History and replacement of the free radical theory of aging
The biological mechanisms governing the aging process have been the subject of intensive scientific inquiry for over a century. Throughout the latter half of the twentieth century, the prevailing paradigm to explain the progressive, time-dependent decline in physiological integrity was the free radical theory of aging (FRTA). Originally formulated in the 1950s and subsequently refined into the mitochondrial free radical theory of aging in the 1970s, this hypothesis posited a linear, causal relationship between the accumulation of oxidative damage and organismal senescence. For decades, the theory profoundly influenced both basic biological research and public health recommendations, driving billions of dollars in funding and facilitating the creation of a massive global antioxidant supplement industry.
However, as molecular biology, genome sequencing, and comparative zoology advanced in the late twentieth and early twenty-first centuries, a substantial body of contradictory evidence began to emerge. Observations of exceptionally long-lived species, genetic manipulation of model organisms, and large-scale, randomized human clinical trials consistently failed to support the central tenets of the FRTA. Consequently, the field of biogerontology has undergone a fundamental paradigm shift. The linear damage model has been largely abandoned in favor of complex, systemic frameworks, most notably the "Hallmarks of Aging," which view reactive oxygen species (ROS) not merely as toxic metabolic byproducts, but as critical signaling molecules essential for cellular homeostasis, adaptation, and survival.
Historical Context and Theoretical Genesis
The pursuit of a universal, mechanistic theory of aging in the mid-twentieth century was heavily influenced by earlier physiological concepts. Prominent among these was the "rate of living" theory, originally proposed in the 1920s, which suggested that an organism's lifespan was inversely proportional to its basal metabolic rate and, by extension, its total lifetime oxygen consumption 123. While the rate of living theory provided a macroscopic observation of cross-species lifespans, it lacked a molecular mechanism to explain how cellular degradation actually occurred.
Initial Formulation by Denham Harman
In 1956, Denham Harman, a researcher at the Donner Laboratory of Medical Physics at the University of California, Berkeley, provided this missing mechanism by formally proposing the free radical theory of aging 3456. Harman's hypothesis was deeply inspired by the burgeoning field of radiation chemistry. He specifically drew upon the observation by Rebeca Gerschman that the toxic effects of both hyperbaric oxygen and ionizing radiation could be attributed to a common underlying phenomenon: the generation of oxygen free radicals 12.
Harman noted the striking phenomenological parallels between the effects of acute ionizing radiation and the chronic physiological changes associated with aging, including mutagenesis, cellular membrane degradation, and the development of cancer 36. At the time, early paramagnetic resonance spectroscopy had recently confirmed the in vivo presence of highly reactive hydroxyl radicals in living matter 36. A free radical is defined in chemistry as any atom or molecule possessing a single unpaired electron in an outer orbital shell, rendering it chemically unstable and highly reactive as it seeks to steal an electron from neighboring molecules 12.
Harman theorized that these free radicals, generated continuously as inevitable byproducts of normal aerobic metabolism and biological oxidation-reduction reactions, would relentlessly attack adjacent cellular constituents 26. Over an organism's lifespan, the cumulative deleterious side attacks on lipids (causing lipid peroxidation), proteins (creating carbonyl derivatives), and nuclear DNA would lead to a progressive loss of organismal functionality, culminating in the degenerative diseases of aging and ultimate death 126.
The Mitochondrial Free Radical Theory of Aging
While Harman's original 1956 hypothesis implicated free radicals generally, it did not specify their primary subcellular origin. In 1972, Harman refined and extended his model into the Mitochondrial Free Radical Theory of Aging (MFRTA) 178. This critical revision was driven by the recognition that approximately 95% of cellular energy is produced within the mitochondria via oxidative phosphorylation (OXPHOS), a process that utilizes 97% to 99% of total cellular oxygen consumption 2.
During the electron transport chain (ETC) cycle, specifically at respiratory Complexes I and III, a small fraction of electrons prematurely "leak" and react with molecular oxygen to form the superoxide anion ($O_2^{\bullet-}$). Historically, this leakage was estimated at 1% to 3% of total oxygen consumed, though modern in vivo estimates frequently place it lower, between 0.15% and 5% depending on the metabolic state of the cell 2789. Superoxide serves as the progenitor for a cascade of other reactive oxygen species (ROS), including hydrogen peroxide ($H_2O_2$) and the highly reactive hydroxyl radical ($^{\bullet}OH$) 71011.
The MFRTA posited a self-amplifying cycle of structural destruction. Because mitochondria are the primary intracellular producers of ROS, they are simultaneously the primary targets of oxidative damage 1011. Furthermore, mitochondrial DNA (mtDNA) is situated in close physical proximity to the ROS-generating ETC complexes, lacks the protective histones found in nuclear DNA, and possesses limited repair capacity 11011. According to the theory, accumulating mtDNA mutations would result in the synthesis of defective respiratory chain subunits. These defective subunits would further impair ETC efficiency, thereby increasing the rate of electron leakage and ROS production. This "vicious cycle" of escalating oxidative stress and exponential mitochondrial dysfunction was widely accepted for decades as the primary biological clock dictating maximum lifespan potential 31011.
Accumulation of Contradictory Evidence
By the turn of the twenty-first century, the tools available for genetic sequencing, molecular manipulation, and comparative zoology had advanced significantly. As researchers attempted to validate the MFRTA through rigorous, targeted experimentation, the empirical data increasingly conflicted with the theory's foundational predictions. The core assumption of the MFRTA was simple: if the accumulation of ROS-induced damage was the fundamental cause of aging, then suppressing ROS or enhancing antioxidant defenses should universally extend lifespan, while increasing ROS should prematurely accelerate aging. Systematic testing revealed that neither prediction held true consistently across species.
Genetic Manipulations in Model Organisms
Experimental models utilizing yeast, the nematode Caenorhabditis elegans, Drosophila melanogaster (fruit flies), and laboratory mice were genetically engineered to test the MFRTA. Initial, less sophisticated studies seemed promising, as the overexpression of certain antioxidant enzymes in Drosophila occasionally yielded modest lifespan extensions 8. However, systematic evaluations in complex mammalian models proved devastating to the theory.
Researchers generated 18 distinct genetic alterations in mice designed to selectively block or severely compromise endogenous antioxidant defenses 1. According to the MFRTA, these mice should have experienced catastrophic, unregulated oxidative damage and dramatically shortened lifespans. However, empirical observation showed that only one of the 18 alterations - the deletion of the SOD-1 (superoxide dismutase 1) gene - resulted in a shortened lifespan 1. In other experiments, heterozygous mice with reduced expression of the specific mitochondrial antioxidant SOD2 accumulated severe oxidative damage to their DNA and developed high incidences of cancer, yet their biological aging rate did not accelerate, and their lifespans were statistically indistinguishable from wild-type mice when cancer mortality was excluded 812. Conversely, the transgenic overexpression of antioxidant enzymes successfully reduced cellular oxidative stress biomarkers but reliably failed to increase the maximum lifespan of mice 81213.
Even more perplexing were findings generated using C. elegans. Mutations in the isp-1 and nuo-6 genes, which affect the structural components of the mitochondrial electron transport chain, resulted in an overall enhanced respiration rate and a significant increase in mitochondrial superoxide levels and overall ROS formation 1014. Rather than dying prematurely from oxidative toxicity, these mutant worms exhibited profound lifespan extensions 1014. When researchers treated these long-lived, high-ROS worms with chemical antioxidant compounds that neutralized the ROS, their lifespan extension was completely abolished 1014. This suggested a strong positive correlation between specific forms of mitochondrial oxidative stress and longevity, a finding that directly inverted the MFRTA paradigm.
Comparative Biology and the Naked Mole-Rat Paradox
The study of comparative biogerontology delivered another profound challenge to the oxidative stress theory through the East African naked mole-rat (Heterocephalus glaber). In mammalian biology, maximum lifespan generally scales predictably with body mass. The naked mole-rat, despite being roughly the size of a standard laboratory mouse, boasts a maximum recorded lifespan exceeding 30 years in captivity - approximately eight to ten times longer than a comparative mouse 121516.
If the MFRTA were correct, the naked mole-rat's exceptional longevity would be explained by remarkably low ROS production, exceptionally robust antioxidant defenses, and minimal baseline oxidative damage. Rigorous empirical assessments revealed the exact opposite. Naked mole-rats generate ROS at rates similar to, or slightly higher than, mice 131516. Their endogenous antioxidant defense mechanisms are unexceptional and not demonstrably superior to those of shorter-lived, physiologically age-matched rodents 1315. Most strikingly, even under constitutive conditions from a very young age, naked mole-rats tolerate a pro-oxidant intracellular environment and carry extremely high, steady-state levels of oxidative damage to their cellular lipids, proteins, and DNA - damage levels that would be highly pathological or lethal in mice 12131517.
These observations created a profound paradox for geriatric researchers 1517. The data indicated that neither the absolute amount of ROS produced nor the sensitivity of the organism in neutralizing those ROS dictates longevity 1213. Instead, longevity appears highly dependent upon downstream cytoprotective mechanisms and robust homeostasis-maintaining activities that allow an organism to tolerate severe oxidative damage without it translating into fatal age-associated diseases, such as cancer or neurodegeneration 121315. Theoretical models based on these observations suggest that the naked mole-rat's extended lifespan may be a function of an evolutionary energy tradeoff, allocating metabolic resources away from rapid growth and heavily toward somatic maintenance and damage repair tolerance 1517.
Clinical Trials of Antioxidant Supplementation
The widespread belief that neutralizing free radicals could delay human aging and prevent chronic degenerative diseases spurred the initiation of massive, government-funded clinical trials in the 1980s and 1990s. Early epidemiological data had shown that human populations consuming diets rich in fruits and vegetables - which inherently contain high levels of naturally occurring antioxidants - enjoyed lower rates of cancer and cardiovascular disease 1819. Based directly on the mechanistic logic of the FRTA, researchers hypothesized that supplementing individuals with isolated, high-dose antioxidant vitamins would replicate and even enhance these protective physiological effects.
The results of these double-blind, placebo-controlled trials were not merely statistically disappointing; they revealed active harm in treated cohorts, delivering the final, definitive blow to the clinical applicability of the free radical theory.
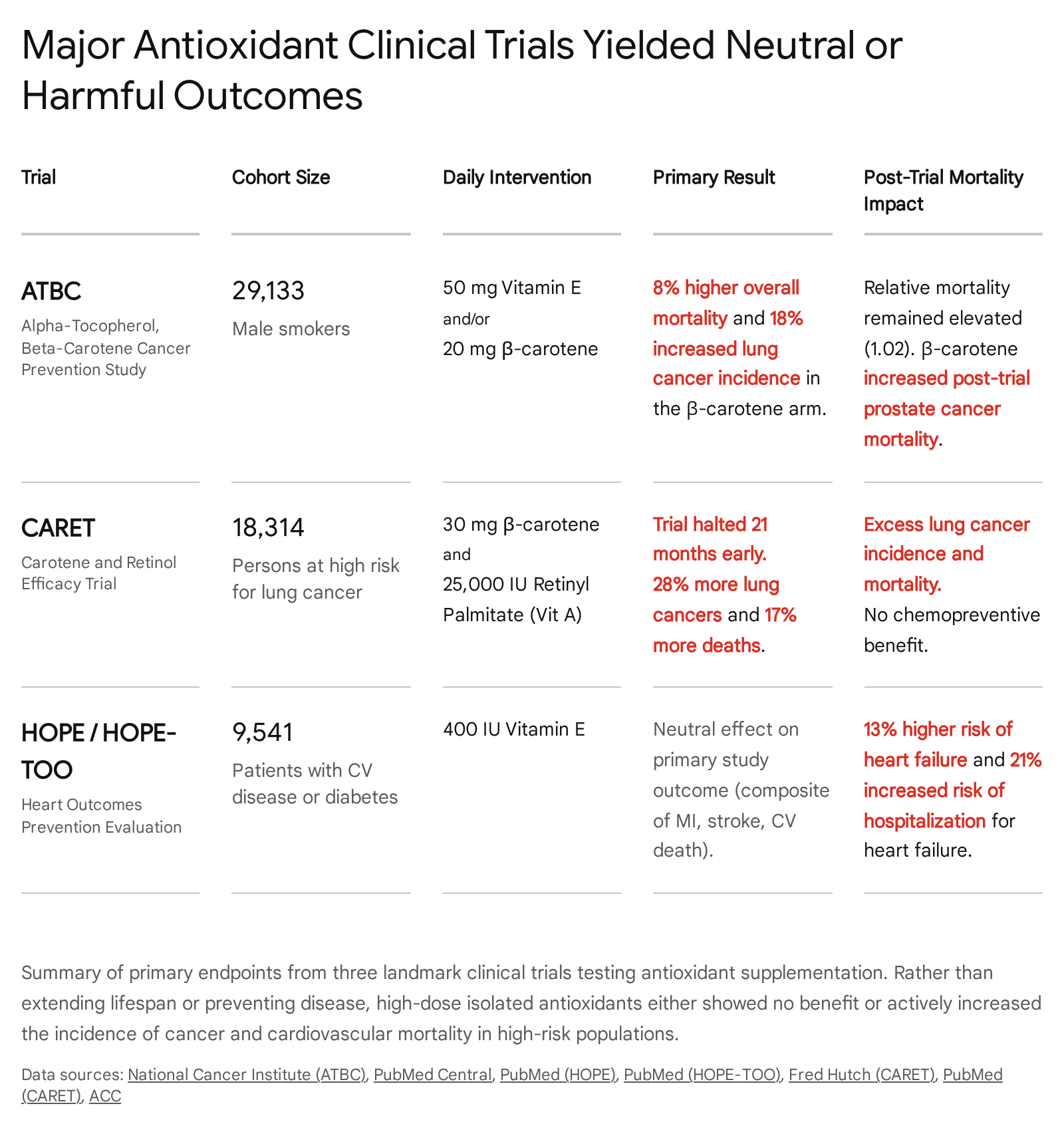
The Alpha-Tocopherol, Beta-Carotene Cancer Prevention (ATBC) Study
Initiated in 1985 as a joint project between the U.S. National Cancer Institute and the National Public Health Institute of Finland, the ATBC study sought to determine whether daily supplementation with specific vitamins could prevent lung and other incident cancers 1820. The trial enrolled an massive cohort of 29,133 Finnish men, aged 50 to 69, who were active smokers 182021. Participants were randomized in a strict 2x2 factorial design to receive 50 milligrams of vitamin E (alpha-tocopherol), 20 milligrams of beta-carotene (a vitamin A precursor), both supplements, or a placebo capsule daily for a median duration of 6.1 years 182021.
The trial results stunned the preventive medicine community. Rather than exerting a protective effect against cellular oxidation, beta-carotene supplementation resulted in an 18% increase in the incidence of lung cancer and an 8% increase in overall mortality among the treatment arm 1819. While vitamin E supplementation did demonstrate a 32% lower incidence of prostate cancer during the trial phase, it had absolutely no significant beneficial effect on total mortality 1822. Long-term, post-intervention follow-up spanning 18 years, utilizing the Finnish Cancer Registry, confirmed that the antioxidant supplements had no positive late effects on overall cancer incidence or lifespan extension; post-trial relative mortality remained persistently elevated at 1.02 (95% CI, 0.99 - 1.05) for beta-carotene recipients compared to nonrecipients 19.
The Carotene and Retinol Efficacy Trial (CARET)
Running concurrently in the United States, the CARET trial evaluated the efficacy of 30 mg of beta-carotene and 25,000 IU of retinyl palmitate (vitamin A) administered daily 2324252627. The cohort consisted of 18,314 individuals determined to be at high risk for lung cancer, specifically comprising heavy smokers and men with extensive occupational asbestos exposure histories 232425.
In January 1996, the CARET intervention was abruptly halted 21 months ahead of schedule by its safety monitoring board 23242526. The data monitoring committee found definitive evidence of no clinical benefit, coupled with substantial, statistically significant evidence of severe harm: participants receiving the active antioxidant combination exhibited a 28% higher incidence of lung cancer and a 17% increase in total mortality from all causes compared to the placebo control group 2324252627. Furthermore, deaths specifically attributable to cardiovascular disease were 26% higher in the treatment arm (Relative Risk = 1.26; 95% CI, 0.99 to 1.61) 27.
The Heart Outcomes Prevention Evaluation (HOPE) Trial
While the ATBC and CARET trials focused heavily on chemoprevention and cancer incidence, the HOPE trial explicitly investigated the cardiovascular benefits of antioxidant supplementation. The trial enrolled 9,541 high-risk patients, aged 55 or older, who had preexisting vascular disease or diabetes complicated by at least one additional coronary risk factor 28293031. Participants received either 400 IU of natural-source vitamin E or a placebo daily, with a mean follow-up of 4.5 years 282930.
Vitamin E supplementation had a strictly neutral effect on the primary composite outcome of myocardial infarction, stroke, or cardiovascular death (Relative Risk = 1.05, p=0.33) 283031. However, an extended follow-up phase of the trial, known as HOPE-TOO (The Ongoing Outcomes), which tracked 7,030 patients for a median of 7.0 years, revealed alarming secondary outcomes 2932. Patients in the long-term vitamin E arm experienced a statistically significant 13% higher risk of incident heart failure (p=0.03) and a 21% increased risk of hospitalization for heart failure (p=0.045) 2931. The trial's chief investigator publicly stated that the extended data proved vitamin E provided no benefit and caused active harm, suggesting that high-dose vitamins should be regulated with the stringency of pharmaceutical drugs 32.
Systematic Reviews and Meta-Analyses on Mortality
To systematically synthesize the vast amount of clinical data generated over two decades, the Cochrane Collaboration conducted rigorous meta-analyses assessing antioxidant supplements for the primary and secondary prevention of mortality 3334343536. An updated comprehensive review evaluated 78 randomized clinical trials encompassing 296,707 total participants, including both healthy individuals and patients with stable chronic diseases 333436.
The findings were unequivocal and effectively closed the debate regarding the clinical utility of the FRTA. Antioxidant supplements do not prolong life or reduce mortality 333435. When the analysis was restricted to the 56 trials characterized by a low risk of methodological bias, the data showed that antioxidant supplementation significantly increased mortality by 4% to 5% (Relative Risk = 1.04; 95% CI 1.01 to 1.07) 33343436. Specifically, beta-carotene, vitamin E, and higher doses of vitamin A were all independently associated with an increased risk of death 33343435. The review concluded with a stark recommendation that these antioxidant supplements should be considered medicinal products requiring stringent safety evaluations before commercial marketing 333435.
Summary of Key Antioxidant Clinical Trials and Interventions
| Trial Name | Cohort Profile | Daily Intervention | Primary Outcomes and Post-Trial Impact |
|---|---|---|---|
| ATBC (1985 - 1993) | 29,133 male smokers (Finland) | 50 mg Vitamin E, 20 mg Beta-carotene | 8% increase in overall mortality; 18% increase in lung cancer (beta-carotene arm). |
| CARET (1985 - 1996) | 18,314 asbestos workers and smokers (US) | 30 mg Beta-carotene, 25,000 IU Retinyl palmitate | Halted early. 17% increase in total mortality; 28% increase in lung cancer. |
| HOPE / HOPE-TOO (1993 - 2003) | 9,541 high cardiovascular risk patients | 400 IU Vitamin E | No reduction in cardiovascular events; 13% increase in heart failure; 21% increase in heart failure hospitalizations. |
| Cochrane Review (2012 update) | 78 trials, 296,707 participants | Various (Beta-carotene, Vitamins A, C, E, Selenium) | 4% increase in mortality across 56 low-bias trials. No evidence to support primary or secondary prevention. |
The Modern Paradigm of Redox Biology
The abject failure of the FRTA to withstand experimental and clinical scrutiny forced a comprehensive reevaluation of redox biology. The fundamental scientific error of the free radical theory lay in its overly simplistic characterization of ROS as purely destructive, unwanted metabolic waste products. Modern molecular biology has revealed that this view is fundamentally flawed.
Reactive Oxygen Species as Essential Signaling Molecules
Rather than acting merely as stochastic agents of cellular degradation, reactive oxygen species - particularly hydrogen peroxide and superoxide - are essential, highly regulated secondary signaling molecules 791037. Mitochondria utilize the precise, localized production of ROS at Complexes I and III to communicate their energetic and metabolic status to the nucleus, a critical biological process known as mitonuclear communication 91038.
These redox signals govern critical physiological processes, including stem cell differentiation, immune system activation against pathogens, insulin sensitivity, and adaptive stress responses 791038. For example, the transient bursts of ROS generated by mitochondria during physical exercise act as vital molecular signals that trigger beneficial adaptations, such as improved fatty acid metabolism and muscle repair 7939. High doses of synthetic antioxidants, such as high-dose vitamin C and E, actively scavenge and neutralize these vital signaling molecules, effectively "blinding" the cell to metabolic stress and preventing the beneficial adaptations of exercise and dietary restriction 791440. Therefore, the age-related loss of precise, localized redox signaling is now considered a significant driver of aging, and indiscriminate antioxidant supplementation exacerbates, rather than resolves, this signaling decay 940.
The Concept of Mitohormesis
The recognition of ROS as critical signaling molecules gave rise to the concept of mitochondrial hormesis, or "mitohormesis." Hormesis describes a universal biological phenomenon characterized by a non-linear, biphasic dose-response relationship: exposure to a low or moderate dose of a stressor provokes a compensatory biological defense response that ultimately leaves the organism healthier and more resilient than it was prior to the stress exposure 7143841.
The theoretical relationship between ROS and mortality shifts from a linear progression - where any increase in ROS accelerates aging, as proposed by the FRTA - to a J-shaped or U-shaped biphasic curve. In this mitohormetic model, low to moderate levels of ROS decrease mortality by triggering adaptive responses, whereas only chronically elevated, unregulated ROS levels overwhelm cellular defenses and lead to pathology 741.
In the specific context of aging, mitohormesis occurs when mild mitochondrial dysfunction or mildly elevated ROS levels act as an initial stressor 3841. This stress triggers the activation of retrograde signaling pathways to the nucleus, aggressively upregulating gene expression related to the unfolded protein response (UPR), DNA repair mechanisms, and the organism's endogenous antioxidant network (such as superoxide dismutase and catalase) 7838. This systemic upregulation of defense mechanisms confers broad stress resistance and reliably extends lifespan in model organisms 738. It is precisely this mitohormetic response that mediates the well-documented, life-extending benefits of caloric restriction, physical exercise, and transient hypoxia 7. Thus, ROS increase with age not primarily because they are the root cause of aging, but because they are deliberately generated by the cell to combat the deteriorating cellular environment and to stimulate survival pathways 14.
Integrated Frameworks Replacing the Free Radical Theory
With the obsolescence of the FRTA, researchers sought a framework capable of encompassing the vast, interconnected complexity of senescence. The result was a definitive shift away from single-cause (monocausal) theories of aging toward integrated, systems-biology approaches that recognize aging as a network of failing homeostatic systems.
The Hallmarks of Aging
The definitive conceptual replacement for the free radical theory is the "Hallmarks of Aging" paradigm, first articulated by Carlos López-Otín and colleagues in a landmark 2013 paper and comprehensively updated in 2023 4243. This framework categorizes the biological drivers of aging into an interconnected network of time-dependent cellular manifestations. To be classified as a hallmark, a biological process must fulfill three criteria: it must manifest during normal physiological aging, its experimental aggravation must accelerate aging, and its therapeutic alleviation must decelerate or reverse aging 4245.
The 2023 update recognizes twelve distinct hallmarks, structurally divided into three hierarchical categories based on their role in the sequence of physiological decline 424544:
| Category | Specific Hallmarks | Biological Role |
|---|---|---|
| Primary Hallmarks (The Initiators) | Genomic Instability, Telomere Attrition, Epigenetic Alterations, Loss of Proteostasis, Disabled Macroautophagy. | Intrinsically negative damages that accumulate over time. These are the root causes of age-related cellular decline. 414547 |
| Antagonistic Hallmarks (The Responses) | Deregulated Nutrient-Sensing, Mitochondrial Dysfunction, Cellular Senescence. | Evolved as protective, compensatory mechanisms to mitigate primary damage. Beneficial at low levels in youth, but become deleterious when hyperactivated or chronically sustained. 414547 |
| Integrative Hallmarks (The Phenotypes) | Stem Cell Exhaustion, Altered Intercellular Communication, Chronic Inflammation (Inflammaging), Dysbiosis. | The downstream, systemic consequences that arise when tissue homeostasis completely fails due to the accumulated weight of the previous hallmarks. 42454447 |
By reclassifying mitochondrial dysfunction as an antagonistic hallmark rather than a primary driver, the framework resolves the contradictions that collapsed the FRTA 4147. It acknowledges that mitochondrial ROS production and the subsequent senescence of damaged cells are vital defense mechanisms early in life that prevent the unchecked proliferation of mutated, cancerous cells 394147. It is only later in life, when the clearance of these senescent cells fails and mitochondrial damage becomes pervasive and unregulated, that the compensatory mechanisms tip into pathology, driving chronic inflammation and accelerating systemic decline 39414845.
Epigenetic Alterations and Cellular Reprogramming
Among the primary hallmarks, epigenetic alterations have emerged as a profound mechanism of aging, significantly expanding the field's understanding beyond the structural DNA damage model of the FRTA 342464748. The epigenome acts as the regulatory software of the cell, determining which specific genes are expressed and which are silenced via mechanisms such as DNA methylation, non-coding RNAs, and histone modification 464748. During aging, the enzymes responsible for maintaining this code (such as DNMT1 and DNMT3a) become less efficient 46.
During aging, cells experience "epigenetic drift" - a progressive, predictable degradation of these precise methylation patterns involving both global hypomethylation and localized hypermethylation 4648. This leads to a gradual loss of cellular identity, where cells forget their specialized functions and begin expressing inappropriate genetic sequences 4748. The precision of this degradation has allowed researchers like Steve Horvath to create "epigenetic clocks," algorithms that utilize DNA methylation data to calculate a tissue's chronological and biological age with remarkable accuracy, predicting mortality risk independent of standard blood markers 4648. Because epigenetic modifications do not alter the underlying nucleotide sequence of DNA, they are theoretically reversible 4748. Experimental interventions utilizing cellular reprogramming (e.g., exposing cells to Yamanaka factors) have successfully reversed epigenetic age profiles in model organisms, demonstrating that aging is heavily governed by reversible gene regulation errors rather than strictly irreversible oxidative physical damage 48.
Nutrient Sensing, Autophagy, and the mTOR Pathway
Another dominant framework that has fundamentally superseded the FRTA is the study of the mechanistic Target of Rapamycin (mTOR) signaling pathway, which acts as the master regulator of cellular metabolism 49505152. Operating primarily within the "deregulated nutrient sensing" hallmark, mTOR integrates signals from nutrient availability, insulin, and environmental stress to balance cellular growth (anabolism) against maintenance and repair (catabolism) 495051.
When nutrients, particularly amino acids, are abundant, mTOR Complex 1 (mTORC1) is highly activated, driving protein synthesis and cellular proliferation 495051. Concurrently, activated mTORC1 aggressively suppresses macroautophagy - the vital cellular mechanism responsible for engulfing and recycling misfolded proteins and dysfunctional organelles, including damaged mitochondria (a process specifically termed mitophagy) 41495052. While this growth-oriented state is highly beneficial during developmental stages, the chronic hyperactivation of mTOR in adulthood accelerates aging. It causes a toxic accumulation of cellular debris, depletes stem cell pools, and drives viable cells into a state of permanent senescence (geroconversion) 495052.
Inhibition of the mTOR pathway - whether through dietary interventions like caloric restriction or pharmacological agents like rapamycin - is currently the most robust and highly conserved method known to extend lifespan across multiple species, from yeast to mammals 4950515257. By downregulating mTOR activity, cells are forced into a state of metabolic conservation, which drastically upregulates autophagy and enhances cellular resistance to all forms of stress, including oxidative damage 495057. This mechanism explains why reducing energy intake extends life, completely shifting the gerontological focus from attempting to mitigate free radical production to enhancing cellular maintenance, quality control, and recycling networks 1549.
Commercial and Regulatory Divergence
Despite the overwhelming clinical evidence invalidating the free radical theory of aging as a basis for high-dose antioxidant therapy, the scientific consensus has failed to deeply penetrate the broader consumer market.
The Global Antioxidant Supplement Market
The global antioxidant supplement market continues to expand at a rapid and accelerating pace. Valued at approximately $5.34 billion in 2024, the market is projected to reach over $10.02 billion by 2032, growing at a compound annual growth rate (CAGR) of over 8% 535455. This exponential economic growth is fueled largely by an aging global population seeking over-the-counter anti-aging remedies, coupled with intense marketing campaigns that continue to promote the outdated, intuitively appealing linear damage model of the FRTA 545561.
Commercial messaging successfully exploits the simplicity of the FRTA - framing free radicals as absolute toxins and antioxidants as absolute biological cures 56. This widespread consumer adoption leads to the physiological phenomenon of "antioxidative stress," wherein health-conscious individuals ingest excessive, unregulated levels of synthetic vitamins (such as A, C, and E), unwittingly suppressing their endogenous mitohormetic signaling pathways and potentially increasing their risk of mortality, exactly as demonstrated in the Cochrane meta-analyses 32334056.
Regulatory Landscapes: FDA versus EFSA
The persistence of the antioxidant supplement industry is heavily facilitated by a highly fragmented global regulatory landscape, particularly the divergent legal and evidentiary standards between the United States and the European Union 57585960.
Comparison of Regulatory Frameworks for Antioxidant Claims
| Regulatory Body | Legal Framework | Pre-Market Approval Required | Allowable Claims & Evidentiary Standards |
|---|---|---|---|
| U.S. Food and Drug Administration (FDA) | Dietary Supplement Health and Education Act (DSHEA) of 1994. | No pre-market approval required for dietary supplements. | Allows "Structure/Function" claims (e.g., "supports cellular health") with a disclaimer. Allows "Nutrient Content" claims (e.g., "High in Antioxidant Vitamin C") if the product contains an established Daily Value of recognized nutrients. 58596162 |
| European Food Safety Authority (EFSA) | Regulation (EC) No 1924/2006. | Strict pre-market scientific evaluation required by the NDA Panel. | Prohibits general "anti-aging" claims. Rejects in vitro antioxidant capacity (e.g., ORAC scores) as proof of health benefit. Requires in vivo human trials proving protection of specific molecules to authorize a health claim. 57586364 |
In the United States, the FDA regulates dietary supplements under the DSHEA, which treats supplements more akin to food products than pharmaceuticals, relying primarily on post-market surveillance 59. Under this framework, manufacturers are permitted to make broad "structure/function" claims provided they do not explicitly claim to cure or prevent specific diseases 585961. This leniency allows companies to legally market antioxidants for generic "anti-aging" benefits without conducting clinical trials to prove actual physiological efficacy 58596165.
Conversely, the EFSA enforces a considerably stricter, science-first regulatory regime 575859. Under EU regulations, any claim - whether it relates to basic nutrition, specific organ functions, or disease risk reduction - must undergo rigorous scientific substantiation by the EFSA before it can be legally utilized 5758596466. The EFSA has explicitly addressed the scientific failure of the free radical theory in its regulatory guidance. When assessing applications for antioxidant health claims, the EFSA has consistently ruled that demonstrating "antioxidant activity" in vitro (in a test tube, using assays like ORAC or TRAP) is biologically irrelevant and cannot be used to substantiate a human health claim 616364. Furthermore, the EFSA has concluded that claims referring generally to the "protection of cells from premature aging" are scientifically meaningless and do not comply with EU criteria 6364. Due to the consistent failure of antioxidants to demonstrate objective physiological benefits in human clinical trials, the vast majority of antioxidant health claim applications to the EFSA have been rejected 6166.
Ultimately, the free radical theory of aging represents one of the most prominent cautionary tales in modern biology. The transition from the FRTA's linear damage model to the nuanced, systems-level understanding of the Hallmarks of Aging and the mitohormesis framework highlights the profound evolutionary complexity of biological systems, where stress, damage, and reactive molecules are inextricably linked to survival and adaptation.