prophylactic anti-aging therapies 3437. Moving forward, the successful clinical application of senolytics will require the development of highly sensitive, universal molecular biomarkers. Accurately quantifying the precise senescent cell burden before and after intervention will allow clinicians to personalize intermittent dosing protocols, tailoring interventions to individual aging trajectories to safely extend human healthspan 21137.