Hormesis and lifespan extension
Hormesis describes a highly conserved, biphasic dose-response phenomenon wherein exposure to low doses of an otherwise toxic or disruptive stressor induces a beneficial, adaptive response in biological systems. In the context of biological aging, the hormesis paradigm provides a mechanistic framework explaining why mild, intermittent stressors - such as physical exertion, thermal extremes, caloric scarcity, and exposure to specific plant-derived secondary metabolites - can enhance cellular resilience, delay age-related degeneration, and extend the functional healthspan.
Rather than viewing aging exclusively as a linear trajectory of passive molecular wear-and-tear, the hormetic model suggests that the rate of functional decline is heavily dictated by the robustness of an organism's endogenous stress-response networks. Over time, as these adaptive mechanisms are progressively down-regulated, the body experiences a shrinkage of its "homeodynamic space" - defined as the physiological capacity to absorb internal and external metabolic fluctuations without sustaining permanent cellular damage 12. Deliberately triggering latent repair pathways through acute hormetic conditioning expands this homeodynamic space, effectively re-engaging the organism's evolutionary maintenance machinery.
Conceptual Foundations of the Hormetic Dose-Response
The defining characteristic of hormesis is its non-linear, biphasic curve, which challenges traditional linear no-threshold (LNT) models historically utilized in toxicology and risk assessment 34. The specific shape of the hormetic curve depends on the biological endpoint being measured. In an inverted U-shape model representing longevity, systemic fitness, or cellular growth, the physiological response peaks in a stimulatory "hormetic zone" before declining through the zero-effect axis into a toxic zone of inhibition 24. Conversely, when endpoints track adverse outcomes such as disease incidence or mutation rates, the curve manifests as a U-shape, dipping significantly below the control baseline at low doses before rising sharply at higher exposures 4.
Biological plasticity imposes strict quantitative limits on the magnitude of the hormetic response. Across tens of thousands of measured endpoints in diverse species, the maximal stimulatory response consistently caps at approximately 30% to 60% above control baseline levels 567. This limit reflects a fundamental evolutionary constraint. An organism cannot perpetually over-allocate its finite metabolic resources to somatic repair pathways without compromising other essential functions, such as reproduction and immediate survival 78.
A critical distinction within this framework is the differentiation between hormetic stress and toxic stress. Hormetic stress is characterized by its acute, mild, and intermittent nature, which provides the organism with adequate recovery periods to enact compensatory transcription and up-regulate cytoprotective enzymes 91011. In contrast, toxic stress - whether physical, chemical, or psychological - is severe or chronic. Prolonged exposure prevents a return to basal homeostasis, ultimately overwhelming the repair machinery, inducing allostatic load, and accelerating the macroscopic tissue damage associated with biological aging 910.
Primary Effectors of Cellular Stress Resilience
The translation of external environmental stressors into systemic life-extending adaptations relies on a matrix of highly conserved intracellular signaling networks. These interconnected pathways detect shifts in energetic demand, thermal stability, and redox balance, orchestrating a shift from anabolic growth to somatic maintenance.
Mitohormesis and Reactive Oxygen Species Signaling
Historically, the free radical theory of aging posited that reactive oxygen species (ROS) - volatile byproducts of mitochondrial oxidative phosphorylation - were purely destructive agents driving age-related macromolecular damage 511. The hormesis paradigm substantially revises this model through the concept of mitohormesis. Under mitohormetic conditions, mild, transient increases in mitochondrial ROS act as indispensable secondary messenger molecules rather than mere toxic byproducts 5611.
When cells are subjected to metabolic or physical stressors, mitochondrial ROS production briefly elevates by approximately 15% to 35% above the basal resting state 6. This acute oxidative burst triggers a retrograde signaling cascade from the mitochondria to the nucleus, commanding the up-regulation of endogenous antioxidant defense systems. This process culminates in a durable state of stress resistance, ultimately reducing chronic oxidative stress well below the original baseline over the long term. Experimental evidence indicates that if this initial ROS signal is artificially suppressed - such as through the administration of high-dose, non-specific antioxidant supplements - the downstream adaptive benefits of the stressor are frequently blunted or entirely abolished 111213.
The Nrf2 Pathway and the Vitagene Network
The primary transcriptional effector of ROS-mediated signaling is Nuclear factor erythroid 2-related factor 2 (Nrf2). Under basal, non-stressed conditions, Nrf2 is sequestered in the cytoplasm by its repressor protein, Keap1, which targets it for continuous proteasomal degradation 61014. However, the introduction of mild electrophilic or oxidative stress oxidizes specific sensory cysteine residues on Keap1, altering its conformation and releasing Nrf2. Once liberated, Nrf2 translocates to the nucleus and binds to Antioxidant Response Elements (AREs) across the genome, initiating the massive transcription of cytoprotective genes 614.
This downstream target network is frequently termed the "vitagene" network. It includes the production of heat shock proteins (such as Hsp32 and Hsp70), thioredoxin, and various sirtuin protein systems 56. These gene products act collectively to stabilize unfolded proteins, detoxify accumulating metabolic byproducts, and restore the cellular redox balance. Because Nrf2 sits at the intersection of numerous stress inputs, it represents a centralized molecular hub through which diverse hormetic stressors confer cross-modal resilience against age-related pathologies 14.
AMPK, mTOR, and Autophagic Regulation
Autophagy is a catalytic cellular self-cleaning process responsible for degrading and recycling damaged organelles, oxidized lipids, and misfolded proteins. During biological aging, autophagic efficiency progressively declines, leading to the toxic accumulation of protein aggregates that drive neurodegenerative and metabolic diseases 1516. Mild environmental stress systematically up-regulates autophagic flux through the dual modulation of 5' AMP-activated protein kinase (AMPK) and the mechanistic target of rapamycin (mTOR) 161718.
When intracellular ATP levels drop as a result of energetic stress - such as during fasting or exercise - AMPK acts as an acute energy sensor. Activated AMPK immediately halts energy-consuming anabolic processes by inhibiting mTOR, simultaneously initiating autophagic recycling pathways to generate alternative substrates for energy production 171819. Crucially, the autophagy induced by an acute hormetic stressor not only clears the immediate damage caused by that specific stress event but also incidentally clears coincidental, pre-existing age-related damage, driving a net rejuvenation of the cellular environment 21516.
Physiological Stressors and Organismal Adaptation
The biological adaptations that mitigate the hallmarks of aging can be activated through multiple distinct external modalities. While these stressors differ vastly in their immediate physiological impact, they converge on the same downstream longevity-promoting networks.
| Hormetic Modality | Primary Physiological Stressor | Key Molecular Effectors | Target Age-Related Pathology Mitigated |
|---|---|---|---|
| Physical Exercise | ATP depletion, mechanical shear, mild ROS burst | AMPK, PGC-1α, IL-6, Nrf2 | Sarcopenia, cardiovascular disease, metabolic syndrome 121820 |
| Thermal (Cold) | Norepinephrine surge, ATP demand for thermogenesis | RBM3, SIRT1, UCP1, Nrf2 | Neurodegeneration, immune decline, metabolic inflexibility 192122 |
| Thermal (Heat) | Protein unfolding, mild acute inflammation | Heat Shock Proteins (Hsp70, Hsp32), FOXO | Proteotoxicity, cardiovascular stiffness, aggregate accumulation 2523 |
| Cognitive Stress | Elevated neuronal metabolic demand, environmental complexity | BDNF, CREB, neuronal autophagy | Age-related cognitive impairment (ARCI), dementia 101516 |
Physical Exercise as an Aging Mimetic
Physical exercise remains the most universally validated hormetic intervention in human populations. During moderate-to-vigorous physical activity, skeletal muscle tissues experience transient hypoxia, severe ATP depletion, structural micro-tears, and massive ROS generation 1213. Paradoxically, this acute physiological disruption closely mimics the structural and metabolic deterioration seen in advanced biological aging.
By simulating a state of rapid functional decline, exercise acts as an "aging mimetic," forcing the body into a state of dramatic overcompensation 24. This stress triggers the up-regulation of Peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α), the master regulator of mitochondrial biogenesis, which expands the cellular energy grid 1218. Furthermore, contracting muscles function as an endocrine organ, releasing myokines such as interleukin-6 (IL-6). While chronically elevated IL-6 is a marker of pathological inflammation, the acute, exercise-induced pulse of IL-6 exerts systemic anti-inflammatory effects and promotes structural tissue remodeling 1218. Beyond skeletal muscle, exercise also actively preserves chromosomal stability by altering telomerase activity and enhancing satellite cell content, counteracting telomere attrition, a primary hallmark of cellular aging 18.
Thermal Conditioning: Cold Shock and Heat Stress
Exposure to temperature extremes rapidly disrupts homeostasis, demanding immediate metabolic compensation. Acute cold exposure triggers an instantaneous release of norepinephrine, which sharpens neurological focus, reduces systemic inflammation, and activates uncoupling protein 1 (UCP1) in brown adipose tissue (BAT) to initiate non-shivering thermogenesis 19. Molecular analyses demonstrate that cold stress induces the expression of RNA-binding motif protein 3 (RBM3), a cold-shock protein tightly linked to neuroprotection and synaptic regeneration in animal models 19. However, clinical data emphasizes the necessity of the hormetic recovery window. While short-term, intermittent cold exposure activates Nrf2 and enhances intestinal barrier integrity, chronic, uninterrupted cold exposure induces severe endoplasmic reticulum stress. This chronic state actively inhibits SIRT1, driving systemic inflammation and destroying colonic epithelial tight junctions 2122.
Repeated mild heat stress (RMHS), such as sauna utilization, induces a highly divergent profile of hormetic adaptation. Heat stress physically forces the unfolding of cellular proteins, which poses an immediate threat of proteotoxicity. This thermal shock rapidly activates the transcription of Heat Shock Proteins 21623. These molecular chaperones surround denatured proteins, facilitating their proper refolding, while simultaneously targeting irreversibly damaged proteins for rapid proteasomal degradation 223. In isolated human skin fibroblasts and keratinocytes, periodic RMHS successfully prevents the accumulation of damaged macromolecules and extends replicative lifespan 23. Additionally, emerging research indicates that atmospheric physical stressors, such as exposure to Negative Air Ions (NAIs) generated via cold atmospheric plasma, can elicit similar cross-modal hormetic responses, enhancing glutathione metabolism, reducing lipid peroxidation, and improving mitochondrial efficiency in blood and hepatic tissues 25.
Cognitive Stress and Neuronal Maintenance
The mammalian brain also undergoes structural hormetic adaptation in response to demanding stimuli. Processing complex digital information, mastering new skills, or navigating an enriched environment exerts a continuous, mild energetic stress on neuronal networks 1516. This "information-that-requires-action" acts as a cognitive stressor, triggering the localized expression of brain-derived neurotrophic factor (BDNF) and heavily up-regulating neuronal autophagic flux 1015.
Over the lifespan, this cognitive stress expands neural connectivity and builds synaptic resilience, protecting the brain against age-related cognitive impairment (ARCI) and neurodegenerative pathology. Certain evolutionary frameworks propose a neuron-germline conflict, wherein cognitive stress forces the organism to preferentially allocate its finite repair resources to the brain to process environmental complexity. This localized maintenance effectively extends somatic healthspan, albeit theoretically at a slight metabolic cost to reproductive capacity 1526.
Nutritional Hormesis and the Limitations of Caloric Restriction
Caloric restriction (CR) - defined as a sustained 10% to 40% reduction in ad libitum caloric intake without incurring essential nutrient malnutrition - is the most robust non-genetic intervention documented to extend maximum lifespan across multiple model organisms, ranging from yeast to non-human primates 5272829. The underlying mechanism is classically hormetic: an imposed energy deficit signals severe environmental scarcity, prompting the organism to suspend reproductive and growth programs in favor of investing heavily in somatic stress resistance 2728.
This energetic stress fundamentally alters intracellular metabolism. CR elevates the cellular ratio of NAD+ to NADH, an enzymatic shift that directly activates the sirtuin family of NAD-dependent deacetylases, particularly SIRT1. Sirtuins modulate global chromatin structure, enhance DNA damage repair efficiency, and aggressively inhibit pro-inflammatory pathways such as NF-κB 52328. Concurrently, CR blunts the insulin and insulin-like growth factor 1 (IGF-1) signaling axis, which leads to widespread inhibition of mTOR and the activation of longevity-associated transcription factors such as FOXO, driving deep systemic autophagy 52728.
Skeletal Pathologies in Lean Populations
Despite definitive longevity success in animal models, the translation of long-term caloric restriction to human populations has revealed significant, potentially dangerous clinical complexities. The CALERIE trial (Comprehensive Assessment of Long-term Effects of Reducing Intake of Energy) evaluated a 12% reduction in daily caloric intake over two years in healthy, non-obese human adults 2930. Participants exhibited a 10.4% sustained weight reduction, significantly decreased circulating inflammatory markers, and a demonstrably slower pace of biological aging as measured by composite epigenetic clocks 29. Crucially, while absolute muscle mass declined due to the energy deficit, lower-body muscle strength was fully preserved, indicating a profound improvement in specific muscle quality and underlying mitochondrial density 29.
However, the trial data and subsequent longitudinal assessments highlighted the severe physiological limits of CR in non-obese humans, where the intervention frequently crosses the boundary from hormetic stimulus into toxic structural deterioration. In lean subjects, sustained negative energy balance leads to clinically significant reductions in bone quantity and bone mineral density (BMD) at the femoral neck, lumbar spine, and total hip 303131.
Furthermore, human CR models induce a paradoxical shift in adipose storage. Unlike peripheral white adipose tissue, which is mobilized and burned during starvation, bone marrow adipose tissue (BMAT) actively expands during caloric restriction 3032. This expanded BMAT releases high levels of Receptor Activator of Nuclear Factor κB Ligand (RANKL), an inflammatory cytokine that aggressively stimulates bone-resorbing osteoclasts while simultaneously suppressing the formation of new bone-building osteoblasts 30. Consequently, while prolonged CR undoubtedly extends metabolic healthspan and suppresses systemic inflammation, its induction of severe osteopenia and potential sarcopenia in lean individuals severely complicates its viability as a broad, unsupervised human anti-aging intervention 3031.
Xenohormesis and Phytochemical Signaling
Because chronic caloric restriction is difficult to sustain behaviorally and carries severe structural risks for lean populations, pharmacological research has increasingly sought molecules capable of mimicking the physiological state of starvation or physical stress without requiring actual physical or nutritional deprivation. This search relies heavily on the evolutionary biological concept of "xenohormesis" 3334.
The xenohormesis hypothesis posits that heterotrophs (animals and fungi) have evolved the highly specific ability to sense and respond to the chemical stress signals produced by autotrophs (plants). Because plants are physically stationary, they cannot flee harsh environmental conditions; they must survive extremes - such as drought, severe UV radiation, microbial infection, or nutrient deprivation - by synthesizing a complex array of secondary defense metabolites, notably polyphenols 333435. When animals consume these environmentally stressed plants, these secondary metabolites bind to highly conserved cellular receptors (such as sirtuins and AMPK) within the animal's tissues, triggering preemptive defense and repair responses. The animal effectively "piggybacks" on the plant's stress response, gaining survival benefits from an impending environmental deterioration before it actually impacts the animal directly 273336.
Traditional Diets as Xenohormetic Vectors
The xenohormetic framework provides a compelling biological explanation for the longevity benefits consistently observed in human populations consuming regional, plant-rich traditional diets. Phytochemicals such as resveratrol (found in environmentally stressed grape skins), quercetin (in onions and apples), and epicatechin (in cocoa) do not serve as direct antioxidants in the trace quantities typically consumed; rather, they act as mild xenobiotic stressors that activate Phase I and II cellular detoxification pathways and powerfully up-regulate the Nrf2 vitagene network 2737.
Epidemiological evidence from populations adhering to the Mediterranean and the New Nordic diets strongly supports this signaling dynamic. The New Nordic diet, which relies heavily on root vegetables, cruciferous plants, and highly stressed berries grown in cold, harsh climates, exposes the consumer to exceptionally high levels of cold-stress phytochemicals. Adherence to this diet is clinically associated with a 22% lower risk of premature all-cause mortality, a 58% lower risk of developing non-alcoholic fatty liver disease, and significant reductions in cardiovascular and metabolic disease risk 3839. These metabolic benefits emerge even when aggregate caloric intake remains constant, implying that the specific molecular structure of the diet initiates systemic hormetic signaling cascades entirely independent of simple weight loss or macronutrient ratios 3839.
Pharmacological Hormesis and the Interventions Testing Program
To systematically evaluate the efficacy of xenohormetic compounds and synthetic longevity drugs, the National Institute on Aging (NIA) established the Interventions Testing Program (ITP). Designed to correct the historical lack of reproducibility in aging biology, the ITP utilizes genetically heterogeneous UM-HET3 mice, ensuring that successful interventions are not simply artifacts of inbred genetic quirks. Furthermore, every candidate compound is tested simultaneously across three independent testing sites (The Jackson Laboratory, the University of Michigan, and UT Health San Antonio) using standardized operating procedures, with sufficient statistical power to detect a 10% increase in lifespan 404241.
Lifespan Extension and Sexual Dimorphism
Over two decades of rigorous testing, the ITP has identified numerous compounds that reliably extend median and maximal mammalian lifespan. The most potent pharmaceutical intervention validated to date is rapamycin, an FDA-approved immunosuppressant derived from soil bacteria that acts as a highly specific inhibitor of the mTOR pathway, essentially functioning as a chemical mimetic of caloric restriction 404442. Rapamycin has successfully extended lifespan even when administration begins late in life, at an age equivalent to a 60-year-old human 4442.
A striking and consistent finding from the ITP data is the extreme prevalence of sexual dimorphism in pharmacological hormesis. Many successful compounds extend lifespan exclusively, or predominantly, in only one sex - most frequently male mice 424347.
| ITP Validated Compound | Target / Biological Mechanism | Median Lifespan Effect (Male) | Median Lifespan Effect (Female) |
|---|---|---|---|
| Rapamycin | mTOR inhibition (CR mimetic) | +9% to +23% | +14% to +26% 4447 |
| Acarbose | Alpha-glucosidase inhibition (Glucose blunting) | +22% | +5% 4247 |
| 17α-estradiol | Non-feminizing estrogen, metabolic regulation | +19% | No effect 424144 |
| Astaxanthin | Marine carotenoid (ROS modulation) | +12% | No effect 4447 |
| Meclizine | Antihistamine, theorized mTOR interaction | +8% | No effect 4443 |
| Epicatechin | Cocoa flavanol (Xenohormetic stressor) | +5% | No effect 4347 |
The profound variance in these longevity responses highlights that the metabolic processing of xenobiotics, and the subsequent activation of cellular maintenance pathways, differs significantly by sex. This dimorphism underscores the immense complexity of translating broad pharmacological interventions into generalized clinical practice for human populations 424143.
Targeted Mitophagy and Immune Rejuvenation
A major breakthrough in translating targeted postbiotic and xenohormetic principles directly to human clinical endpoints occurred with the investigation of Urolithin A (UA). UA is not found natively in any food source; rather, it is a secondary metabolite produced exclusively by specific strains of the human gut microbiome following the ingestion of dietary ellagitannins (polyphenols found abundantly in pomegranates, berries, and walnuts). Once metabolized, UA functions systemically as a highly potent inducer of mitophagy - the specialized autophagic clearance and recycling of defective, ROS-leaking mitochondria 444950.
In a pivotal 2025 double-blind, placebo-controlled clinical trial published in Nature Aging (the MitoImmune trial), researchers demonstrated UA's remarkable capacity to counteract immunosenescence, the progressive age-related decline of the immune system. Fifty healthy adults (aged 45 - 70) were administered 1000 mg of UA daily for 28 days 495051. The postbiotic treatment induced a profound functional and structural shift within the peripheral immune compartment. Specifically, UA supplementation triggered the massive expansion of the naïve CD8+ T cell population - the critical, highly adaptable immune sentinels that steadily deplete with advanced age - while simultaneously reducing the expression of cellular markers associated with terminal T cell exhaustion 495152.
Crucially, UA fundamentally reprogrammed the baseline metabolism of these immune cells. While aged, exhausted immune cells typically rely heavily on inefficient glycolysis for rapid energy, SCENITH analysis revealed that the T cells and natural killer (NK) cells in the UA-treated group shifted decisively toward fatty acid and amino acid oxidation 495245.
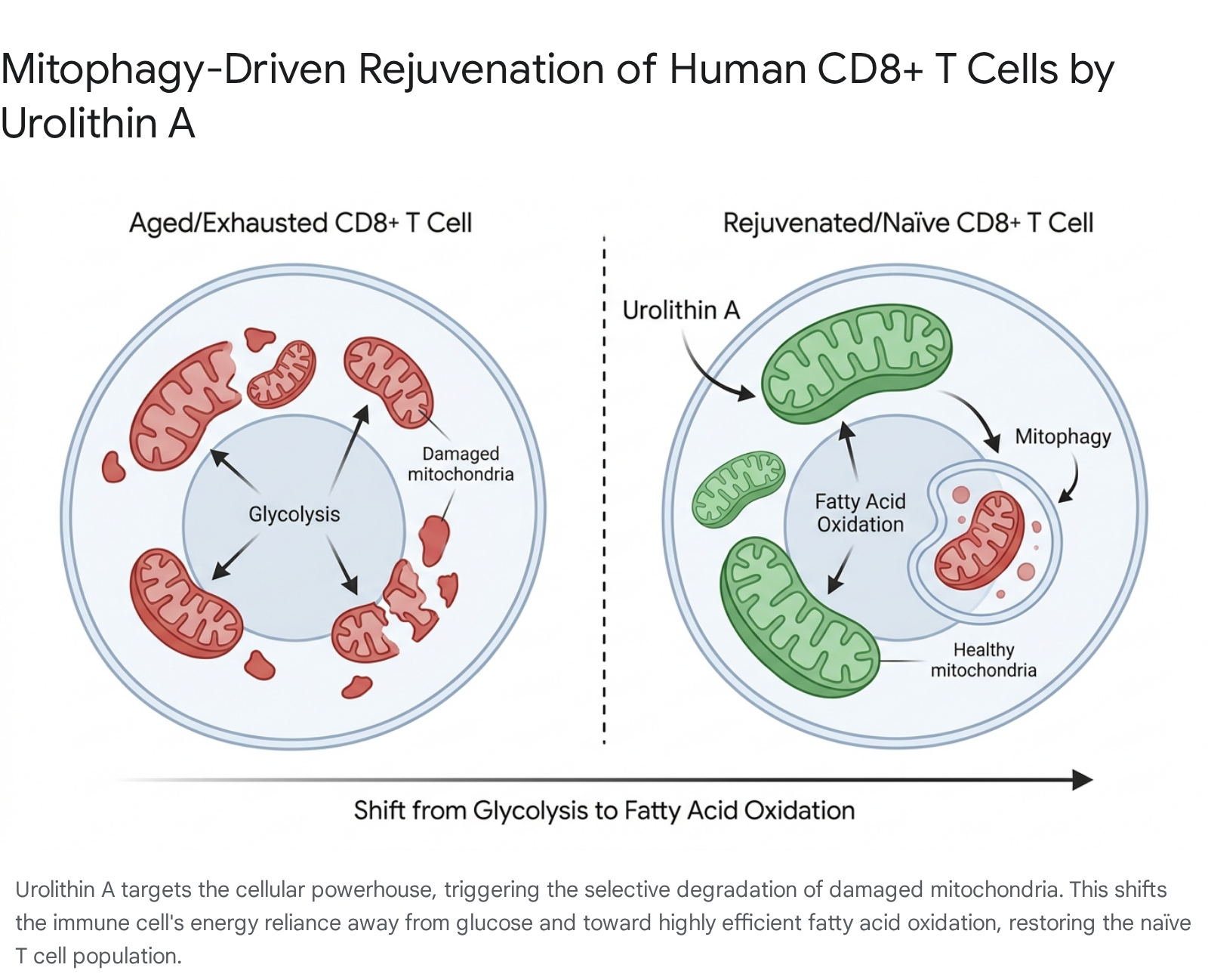
This metabolic pivot reflects a much cleaner, mitochondrial-driven energy architecture. This data provides definitive clinical evidence that up-regulating mitophagy through a specific, microbiome-derived molecular stressor can functionally rejuvenate human immune profiles at a fundamental metabolic level.
Criticisms and Limitations of the Hormetic Framework
Despite the biological elegance of the hormesis concept and its efficacy in highly controlled animal models, the framework remains the subject of intense debate, particularly regarding its application in toxicology, public health regulation, and human clinical trial design.
Toxicological Controversies and the Damage Paradox
Within traditional environmental risk assessment and regulatory science, the dominant models for establishing toxic exposure limits remain the Threshold Dose-Response and the Linear No-Threshold (LNT) models 34. Critics of hormesis argue that the biphasic response is not a universal biological law; because hormetic curves cannot be reliably or consistently predicted across all chemical classes, human cell lines, and diverse environmental conditions, relying on hormesis as a default assumption in public health regulations poses a severe risk of under-regulating hazardous toxicants 46. Some toxicologists caution that many purported hormetic datasets might simply represent post hoc data fitting, random statistical variations, or the artifactual summation of multiple, entirely unrelated toxic mechanisms rather than a unified, beneficial adaptive response 46. Furthermore, an agent that provides a demonstrable hormetic benefit for one physiological endpoint (e.g., preserving cellular growth) might simultaneously induce severe toxicity in another biological system within the same organism 1146.
A persistent theoretical critique within the biology of aging is the "damage paradox." If biological aging is strictly defined by the lifetime accumulation of random molecular damage, introducing further physiological damage via a chemical toxicant or physical stressor should invariably accelerate the overall aging process 47. Proponents of the damage theory argue that the very concept of "damage curing damage" through the activation of repair pathways is inherently paradoxical. However, emerging biogerontological perspectives largely reject the premise that aging is merely the passive accumulation of mechanical wear-and-tear. Instead, aging is increasingly viewed as the progressive dysregulation of vital nutrient-sensing pathways (such as mTOR) and the age-related decline of programmed repair responses. By this logic, acute hormetic stressors correct this signaling dysregulation, forcing the cell into an active, protective state that drastically slows the rate of future damage, yielding a net positive effect on functional longevity despite the initial, acute physiological insult 4748.
Biological Plasticity Limits in Clinical Trials
The quantitative constraints of hormesis pose severe logistical and statistical challenges for modern clinical research. Because human biological plasticity generally limits true hormetic improvements to a maximum of 30% to 60% above the control baseline, these relatively modest physiological effects are highly vulnerable to being masked by the statistical noise inherent in large, heterogeneous human trials 678. Modern clinical trials are fundamentally designed and powered to detect massive, acute pharmaceutical effects - often requiring responses 200% to 300% above control levels to achieve statistical significance 78.
Consequently, researchers suggest that the rigid statistical structure of standard randomized controlled trials routinely produces a high rate of "false negatives" regarding hormetic lifestyle interventions or low-dose supplements. A compound might successfully engage cellular maintenance pathways and provide a genuine 25% physiological improvement in target tissues, but fail to meet the trial's aggressively calculated primary endpoints, leading to the erroneous clinical conclusion that the intervention is entirely ineffective 78.
Precision Hormesis and Computational Aging Clocks
The realization that human physiological responses to stress interventions are vastly heterogeneous has birthed the emerging field of precision hormesis. The ultimate goal of this discipline is to move beyond generic, population-level health recommendations and to precisely calibrate the specific type, dose, and frequency of a stressor based on an individual's unique molecular profile, genetic background, and current biological age 495850.
Single-Cell Multi-Omics and Biomarker Identification
The deployment of single-cell multi-omics (SCMO) has fundamentally transformed the cellular resolution of aging research. Techniques such as single-cell RNA sequencing (scRNA-seq), Cellular Indexing of Transcriptomes and Epitopes by Sequencing (CITE-seq), and single-cell metabolomics allow researchers to decode deep metabolic heterogeneity across diverse cell populations within the exact same tissue sample 2049. Rather than measuring a blunt, bulk tissue average, researchers can now detect highly subtle molecular shifts - such as the gradual expansion of CD16+ monocytes, the exhaustion of specific CD8+ T cells bearing CD57 and PD-1 markers, or the epigenetic priming of stress-related genes in skeletal muscle stem cells - years before macroscopic clinical pathology physically manifests 2049.
This high-resolution molecular profiling allows for the establishment of highly complex composite biomarkers. For example, identifying elevated levels of specific polyamines like spermine (tightly associated with chromatin structural protection) or mapping specific NAD+ and glutathione metabolite signatures can pinpoint exactly where an individual currently resides on the continuum of cellular senescence 20.
Artificial Intelligence and Deep Aging Clocks
To successfully translate this massive volume of high-dimensional omics data into actionable clinical utility, the field is rapidly integrating deep learning (DL) algorithms and Generative Artificial Intelligence (GenAI). These advanced computational models ingest massive, diverse multimodal datasets - ranging from granular DNA methylation patterns to routine clinical blood panels - to generate highly accurate "deep aging clocks" (DACs) 5152.
For instance, composite clinical biomarkers like the DNAm PhenoAge clock combine advanced epigenetic methylation analysis with 10 standard, easily accessible physiological markers (including chronological age, albumin, creatinine, glucose, C-reactive protein, lymphocyte percentage, mean corpuscular volume, red blood cell distribution width, alkaline phosphatase, and white blood cell count). This algorithmic synthesis provides a highly precise estimation of an individual's true biological age versus their chronological age 17. Furthermore, AI-driven natural language processing (NLP) models, utilizing techniques like Latent Dirichlet Allocation (LDA) and term frequency-inverse document frequency (TF-IDF) analysis, are currently parsing nearly a century of historical aging research. These models consistently highlight critical translational gaps, demonstrating that clinical geriatric outcomes in human trials frequently fail to map back to foundational cellular mechanisms - like autophagy or mitohormesis - without the aid of explicit, AI-driven predictive modeling bridging the gap 5354.
By establishing real-time data feedback loops via advanced clinical biomarker panels, precision hormesis aims to accurately map the exact boundaries of an individual patient's inverted-U dose-response curve 58. An older patient presenting with high basal levels of systemic inflammation (inflammaging) may have a severely compressed homeodynamic space; therefore, a standard dose of intense physical exercise or extreme thermal stress that is highly beneficial for a younger adult could easily cross the threshold into severe toxicity for them 21058. Tailoring the anti-aging intervention - perhaps by utilizing specific pharmacological CR mimetics or careful xenohormetic dosing to build basal resilience before attempting aggressive physical stressors - offers a precise, data-driven pathway to safely re-expand their homeodynamic resilience and effectively close the translational gap between maximum lifespan and active healthspan 205850.